Abstract
The coronavirus disease 2019 (COVID-19) pandemic has left the world in a state of desolation with overburdening public health systems in a short period. Finding possible preventative and therapeutic measures to counter severe respiratory syndrome coronavirus 2 (SARS-CoV-2), the causative agent of COVID-19, has been the priority. A possible solution is convalescent blood products (CBP), primarily convalescent plasma (CP) and immunoglobulins, as an adjunctive therapy. CBP has been tried on the previous coronavirus epidemics with severe acute respiratory syndrome coronavirus (SARS-CoV) and the Middle East Respiratory Syndrome Coronavirus (MERS-CoV). Therefore, we reviewed the clinical utility of CBP and available evidence in COVID-19. We found some of the current anecdotal studies demonstrate promising therapeutic potential, but many of these studies do not meet the academic rigours to substantiate its use with confidence. However, the compassionate use of CBP in critically ill COVID-19 patients can be an option while we await a definitive answer from ongoing randomised clinical trials.
Introduction
Since the coronavirus disease 2019 (COVID-19) was first reported in December 2019 in Wuhan, China, the global pandemic has resulted in over 12.4 million confirmed cases, including 559,047 deaths as of 12 July 2020. 1 While dexamethasone and remdesivir have shown some promising results recently with a reduction in mortality and recovery time, we are still awaiting a definitive therapy against COVID-19.2,3 An alternative therapeutic option for severe respiratory syndrome coronavirus 2 (SARS-CoV-2), 4 the pathogen responsible for COVID-19, could be the use of convalescent blood products (CBP), primarily convalescent plasma (CP) and immunoglobulins, as an adjunctive therapy based on previous experience with coronavirus epidemics with severe acute respiratory syndrome coronavirus (SARS-CoV) and the Middle East Respiratory Syndrome Coronavirus (MERS-CoV). Therefore, this study aims to discuss the clinical usage of CBP and review the existing evidence in COVID-19.
Method
A review of the literature was done through PubMed and Google Scholar databases to identify all relevant English language scientific studies based on the study objectives. Unspecified combinations of the search strings included, ‘Coronavirus’ OR ‘Severe Acute Respiratory Syndrome’ OR ‘SARS’ OR ‘SARS-CoV’ OR ‘Middle Eastern Respiratory Syndrome’ OR ‘MERS’ OR ‘MERS-CoV’ OR ‘Severe Acute Respiratory Syndrome Coronavirus 2’ OR ‘SARS-CoV-2’ OR ‘2019-nCoV’ OR ‘Coronavirus disease’ OR ‘COVID-19’ AND ‘convalescent blood products’ OR ‘CBP’ OR ‘convalescent plasma’ OR ‘CP’ AND ‘intravenous human immunoglobulin’ OR ‘IVIG’ in association with related physiology and efficacy. Included studies assessed the efficacy and safety of CBP in SARS, MERS, and COVID-19, including in vivo models. Owing to the scarcity of randomised control trials (RCT) and other relevant studies, including observational studies, case reports, case series and review articles were also involved. Studies carried out in the paediatric population were excluded. A separate assessment of the studies was executed for a secondary reference search on applicable supplementary research studies.
Convalescent blood products
CBP consists of 5 :
convalescent whole blood or convalescent plasma or convalescent serum;
pooled human immunoglobulin for intravenous or intramuscular administration;
high-titre human immunoglobulin; and
polyclonal or monoclonal antibodies.
CBP is obtained from an individual who has developed humoral immunity and recovered from an infection. It is suppose to have high-titres of neutralising antibodies (NAb) used either for prophylactic passive inoculation or supportive treatment with the intention of reducing symptoms and mortality.
CBP has been used throughout history to confer artificially acquired passive immunity against various pathogens. The first use can be traced back between 1918 and 1920 during the Spanish Flu pandemic.6–11 A meta-analysis was conducted by Luke et al. regarding the effectiveness of CBP against Spanish influenza that involved eight studies and 1703 patients. 12 The study revealed significantly reduced mortality in CBP groups compared with untreated patients [16% versus 37% fatality; absolute risk difference 8% to 26%; pooled risk difference 21%, 95% confidence interval (CI): 15–27%].
