Abstract
Introduction:
Anti-snake venom (ASV) is the standard therapy for the management of snakebite envenoming (SBE). Therefore, the knowledge of ASV among healthcare practitioners (HCPs) is essential for achieving optimal clinical outcomes in snakebite management. This study aimed to assess knowledge of ASV among the HCPs in northern Nigeria.
Methods:
We conducted a cross-sectional study involving eligible HCPs from different healthcare settings in northern Nigeria. The participants were recruited into the study using a combination of online (via Google Form) and face-to-face paper-based survey methods. The ASV knowledge of the respondents was measured using a validated anti-snake venom knowledge assessment tool (AKAT). Inadequate overall knowledge of ASV was defined as scores of 0–69.9%, and 70–100% were considered adequate overall knowledge scores. The predictors of ASV knowledge were determined using multiple logistic regression.
Results:
Three hundred and thirty-one (331) eligible HCPs were included in the study analysis (310 from online and 21 from paper-based survey). Overall, an estimated 12.7% of the participants had adequate knowledge of ASV. Adequate ASV knowledge was higher among physicians compared with other HCPs (21.7%; χ2 = 8.1; p = 0.04). Those without previous training on ASV (adjusted odds ratio [aOR], 0.37; 95% confidence interval [CI], 0.18–0.73; p = 0.004) and who have not previously administered/dispensed ASV (aOR, 0.31; 95% CI, 0.15–0.63; p < 0.001) were less likely to have adequate knowledge of ASV.
Conclusion:
The knowledge of ASV among healthcare practitioners in northern Nigeria is grossly inadequate. Experience with administering or dispensing ASV predicts ASV knowledge. Therefore, appropriate interventions are needed to improve ASV knowledge, particularly among the HCPs, for optimal healthcare outcomes.
Introduction
Snakebite envenoming (SBE) is an important occupational and public health hazard. 1 Annually, about 5.4 million people mainly in Asia and Africa, suffer from SBE-related mortality and morbidity due to a lack of or inadequate access to appropriate treatment. 2 The crisis has reached an alarming level in sub-Saharan Africa, and in 2017 the World Health Organization (WHO) enlisted SBE as a high priority (category A) neglected tropical disease. 3
Nigeria is reported to have one-fifth of all West African regional snakebite cases, with 174 cases in every 100,000 hospital admissions. 4 The burden associated with it is on the increase in the northern part of Nigeria.5,6 This region was reported to have the highest number of snakes than other regions of the country put together.4,5 The sub-Saharan African burden was estimated at 1.03 million disability-adjusted life years per annum, out of which Nigeria has the highest burden with 43% of the total burden in West Africa.7,8 A 19-year Federal Ministry of Health hospital record showed that 76,310 cases of snakebite with 910 cases of death have been recorded in some snakebite hospitals in northern Nigeria. 9
Despite high numbers of snakebites in Africa, there has been a decline in production and loss of product confidence of antivenom products. Good quality anti-snake venom (ASV) made by proven, long-established manufacturers has become unaffordable to all except a few. 10 These result in the proliferation of ASV with questionable efficacy manufactured mainly not for Nigerian snakes.11,12 ASV is the only standard antidotal treatment available for SBE, and it has been in clinical use for over a century. 13 High-quality ASVs against the most medically important snakes exist. However, some regions where snakebite incidence is common are lacking such adequate ASV and storage facilities. 12 ASVs are recommended to be stored at a temperature within the range that assures stability; this is particularly critical for liquid formulations, which usually require storage at 2–8°C. Therefore, deviations from this temperature range due to interruptions in the cold chain during transportation or storage are likely to result in product deterioration. 13 However, handling of ASV such as distribution, storage, and use has been recognized to be challenging in sub-Sahara Africa where the temperature is high. There are also possible problems related to logistics, including lack of proper storage facilities, inadequate electricity supply, and absence of technical capacity. 14
Despite the increasing incidence of SBE in tropical regions, the literature suggests poor knowledge of snakebite management among HCPs in Africa and Asia.15,16 A study by Michael and colleagues reported a low overall knowledge of venomous snakes, snakebite first aid, treatment, and prevention among physicians in northern Nigeria. 17 Similarly, a study in Asia revealed a gap in baseline knowledge and confidence of therapy among physicians in Hong Kong. 16 It was also reported that knowledge of snakebite management among physicians and nurses in the Savannakhet Province of Laos was insufficient to ensure snakebite patients’ professional care in the province. 18 The authors also identified that ASV knowledge deficits related to the dosage and appropriate administration have also been identified among physicians and nurses. 18 Intern physicians were also found lacking in general knowledge of snakebite management in West Bengal. 19 Therefore, there is a considerable need to continually evaluate ASV knowledge among other HCPs to guide interventions for improving the expertise and subsequent optimal clinical outcomes.
