Abstract
Spinal tuberculosis (TB) is a rare cause of vertebral osteomyelitis in the developed world. Co-infections with other microorganisms are seldom reported in the literature. Here we report a case of Mycobacterium tuberculosis and Streptococcus anginosus causing acute on chronic vertebral osteomyelitis with an epidural abscess.
Introduction
Spinal tuberculosis (TB) was first described by Sir Percival Pott in 1782, from whom it garnered the name of Pott’s disease. 1 Spinal TB accounts for 1–3% of all TB cases.2,3 It is most often the result of hematogenous spread from a primary pulmonary, or less frequently, non-pulmonary source and accounts for about 10% of extra-pulmonary TB cases.2,4
Mycobacterium tuberculosis co-infections with other organisms are rare events in non-HIV patients. Despite being described affecting other tissues, its occurrence in vertebral osteomyelitis is unusual.
Case presentation
A 45-year-old HIV negative male immigrant from Mexico presented to our emergency department complaining of progressive bilateral lower extremity weakness for one month with increasing difficulty walking and decreased sensation below the nipple level over the preceding two weeks. He reported urine and stool retention over the preceding one week. He denied cough, shortness of breath, or fevers/chills but did complain of night sweats for the past one month. On physical exam he was found to have profound diffuse bilateral lower extremities weakness (2/5) and diminished sensation to light touch and pinprick below the level of T4–T5. There was no lymphadenopathy appreciated on examination. Initial work up included a white blood cell count of 12.6, platelet count of 494, C-reactive protein of 2.6 mg/dl, erythrocyte sedimentation rate of 20, and a positive QuantiFERON (cutoff value of 0.35 IU/ml). A set of blood cultures was negative. HIV immunoassay testing was negative. Gadolinium enhanced magnetic resonance imaging (MRI) of the thoracic spine demonstrated evidence of T3–T4–T5 osteomyelitis with epidural abscess and bony destruction of the T4 and T5 vertebral bodies, resulting in severe spinal canal stenosis and spinal cord compression (Figures 1 and 2).

Magnetic resonance image with gadolinium, T2 sagittal view. Bone marrow edema at T3–T4. Large epidural abscess at T3–T4–T5 complicated with severe spinal canal stenosis.
