Abstract
Background:
A better understanding of the epidemiology of cryptococcal infection in HIV-negative individuals is an international research interest. Immune dysfunction in diabetes mellitus (DM) significantly increases the risk of acquiring and reactivation of infection due to Cryptococcus neoformans. Risk factors and outcomes of cryptococcosis in DM are not well documented.
Objective:
The objective of this study was to determine the clinical characteristics and outcomes of cryptococcal infections in persons living with DM.
Methods:
MEDLINE (via PubMed), EMBASE, and the Cochrane Library databases were searched in November 2020. The searches covered the period between 1980 and 2020.We included studies that reported confirmed cryptococcosis in patients with DM. Reference lists of included articles were also searched, and additional studies were included if appropriate. No language restriction was applied. Single case reports, case series and original articles were included whereas review articles were excluded.
Results:
A total of 28 studies (24 single case reports, 4 retrospectives) were included involving 47 unique patients from Asia (17 cases), North America (six cases), South America (three cases) and Africa (two cases). Men constituted 75% (n = 18) of the cases. Median age was 60.5 (range: 27–79) years. The majority of the patients had cryptococcal meningitis (68.1%, n = 32) followed by disseminated cryptococcosis (6.4%, n = 7), and others (isolated cutaneous disease one, peritonitis one, pleural one, thyroid one, adrenal one). Diagnosis was achieved through either culture and microscopy (38/47), cryptococcal antigen tests (9/47) or histopathology (9/47) singly or in a combination. All-cause mortality was 38.3% (n = 18). Among those with meningitis mortality was 36.2%.
Conclusion:
A wide spectrum of cryptococcal infections with varying severity occurs in DM. Mortality remains unacceptably high. There is a need for more studies to characterize better cryptococcal disease in DM.
Introduction
Cryptococcosis is an opportunistic, multi-systemic fungal infection with a predilection for the central nervous system (CNS). It is acquired through inhalation of airborne yeast cells (basidiospores) and subsequent pulmonary infection of mainly two etiological agents, Cryptococcus neoformans and Cryptococcus gattii. 1 Soil containing pigeon droppings are the main reservoir for C. neoformans, whereas eucalyptus trees and decaying hollows in living trees are the major environmental source for C. gattii.2–4 C. neoformans is the most predominant cause of cryptococcosis and accounts for 80% of cases globally. Immune suppressive states such as HIV/AIDS, immunosuppressive drugs, and solid organ transplant recipients are key risk factors for acquisition and reactivation of the infection. Conversely, C. gattii is less frequent (20%), and a growing number of cases occur mainly in immunocompetent individuals.5–7
There is growing data on the epidemiology of cryptococcosis among HIV-negative populations. 8 In a recent prospective study conducted among HIV-negative people in the United States, CNS cryptococcosis was found to be present in 50% of 145 study subjects with underlying immunosuppression, and was associated with recurrent neurological morbidities and pressure-related complications. 9 In a recent review of isolated pulmonary cryptococcosis among HIV-negative patients, men were found to be more prone than women, and isolated pulmonary cryptococcosis was associated with underlying diseases and immune dysfunction. 10 The key risk factors of cryptococcosis in HIV-negative individuals are systemic lupus erythematosus and other autoimmune rheumatic diseases, smoking, corticosteroid use, and chronic obstructive pulmonary disease, 7 cirrhosis, 11 pregnancy and diabetes mellitus (DM). 12
DM is one of the most common predisposing underlying diseases for cryptococcosis in HIV-negative individuals, accounting for 10–20% of cryptococcosis patients with underlying diseases.10,13–15 Cryptococcal meningitis in patients with DM is associated with poor clinical outcomes.16,17 Hyperglycemia seen in DM patients causes defects in the host’s immune system which predisposes DM patients to opportunistic infections such as cryptococcosis. 18 In mainland China, a retrospective study among Chinese individuals with type 2 DM found a hospital-based cryptococcal prevalence of 0.21%, with 62% having DM before infection. 19 In a study in Taiwan, in addition to DM being associated with the occurrence of cryptococcosis in HIV-negative patients, it was also found to be associated with a 1-year and overall mortality from cryptococcosis and cryptococcal meningitis. 20
There have been few data available to understand cryptococcosis clinical manifestations, management, and outcomes of cryptococcosis in people without HIV. 21 In addition, the role of the different types of DM in the development of cryptococcosis in DM patients is unclear. We undertook this study to describe the epidemiology, clinical manifestations and treatment outcomes of cryptococcosis in patients with DM through a scoping review of case reports, case series and studies of the epidemiology and clinical characteristics of DM patients with cryptococcosis.
