Abstract
The coronavirus disease of 2019 (COVID-19) has caused significant morbidity and mortality among infected individuals across the world. High transmissibility rate of the causative virus – Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) – has led to immense strain and bottlenecking of the health care system. While noteworthy advances in vaccine development have been made amid the current global pandemic, most therapeutic agents are repurposed from use in other viral infections and are being evaluated for efficacy in COVID-19. Favipiravir, an orally administered drug originally developed in Japan against emerging influenza viral strains, has been shown to have widespread application and safety across multiple ribonucleic acid (RNA) viral infections. With a strong affinity toward the viral RNA-dependent RNA polymerase (RdRp), favipiravir could be a promising therapy against SARS-CoV-2, by targeting downstream viral RNA replication. Initial trials for usage in COVID-19 have suggested that favipiravir administration during initial infection stages, in individuals with mild to moderate infection, has a strong potential to improve clinical outcomes. However, additional well-designed clinical trials are required to closely examine ideal timing of drug administration, dosage, and duration, to assess the role of favipiravir in COVID-19 therapy. This review provides evidence-based insights and throws light on the current clinical trials examining the efficacy of favipiravir in tackling COVID-19, including its mechanism, pharmacodynamics, and pharmacokinetics.
Introduction
SARS-CoV-2 and its anatomy
The coronavirus disease of 2019 (COVID-19) has infected over 242 million people, leading to over 4.9 million deaths worldwide, at the time of this publication.
1
It is caused by the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2),2,3 closely related to Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV) and Middle East Respiratory Syndrome Coronavirus (MERS-CoV).3,4 SARS-CoV-2 is a single-stranded positive sense ribonucleic acid (RNA) virus, 50–200 nm in diameter, consists of four structural proteins (about 33% of viral genome) – Spike (S), Envelope (E), Membrane (M), and Nucleocapsid (N).
5
The S protein, comprising of S1 and S2 subunits, mediates viral entry into the host cell membrane
COVID-19 pathogenesis and symptoms
COVID-19 is a multisystemic viral disease and commonly affects the respiratory tract. 13 SARS-CoV-2 is transmitted mainly through droplets and exhibits pulmonary tropism, which is attributable to ACE2 receptors being widely distributed across lung epithelial cells. 14 Clinical manifestations in human hosts can be wide ranging from asymptomatic to symptomatic disease with potential for sudden symptom evolution.12,14 Symptoms of mild disease may include fever, myalgia, cough, fatigue, anosmia, dysgeusia, and shortness of breath while moderate-severe or critical illness may lead to prolonged hospitalization, cytokine storm, viral pneumonia, secondary bacterial or fungal pneumonia, arrhythmias, acute kidney injury, neurological illness, and so on. 12 The COVID-19 response in some individuals can lead to a complex immunological cascade leading to a cytokine storm with damage to multiple organs.13,15 Symptomatic disease could eventually lead to post-acute COVID-19 syndrome.16,17 The exact duration for which these symptoms persist after resolution of acute disease remains to be fully understood.
COVID-19 management
While COVID-19 prevention is largely associated with targeting viral spike and structural proteins by highly efficacious vaccination – generating robust humoral antibody response – hospitalization-associated patient management and therapy is critically symptom associated. Identification of clinical disease staging – clinical symptoms combined with laboratory and radiologic assessment – are critical for risk stratification, triage, and early therapeutic intervention of patients who progress to severe disease, requiring hospitalization.13,18 In hospitalized patients, mechanical ventilation, oxygen therapy, and symptomatic treatment have been the mainstay of therapy. 19 While there is no clinically effective therapy for COVID-19 in singular, several therapeutic agents such as corticosteroids, antivirals, monoclonal antibodies, and convalescent plasma are currently being administered and evaluated, many a times, in conjunction. Antivirals have a special role to play, repurposed through their role in other viral infections, preventing viral entry into the host cells and causing suppression of viral replication at various steps. 20 Some key antivirals of interest are listed in Table 1.
