Abstract
Chagas disease (CD), caused by the protozoan Trypanosoma cruzi, is a public health concern, mainly among countries in South and Central America. However, despite the large number of immigrants from endemic countries living in the USA, awareness of CD is poor in the medical community, and therefore it is significantly underdiagnosed. To avoid the catastrophic cardiac complications of CD and to prevent maternal–fetal transmission, widespread educational programs highlighting the need for diagnosis are urgently needed.
Introduction
Chagas disease (CD) is a public health concern in endemic and nonendemic countries. Although many efforts have been made across different fronts to mitigate the burden of this disease (i.e. fumigation strategies, antiparasitic treatment, housing improvement, and vaccine development), the complexities surrounding this disease have not been successfully addressed. The epidemiology of CD in the USA is incompletely defined 1 owing to a lack of awareness in the at-risk and healthcare communities and the poor diagnostic profiles of commercially available US assays, resulting in clinical diagnostic challenges in identified at-risk populations. 2 This paper represents the outcome of a 2017 multidisciplinary experts meeting to evaluate opportunities to improve the detection of CD. Using this initial discussion as a starting point, we provide a brief overview of CD and highlight current challenges and potential solutions to screening and prevention.
Disease background
CD is a parasitic zoonosis transmissible to many mammals, including humans, caused by the protozoan Trypanosoma cruzi.1,3 T. cruzi consists of seven main genetic subtypes, classified as discrete typing units (DTUs) TcI–VI and TcBat, whose prevalence varies geographically.3,4 Over 140 competent triatoma vectors (Triatominae insects, a subfamily of Reduviidae) of T. cruzi exist, and 11 species of the ‘kissing bugs’, also known as cone-nose bugs, chinches, vinchucas, pitos, piks, and barbeiros, capable of transmitting CD, are found throughout the southern half of the USA.5,6 Several mammals, including dogs, raccoons, opossums, wood rats, and armadillos, can act as reservoirs. 7
Due mainly to a lack of provider knowledge or limited insurance coverage, less than 1% of the estimated patient population with CD in the USA has access to diagnosis and treatment. 8 The majority of affected people are unaware they are infected because of the long asymptomatic period associated with CD. Further, the familiarity of CD within the general public, as well as in at-risk groups and among healthcare providers, is low. 9
However, physician education campaigns have proven to be successful in the USA, and future efforts should work on expanding these targeted campaigns among physicians who work closely with Hispanic populations. 10
Intraspecies genetic diversity of the parasite could influence the variation in disease manifestations and diagnostic performance across different geographic regions. TcII, TcV, and TcVI are more prevalent in the Southern Cone (Uruguay, Chile, and Argentina), while TcI is more common in the Northern Cone. 5 In the USA, TcI and TcII have been identified in autochthonous cases, and type TcIV has been identified in vector species suggesting its potential in humans. While the underlying geographic origin is relevant, the likelihood of having a patient in the USA with any DTU is great, highlighting the need to have an all-encompassing diagnostic.11,12
Epidemiology
With over 70 million people at risk of infection, an estimated 6 million people are affected globally, with an estimated annual mortality of 11,000–13,000.13,14 Approximately 250,000 infected individuals reside in the USA. 15 Endemic countries traditionally include the 21 countries from the 42°N to the 40°S latitude of the Americas, excluding the Caribbean islands and the USA.16,17 The worldwide distribution of CD is driven by the presence of the vector, living conditions, climate change, and migration.18–20 Prevalence rates within endemic countries vary on a regional basis, given different climate conditions, housing, socioeconomic status, and screening and control efforts. 20 Country-wide estimates are shown in Table 1.
Countrywide estimates of CD in endemic countries.