To our knowledge, there is no published data on ASV knowledge among the HCP from northern Nigeria. The region has a high incidence of SBE in the country,4,5 thus making it a suitable setting for this study. Therefore, we aimed to assess the baseline ASV knowledge and determinants of ASV knowledge among HCPs in northern Nigeria.
Methods
The survey was conducted and reported based on the Checklist for Reporting Results of Internet E-Surveys (CHERRIES) statement 20 and it was followed by using the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines as attached in Supplemental Appendix 1. 21
Ethics statement
The study was approved by the ethics committee of the College of Health Sciences, Bayero University, Kano, with approval number (no. BUK/CHS/REC/VII/59). The study was conducted based on the Declaration of Helsinki 2018.
Study design/population
This cross-sectional study was conducted among selected HCPs, namely doctors, pharmacists, nurses, and pharmacy technicians. The study protocol has been published elsewhere. 22
Sample size determination
The sample size (n) for the study was calculated using two mean sample estimations with the aid of OpenEpi version three software (www.openepi.com). Assuming a finite population, a standard deviation (SD) of 10, and the mean difference between groups was set at two, with a power of 80% and a confidence interval of 95%. The attrition rate of 10% was assumed. We calculated a minimum sample of 304.
Inclusion criteria
The eligibility criteria included HCPs working at healthcare facilities in northern Nigeria. The HCPs with more than one year post-internship experience practising in primary, secondary, and tertiary health institutions of urban and rural settings of the study locations were included in the study.
Exclusion criteria
The HCPs under training, such as interns and those undergoing the National Youth Service Corps (NYSC) were excluded from the study.
Study settings
The study was among HCPs drawn from urban and rural healthcare facilities in northern Nigeria, including secondary healthcare facilities dedicated to the management of SBE. The target facilities were hospitals, community pharmacies, government-owned medical stores, and relevant drug distribution companies. Northern Nigeria comprises three geo-political zones: north-west, north-east, and north-central, with an estimated population of 90 million and an average temperature of 37°C. 23
ASV assessment tool
We utilized a validated anti-snake venom knowledge assessment tool (AKAT) to conduct the knowledge assessment of ASV among HCPs which is presented in the attached Supplemental Appendix 2. 24 The AKAT was validated using extensive psychometric tests as recommended in previous studies.25–27 Both the AKAT and the study protocol have been published elsewhere by the same authors.22,24
The ASV tool consists of a total of 28 items covering knowledge (19 items), availability, cost, and logistics (9 items) of ASV in northern Nigeria. The availability, cost, and logistics were presented as frequencies and percentages. For the ASV knowledge assessment, the items were scored as 0 for a wrong answer and 1 or 2 for a correct answer. From the 19 items covering knowledge, 15 items were scored 1 point each, while the remaining four items were scored 2 points each. This implies that a respondent could score from zero to a maximum of 23 points. We calculated the percentage of correct responses among the respondents for each item. The respondents’ overall knowledge score was calculated as the percentage of their scores from the total of the 23 knowledge-based scores. We grouped the respondents into two as those with inadequate and adequate ASV knowledge. Inadequate overall knowledge was defined as scores of 0–69.9%, and 70–100% were considered adequate overall knowledge scores, based on a similar study involving physicians only. 17
Study procedure/recruitment
The study was conducted using a combination of an online survey platform (via Google Forms) and a face-to-face survey platform (paper-based). The AKAT was designed in the form of a Google survey. The survey link consisting of a hyperlink to the survey page, study information, a statement about implied consent, and an invitation to participate was developed using Google Forms. The link was then shared with the HCPs via social media platforms (personal and professional group accounts) such as Facebook and WhatsApp, belonging to the HCPs. The link was also shared on the Twitter handle and LinkedIn profile of the authors. For the face-to-face survey, two research assistants were involved in the data collection, especially in the rural areas with limited internet access. The answers to the questions on the tool were only made available to the participants on request after the data collection period.
