Abstract
Objective:
To review the mechanism of action, mechanisms of resistance,
Data sources:
A PubMed search was performed from January 2006 to August 2019 using the following search terms: plazomicin and ACHN-490. Another search was conducted on clinicaltrials.gov for published clinical data. References from selected studies were also used to find additional literature.
Study selection and data extraction:
All English-language studies presenting original research (
Data synthesis:
Plazomicin has
Relevance to patient care and clinical practice:
Plazomicin will likely be used in the treatment of multi-drug-resistant cUTIs or in combination to treat serious carbapenem-resistant
Conclusions:
Plazomicin appears poised to help fill the need for new agents to treat infections caused by multi-drug-resistant
Keywords
Introduction
Antimicrobial resistance has positioned itself as a serious threat to patient care with global reach. A recent projection from the World Health Organization stated that mortality due to antimicrobial-resistant infections could reach 10 million by 2050, up from ~700,000 currently.
1
Some groups have suggested that this projection is a bit inflated,2,3 but regardless, our current situation remains dire. In an effort to raise awareness of this growing crisis, the Centers for Disease Control and Prevention released the Antibiotic Resistance Threats in the United States, 2013.
4
Many of the more serious threats listed were multi-drug-resistant (MDR) Gram-negative organisms, including carbapenem-resistant
The signing of the 21st Century Cures Act and the GAIN (Generating Antibiotic Incentives Now) ACT, which created the qualified infectious disease product (QIDP) indication, has helped to rejuvenate innovation to address antibiotic resistance. Examples of successful QIDP antimicrobials are ceftazidime/avibactam (Avycaz®), meropenem/vaborbactam (Vabomere®), imipenem/cilastatin/relebactam (Recarbrio®), eravacycline (Xerava®). All of these have documented activity against organisms possessing many different resistance phenotypes, including CRE. Another success of the QIDP indication, plazomicin (Zemdri®), looks to add yet another viable option. This article will review the pharmacokinetics/pharmacodynamics, available pre-clinical data, and clinical trials for plazomicin, and discuss its role in therapy.
Search methods
A PubMed search was completed from January 2006 to August 2019 using the search “ACHN-490” or “plazomicin”. All English-speaking studies were collected and evaluated for inclusion in the review. The addition of other search terms, namely resistance phenotypes like “extended-spectrum beta-lactamase”, “carbapenem-resistant