Methods
We systematically explored international medical databases: MEDLINE (via PubMed), Embase, and the Cochrane Library in November 2020. The searches covered the period between 1980 and 2020. The search terms used were ‘disseminated cryptococcosis’, ‘cryptococcal meningitis’, ‘cryptococcal pneumonia’, ‘diabetic mellitus’, ‘organ transplant’. Boolean operators AND, OR and NOT were used appropriately. No language restrictions were applied.
Reference lists of included articles were also searched, and additional studies were included if appropriate. We included single case reports, case series and original articles and excluded review articles and studies including DM and other known risk factors for cryptococcosis such as HIV/AIDS and solid organ transplant (Figure 1). We included studies that reported confirmed cryptococcosis in DM patients. The confirmation of cryptococcosis was based on culture and microscopy, positive Indian ink staining, histopathological examination and/or cryptococcal antigen screening (CrAg). The diagnosis of DM should have been made prior to cryptococcosis diagnosis. We collected the following data: publication data, age, gender, study type, clinical characteristics, other underlying conditions, diagnostic modalities, and disease outcomes.
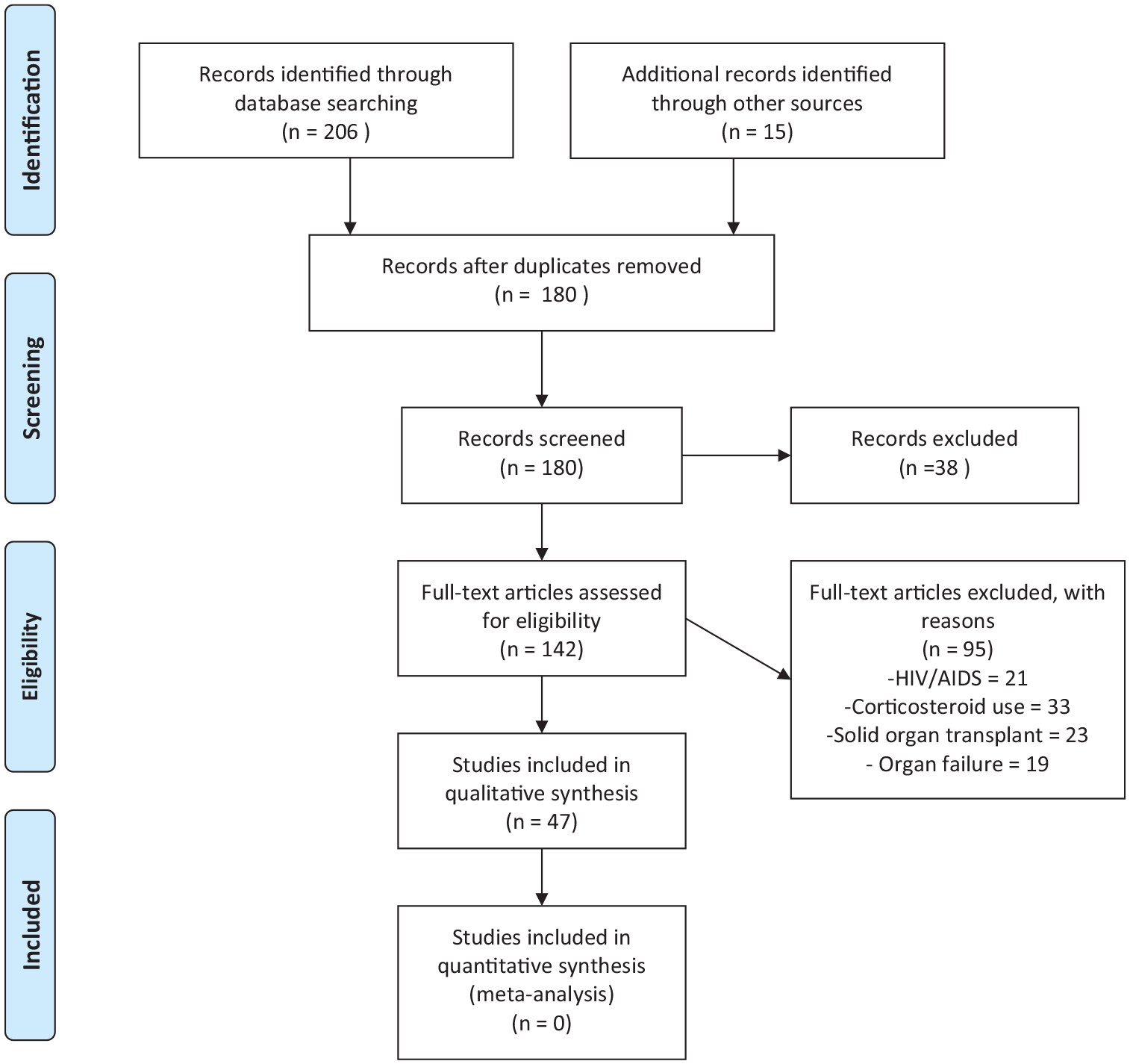
PRISMA flow diagram.