Consent statement
Face-to-face data collection
Consent of the participants was obtained by sharing the consent form on the first page of the survey. The consent statement contains a brief introduction to the study, confidentiality statement and an invitation for a voluntary participation. The participants were asked to sign the consent form before proceeding.
Online data collection
The survey link consisting of a hyperlink to the survey page, study information, confidentiality statement, consent statement, and an invitation to participate was developed using Google Forms. The participants were asked to click on the consent statement before proceeding.
Data preparation and analysis
The data collected from the Google form and paper-based responses were transferred into a Microsoft Excel sheet. The final data in Microsoft Excel format were exported into IBM SPSS Statistics for Windows, version 24.0. Armonk, NY, USA: IBM Corp. The data were then cleaned and analyzed. The numerical variables were presented as mean (SD) or median (interquartile range) depending on the normality distribution of the data. Categorical variables were reported as frequencies and percentages. The factors associated with knowledge of ASV were determined using multiple logistic regression (MLR). The chi-square test was used to screen each variable for multivariable analysis and assess the association between variables and ASV knowledge. From the results of chi-square, variables that had a p < 0.25 were included in the MLR. 28
The MLR was performed using the backward likelihood ratio method to determine the adjusted odds ratios (aORs) of the potential independent predictors. Multicollinearity and interactions between the variables were checked. Assumptions were checked using Hosmer–Lemeshow and omnibus tests of model coefficients. The final model was presented as aOR, 95% confidence interval (CI), and corresponding p-values; p < 0.05 was considered statistically significant.
Results
Recruitment of the study respondents