Chemistry and mechanism of action
As the name suggests, aminoglycosides are amine-containing sugars linked together by glycosidic bonds. The most clinically relevant aminoglycosides (gentamicin, tobramycin, and amikacin) contain three sugars. Plazomicin is a semi-synthetic aminoglycoside, created in an eight-step synthesis from sisomicin. During this synthesis, a hydroxy-aminobutyric acid (HABA) group is added to the amine at C-1, similarly to amikacin. Uniquely, an additional hydroxyethyl group is added to the amine at C-6′. 6 These structural features allow plazomicin to evade almost all clinically relevant aminoglycoside-modifying enzymes (AMEs), as demonstrated in Figure 1.
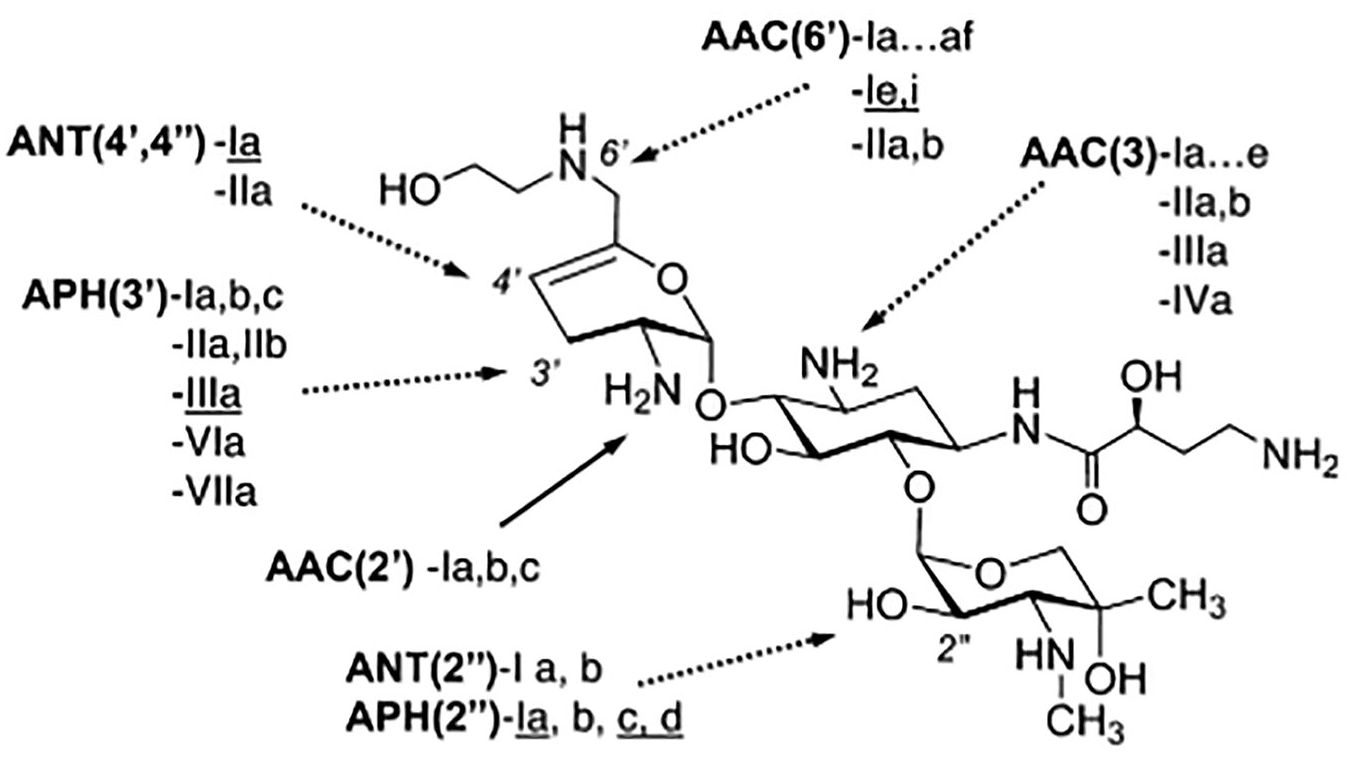
Plazomicin structure shown with clinically relevant aminoglycoside-modifying enzyme (AMEs) from both Gram-negative and positive (underlined) organisms. AMEs with a dotted line cannot modify plazomicin. Reproduced from Aggen et al with permission from the American Society of Microbiology. 6
Aminoglycosides bind to the aminoacyl-tRNA recognition site (A-site) of the 16S rRNA, which is a component of the 30S ribosomal subunit. This interrupts the elongation of the nascent protein sequence during the translation phase and therefore inhibits ribosomal protein synthesis.
7
Since they are cationic, hydrophilic molecules, aminoglycosides are thought to enter into Gram-negative bacterial cells
In vitro studies
Plazomicin has been assigned susceptibility breakpoints from the U.S. Food and Drug Administration (FDA) and U.S. Committee on Antimicrobial Susceptibility Testing for

Food and Drug Administration susceptibility breakpoint is used for plazomicin (⩽2 µg/mL). 10
Study reported % susceptibility using a susceptibility breakpoint of ⩽4 µg/mL.
MIC50/90 – minimum inhibitory concentration needed to inhibit 50% and 90% of the included isolates, respectively
Although plazomicin only has susceptibility breakpoints assigned for
Aminoglycoside resistance
Aminoglycoside resistance can be mediated by three types of mechanisms: enzymatic modification, target site modification, and porin channel/efflux pump expression changes. The most common mechanism in
As mentioned before, plazomicin is protected from nearly all clinically relevant AMEs due to structural differences. The lack of -OH groups at the C-3′ and 4′ positions prevents activity from APH(3′) and ANT(4′). The addition of a HABA at the C-1 position protects against AAC(3), ANT(2′′), and APH(2′′). Furthermore, the addition of the hydroxyethyl group to the amine at the C-6′ position protects against AAC(6′). The only AME currently identified amongst Gram-negative organisms with activity against plazomicin is AAC(2′)-I, which is chromosomally expressed in some