Repurposed antivirals being evaluated for therapeutic activity in COVID-19.

ACE2, angiotensin converting enzyme 2; HIV, Human Immunodeficiency Virus; MERS, Middle East Respiratory Syndrome; RNA, ribonucleic acid; RSV, Respiratory Syncytial Virus; SARS, Severe Acute Respiratory Syndrome.
Several clinical trials recruiting patients across the disease stages are trying to identify the most efficacious drug(s) for efficacious COVID-19 therapy, and retroviral therapies have been of most interest.21,23 Remdesivir has been used under emergency use authorization in several countries, demonstrating some efficacy in shortening recovery time in COVID-19 patients. 27 The combination of lopinavir with ritonavir showed some favorable outcome, but a clinical trial was discontinued due to side effects. 19 Ribavirin, a guanine analog targeting viral RdRp was found to be ineffective in improving COVID-19 recovery outcomes, although in combination triple therapy with lopinavir and ritonavir, with interferon beta-1b, some improvement in efficacy was observed to decrease recovery in hospitalized patients with mild to moderate COVID-19. 25 In addition, singular therapy with umifenovir (arbidol) did not improve patient outcomes. 26 Favipiravir, similar in action to remdesivir, inhibits transcription and replication of multiple RNA viruses by targeting RdRp. 28
Favipiravir
Drug development and pharmacodynamics
In the process of viral replication, RdRp is a critical element, conserved across RNA viruses but absent in human cells, making it a promising antiviral target.
12
Favipiravir was developed by Toyama Chemical Company Limited and was first approved for clinical use in Japan in 2014, for novel and re-emerging influenza virus infections. It was discovered through a library screen of chemical candidates utilizing a plaque reduction assay.28,29 Understanding efficacy of favipiravir in COVID-19 is of particularly high interest,30,31 as it has been shown to have effective

Schematic showing the mechanisms of action associated with favipiravir.
Pharmacokinetics and safety
Favipiravir is metabolized mainly by aldehyde oxidase (AO) and partly metabolized to a hydroxylated form by xanthine oxidase (XO). A glucuronate conjugate was observed in human plasma and urine as a metabolite other than the hydroxylated form. It is predominantly excreted into the urine as a hydroxylated form, with minimal unchanged drug. The pharmacokinetic profile of favipiravir has been studied through dose-escalated studies, with oral dosage for adults at 1600 mg twice daily on day 1 (loading dose), followed by 600 mg twice daily for 4 days, with the total duration of therapy being 5 days.
29
The maximum plasma concentration was achieved within 2 h after administration of a single dose, although plasma concentration quickly decreased, with half-life between 2 and 6 h.29,42 Interestingly, administration of multiple doses led to an increase in the time to maximum plasma concentration and half-life.
42
Favipiravir is associated with dose- and time-dependent pharmacokinetics, possibly explainable by saturation of the main enzymatic pathway as it was shown to inhibit AO
The literature suggests that favipiravir is a well-tolerated drug. 32 The most frequently reported adverse effects of favipiravir when used for influenza treatment, are mild to moderate diarrhea, asymptomatic increase of blood uric acid and transaminases, and a decrease in the neutrophil count. 29 In a study evaluating the inhibition properties of favipiravir against human and mouse norovirus RNA polymerases, it was found that favipiravir ribonucleoside triphosphate was recognized as a substrate of the human mitochondrial RNA polymerase, but did not result in inhibition of the DNA-dependent RNA polymerase nor cause mitochondrial toxicity in cells.32,40 Despite good tolerance observed, a major concern to safety of clinical favipiravir use is related to its teratogenic tendencies. In studies with fetal/embryo models of multiple animal species, teratogenicity was observed across all. 44 The exposure dosage causing teratogenicity was comparable with the proposed human use dosage. Therefore, it is strongly recommended that favipiravir not be administered to pregnant or possibly pregnant individuals.29,44 Due to this high risk of teratogenicity and embryotoxicity, favipiravir is conditionally marketed with strict regulation in Japan. 32 Hence the clinical use of favipiravir is restricted to novel or re-emerging influenza viruses, only when that virus is resistant to other influenza antivirals, while being manufactured and distributed only upon request by the Minister of Health, Labor and Welfare in Japan.29,32