CBP continued to be used prophylactically against viral infections such as measles, varicella-zoster, and cytomegalovirus to variable effects due to the nature of its preparation.13–15 Pooled human plasma tends to have inconsistent titres of NAbs. However, in 1944, Stokes et al. argued that the efficacy of CBP could be improved through more potent preparations and intravenous administration. 16 But, as definitive preventative tactics, such as vaccines, emerged, CBP became marginalised. Presently, CBP is used as a replacement therapy for congenital or acquired immunoglobulin production deficiencies.
CBP against SARS
During the 2002 SARS-CoV epidemic, there were no specific therapies/vaccines available for treatment. CBP therapeutic potential was first observed by Wong et al., 17 when a 57-year-old SARS-CoV infected woman received 200 ml CBP + 500 mg methylprednisolone and made an unremarkable recovery with no adverse drug reactions (ADRs). Similar recoveries were reported in three case series, including one pregnant female, and another woman, who subsequently became pregnant, without complications.18–20 Separately, Soo et al. 21 found that patients (n = 19) receiving CBP + ribavirin-steroid had reduced admission lengths (p = 0.001) and mortality (p = 0.049) compared with patients (n = 21) receiving 1.5 g pulsed methylprednisolone. However, patients had a poor clinical response when CBP was given beyond 16 days post-symptom onset. Cheng et al. reported similar findings in 80 SARS-CoV infected patients with 279.3 ± 127.1 ml CBP (range:160–640 ml), with a greater 22-day discharge rate when CBP was given within 14 days of illness (58.3% versus 15.6%; p < 0.001). 22 A systematic review by Mair-Jenkins et al. analysed 32 studies regarding the efficacy of CBP against SARS and influenza and reported that mortality declined significantly in CBP patients [odds ratio (OR): 0.25; 95% CI: 0.14–0.45; I2 = 0%]. 23 They concluded that CBP is a safe therapeutic option with decreased mortality against SARS-CoV.
CBP against MERS
Similar to the 2002 SARS-CoV epidemic, no proven therapy was found during the 2012 MERS-CoV outbreak. By July 2014, 12 MAbs were identified to have prophylactic or therapeutic potential against MERS-CoV.24–26 Subsequent rapid and affordable production of NAb LCA60, exhibiting MERS-CoV S protein interaction, demonstrated MAb/NAb as a viable option to combat emerging viral infections with low ADR risk.27,28 Of the 12 NAbs identified, m336 underwent further in vivo studies. Houser et al. reported that m336 significantly reduced viral titres (40–9000 fold) in rabbit lung tissue, 29 and these results were replicated in mouse models by Agrawal et al. 30 However, the therapeutic predictive value remains limited compared with non-human primate models. 31 As such, Doremalen et al. began examining MERS-CoV infected common marmosets, given either high-titre hyperimmune plasma (1:3840) or m336. 32 The m336 conferred better clinical recovery, and hyperimmune plasma reduced respiratory tract viral load. Separately, Ko et al. examined three MERS-CoV infected patients, who received four CBP transfusions. 33 They showed significant serologic CBP response with plaque reduction neutralisation test (PRNT) titres of 1:80, and concluded that PRNT titre >1:80 could be effective against MERS-CoV. However, as per Choe et al., 34 it was challenging to obtain high CBP titre. They followed 11 MERS-CoV patients (five severe and six mild) for 12 months post-recovery, and found that severe patients tend to exhibit higher acute (0–6 months) and maintenance (6–12 months) PRNT90 titres, suggesting the possibility of obtaining effective high titre CBP only within the initial months of severe disease.
Overall, the quality of clinical evidence of CBP in SARS and MERS was low and lacked substantial support from RCTs for a definitive conclusion.
CBP against COVID-19
CBP has been used frequently against COVID-19 due to the existing therapeutic inertia. Here, we discuss the evidence concerning CBP in COVID-19 (summarised in Table 1).35-48 On 2 February 2020, a study by Zhou et al. examined the full-length genome sequences of SARS-CoV-2 isolated from five patients. 49 SARS-CoV-2 was 79.6% and 96.0% similar to SARS-CoV and a bat coronavirus, respectively. This study encouraged clinicians to repurpose the therapeutic options tried and tested in SARS and MERS outbreaks. Subsequently, Zhou et al. conducted a serum-neutralisation assay in Vero E6 cells using IgG-positive sera from five human donors. 49 At a dilution of 1:40–1:80, the sera were able to neutralise SARS-CoV-2 samples of 100 TCID50 (50% tissue-culture-infective dose). Also, they found horse anti-SARS-CoV-2 serum to effectively cross-neutralise the virus, indicating the therapeutic potential of CBP against COVID-19.