Dosing and administration
Plazomicin is administered as a 15 mg/kg intravenous (IV) infusion over 30 min once daily and is dosed using total body weight (TBW) for patients with TBW <125% ideal body weight (IBW). For patients with TBW >125%, adjusted body weight (ABW) should be utilized and is calculated using the following equation: ABW = IBW + 0.4 (TBW - IBW). Plazomicin is supplied as 10 mL, 50 mg/mL vials. For administration, the desired dose of plazomicin should be diluted to a final volume of 50 mL in either 0.9% sodium chloride, USP, or lactated Ringers, USP. Sterilely compounded products between 2.5 and 45 mg/mL are stable at room temperature for 24 h. 26
Pharmacokinetics/pharmacodynamics
Clinically relevant pharmacokinetic parameters from phase I clinical trials may be found in Table 2. Plazomicin displayed linear and dose-proportional pharmacokinetics following a single dose or multiple doses across a range of doses. These relationships can be seen when comparing the results of P1-01, which used half the normal dose of plazomicin (plazomicin 7.5 mg/kg), with other studies in Table 2. The maximum concentration (Cmax) and area under the curve 0–∞ (AUC0–∞) for P1-01 are approximately half of those seen in other studies. Overall, Cmax ranged from 161 ± 31 to 76.0 ± 19.6 mg/L and was reached immediately following the infusion in most studies. The wide range of measured values stems from the use of two different infusion times across phase I studies (30 and 10 min). Not surprisingly, the studies using a 10-min infusion measured higher Cmax and had a lower time to max (Tmax). AUC0–∞ ranged from 246 ± 39 mg*h/L to 309 ± 45 mg*h/L. The volume of distribution (Vd) of plazomicin ranged from 36.0 ± 7.8 to 11.3 ± 1.4 L across phase I studies and approximated total body water volume, similar to other aminoglycosides.27–32 Two studies calculated Vss using non-compartmental models, which resulted in higher reported values, as seen in Table 2.27,30 Protein binding appears to be relatively low in plazomicin at 16% ± 5. 31 Plazomicin was also found to penetrate the lungs to a similar degree as amikacin in non-inflamed lungs (ELF: plasma AUC 13% and 14% for plazomicin and amikacin, respectively). 32 Plazomicin is almost exclusively renally excreted. Following a single dose of plazomicin 15 mg/kg, 97.5% of the administered dose was recovered unchanged in the urine (56% within the first 4 h), while <0.2% was recovered from feces. 30
Pharmacokinetics in healthy volunteers from phase I clinical trials of plazomicin.
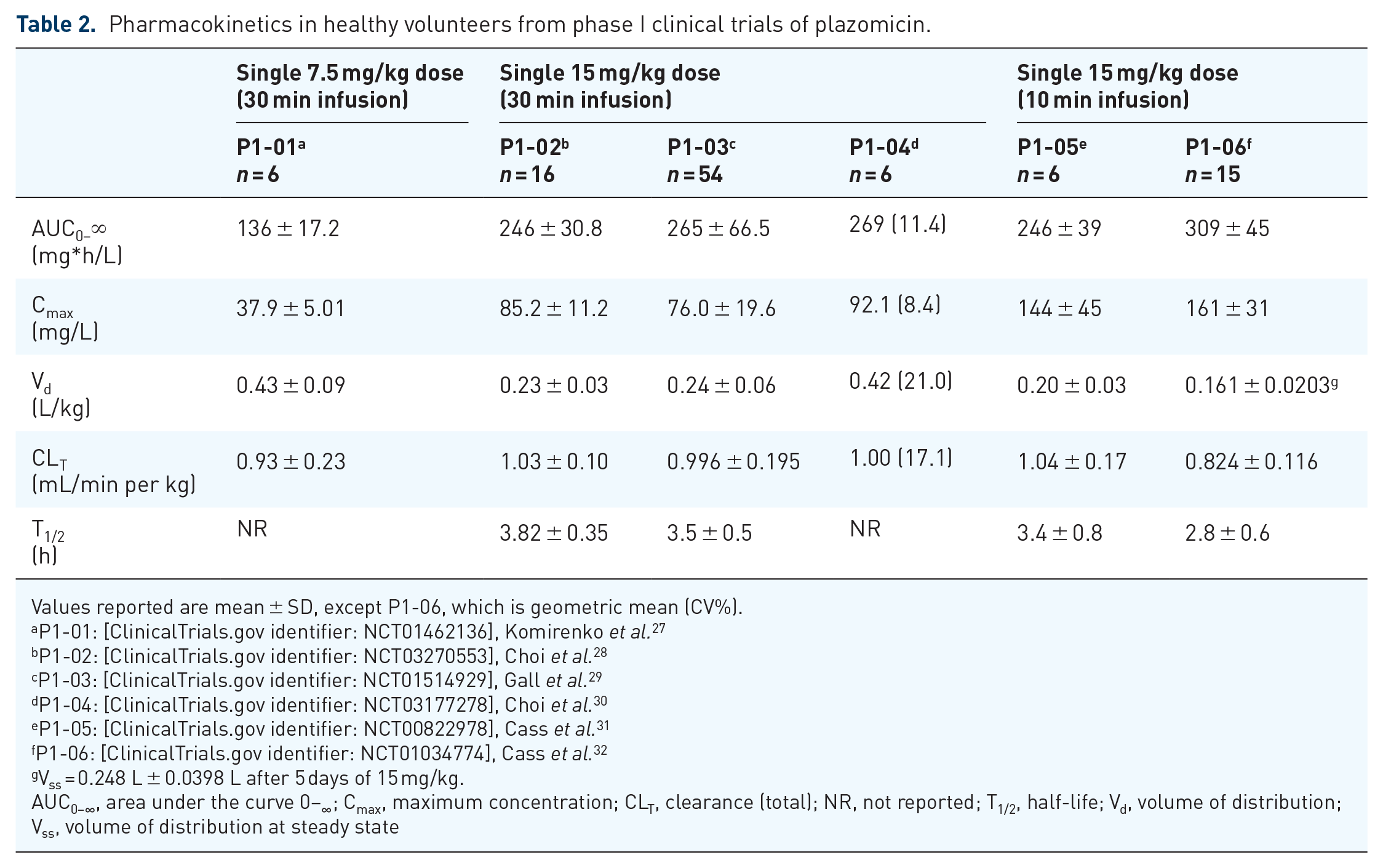
Values reported are mean ± SD, except P1-06, which is geometric mean (CV%).