Use in influenza
Early literature on favipiravir dates to 2001. Also known as T-705, favipiravir was tested
The approved favipiravir drug regimen in Japan for the treatment of Influenza is 1600 mg (loading doses) on day 1 followed by 600 mg twice a day, with the total duration of therapy being 5 days.29,52 Other experimental clinical studies in influenza53–56 used a regimen of 1800 mg (loading doses) followed by 800 mg twice a day, for a total duration of therapy of 5 days. Evidence from clinical trials assessing over 2000 patients suggests that favipiravir is well-tolerated.51,53–56 Adverse drug reactions reported include elevation in serum uric acid levels, diarrhea, elevation of liver enzymes, and reduction in neutrophil count. 52
Use in other viral diseases
Viral hemorrhagic fevers
The causative agents of viral hemorrhagic fevers (VHFs) belong to
Favipiravir has been tested
Yellow fever is a VHF caused by the yellow fever virus (YFV), a flavivirus. The YFV distribution runs through tropical and subtropical Africa and South America. Clinical disease could be asymptomatic or may manifest with fever, chills, headache, myalgia, jaundice, bleeding, shock, and organ failure. The mortality rate of those with severe symptomatic disease can be > 30%. Julander
West Nile Virus
West Nile Virus (WNV) is a vector-borne (mosquito) disease caused by the West Nile Virus, a flavivirus. The spectrum of disease can vary from asymptomatic infection to mild febrile illness to severe viral meningitis or encephalitis. To date, there is no approved antiviral for the treatment of WNV. Morrey
Nipah virus
Nipah virus is an emerging zoonotic infection caused by the Nipah virus which belongs to the
Respiratory viral diseases (other than influenza and COVID-19)
Human metapneumovirus (hMPV) and RSV, previously classified under
Use in COVID-19
Early genomic sequencing of SARS-CoV-2 virion suggested over 90% homology, across several essential enzymes, with other coronaviruses, particularly those causing SARS and MERS. 66 In a quest for identifying effective therapy amid a catastrophic global pandemic, this knowledge helped trigger the ‘repurposing’ of multiple antiviral drugs including broad-spectrum antivirals and more specifically, drugs previously directed toward therapy of SARS, MERS, and other viral infections. The mechanism of action of favipiravir against SARS-CoV-2 is similar to that previously described in literature. After viral incorporation of T-705 RTP by RdRp, the nascent coronaviral RNA undergoes slowed RNA synthesis, chain termination, and viral mutagenesis, while evading the viral RNA repair mechanism. The SARS-CoV-2 RdRp has been described to have 10-fold higher activity than other viral counterparts, while simultaneously being prone to high nucleotide incorporation and error rates. 67 These processes significantly bottleneck the RNA replication by causing nucleotide shifts in an unbalanced viral environment with already low cytosine levels. 67 These critical hallmarks of the SARS-CoV-2 virion machinery make it vulnerable to RdRp inhibitors.
Alongside an effective vaccine, antivirals have a critical role in containing local spread of infection by curtailing viral load and shedding, at an early infection stage. 68 Studies suggest viral load in the upper and lower respiratory tracts in patients with mild infection peak in the early stage of infection, days 4 and 6, respectively, while in moderate-severely infected patients they peak in later stages, days 8 and 11. 69 Thus, the role of antivirals like favipiravir may be higher in mild-moderate infections at an early stage of the disease, to control the viral load, shedding, and infectivity rates, 70 to help reduce the burden of hospitalization and associated patient care on an already overwhelmed system.