Summary of evidence on CBP usage in COVID-19.
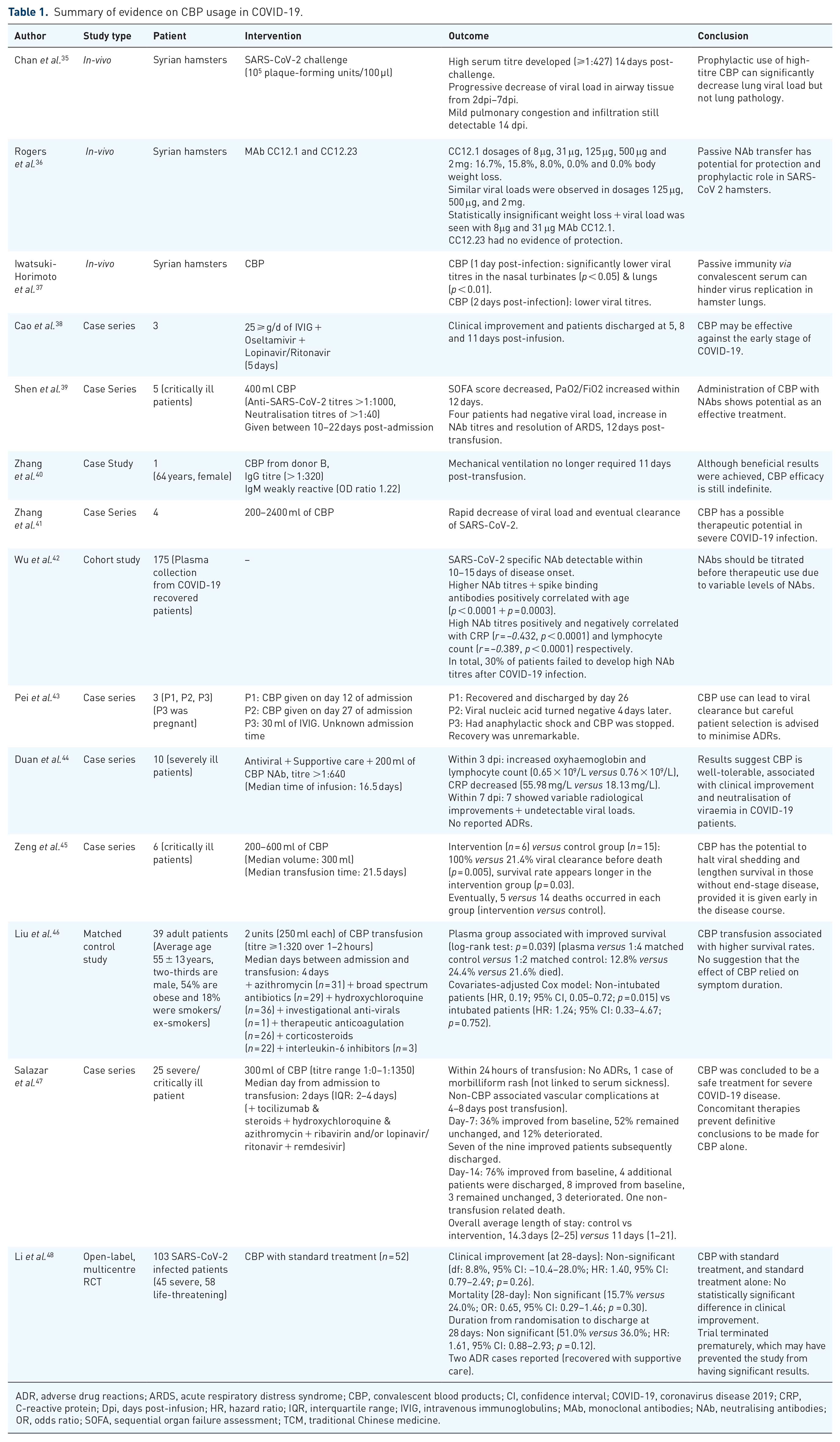
ADR, adverse drug reactions; ARDS, acute respiratory distress syndrome; CBP, convalescent blood products; CI, confidence interval; COVID-19, coronavirus disease 2019; CRP, C-reactive protein; Dpi, days post-infusion; HR, hazard ratio; IQR, interquartile range; IVIG, intravenous immunoglobulins; MAb, monoclonal antibodies; NAb, neutralising antibodies; OR, odds ratio; SOFA, sequential organ failure assessment; TCM, traditional Chinese medicine.