P1-01: [ClinicalTrials.gov identifier: NCT01462136], Komirenko
P1-02: [ClinicalTrials.gov identifier: NCT03270553], Choi
P1-03: [ClinicalTrials.gov identifier: NCT01514929], Gall
P1-04: [ClinicalTrials.gov identifier: NCT03177278], Choi
P1-05: [ClinicalTrials.gov identifier: NCT00822978], Cass
P1-06: [ClinicalTrials.gov identifier: NCT01034774], Cass
Vss = 0.248 L ± 0.0398 L after 5 days of 15 mg/kg.
AUC0–∞, area under the curve 0–∞ ; Cmax, maximum concentration; CLT, clearance (total); NR, not reported; T1/2, half-life; Vd, volume of distribution; Vss, volume of distribution at steady state
In a study that recruited patients with various degrees of renal dysfunction at baseline, plazomicin AUC0–∞ was higher in patients with lower creatinine clearance (CLCr) as expected. AUC0–∞ in patients with normal (CLCr ⩾90 mL/min) and mild renal impairment (CLCr <90 and ⩾60 mL/min) were negligible, but were 1.98- and 4.42-fold higher on average in patients with moderate (CLCr <60 and ⩾30 mL/min) and severe renal impairment (CLCr <30 and ⩾15 mL/min), respectively. 27 In order to maintain similar exposures in patients with normal and impaired renal function, which may occur during complicated urinary tract infection (cUTI), the FDA package insert recommends alternate dosing regimens of 10 mg/kg once daily in patients with CLCr ⩽30 and <60 mL/min and 10 mg/kg every 48 h in patients with CLCr ⩽15 and <30 mL/min. Following the initial dose, the dosing interval may be adjusted 1.5-fold based on therapeutic drug monitoring (TDM) to maintain plasma trough concentrations <3 µg/mL. At the adjusted dosages, the AUC0–24h for patients with cUTI and mild or moderate renal impairment was 261 ± 102 mg*h/L and 224 ± 147 mg*h/L, respectively. 26 In an attempt to protocolize TDM for plazomicin, the group from Hartford Hospital adapted their aminoglycoside dosing nomogram for dosing interval selection to better assess patients in need of renal adjustment.33,34 Importantly, patients with CLCr <15 mL/min or who are on renal replacement therapy were excluded from phase I studies, so recommendations for dosing adjustments in these patients are currently unavailable.
There are currently three pharmacodynamic parameters that determine efficacy of antimicrobial agents: ƒ%T > MIC, ƒAUC:MIC, and ƒPeak:MIC. These parameters are often elucidated in dose fractionation studies conducted in murine models of infection. The groups of infected rodents are exposed to increasing doses of a drug using multiple dosing intervals. Non-linear regression analyses are performed between each pharmacodynamic parameter and the bacterial concentrations, colony-forming units (CFU)/mL, with the best fitting parameter being chosen.