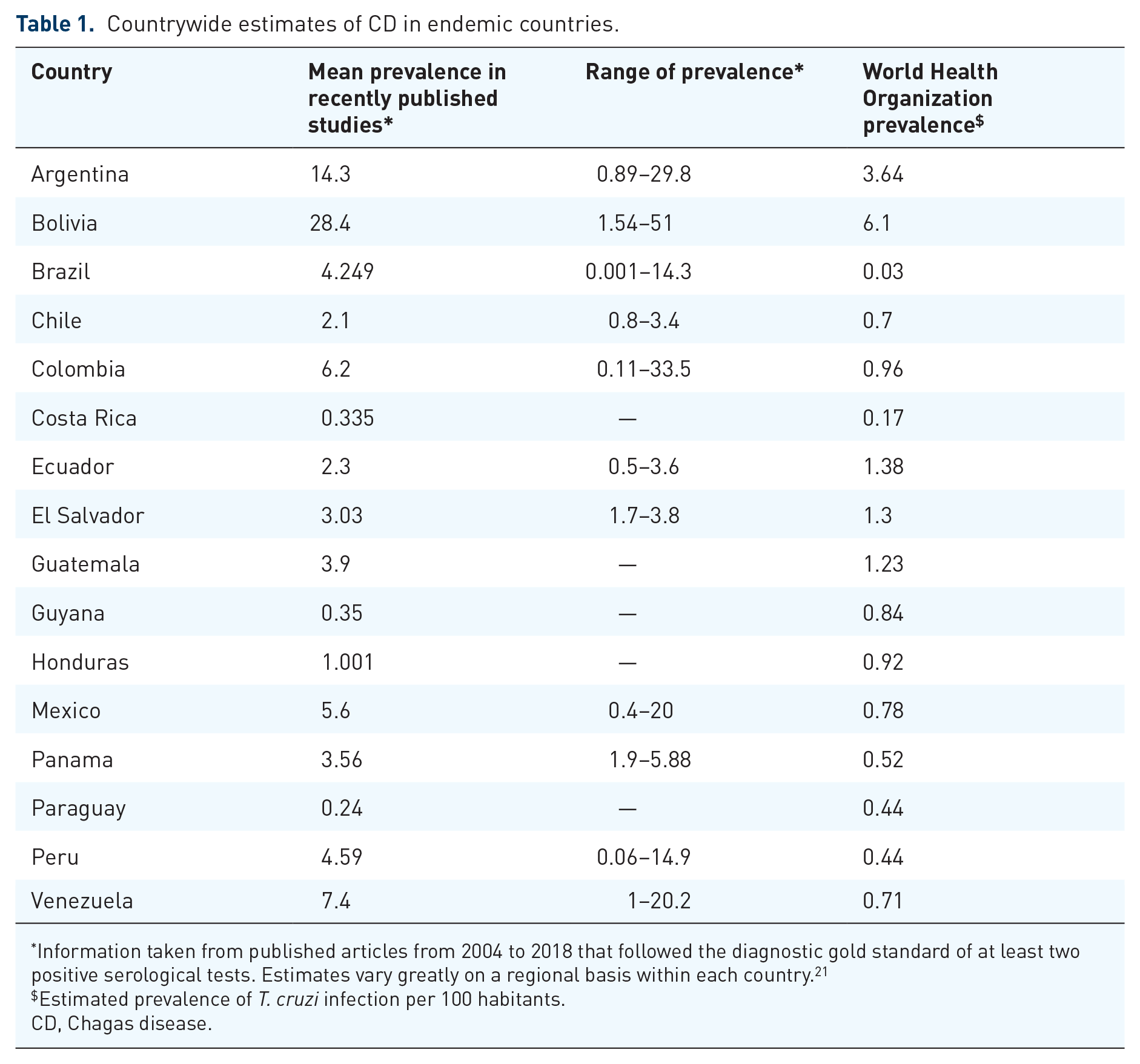
Information taken from published articles from 2004 to 2018 that followed the diagnostic gold standard of at least two positive serological tests. Estimates vary greatly on a regional basis within each country. 21
Estimated prevalence of T. cruzi infection per 100 habitants.
CD, Chagas disease.
Most reported cases are of chronic CD, predominantly in older people, as the aging population infected with the parasite develops symptoms.13,22 As successful vector control methods have been implemented in endemic countries, congenital and oral transmission have become more important mechanisms of transmission. T. cruzi may also be transmitted to a lesser extent through uncontrolled blood donations and organ transplants, laboratory accidents, and needle sharing. 13
CD is considered by the World Health Organization as a ‘neglected tropical disease’13,23 and affects people of low socioeconomic status. The condition is more likely to be present where housing is substandard, sanitation is poor, insect vectors are widespread, and there is restricted access to healthcare. 13 Table 2 shows known environmental factors, mostly socioeconomic surrogates of poverty, that increase the pretest probability of a patient to have a positive test for CD. Geographic distribution varies within each country. The two factors that are determinant for an area to become endemic are (1) the natural presence of an aggressive domicile vector and (2) socioeconomic factors resulting in humans living with the vector species inhabiting their homes.