On 21 March 2020, a case series by Cao et al. investigated the use of intravenous immunoglobulins (IVIG) as a therapy against COVID-19 patients. 38 They described three case studies reporting administration of 25 g/d of IVIG for 5 days along with empiric treatment (oseltamivir and lopinavir-ritonavir). All three cases showed clinical improvement after the infusion and were subsequently discharged at 8, 5 and 11 days post infusion. The outcome suggested that CBP may be effective against early-stage COVID-19, inhibiting disease progression.
On 26 March 2020, an in vivo study by Chan et al. assessed the use of convalescent serum as an immunoprophylactic agent against COVID-19. 35 Groups of Syrian hamsters were either given SARS-CoV-2 suspended in phosphate-buffered saline (PBS) (105 plaque-forming units/100 µl) or just 100 µl PBS. Compared with the control group, the SARS-CoV-2 challenged group had a weight loss of up to 11%, 1–6 days post inoculation (dpi). By 14 dpi, a high serum NAb titre >1:427 was achieved, weight was regained, but mild pulmonary congestion and infiltration were still detectable. Viral loads progressively declined in airway tissues between 2 and 7 dpi. The authors concluded that the prophylactic use of this high-titre CBP resulted in a significant decrease in viral lung load but no pathological changes in the lung.
By 27 March 2020, a case series by Shen et al. assessed the benefit of the CBP transfusion in five critically ill SARS-CoV-2 infected patients. 39 Each patient was infused with 400 ml CBP with a SARS-CoV-2 specific antibody binding titre >1:1000 and a neutralisation titre >1:40, between 10 and 22 days after admission. Following CBP therapy, the sequential organ failure assessment (SOFA) score decreased, and the ratio of arterial oxygen partial pressure to fractional inspired oxygen (PaO2/FiO2) increased within 12 days. Furthermore, 12 days post transfusion, the viral load became negative, NAb titres increased, and there was a resolution of acute respiratory distress syndrome (ARDS) in four patients. They concluded that administration of CBP with NAbs showed some potential as an effective treatment; however, their study was limited by small sample size and subordinate design. Subsequently, Zhang et al. 40 examined anti-SARS-CoV-2 antibody levels in six CBP donors and all donors had high IgG titres (>1:320) except for one (< 1:40). Subsequently, 200 ml of CBP from one of the donors was given to a SARS-CoV-2 infected female. Following CBP transfusion, the CBP recipient exhibited clinical improvement and no longer required mechanical ventilation 11 days post transfusion. It was concluded that, although the outcome was favourable, further research is needed with an emphasis on long-term complications and safety profile.
On 31 March 2020, a case series by Zhang et al. described clinical improvement in four COVID-19 patients following CBP administration. 41 Initially, all four patients were unresponsive to empiric treatment; however, after CBP transfusion (range: 200–2400 ml), the viral load decreased rapidly and all the patients were subsequently cleared of SARS-CoV-2 infection.
On 6 April 2020, Wu et al. reported the characteristics of SARS-CoV-2 specific NAb from plasma collected from 175 recovered COVID-19 patients in a preprint cohort study. 42 It was revealed that the detection of such NAbs is possible within 10–15 days after disease onset. Furthermore, higher plasma NAb titres and spike-binding antibodies were positively correlated with age (p < 0.0001 and p = 0.0003, respectively). NAb titres were also correlated to C-reactive protein (CRP) [correlation coefficient (r) = –0.432, p < 0.0001] and lymphocyte count (r = –0.389, p < 0.0001), respectively. However, 30% of patients failed to develop high NAbs titres after COVID-19 infection. It was concluded that CBP from donors should be titrated before use as a passive antibody therapy due to the highly variable levels of NAbs.