Data were analyzed using Microsoft Excel 2016 and descriptive statistics (mean or median for numerical data; frequency and percentages for categorical variables) were employed. This study did not require approval by the institutional review board.
Results
Epidemiology of cryptococcosis among patients with DM
We identified 28 studies, including 47 patients from across four different continents: Asia (60.7%, n = 17), North America (21.4%, n = 6), South America (10.7%, n = 3), and Africa (7.1% n = 2). Most of the studies were single case reports (85.7%, n = 24) and retrospective (14.3%, n = 4) studies. Male patients (75%, n = 18) were recorded more than female patients (25%, n = 6). The median age was 60.5 years (27–79) (Table 1).
Study characteristics.
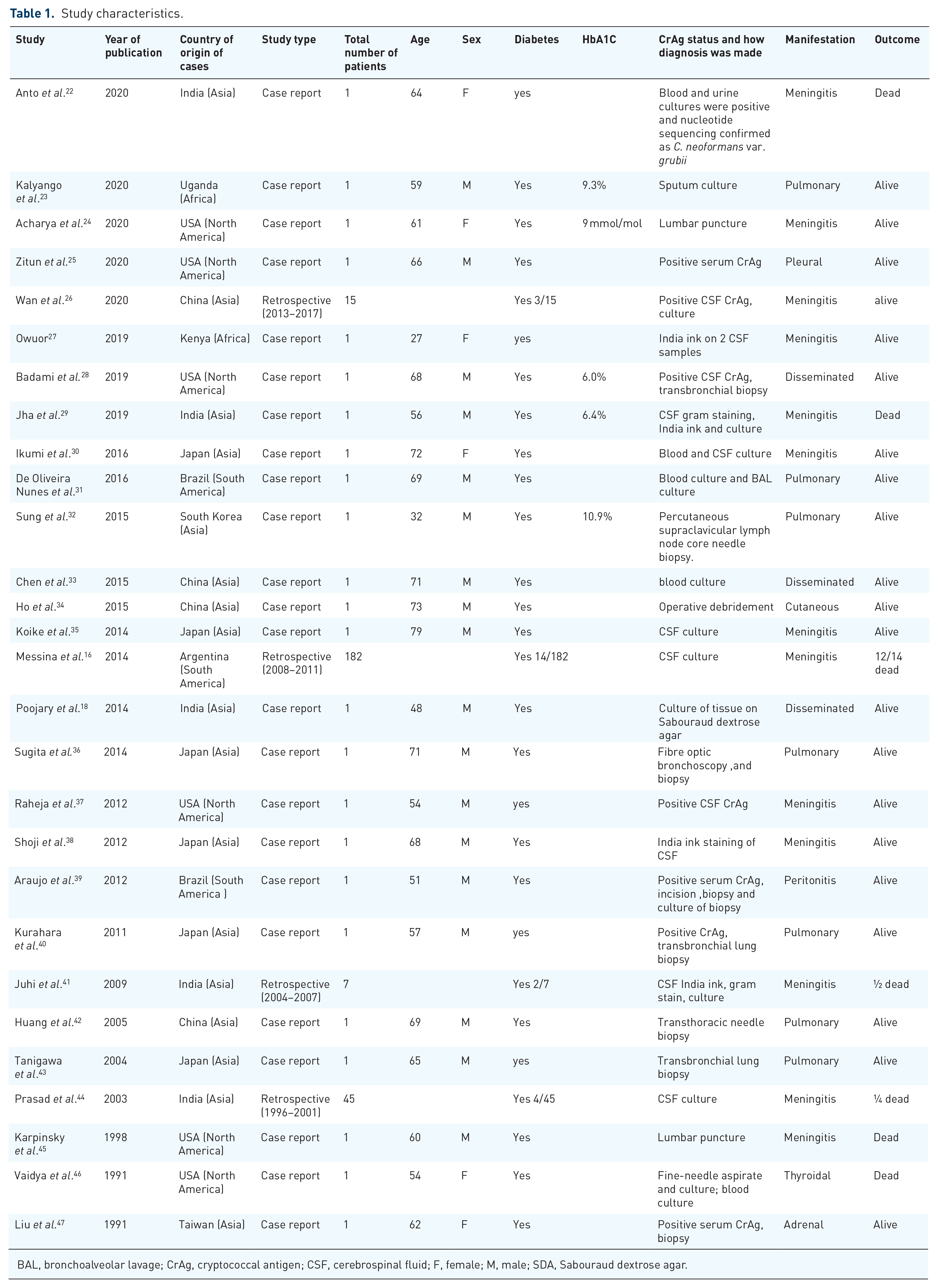
BAL, bronchoalveolar lavage; CrAg, cryptococcal antigen; CSF, cerebrospinal fluid; F, female; M, male; SDA, Sabouraud dextrose agar.
Clinical manifestations of cryptococcosis among patients with DM
The majority of the patients had cryptococcal meningitis (68.1%, n = 32), followed by disseminated cryptococcosis (6.4%, n = 3), isolated pulmonary disease (14.9%, n = 7), and others (10.6%, n = 5) (Table 1). The most reported presenting complaints among cryptococcal meningitis were headache (57.1%, n = 8), fever (57.1%, n = 8), altered mental status (35.7%, n = 5), and nausea and vomiting (28.6%, n = 4). Isolated pulmonary cryptococcosis common symptoms included fever (42.9%, n = 3), shortness of breath (42.9%, n = 3), and cough (100%, n = 7). Although patients with disseminated disease presented with varying symptoms, fever was the most common among the cases.
Diagnosis of cryptococcosis among patients with DM
The diagnosis of cryptococcal meningitis was made by culture and microscopy of cerebrospinal fluid (CSF) 81.3% (n = 26), India ink 21.9% (n = 7), and CSF cryptococcal antigen 12.5% (n = 4), whereas the diagnosis of isolated pulmonary disease was made by culture 42.9% (n = 3) and histopathology 71.4% (n = 5). Disseminated disease was diagnosed by culture and microscopy culture 66.7% (n = 2), and other tests listed (Table 2).
Diagnostic yield stratified by sample type and clinical syndrome of cryptococcosis in patients with diabetes mellitus.

BAL, bronchoalveolar lavage; CrAg, cryptococcal antigen; CSF, cerebrospinal fluid.
Treatment outcomes of cryptococcosis among patients with DM
Of the 47 patients, 38.3% (n = 18) patients died on therapy. Mortality in patients with meningeal disease was 36.2% (n = 17) and thyroid disease 0.21% (n = 1), whereas patients with pulmonary disease and disseminated disease reported recoveries (Table 1).
Discussion