An early study (February 2020) showed that favipiravir at higher concentrations (EC50 = 61.88 μM) was able to minimize viral infection in vero E6 cells
Subsequently, other studies have been conducted to understand the efficacy of favipiravir in COVID-19. Studies have suggested that favipiravir could lead to viral clearance by day 7 and improve clinical outcome by 2 weeks when administered in early stages of infection.76,77 In one study, the median time to viral clearance was about 12.8 days in patients when favipiravir therapy was initiated on day 1, and increased to 17.8 days when therapy was started on day 6.
78
In addition, there were significant differences in duration of hospitalization, with need for oxygenation and mechanical ventilation, in patients with mild or asymptomatic infection when favipiravir therapy initiated late stage
The use of favipiravir on a compassionate or approved basis for the indication of COVID-19 treatment has been put through clinical trials across the globe. Table 2 summarizes key published clinical trials (including from preprint servers) to date. The drug was trialed in cases of asymptomatic and mild-moderate disease as well as in moderate-severe cases of COVID-19 pneumonia. While most trialed a regimen of 1600 mg loading dose (two doses) on day 1 followed by 600 mg twice daily, other regimens used 1800 mg loading dose (two doses) followed by 800 mg twice a day. The length of drug therapy with favipiravir was varied and ranged from 7 to 14 days. At the present time, favipiravir is not approved for use in COVID-19 by the United States Food and Drug Administration or its European counterpart, the European