Three hundred and sixty-seven respondents responded to the survey (346 online and 21 paper-based). Of these, 28 (7.6%) did not complete the survey and were excluded. Similarly, five (1.4%) were excluded because they were not HCPs, while three (0.8%) were excluded because they were considered careless responders (many serialized items with a response of ‘first option’). Finally, 331 (90.2%) participants with complete responses were included in the final analysis. Figure 1 summarizes the recruitment process of the study participants.
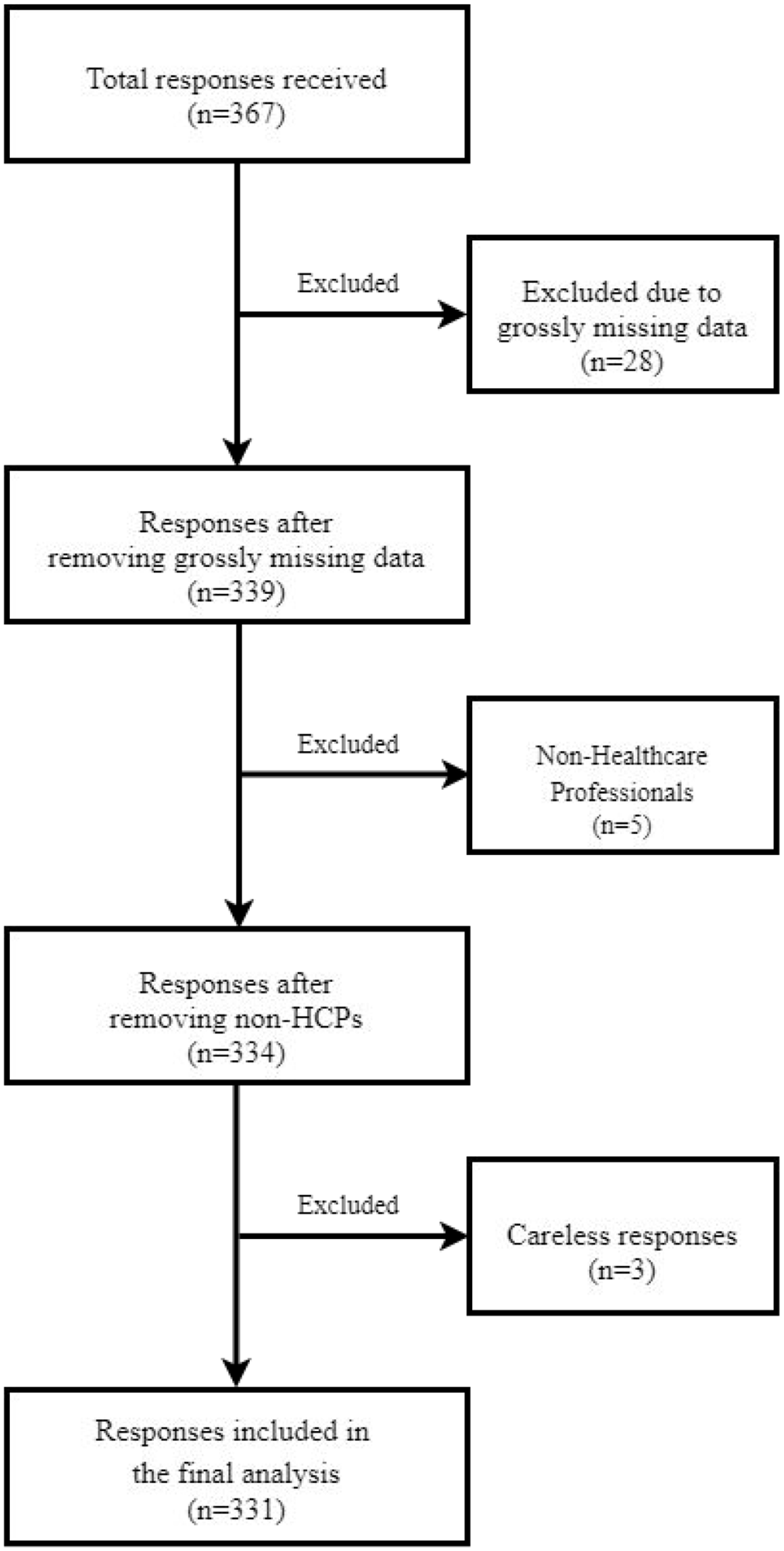
The flowchart of the recruitment process of the study participants.
Sociodemographic characteristics of the respondents
Of the 331 respondents included in the final analysis, the majority were men 223 (67.4%), pharmacists 211 (63.7%), those working in healthcare facilities located in the northwest region (54.2%). The mean age (SD) of the respondents was 34.4 (8.5) years. The mean (SD) year of post-graduation working experience was 9 (8.2) years. The majority have had previous training on ASV 211 (63.7%). Most of the respondents, 268 (81.0%), did not report a prior history of administering/dispensing ASV to patients. The details of the sociodemographic characteristics of the HCPs are shown in Table 1.
Sociodemographic characteristics of respondents (n = 331).

ASV, antisnake venom; SD, standard deviation.
Assessment of ASV knowledge among respondents
Only 42 (12.7%) of the HCPs had overall adequate ASV knowledge scores. A higher proportion of physicians (21.7%) had an adequate knowledge score than other HCPs (χ2 = 8.1, p = 0.04). Among respondents who did not previously administer/dispense ASV, those with adequate knowledge scores were more (9.0% versus 91.0%; χ2 = 17.7, p < 0.001) than those without adequate knowledge. A lower proportion of respondents with adequate knowledge (21.7% versus 78.3%; χ2 = 13.7.0; p < 0.001) was also identified among participants with previous training on ASV compared with those who did not have previous training. The distribution of ASV knowledge is presented in Table 2.
Distribution of ASV knowledge scores among the respondents.

Scores 0–69.9% = inadequate knowledge and 70–100% = adequate knowledge; N = 310.
ASV, antisnake venom; NGO, non-governmental organization; SD, standard deviation.