Clinical markers that increase the risk of CD.

CD, Chagas disease.
The term ‘autochthonous Chagas’ in the USA is reserved for residents who acquire infection with T. cruzi in the USA. There are at least 76 well-documented cases 24 of autochthonous CD established by clinical findings with or without a positive serology. Low US physician awareness of CD as a whole, including a lack of awareness that CD can occur in those without a history of travel, 25 and low diagnostic testing rates likely result in misdiagnosis and underreporting of autochthonous CD cases. In the USA, the epidemiologic profile of infected cases is still unclear. In Texas, for example, a higher burden of autochthonous cases identified through the blood bank has been documented.26–28 However, a larger seroprevalence study of Texas game hunters and a study of two cities in Arizona that included evaluation of domestically captured kissing bugs noted that despite an aggregate of thousands of triatomine nocturnal bites, no residents tested positive for T. cruzi infection.29,30 In contrast, case reports have documented T. cruzi-positive persons in Texas without any known history of triatomine bites.26,27 A total of 11 different triatomine species are found in the USA, and the presence of specific triatomine species in an area likely impacts the risk of transmission. 6 In summary, autochthonous human CD in the USA is worthy of continued study, but evidence of its existence remains limited.
Critically important, but unrecognized in the USA, is the presence of CD in immigrants. Due to large-scale migration of individuals born in areas of rural poverty in Latin America, many infected individuals live in the USA. A summary of reported prevalence in immigrants is shown in Table 3. More than 10 million foreign-born immigrants reside in California, and 50% of those come from Latin America. 31 In testing 4755 Latin America-born residents of Los Angeles (LA) county for the presence of T. cruzi antibodies, the prevalence of antibodies was 1.24%, which implies that tens of thousands of people in the LA area are potentially infected. 2 CD transmission has been reported in heart transplantations and blood banks in LA.32–34 In addition, it has been diagnosed in infants in California, suggesting possible ongoing congenital transmission of CD in at-risk pregnant women. 35 A prevalence study in LA county found the highest prevalence among Salvadorans (3.45%), seconded by Mexicans (0.79%), predominantly from Oaxaca (4.65%) and Zacatecas (2.2%). 1 In a primary care sample in East Boston, 5125 immigrants were screened, with an overall seroprevalence of 0.97%. 36 Finally, in a community-based seroprevalence study in the metropolitan Washington DC area, 3.8% of 1500 immigrants tested positive for T. cruzi infection, with 2% of Central American and 23% of South American participants testing positive. 37
CD prevalence reported in immigrants from Latin America in nonendemic countries.*

Country of origin of those screened and with positive serology was mainly Bolivia.
CD, Chagas disease.
Clinical course of CD
Acute phase of CD infection
While several modes of acute disease can occur, vector-borne transmission is the most common. Over 140 different kissing bug species are competent vectors, however, a select few with specific geographic regions are considered of principal public health importance. 5 These nocturnal hematophagous vectors typically defecate during the blood meal process, and T. cruzii-infected excrement can enter the body either through mucosal membranes or incisions in the skin (i.e. from scratching a bite). The acute phase of vector-borne CD usually manifests 1–2 weeks after vector transmission of the parasite and is most often asymptomatic. Clinical manifestations can include fever and/or enlargement of the liver, lymph nodes, or spleen. Occasionally, a swollen area, ‘chagoma’, may be observed near the site of the bite. Some individuals with acute infection may exhibit a swelling of the eyelid, called Romaña sign. Rarely, acute CD involves potentially life-threatening meningoencephalitis and/or myocardial damage. Typically, the acute phase goes unrecognized and resolves spontaneously in >90% of patients. 50
Chronic manifestations of CD
Chronic CD is an indeterminate phase that lasts for the life of the patient unless antiparasitic treatment is provided. Of those who progress into chronic CD, most individuals remain asymptomatic, but in endemic countries, the annual rate of progression to cardiomyopathy is 2% among the indeterminate form and as high as 4% for the acute phase. 51 Overall, up to 30% of those infected progress to the chronic phase with cardiac manifestations or digestive involvement. 52 The underlying factors associated with disease progression and discriminant, progressive cardiac biomarkers are largely unknown. Of the chronic cardiac complications, electrocardiogram (ECG) abnormalities are generally the first markers of cardiac damage. Right bundle branch block combined with left anterior fascicular block is a common manifestation, but a wide range of ECG alterations may occur.50,51,53,54 Around 20–30 % develop significant symptomatic cardiac disease with varying degrees of fibrosis of the myocardium. These abnormalities contribute to the clinical manifestations that include symptomatic bradyarrhythmia, tachyarrhythmia, thromboembolic phenomenon from left ventricular aneurysms, sudden death, and congestive heart failure.50,55 Unfortunately, several of these complications can occur with minimally reduced ejection fraction, a feature that distinguishes CD from other cardiomyopathies. In addition, CD cardiomyopathy is associated with worse outcomes than other cardiomyopathies. 55