Medicines Agency. The formally recommended dose and duration of therapy remain awaited based on results of the ongoing clinical trials. However, it is noteworthy that 600 mg twice daily maintenance dose has been used to manage COVID-19 in some international markets.
79
Doi
Summary of published (including preprint) literature from favipiravir clinical trials to date.
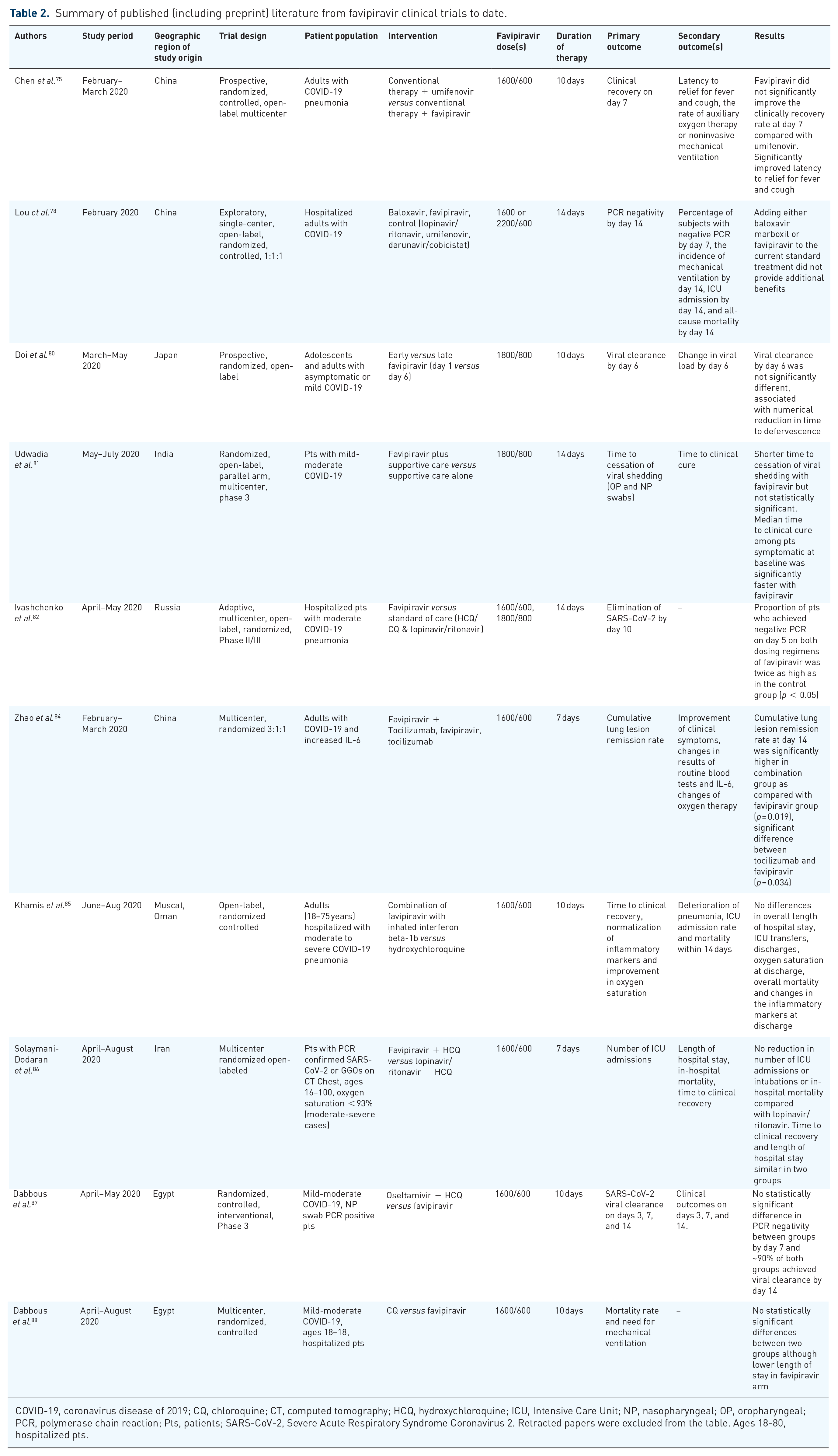
COVID-19, coronavirus disease of 2019; CQ, chloroquine; CT, computed tomography; HCQ, hydroxychloroquine; ICU, Intensive Care Unit; NP, nasopharyngeal; OP, oropharyngeal; PCR, polymerase chain reaction; Pts, patients; SARS-CoV-2, Severe Acute Respiratory Syndrome Coronavirus 2. Retracted papers were excluded from the table. Ages 18-80, hospitalized pts.
Future directions
Table 3 summarizes publicly available information about ongoing favipiravir clinical trials.89–95 The GETAFIX, FLARE, COVERAGE, and VIRCO trials are focused on drug efficacy early in the treatment of COVID-19 and the COVERAGE trial will be looking specifically at geriatric patients. The PRINCIPLE trial collaborative group evaluated the use of azithromycin among adults 50 years of age or more with medical comorbidities and all adults above 65 years of age with COVID-19 and found that its use did not shorten the time to clinical recovery or reduce the risk of hospitalization.
96
Interim results of the PRINCIPLE trial also reported clinical utility of inhaled budesonide among patients with COVID-19 at risk for adverse outcomes.
97
The findings from the PRINCIPLE trial about favipiravir’s clinical utility remain awaited. Bosaeed
Summary of ongoing favipiravir clinical trials.