On 11 April 2020, a case series by Pei et al. described the effects of CBP in three COVID-19 patients. 43 The first patient received CBP on day 12 of admission and was discharged by day 26. The second patient received CBP by day 27 and the viral nucleic acid turned negative after 4 days. The third patient suffered severe anaphylactic shock after receiving 30 ml of convalescent plasma from a 51-year-old pregnant woman. CBP treatment was immediately stopped, and the patient ultimately recovered and was discharged. This case highlights the importance of patient selection. Overall, the study showed that CBP use could lead to viral clearance as demonstrated by a negative viral nucleic acid test with a significant reduction in symptoms; however, careful patient selection should be conducted to prevent any adverse reactions.
On 28 April 2020, a case series by Duan et al. investigated the efficacy of CBP therapy in 10 severely ill COVID-19 patients. 44 In addition to antivirals and supportive care, patients were given 200 ml of CBP, NAb titre >1:640, with a median time of infusion at 16.5 days of symptom onset. The primary and secondary endpoints were the safety profile of CBP use and symptomatic improvement, respectively. Within 3 days post transfusion, oxyhaemoglobin increased, lymphocyte count increased (0.65 × 109/l versus 0.76 × 109/l) and CRP decreased (55.98 mg/l versus 18.13 mg/l). Within 7 days post transfusion, a variable radiological improvement was observed, and viral load was undetectable in seven patients. No ADRs were observed. The authors concluded that CBP was well tolerated, and its use was associated with clinical improvement and neutralisation of viremia in COVID-19 patients.
On 29 April 2020, a case series by Zeng et al. assessed CBP efficacy in six critically ill COVID-19 patients. 45 The intervention group received 200–600 ml CBP (median volume: 300 ml) at a median infusion time of 21.5 days, following the first detection of viral shedding. The control and intervention group achieved 21.4% and 100% viral clearance before death (p = 0.005), with the intervention group all testing negative for SARS-CoV-2 RNA within 3 days of transfusion. Furthermore, the survival rate appeared to be longer in the intervention compared with the control group (p = 0.03); however, there were 5 deaths in the intervention and 14 in the control groups. Death in the intervention group was attributed to late CBP transfusion, and no immediate ADRs were reported. They concluded that CBP therapy has the potential to halt viral shedding and lengthen survival in patients without the end-stage disease if administered early in the course of the disease.
Rajendran et al. conducted a systematic review on 1 May 2020, with five eligible studies. 50 Based on the existing clinical evidence, they concluded that: (i) elevation in NAb titres with the disappearance of viral RNA was noted in nearly all patients following CBP administration, (ii) CBP had a beneficial effect on the clinical picture, and (iii) CBP might decrease mortality in critical COVID-19 patients. CBP was found to be safe and clinically effective, with a reduction in mortality. However, they recommended more extensive multicentre clinical trials to have conclusive evidence for efficacy.
On 22 May 2020, a preprint by Liu et al. described a matched control study comparing the outcomes of severe to life-threatening COVID-19 cases receiving CBP (n = 39). 46 All 39 patients were given two CBP units (250 ml per unit), with a SARS-CoV-2 anti-spike antibody titre of >1:320, over 1–2 h at a median of 4 days from admission to transfusion. Patients were also receiving concomitant therapy that included azithromycin (n = 31), broad-spectrum antibiotics (n = 29), hydroxychloroquine (n = 36), non-specified investigational anti-virals (n = 1), therapeutic anticoagulation (n = 26), corticosteroids (n = 22), and interleukin-6 inhibitors (n = 3). By day 14, clinical conditions deteriorated in both groups (18.0% intervention versus 24.3% control, p = 0.167). The effects of CBP seemed to be confounded by therapeutic anticoagulants (unadjusted versus adjusted OR: 0.90 versus 0.84). Furthermore, the plasma group was associated with improved survival (log-rank test: p = 0.039) (plasma versus 1:4 matched control versus 1:2 matched control: 12.8% versus 24.4% versus 21.6% mortality). In a covariates-adjusted Cox model, CBP transfusion significantly improved survival in non-intubated patients [hazard ratio (HR): 0.19; 95% CI: 0.05–0.72; p = 0.015] but not in intubated patients (HR: 1.24; 95% CI: 0.33–4.67; p = 0.752). There was no evidence to suggest that the effect of CBP relied on symptom duration. Finally, it was concluded that CBP transfusion might be associated with higher survival rates; however, they also recommended a larger RCT to establish these findings.