Gastrointestinal (GI) complications occur in about 10–15% of patients. Damage to the GI system, including megaesophagus and megacolon, is more common among patients from the Southern Cone of South America. GI complications can lead to profound malnutrition or obstipation. 50 The central or peripheral nervous system may also be impacted, particularly in immunocompromised patients. T. cruzi acts as an opportunistic infection in patients who are coinfected with HIV. 56 Patients with HIV and chronic T. cruzi infection may experience reactivation with a presentation that can be confused with cerebral toxoplasmosis.
Clinical aspects of congenital T. cruzi infection
Most infected women will be asymptomatic during pregnancy. These women will likely remain asymptomatic for decades, but eventually one third of them will develop cardiac or GI complications and will transmit the infection in 1–5% of pregnancies.50,57
While most commonly asymptomatic, congenital T. cruzi infection can cause a spectrum of clinical manifestations. Transmission early in pregnancy may increase risks of spontaneous abortion, 58 whereas infection after 22 weeks may be associated with stillbirth, premature labor, low birthweight, or an infected live-born infant. 59 Infected live-born infants are placed into three clinical categories: (1) severe disease at birth with high risk of neonatal death; (2) apparently well at birth with progression to serious complications in the first weeks or months; (3) asymptomatic throughout infancy, but with a 20–30% risk of symptomatic cardiac or GI disease later in life. Most infected newborns fall into categories 2 and 3, and the disease is rarely detected unless actively searched for.
Determinants of vertical transmission
The rate of congenital T. cruzi transmission varies widely between populations studied, ranging from ~1% to >10%.60–64 Pregnant women in regions where the T. cruzi vector is endemic have higher parasitemia levels than those in nonendemic settings, and pregnant women who transmit have higher parasitemia levels than those who do not. 63 Disruption of the placental barrier in seropositive women is also a risk factor for congenital transmission, particularly if it occurs after week 12 of gestation. 60 In animal models, the T. cruzi strain affects the risk of congenital infection, however, the effect of genotype in T. cruzi human congenital infection is not clear.62,65,66
Challenges and opportunities in the identification of CD in the USA
Diagnosis and ongoing assessment
Diagnosis of CD in the acutely infected patient or in reactivation disease is made by either direct visualization of the parasite on buffy coat preparation, or by polymerase chain reaction (PCR) evidence of infection.58,67 The conventional assays are enzyme-linked immunosorbent assay based on epimastigote lysate or recombinant antigens, indirect hemagglutination assay, indirect immunofluorescence tests, and immunoblotting with trypomastigote excreted-secreted antigens of T. cruzi. Two positive tests from different diagnostic platforms or targets are required to confirm the diagnosis. 67 These serum immunoglobulin (IgG) serologic tests are also recommended for chronic CD diagnosis in adults and older children and can be used with pregnant women to identify which infants should be screened at birth. 58
Once diagnosis of CD is confirmed, evaluation for the presence of cardiac or digestive complications is imperative. A patient with no evidence of cardiac manifestation should then be followed on a yearly to biyearly basis at a minimum with ECG to assess for newly developed cardiomyopathy. 67 An echocardiogram (ECHO) should be routinely performed at least on initial evaluation, and repeated in the setting of clinical change. Cardiac magnetic resonance imaging (MRI) is the most sensitive cardiac imaging assay 68 and can detect early myocardial damage before ECG or ECHO alterations are present. However, the cost and logistical aspects of cardiac MRI can be prohibitive in rural or impoverished populations. Due to its poor side-effect profile,69,70 antiparasitic therapy is recommended on a case-by-case basis, and several factors should be considered before treatment, such as age of the patient, comorbidities, and previous history of adverse reaction to medications. Even in patients with advanced CD cardiomyopathy, diagnosis and monitoring are still clinically important, as antiarrhythmics, implantable cardioverter-defibrillator placement, and heart transplant are still recommended. 55 Cardiac evaluation and treatment of patients with more significant disease should recognize the unusually high risk of adverse outcomes, particularly stroke 71 and sudden death, 72 in patients with CD cardiomyopathy.