Item-by-item analysis of the knowledge of ASV among respondents
Table 3 illustrates the correct responses of the respondents to each knowledge item in the AKAT. Of the included respondents, 25.4% correctly answered items related to the conventional dosage formulations, 18.7% common side effects, and 27.5% treatment of ASV reactions. The majority of the respondents correctly reported its major components (82.2%), monovalent ASV (86.4%), ASV for snakebites of unknown species (91.2%), administered orally (95.8%), and administered intravenously (87.9%).
Item-by-item analysis of the knowledge of ASV.

ASV, antisnake venom.
Factors associated with ASV knowledge among HCPs
Of all the variables analyzed in the bivariate analysis, three had a p < 0.25 and were included in the MLR. The final model of the MLR demonstrates that respondents without prior training (aOR, 0.365; 95% CI, 0.182–0.729; p = 0.004) and who have not previously administered/ dispensed ASV (aOR, 0.306; 95% CI, 0.150–0.625; p < 0.001) were less likely to have adequate ASV knowledge. No possible collinearity in the included variables was observed. Interactions between variables were also assessed, and none were identified. The model fit for the data was excellent; the Hosmer–Lemeshow test was significant (p = 0.428). The final model was presented as adjusted ORs with 95% CIs and corresponding p-values in Table 4.
Multiple logistic regression – final model.

Hosmer and Lemeshow test, p = 0.428; Omnibus tests of model coefficients, p < 0.001; classification table, 87.3%.
Adjusted for training on ASV and administration or dispensing of ASV.
ASV, snake antivenom; CI, confidence interval; OR, odds ratio.
Availability, cost, and logistics
Data show that most of the HCPs’ facilities did not distribute ASV in the last 24 months (69.4%), from July 2017 to May 2019. The common brands of ASV distributed were EchiTab (36.8%), Premium Pan Africa (27.5%), and Vins (16.8%). The quantities distributed mainly were less than 10 (68.3%) vials. The majority were polyvalent (73.7%), liquid preparations (72.6%), obtained mainly from the government (31.6%), wholesalers (35.8), and retail pharmacies (24.2%) with a cost ranging between NGN 5000 and 35,000, which was equivalent to 15–95 USD at the time of data collection (2019). The details of the availability, cost, and logistics of ASV are shown in Table 5.
Availability, cost, and logistics.

Discussion
Our study assessed the baseline ASV knowledge among HCPs in northern Nigeria. We found a grossly inadequate knowledge of ASV among the respondents. Although profession, previous training on ASV, and history of administering/dispensing ASV were found to be associated with knowledge, the predictors of ASV knowledge were found to be previous training on ASV and history of administering/dispensing of ASV. These findings would guide the development and implementation of interventions for improving ASV knowledge among HCPs and subsequent optimal outcomes.
Our study found that about 12.7% of the HCPs had adequate overall ASV knowledge. This finding corroborates previous studies that reported inadequate knowledge of SBE management among some HCPs in Hong Kong and South Africa. The study in Hong Kong reported that only 29.0% of physicians had the confidence to treat snakebite patients. Similarly, only 17.0% of general practitioners in Free State, South Africa, knew enough to treat snakebites.8,29 Conversely, other studies reported that 45.4% of healthcare workers had adequate knowledge to manage snakebites in Lao PDR 9 , and 50.8% of physicians in northern Nigeria had adequate knowledge of ASV treatment. 17 Our findings concerning these studies are a clear indication that knowledge of ASV in northern Nigeria, where there is a high incidence of SBE, is grossly inadequate.
Similarly, a study in Cameroon found that HCPs have inadequate knowledge of SBE first aid and treatment. 7 There is likely inadequate knowledge of ASV among HCPs in tropical West Africa. The lack of adequate knowledge may be related to the fact that SBE has been neglected for years until the June 2017 declaration by the WHO of SBE as a high priority neglected tropical disease. 3 This highlights the need for proper training of HCPs on SBE management.
Despite the overall lack of adequate knowledge among HCPs, respondents with previous training on ASV and those with a history of administering/dispensing ASV demonstrated a higher overall knowledge score than those with no prior training or administration/dispensing experience. Therefore, it is an indication that administration/dispensing experience and training can improve ASV knowledge among HCPs, as reported by other studies in Asia 16 and Africa. 15 Furthermore, the WHO guidelines recommend that all clinicians should urgently decide to administer ASV following a snakebite. 14 This finding indicates that many HCPs, particularly the clinicians managing snakebites in the region, could improve their knowledge of SBE treatment. This could be achieved through regular snakebite management training.