On 26 May 2020, a case series by Salazar et al. enrolled 25 severe and/or life-threatening SARS-CoV-2 cases to evaluate clinical improvement following CBP therapy. 47 The primary outcome was clinical disease severity measured through modification of the World Health Organisation (WHO) six-point scale. The patients received 300 ml of CBP, with a titre ranging from 1:0 to 1:1350 for the receptor binding and ectodomain of the SARS-CoV-2 spike protein. One patient was noted to receive a second dose 6 days after the initial dose. Concomitantly, patients were also receiving other treatments; tocilizumab, steroids, hydroxychloroquine, azithromycin, ribavirin and/or lopinavir/ritonavir, and remdesivir. On day 7, 36% (n = 9) improved from baseline, 52% (n = 13) remained unchanged, and 12% (n = 3) deteriorated. Seven amongst nine were discharged. On day 14, 76% (n = 19) improved from baseline, and four additional patients were discharged, eight improved from baseline, three remained unchanged, three deteriorated, and one patient died due to non-transfusion related complications. Overall, the average length of hospital stay and post-transfusion length-of-stay were 14.3 days (range 2–25 days) and 11 days (range 1–21 days), respectively. The authors showed that CBP is a safe treatment option for severe COVID-19 disease, but concomitant therapy prevented definitive conclusions on CBP as a monotherapy.
An open-label multi-centre RCT by Li et al., published on 3 June 2020, examined 103 SARS-CoV-2 infected patients to assess CBP efficacy. 48 The primary outcome was time to clinical improvement before day 28. Patients were stratified based on disease severity (45 severe and 58 life-threatening conditions) and separated into two groups: CBP with standard treatment (n = 52) and standard treatment alone as a control (n = 51). Within 28 days, clinical improvement was observed in 51.9% (n = 27; 21 severe and 6 life-threatening groups) and 43.1% (n = 22; 15 severe and 7 life-threatening groups) in both CBP and control group (diff: 8.8%, 95% CI: −10.4%–28.0%; HR: 1.40, 95% CI: 0.79–2.49; p = 0.26). Comparing severe/life-threatening cases against their respective control groups, the hazard ratio was 2.15 (95% CI: 1.07–4.32; p = 0.03) and 0.88 (95% CI: 0.30–2.63; p = 0.83). Two patients developed ADRs; however, they improved with supportive care. CBP with standard treatment, when compared with the control, was not associated with a statistically significant clinical improvement within 28 days. However, the trial was discontinued early due to the drop in overall COVID-19 cases in China.
A systematic review by Pimenoff et al. on 8 June 2020, published as a preprint in medRxiv examined 10 studies evaluating CBP therapy in 61 severely infected COVID-19 patients. 51 Efficacy was measured through the time of recovery and the presence of viral RNA post-transfusion. Upon admission, 72.1% (n = 44) had ARDS and fever, while the remaining 27.9% (n = 17) had mild disease. In addition to CBP therapy, most patients received arbidol, lopinavir/ritonavir, and interferon-alpha-1b and corticosteroids (n = 46; 75%). A total of 52 patients had between 200 ml and 500 ml of CBP each; however, 9 patients received an additional 300 ml (n = 1), 400 ml (n = 4), 600 ml (n = 2), 900 ml (n = 1) and 2400 ml (n = 1) within a week, following non-response to the initial CBP dose and the presence of clinical deterioration. The CBP titres were not specified. In total, 50.8% (n = 31) recovered within the first week and 39.3% (n = 24) recovered between 8 and 29 days following CBP transfusion. There were no available recovery data in 9.8% (n = 6). Regarding prevalence of viral RNA post transfusion, 66.7% (n = 24) and 30.6% (n = 11) were negative within the first week and 8–29 days, respectively. There was no significant difference in recovery rate after CBP therapy between gender, or the presence of underlying comorbidities, receiving CBP within 3 weeks or beyond following symptom onset. It was concluded that CBP therapy was safe and had a positive recovery response in about half of patients, particularly those younger than 60 years. However, a definitive conclusion regarding the effectiveness of CBP was difficult due to the relatively small number of patients and multiple concomitant treatments.