Current diagnostic test options for congenital T. cruzi infection
The gold standard for diagnosis of congenital CD is either seeing the organism in the blood at birth or 1 month or having a positive IgG assay at 8 months of age. This testing protocol is hampered by a lack of sensitivity in the former and poor compliance by subjects in the latter. PCR is also an accepted test to diagnose congenital infection.
The need for increasing awareness in the USA
A key challenge in the diagnosis of CD in the USA is the lack of awareness of the disease. To improve awareness in the medical community, we recommend increased CD-related content in medical education, student global health consortia, and continuing medical education modules, and more CD-related content at national medical meetings. Networks of provider experts in the care of patients with CD can serve as a resource for physicians new to the process of evaluating and treating patients. Guideline drafting committees will be critical to provide support to clinicians who struggle with whom to screen for CD, and will also provide them with support to advocate for implementing a plan to their workplace administrations. Ongoing cataloging of cases and patient outcomes can eventually be used to analyze the value of increased awareness and screening for CD. Social media platforms to discuss cases will help knowledge sharing among clinicians involved in the care of patients with CD.
Educational programming, including posters and pamphlets, that raise awareness in the at-risk populations must be appropriately designed and implemented to reach a community with limited English and limited healthcare literacy and access. A recent physician education pilot campaign in Texas highlighted the value and capacity of targeted education campaigns. 10 In addition, direct-to-patient advertisements in Spanish-language media, as well as in churches and other protected spots, are also viable targets.
The need for improved screening in the USA
While the Pan American Health Organization’s (PAHO) recent diagnostic and treatment guidelines recommend screening methods for blood banks and seroepidemiological surveys, there is a need for adequate guidelines for screening in primary care, obstetrics/gynecology, and pediatric settings. 73 As transfusional transmission has been an important source of infection in endemic countries and in the setting of high prevalence rates in blood donors in the USA (1:7500 in LA, 1:9000 Miami), the Food and Drug Administration (FDA) has mandated screening of all first-time blood donors in the USA for CD.74,75Universal CD screening in blood banks has been effective in controlling transmission through transfusion.76,77 The general challenges to screening for CD include costs, discordance in test results, and risk/benefit ratio regarding treatment. Initial efforts should focus on patients from Central and South America, newborns, women of childbearing age, and pregnant women.
The need for improved serologic testing options in the USA
The currently available tests in the USA utilize antigen from TcII serotypes, which perform very well in South America. In the USA, however, there is less confidence in how these tests perform, particularly in the diverse immigrant population that is heavily weighted towards individuals who are from countries in which TcI is the predominant serotype. Owing to this, there are high rates of false positive tests in Mexican and Central Americans, and the false negative rate is unknown. Development of tests with accuracy in the diverse at-risk population in the USA is critical. At this time, the Centers for Disease Control (CDC) is the only location to obtain validated confirmatory serologic analysis, which, while free, is complicated to order and has a 10–14-day delay. Expanding confirmatory testing options to commercial laboratories is important.
The need for improved access to screening in the USA
Lack of affordable healthcare is the main barrier to CD screening programs in the USA. Decentralization of healthcare delivery to the state level further complicates a federal policy regarding screening. The best but unlikely solution for this problem is universal health coverage in the USA with targeted programs for diseases of immigrants. In the interim, promoting screening as a matter of social justice, while also focusing on the recommendations for screening by the CDC in the prenatal setting and data about the cost-effectiveness of screening newborns, will incentivize providers to participate. 77 Development of an inexpensive point-of-care test will facilitate adoption of screening programs. The current dropout rate between diagnosis and treatment can be as high as 80% based on a study in the northeastern USA. 36 In that study, of 50 patients who tested positive, only 20% finished a course of benznidazole therapy. Community involvement in the creation of self-awareness materials for CD can increase the number of patients seeking to be screened for the disease.