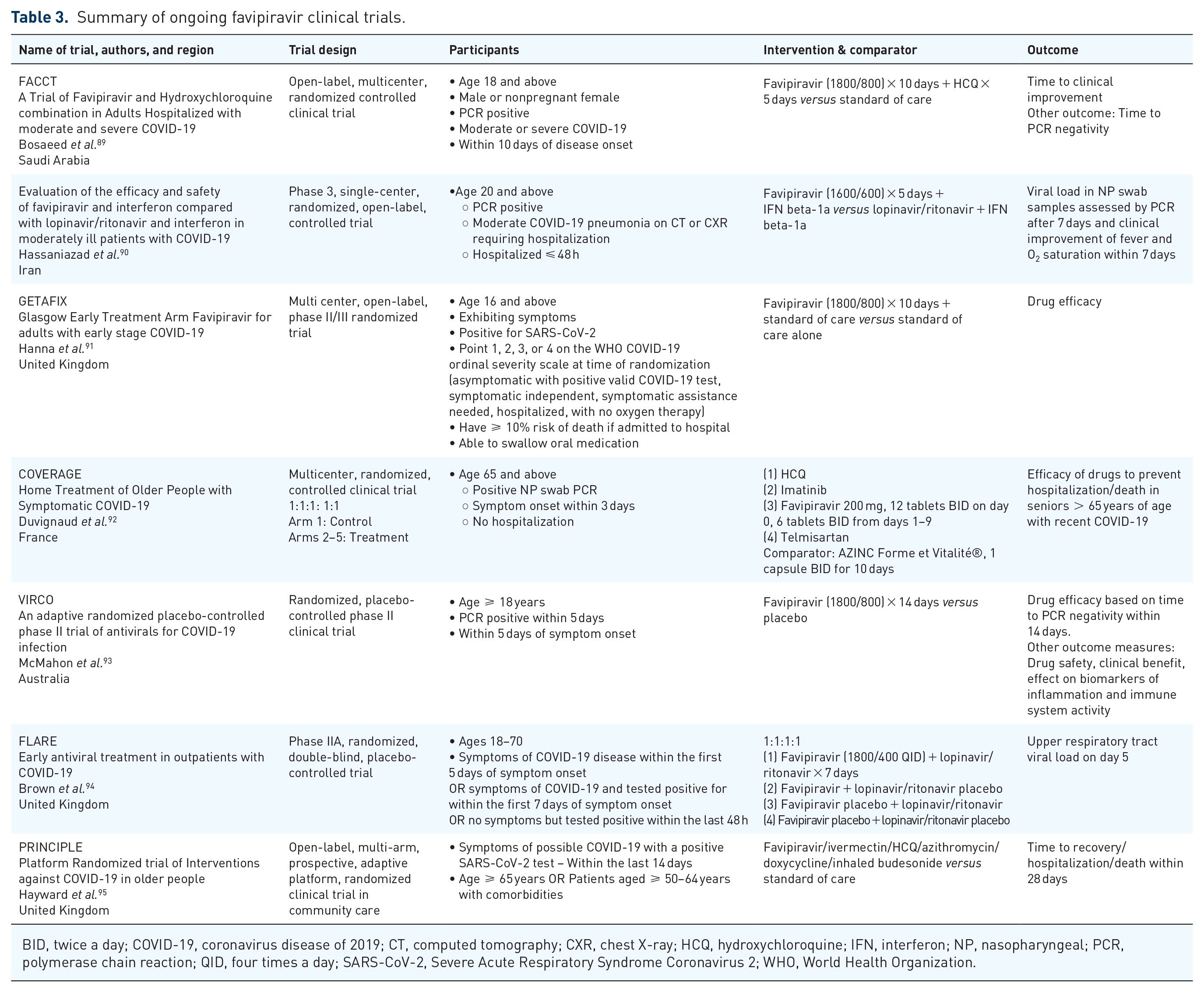
BID, twice a day; COVID-19, coronavirus disease of 2019; CT, computed tomography; CXR, chest X-ray; HCQ, hydroxychloroquine; IFN, interferon; NP, nasopharyngeal; PCR, polymerase chain reaction; QID, four times a day; SARS-CoV-2, Severe Acute Respiratory Syndrome Coronavirus 2; WHO, World Health Organization.
Conclusion
Favipiravir, among other drugs, has gained prominence since 2020 because of the COVID-19 pandemic and its versatility as a broad-spectrum antiviral that inhibits RdRp and targets viral replication. While its utility has shown benefit in a few clinical trials, others yielded mixed results. The drug’s full potential as a therapy for COVID-19 remains to be determined, including its optimal timing of administration, dosage, and duration of therapy. It is fairly well-tolerated with a major safety concern being its teratogenic potential. Adverse drug reactions among patients included hyperuricemia, QTc interval prolongation, and elevation in hepatic enzymes. Finally, favipiravir’s prospect as a post-exposure prophylactic agent in COVID-19 remains to be tested.
Footnotes
Author contributions
KS: Conceptualiztion, manuscript preparation, review and editing.
MR: Conceptualization, manuscript preparation, review, editing and submission.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