On 15 June 2020, an in vivo study by Rogers et al. attempted to discover NAbs through the antibody response against SARS-CoV-2. 36 This was undertaken via two approaches: (i) the development of viral neutralisation assays using a replication-competent virus [SARS-CoV-2 Washington strain (USA-WA1/2020)] and a pseudovirus (PSV) (murine leukemia virus (MLV)-based PSV) in target cells (HeLa-ACE2), and (ii) the establishment of a SARS-CoV-2 cohort of 17 donors based on donor plasma receptor binding domain (RBD) in PSV assays. RBD-binding titres reached EC50 at 103 serum dilutions, with positive correlations observed between RBD binding and PSV neutralisation. A total of 33 antibodies were isolated from three donors (CC6, CC12 and CC25), where, ultimately, lineages MAb CC12.1 and CC12.23 were placed into Syrian hamster animal models. Both anti-SARS-CoV-2 NAbs were given at five different concentrations, starting at 8 μg/animal to 2 mg/animal for the two intervention groups. In contrast, the control group received a single dose of 2 mg anti-dengue virus antibody. Each group was then challenged intranasally with 1 × 106 PFU of USA-WA1/2020 12 h post infusion; 5 days following the SARS-CoV-2 challenge, the hamsters were weighed as a measure of disease, and lung tissue was collected to measure the viral load. The CC12.1 group had an average of 16.7%, 15.8%, 8.0%, 0.0% and 0.0% body weight loss for dosages of 8μg, 31 μg, 125 μg, 500 μg and 2 mg, respectively, against control (13.6%). Similar viral loads were observed for CC12.1 dosages of 2 mg, 500 μg and 125 μg, while the control and CC12.1 doses 8 μg and 31 μg exhibited higher viral loads. MAb 12.23 demonstrated no evidence of protection. Sera collected before the SARS-CoV-2 challenge suggest that approximately 22 μg/ml of NAb (1160 × PSV neutralisation IC50) confers full protection, while 12 μg/ml (630 × PSV neutralisation IC50) is sufficient for 50% reduced disease as measured by weight loss. They concluded that MAb CC12.1 directly competes with angiotensin converting enzyme 2 (ACE-2) with a potential for prophylactic, therapeutic, and vaccine applications. As evident, SARS-CoV-2 shows strong ACE-2 binding affinity due to modification of the functional receptor binding sequence as compared with other coronavirus families, and this improved binding pattern could be utilised to block viral cell entry. 52
On 22 June 2020, another in vivo study by Iwatsuki-Horimoto et al. examined the replicative ability and pathogenesis of SARS-CoV-2 within Syrian hamsters. 37 Post-infection sera were collected from hamsters infected with either high (105.6 PFU) or low (103 PFU) dosages of the SARS-CoV-2 UT-NCGM02 strain. Infected sera were given either 1 or 2 days post-infection while two control groups received uninfected hamster sera. The hamsters receiving CBP 1 day post-infection were found to have significantly lower viral titres in the nasal turbinates (p < 0.05) and lungs (p < 0.01) compared with control groups. Similarly, statistically insignificant lower viral titres were reported in hamsters receiving CBP 2 days post infection compared with control (5.9 ± 1.8 log10 PFU ± SD/g versus 7.8 ± 0.1 PFU ± SD/g). Hamsters with SARS-CoV-2 infection had evoked NAbs resulting in the protection of hamsters from further infection, and viral replication was prevented more effectively in lungs than in nasal turbinates. The study concluded that anti-SARS-CoV-2 polyclonal hyperimmune globulin from CBP derived from COVID-19 patients and MAbs against SARS-CoV-2 could reduce viral loads in patients.
Can we trust existing studies?
As stated by Kalil et al., we must be cautious in a fallacy on unproven drug efficacy. 53 It is not necessarily true that any death must be due to disease, and, likewise, any survival must be due to therapeutic intervention. Past studies relating to SARS and MERS lend no substantial credibility to the efficacy of CBP. They were inconsistent, with vague methodology, and had several limitations like small sample sizes, lacked of large-scale trials, blinding, randomisation and control groups, and tested many concomitant treatments. 54 Although we did not perform quality assessment, we found the majority of existing CBP clinical studies in COVID-19 to be mainly case-based, lacking randomisation and standardisation, with sub-standard quality of methodology, and thus having low applicability and generalisability. Selection bias may also significantly skew the results, contributing to an already difficult interpretation.48,55 Therefore, additional RCTs must be conducted to provide conclusive evidence.