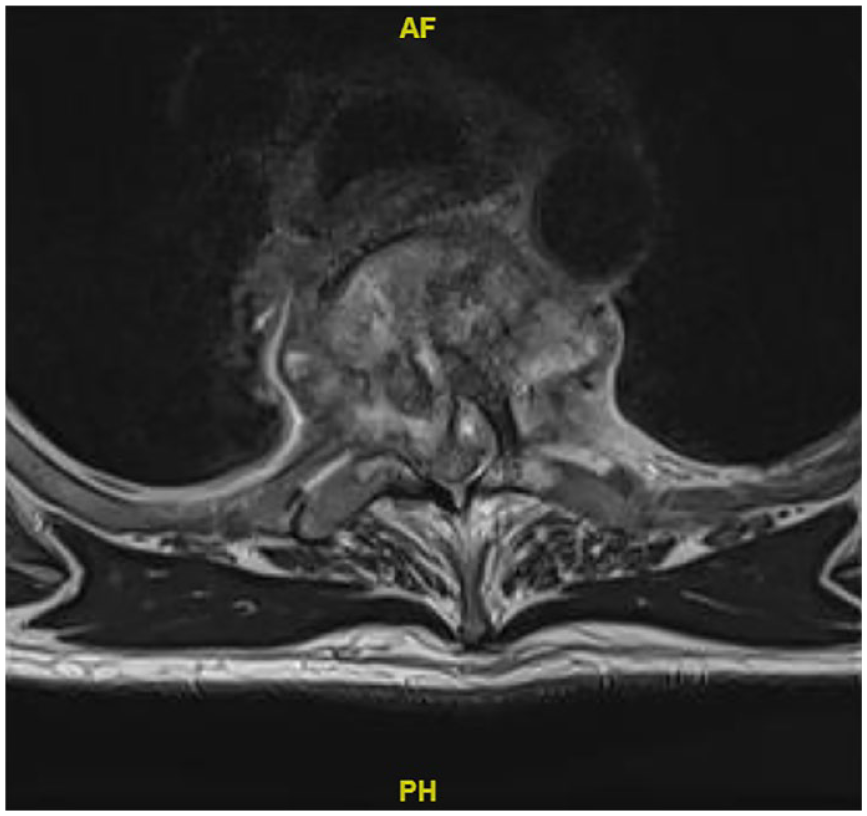
Magnetic resonance image with gadolinium, T2 axial view. Bone marrow edema and abnormal enhancement at the T3–T4 level with large epidural abscess and severe spinal cord compression. There is preservation of the anterior meningovertebral ligament; a finding suggestive of tuberculosis spondylodiscitis. 5
The patient underwent emergency decompression of the epidural abscess with multi-level pedicle screw instrumentation for thoracic vertebral stabilization and interbody cage placement (Figure 3). Intraoperative findings were more consistent with a chronic fibrotic process with a solid mass adherent to the thecal sac with only a small amount of liquid abscess encountered. Epidural tissue biopsies showed caseating and non-caseating granulomas (Figure 4). Further pathology results showed no organisms on acid fast bacilli (AFB) and Grocott methenamine silver stains. M. tuberculosis was not detected on polymerase chain reaction (PCR) (GeneXpert assay) testing of the surgical specimen. The epidural abscess cultures were positive for pan susceptible Streptococcus anginosus at 48 h on multiple tissue samples. A transthoracic echocardiogram did not show evidence of endocarditis. A computed tomography chest showed clear lung parenchyma and multiple sputum samples were negative for AFB stain and culture. Genitourinary testing for TB was not performed given lack of genitourinary symptoms. Approximately 6 weeks after the tissue biopsies were obtained, mycobacterial cultures grew pan susceptible M. tuberculosis. The patient completed six weeks of intravenous ceftriaxone for treatment of S. anginosus and was initiated on rifampin, isoniazid, pyrazinamide, and ethambutol (RIPE) therapy. At hospital discharge the patient had persistent bilateral lower extremity weakness requiring a walker for ambulation, and these deficits improved with outpatient physical and occupational therapy. He was lost to follow-up before completion of RIPE therapy.

Sagittal view computed tomography thoracic spine showing post-operative interval placement of corpectomy device with removal of T4 and the majority of T5. Posterior fusion rods extending T2–T7.
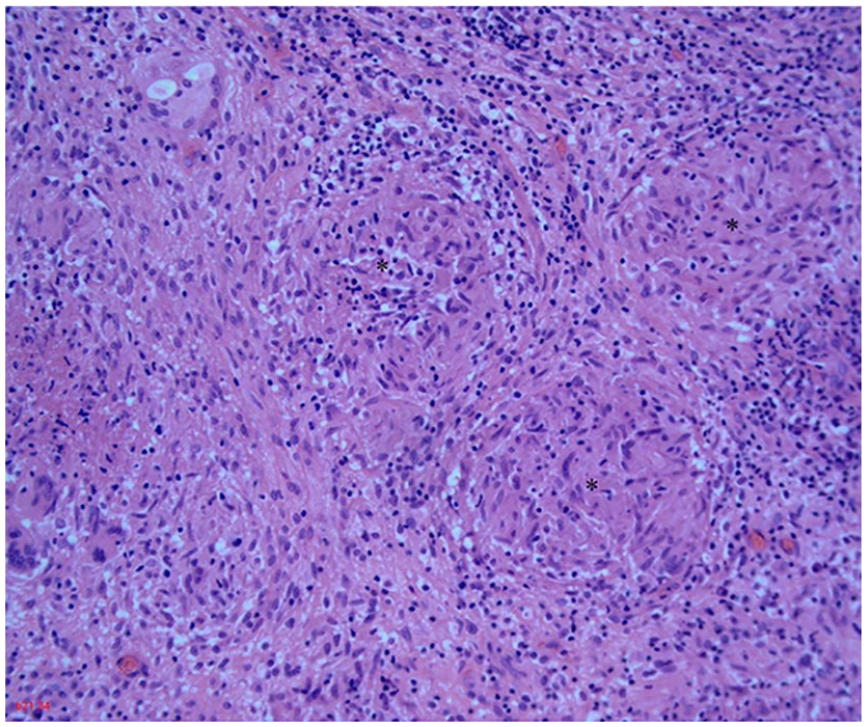
Epidural abscess, hematoxylin–eosin. 10×. Well-formed granulomas (*) with giant cells.