Our recommendations for screening are shown in Table 4 and are based on populations with increased pretest probability, although global screening is preferable to decrease chances of stigmatization. Hispanics with socioeconomic risk factors are the group most vulnerable to the infection, however, we lack studies to assess the cost-effectiveness of universal screening among first-generation Hispanics living in the USA. Clinical tools that allow for further risk stratification within the general population may be helpful to reduce unnecessary testing, i.e. a risk score incorporating known risk factors for disease, such as knowledge of the illness, of the vector, or of a family member who has CD. 35 Health policies making a physician visit and treatment mandatory and covered for patients who are positive for CD can enhance access to care (Table 5).
Recommendations for a good screening program.

CD, Chagas disease.
Proposed national and regional health policies to enhance CD screening and treatment.

CD, Chagas disease.
Maternal screening
Despite an estimate of 40,000 women of childbearing age living in the USA with chronic CD, 80 obstetricians in the USA have little clinical experience with this disease. In 2010, a survey of 400 US obstetricians on their knowledge of CD showed that 68% considered their knowledge as very limited, and only 8.8% knew it could be transmitted vertically. 81 Universal maternal screening of congenital CD is cost-effective in a US model. 81 Similar preventive screening programs for congenital transmission of CD exist in nonendemic regions in Catalonia, Spain.82,83 Mother and infant treatment costs are overcome by the savings of associated morbidity and mortality with congenital transmission rates over 0.001% and maternal prevalence above 0.06%. 77 The PAHO recommends universal serological screening for every pregnant woman during prenatal checkup in the region of the Americas. 73 These recommendations have been previously established by endemic country governments,21,84 and in this setting the CDC recommends screening in at-risk pregnant women in the USA. 57
An effective screening program in at-risk populations would help to identify infected infants and enable early treatment to save lives and ameliorate morbidity. For infants with no or mild symptoms at birth, the sequelae of T. cruzi infection likely could be entirely prevented with early curative antitrypanosomal treatment. In infants, the efficacy of antitrypanosomal drugs is more than 90%, 67 and the side-effect profiles are much better than in older children and adults.
Summary
CD, a potentially debilitating and life-threatening illness, is largely undiagnosed in the USA. Most patients are from endemic countries, and new diagnoses traditionally result from either blood bank surveillance or targeted community/physician outreach in areas of CD-focused clinicians and researchers. Recommendations to increase diagnosis include improving disease awareness in both at-risk and medical communities, as well as removing barriers to diagnoses by improving the accuracy and availability of testing. In addition, generating guidelines about whom to screen and best practices for treatment will encourage physicians to participate in these efforts. All healthcare promotion efforts need to be mindful of the complexity of providing healthcare to this generally marginalized at-risk population, and the complexity of the disease process itself.
We recognize screening represents a significant challenge in the USA, and accordingly, we recommend conducting health economics and outcomes research to justify screening/treatment and drive policy change. In addition, it is appropriate to organize a multidisciplinary group of clinicians and researchers to increase awareness, as well as to draft consensus documents and clinical guidelines.
Footnotes
Acknowledgements
The initial idea for the manuscript was raised at an advisory board organized by Bayer where CD was discussed. This publication and its content are solely the responsibility of the authors. Editorial assistance was provided by Simpson Healthcare, which was funded by Bayer. Simpson Healthcare is committed to adhering to the prevailing standards for ‘Good Publication Practice’ (GPP3), as well as the criteria for authorship established by the International Committee of Medical Journal Editors.
Author contributions
All authors equally contributed to the conception, drafting, and editing of the manuscript.
Conflict of interest statement
AFH-M reported being the recipient of a K12-clinical trial award as a co-principal investigator for the Expanded Access IND Program (EAP) to provide the yellow fever vaccine (Stamaril) to persons in the USA outside the submitted work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for editorial services only was provided by Bayer and conducted by Simpson Healthcare.