There is an emerging body of evidence, mainly from observational studies and case reports, to support DM as an important risk factor for cryptococcosis.39,48–50 In the present study, we sought to describe the clinical characteristics and outcomes of cryptococcosis in people living with DM. We found that the median age was 60.5 years, which is consistent with previous studies that showed a prevalence of cryptococcosis and DM in the older population. 51
According to a population-based study in Taiwan reported by Lin and colleagues, 30.6% of 4587 people who were found to have cryptococcosis had DM as the underlying condition. 20 In that study, it was argued that the higher prevalence of cryptococcosis in DM was attributed to the higher age of DM patients. However, after accounting for the effect of age through case–control matching, a relationship between DM and cryptococcosis was demonstrated. 20
Our data showed a male predominance, which was similar to previous studies.52,53 A proposed explanation for the male gender predominance could be a difference in the host’s immune response between men and women. The immune modulating effect of testosterone makes men more susceptible to IRIS. 54 Furthermore, we found that cryptococcal meningitis was the most frequent form of cryptococcal disease in patients with DM. This is in line with a multicenter study performed in the USA with cryptococcosis in people living with HIV. 55 However, Baddley and colleagues found that in a study of 166 HIV-negative individuals, pulmonary cryptococcosis was the most prevalent form of cryptococcal disease, which is in contrast with our findings. 56
The clinical manifestations of cryptococcal infection vary depending on the patient’s immune status and site of infection, from asymptomatic to having cough, fever, pneumonia, meningoencephalopathy, spread to multiple body sites, severe life-threatening disease, or even death. However, due to the predilection of C. neoformans for the CNS, the most common clinical presentation of cryptococcosis is meningoencephalitis whose signs include meningismus, papilledema, cranial nerve palsies, focal neurological deficits, and depressed level of consciousness. 2 The severity of cryptococcosis depends on the characteristic virulence factors of the Cryptococcus and host immune system responses. A predominant T helper (Th) immune response is associated with protection from cryptococcosis whereas Th-2 responses are associated with susceptibility to cryptococcal infection.7,57 According to this study, common clinical manifestations of cryptococcal disease included: headache, fever, shortness of breath, altered mental status, nausea and vomiting in the respective sites of infection.