Discussion
The case of spinal TB presented here was diagnosed in a low prevalence area and demonstrates the importance of obtaining a thorough travel and social history in all patients presenting with potential osteomyelitis of the spine. Early identification of potential cases of spinal TB can allow for appropriate isolation of the patient and employment of personal protective equipment by healthcare personnel if required. It also prompts a timely initiation of an appropriate diagnostic approach.
Spinal TB is often indolent and without many of the dramatic signs and symptoms typical of other forms of pyogenic vertebral osteomyelitis. Most patients do not have any pulmonary involvement, and therefore lack many of the typical systemic signs and symptoms that accompany pulmonary TB such as weight loss, night sweats, cough, and fever.2,6 Back pain is often the only symptom, and is by far the most common one, with or without accompanying neurologic deficits.7–9 This lack of specific signs and symptoms poses diagnostic challenges, often resulting in delayed diagnosis and neurologic sequelae as demonstrated by a multinational study of 314 patients with spinal TB in whom the mean time of symptom onset to diagnosis was 78 days and neurologic sequelae complicated up to 25% of cases. 10
Aside from the above-mentioned difficulties in diagnosing spinal TB, the case presented here was made more challenging by a negative surgical tissue biopsy AFB smear and TB PCR, despite the abundant presence of granulomas. A positive TB culture is considered the gold standard for diagnosis but can take 6–8 weeks to grow. 11 TB PCR and detection of acid-fast bacilli on tissue smear can provide rapid diagnostic value and expedite clinical decision making and treatment implementation. However, the sensitivity of TB PCR for extrapulmonary specimens is reported to be below 80% 12 and detection of extra-pulmonary organisms on AFB smear has very low sensitivity in paucibacillary disease. Therefore, a significant number of false negative results occur, particularly in extra-pulmonary TB cases as exemplified by the case presented here. Additionally, GeneXpert assay, which has not been validated for non-sputum specimens, was utilized in this case, making the sensitivity of the test results uncertain. When faced with the diagnostic dilemma of negative TB PCR and AFB smear results, clinicians must use their best clinical judgment and consider empiric TB therapy when clinical suspicion is high while awaiting mycobacterial culture results.
Certain imaging findings can be suggestive of TB spondylodiscitis. One such MRI feature evident in this case was relative sparing of the anterior meningovertebral ligament (Figure 2). This finding was sensitive and specific for TB spondylodiscitis in a retrospective cohort study comparing six cases of TB epidural abscess with 35 cases of pyogenic epidural abscess. 5
The growth of S. anginosus on multiple surgical tissue cultures also complicated the clinical picture of the case. The presence of tissue granulomas is not pathognomonic of mycobacterial disease; Brucella, Salmonella, and fungi are other possible etiologies for granulomatous spinal infections. 13 M. tuberculosis and S. anginosus co-infections, although rare, have been previously described in the literature, affecting the adrenal glands and psoas muscles. 14 All co-infections presented as abscesses with negative AFB smears and late positive mycobacterial cultures. Spinal TB co-infection with other microorganisms is an uncommon and infrequently documented event and publications are scarce. Large retrospective studies on vertebral osteomyelitis infectious causes have demonstrated the rarity of spinal TB co-infections despite the high frequency of TB. 15 One of the few spinal TB co-infection publications, performed in Taiwan, reported other organisms in 3/38 (8%) cases. Propionibacterium, methicillin-sensitive Staphylococcus aureus and Candida parapsilosis were co-isolated. 2 One consideration is that the presence of S. anginosus represented a contamination. However, given its growth on multiple tissue cultures, and this species’ well-known predilection for causing abscess formation, its presence most likely represented a true infection.
It is difficult to objectively discern the level of contribution of each of the isolated organisms in our patient’s disease but based on his clinical presentation we hypothesize that the existing thoracic spinal tuberculosis pre-dated and predisposed the patient to hematogenous seeding of the damaged vertebrae with S. anginsosus. This mechanism has been previously suggested to occur in other tissues. 14 Therefore, spinal TB should still be considered in the right clinical setting if the initial pathological picture supports the diagnosis even when other pathogens are isolated first.
Footnotes
Author contributions
WG: co-author of some or all sections of the case report including review of the pertinent literature
MP: co-author of some or all sections of the case report including review of the pertinent literature
MR: co-author of some or all sections of the case report including review of the pertinent literature
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Informed consent
Written informed consent for publication was obtained from the patient.