Perks of CBP transfusion
CBP has the capacity for adverse reactions, similar to regular plasma transfusions. 56 Mild allergy is common, but serious adverse events like transfusion-associated circulatory overload (TACO) are possible, with an elevated risk in the elderly with acute lung damage under mechanical ventilation and those with viral myocarditis. 57 Furthermore, the risk of complement and coagulation pathways activation with CBP is also a concern, due mainly to a potential for complement-dependent antibody enhancement due to COVID-19. 58 Also, the potential of blood-borne infection cannot be ignored. The Pathogen Reduction Technology (PRT) system, using ultraviolet light and riboflavin, have shown an effective reduction of the SARS-CoV-2 in plasma and blood products without decreasing the quality of the blood products, which could be utilised to reduce the risk of transfusion-related viral transmission. 59 However, CBP is thoroughly tested before its use and specially screened for hepatitis B/C, human immunodeficiency virus (HIV), and other infections. Notably, SARS-CoV-2 is not a blood-borne pathogen; therefore, CBP is safe and theoretically the risk of SARS-CoV-2 transmission from donor CBP is not possible. 60
Recommendations
We found some potential limitations of the clinical utility of CBP as a viable therapy in COVID-19, notably, (i) lack of robust clinical evidence, (ii) sufficient production of CBP, and (iii) production of CBP with consistent standard and safety. However, given the antiviral history of CBP and the absence of a definitive therapy against COVID-19, certain authorities such as the United States (US) Food and Drug Administration (FDA) have urgently authorised the therapeutic use of CBP, albeit within the framework of the strict guidelines suggested by scientific entities.58,61,62 This is to standardise treatment, minimise any unforeseen ADRs and allow for subsequent academic investigations.
As of 11 June 2020, 63 interventional trials investigating the efficacy of CBP against COVID-19 were listed on the ‘ClinicalTrials.gov’ (https://clinicaltrials.gov/) (Table 2). Therefore, it would be interesting to wait for their outcomes to have a definitive conclusion.
Summary of ongoing clinical trials of CBP in COVID-19.

Source: ‘Clinicaltrials.gov’ (https://Clinicaltrials.gov/).
CBP, convalescent blood products; COVID-19, coronavirus disease 2019; DB, double-blinded; IV, intravenous; MC, multi-centred; NR; non-randomised; OD, once daily; OL, open label; Ph I-III, phase I-III; QB, quadruple-blinded; RCT, randomised control trial; SARS, severe acute respiratory syndrome; SB, single-blinded; SC, single-centred; TB, triple-blinded; TDS, ter die sumendum; PCD, primary completion data; SCD, study completion date.
Finally, we can say that CBP, either as a monotherapy or in combination with other antiviral drugs, could be an option. Although the optimal antibody titre is unknown, the FDA recommends titer of 1:160, which could be as low as 1:80 if higher titres are unavailable. 63 Similarly, while there are uncertainties regarding the timing of CBP delivery during the course of the disease, studies have shown greater clinical recovery and discharge rate when given within 14 days of illness and poor clinical response after 16 days post-symptoms.19,20 Therefore, the CBP infusion should be of high titre, serologically screened for blood-borne pathogens, administered within 14 days of symptom onset, and the patient should be monitored during and after infusion to observe for any potential ADRs.
Conclusion
The clinical evidence for CBP against COVID-19 is insufficient. Some current anecdotal studies demonstrate promising therapeutic potential, but these studies do not meet the academic rigours to substantiate its use with confidence. However, the compassionate use of CBP in critically ill COVID-19 patients can be an option while we are awaiting a definitive answer from ongoing RCTs.
Study highlights
Currently, clinical evidence on CBP is limited to observational studies and further RCTs or large prospective studies are necessary to provide conclusive evidence.
Given encouraging early in vivo evidence and predicaments in therapeutic options, the compassionate use of CBP against COVID-19 can be an option in the ongoing crisis.
Clinicians and researchers should regularly update and adhere to the available credible evidence and findings of ongoing clinical trials.
Footnotes
Author contributions
KCVL, AS and YA made substantial contributions to the conception and study design, data acquisition and interpretation. All authors were involved in drafting the manuscript as well as the revision process. All authors have approved the final version of the manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