Furthermore, this study indicated an overall mortality of 21.3%, with the death rate in meningeal disease as high as 43.8%. No mortalities were reported among patients with disseminated or pulmonary disease. Cases have been reported of pulmonary cryptococcosis resolving without treatment in immunocompetent individuals. 13 Lin et al. was able to demonstrate a higher 1-year and overall mortality among HIV-uninfected individuals with DM than those without DM in cryptococcosis and cryptococcal meningitis. 20
A study in the USA identified DM as a major risk factor for cryptococcal disease in solid organ transplant and immunocompetent patients. 58 Other predisposing conditions related to cryptococcosis include: recipients of immunosuppressive treatment, this was the most common in this study, HIV/AIDS, chronic renal failure, malignancy, and Cushing’s syndrome. 59 Cell mediated immunity is the most important host response to cryptococcal disease, hence the high incidence of disease among immunosuppressed individuals. DM leads to decreased cell mediated immunity through neutrophilic chemotaxis impairment, abnormal phagocytosis, poor lymphocyte blast transformation, and defective opsonization. Hyperglycemia also influences the microbicidal function of macrophages. This allows for opportunistic infections and is hypothesized to be the relationship between cryptococcal infection and DM.60–62
Culture of appropriate respiratory samples is the gold standard for the diagnosis of cryptococcal infection hence it comes as no surprise that it was the most commonly favored diagnostic modality in this study. The use of CrAg detection for confirming the diagnosis was also widely favored because it is more convenient for all hospital settings and takes a shorter duration of time. 63 In a large validation study in Africa by Boulware et al., 64 CrAg lateral flow assay (LFA) reported a high sensitivity (99.3%) and a high specificity (99.1%) in HIV-positive individuals and a median sensitivity and specificity of 100% [95% confidence interval (CI) 95.6–100%] and 99.5% (95% CI 95.7–100%) whereas culture, although specific, has a low sensitivity (50–80%). 65
The limitations of our study were an inability to have a complete picture on diabetic control and cryptococcal infection because hemoglobin A1C was not recorded for the majority of articles and there were few data on Cryptococcus species typing. Further studies should be done to confirm the relationship between cryptococcosis and other comorbidities and also to document the commonest species of Cryptococcus encountered in DM. Studies concerning diabetic control in cryptococcal infection would be insightful.
Conclusion
There is significant evidence to support the relationship between cryptococcosis and DM. Therefore, health workers should have a higher index of suspicion when presented with elderly patients with DM complaining of headache, fever and shortness of breath. Diagnosis should be ascertained by culture and microscopy, cryptococcal antigen testing or histopathology. Healthcare providers should also give thought to the effect of hyperglycemia in the management of cryptococcal infection and DM. Additional studies on hyperglycemic control with cryptococcosis are recommended.
Footnotes
Author contributions
Felix Bongomin conceptualized the study. Lauryn Nsenga, Jonathan Kajjimu, Ronald Olum, Sandra Ninsiima, Andrew Peter Kyazze, Phillip Ssekamatte, Davis Kibirige, Joseph Baruch Baluku, Irene Andia-Biraro and Felix Bongomin contributed to the drafting of the manuscript. All authors reviewed and approved the final manuscript prior to submission.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Our study did not require ethical board approval because of its design of a literature review of published data. Our study did not require consent because no individual patient data were included.