Our study further showed that the predictors of overall ASV knowledge score were HCPs with no previous training on ASV and those that have not previously administered/dispensed ASV. This substantiates another finding of this study that shows that previous training and administration/dispensing of ASV increased the odds of adequate overall knowledge. 15 This suggests that exposure to ASV is likely to improve their knowledge. Our findings corroborate the previous study that identified experience in snakebite management participation as a determinant that improves overall knowledge.15–17
Although the most common ASV brands were found to be EchiTab Plus, Premium, and Vins ASV, we discovered that other brands were available in the open drug markets, including ASVs meant for Asia from Premium and Vins antivenom manufacturers. The quantities of ASV stored at a given time by healthcare facilities in northern Nigeria were mainly less than 10 (68.3%) vials, and the majority were polyvalent (71.6%), liquid preparations (70.6%). These ASVs were mainly obtained through donations from the government distribution agencies (32.4%), wholesalers (35.3%), and retail pharmacies (22.5%), with a cost ranging between 15 and 95 USD per vial. These findings showed that ASV is still primarily unavailable in desired quantities; it was also found to have a very high cost for the affected communities, as reported by earlier studies. 12 Therefore, policy-makers need to focus more on improving the availability and cost-effectiveness of ASV.
Public health implications
ASV remains the mainstay for the treatment of SBE. However, it contains proteins and can easily get denatured if not handled appropriately, leading to poor therapeutic outcomes. It is also essential for clinicians to be aware of the tendency of ASV to cause a severe life-threatening hypersensitivity reaction. Currently, there are no local manufacturers of ASV in Nigeria, and ASVs are being imported from Asia, Europe, and South America. 12 These factors make knowledge of ASV and its logistics vital to HCPs. Our study found that some of the respondents who have had previous ASV training lack adequate knowledge of ASV 26 (21.7%). The majority of the respondents received training through workshops (53%) and training at school (23%). This indicates that implementation of envenoming management in the healthcare school curriculum will significantly improve the knowledge of SBE management among HCPs. We therefore suggest that interventions should focus on regular training on ASV to HCPs, especially those in areas most affected by SBE.
Furthermore, SBE management, ASV logistics, storage, and administration should be incorporated into the undergraduate and continuing educational development (CED) curriculum of HCPs. ASVs are part of the WHO list of essential medicines, yet the commodities are scarce in our health facilities. Funding for procurement of the commodity is paramount to improve availability and access.
We also found that most respondents (82.2%) are aware of the significant components of ASV, the appropriate type of ASV to be used for an unknown species of snake (91.2%), the appropriate route of administration (87.9%) and that ASV can cause hypersensitivity reaction (94.9%). Yet, there are areas of knowledge that require significant improvement among HCPs that responded to the survey, these include common side effects of ASV (18.7%), treatment of ASV reactions (27.5%), ASV dose (57.1%) and dosage forms (25.4%). This is an indication that more emphasis on ASV as the only drug for the management of SBE needs to be employed during training of HCPs either in schools or during workshops.
Limitations
The present study is not without limitations. Although we conducted a mixed survey method of online and face-to-face data collection, some of the inherent shortcomings of the online survey might prevent us reaching some of the targeted respondents. We acknowledge the limitations of the convenience sampling method employed in the study. Furthermore, the knowledge questions on ASV may not be exhaustive.
Recommendations
We suggest that HCPs should have adequate knowledge of SBE and ASV for optimal clinical outcomes. We therefore suggest an urgent intervention to improve ASV knowledge among the HCPs in northern Nigeria. Accordingly, our findings could guide the development and implementation of educational interventions, modules for CED for HCPs, health promotion through the display of flyers at healthcare facilities, workshops/seminars, and government policies to improve ASV knowledge and positive outcomes.
Conclusion
The knowledge of ASV among healthcare professionals in northern Nigeria is grossly inadequate. We identified predictors of ASV knowledge among the respondents to include experience with administering or dispensing ASV and previous training on ASV. Therefore, there is a need for intervention to improve ASV knowledge through training and continuing education programmes among HCPs.
Supplemental Material
sj-pdf-1-tai-10.1177_20499361211039379 – Supplemental material for Knowledge assessment of anti-snake venom among healthcare practitioners in northern Nigeria
sj-pdf-1-tai-10.1177_20499361211039379 for Knowledge assessment of anti-snake venom among healthcare practitioners in northern Nigeria by Auwal A. Bala, Abubakar I. Jatau, Ismaeel Yunusa, Mustapha Mohammed, Al-Kassim H. Mohammed, Abubakar M. Isa, Abubakar S. Wada, Kabiru A. Gulma, Inuwa Bello, Sani Malami, Godpower C. Michael and Basheer AZ. Chedi in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-pdf-2-tai-10.1177_20499361211039379 – Supplemental material for Knowledge assessment of anti-snake venom among healthcare practitioners in northern Nigeria
sj-pdf-2-tai-10.1177_20499361211039379 for Knowledge assessment of anti-snake venom among healthcare practitioners in northern Nigeria by Auwal A. Bala, Abubakar I. Jatau, Ismaeel Yunusa, Mustapha Mohammed, Al-Kassim H. Mohammed, Abubakar M. Isa, Abubakar S. Wada, Kabiru A. Gulma, Inuwa Bello, Sani Malami, Godpower C. Michael and Basheer AZ. Chedi in Therapeutic Advances in Infectious Disease
Research Data
sj-xlsx-1-tai-10.1177_20499361211039379 – Supplemental material for Knowledge assessment of anti-snake venom among healthcare practitioners in northern Nigeria
sj-xlsx-1-tai-10.1177_20499361211039379 for Knowledge assessment of anti-snake venom among healthcare practitioners in northern Nigeria by Auwal A. Bala, Abubakar I. Jatau, Ismaeel Yunusa, Mustapha Mohammed, Al-Kassim H. Mohammed, Abubakar M. Isa, Abubakar S. Wada, Kabiru A. Gulma, Inuwa Bello, Sani Malami, Godpower C. Michael and Basheer AZ. Chedi in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
The authors would like to thank and appreciate Professor Habib G. Abdulrazaq, Dr. Garba Iliyasu, Dr. Hamza Muhammad (Infectious Disease Unit, Aminu Kano Teaching Hospital) for providing technical support during tool development. We also thank Mr. Dauda Puki (Federal Medical Centre, Azare), Dr. Shuaibu Hamza Auwal and Mr. Abba Rabiu Usman (General Hospital Alkaleri), Dr. Mana Dillos (Abubakar Tafawa Balewa University Teaching Hospital, Bauchi), Mr. Umar Danjuma (Ibrahim Badamasi Babangida Specialist Hospital, Minna), Mr. Yusuf Mohammed (Bauchi State University, Gadau) and Mr. Micah Musa (Snakebite and Research Hospital, Kaltungo) for their recommendations and roles in data collection. Special appreciation to all Young Pharmacist Scholars (YPS) members for their support, guidance, and data collection. We also wish to appreciate the Nigerian Snakebite Research and Intervention Centre (NSRIC) for their contribution and recommendations.
Author contributions
Auwal A. Bala conceptualized the original idea, developed the theory, and co-wrote the manuscript. Abubakar Ibrahim Jatau developed the study methods and co-wrote the manuscript. Ismaeel Yunusa and Mustapha Mohammed designed and conducted the formal analyses. Al-Kassim Hassan Mohammed wrote the introduction, data curation and co-wrote the manuscript. Abubakar Musa Isa and Wada Abubakar Sadiq performed the literature review and data curation. Kabiru Abubakar Gulma and Inuwa Bello designed the questionnaire, co-wrote, and edited the final manuscript. Godpower C. Michael and Sani Malami edited and critically reviewed the manuscript for intellectual content. Basheer Z.A. Chedi gave the supervisory approval and finally revised the manuscript for intellectual content.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online as appendix 1 and 2.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