35
Aminoglycosides have been shown to display optimal activity when the ratio of AUC:MIC is maximized.
36
In addition, studies have shown an independent benefit gained by maximizing the ratio of Cmax:MIC.37–39 For plazomicin, AUC:MIC was the parameter of best fit (
Clinical trials
Highlights from the phase II and III trials for plazomicin can be found in Table 3. Two indications have been pursued for plazomicin: cUTI in a phase II trial and the EPIC trial and serious CRE infection (included blood stream infections (BSIs), hospital-acquired pneumonia (HAP), and ventilator-associated pneumonia (VAP)) in the CARE trial.
Summary table of phase II and III clinical trials of plazomicin.

Dosages for trial drugs were: plazomicin 15 mg/kg intravenously (IV) once daily with therapeutic drug monitoring for maintenance dosing, levofloxacin 750 mg IV once daily, meropenem 1 g IV q 8 h, and colistin 5 mg/kg IV loading dose with 5 mg/kg per day IV divided into 8–12 h dosing intervals maintenance dosing.
P2-01: [ClinicalTrials.gov identifier: NCT01096849], Connolly
Included blood stream infection, hospital-acquired pneumonia, and ventilator-associated pneumonia.
Composite cure defined as both clinical cure and microbiological cure. Clinical cure was defined as reduced symptom severity at day 5/end of the infusion, complete symptom resolution at the TOC visit, or return to patient baseline prior to urinary tract infection. Microbiological eradication was defined as reduction in causative pathogen to <104 CFU/mL.
Given in combination with either meropenem 2 g IV q 8h (3 h extended-interval infusion) or tigecycline 100–200 mg IV loading dose with 50–100 mg IV q 12 h maintenance dosing.
CFU, colony-forming units; CI, confidence interval; CRE, carbapenem-resistant
cUTI
The FDA approved plazomicin for the treatment of cUTIs in July 2018 following the success of Study P2-01 and the EPIC trial. Study P2-01 was a phase II, multicenter, double-blind, randomized, comparator-controlled clinical trial. Patients were randomized 1:1:1 to receive either plazomicin 15 mg/kg IV once daily, plazomicin 10 mg/kg IV once daily, or levofloxacin 750 mg IV once daily. Preference was later given to the plazomicin 15 mg/kg IV once daily and subsequent randomization proceeded 2:1 to receive plazomicin 15 mg/kg IV once daily or levofloxacin 750 mg IV once daily. Patients enrolled in the study were between 18 and 85 years of age, ⩽100 kg, and had a CLCr ⩾60 mL/min based on Cockcroft and Gault. The primary efficacy endpoints in this trial were microbiological eradication (<104 CFU/mL of causative pathogen) in both the modified-intent-to-treat (MITT) and microbiologically evaluable (ME) populations at the test-of-cure (TOC) visit (5 and 12 days post-treatment).
The differences in percent microbiological eradication rate in the MITT and ME populations between groups was 2.2 (95% confidence interval (CI): –22.9 to 27.2) and 7.6 (95% CI: –16.0 to 31.3), respectively, both in favor of plazomicin, though neither result was considered statistically significant. The number of patients experiencing any adverse effect (AE) was similar between groups, with the most common AEs in either plazomicin group being headache (8.3%), dizziness (4.2%), nausea (4.2%), vomiting (4.2%), and diarrhea (4.2%), which is similar to phase I trial data. In addition, an increase in serum creatinine ⩾0.5 mg/dL occurred in 3.2% of patients receiving plazomicin, which did not occur in any patient receiving levofloxacin. 43
The EPIC trial was a phase III multicenter, multinational, double-blind, randomized clinical trial. Patients were randomized 1:1 to receive either plazomicin 15 mg/kg IV once daily or meropenem 1 g IV every 8 h for 7–10 days. The eligibility criteria in the EPIC trial were similar to the previous trial; although, patients with a CLCr ⩽30 mL/min were included in the EPIC trial. The primary efficacy endpoints were composite cure (both clinical cure and microbiological eradication) at day 5 of therapy and at the TOC visit (15–19 days following initiation of IV therapy) in the MITT population. Clinical cure was defined as reduced symptom severity at day 5 or end of the infusion, complete symptom resolution at the TOC visit, or return to patient baseline prior to urinary tract infection and microbiological eradication as reduction in causative pathogen to <104 CFU/mL.
At day 5 of therapy, the difference in percent composite cure between groups was not statistically significantly different; however, at both the TOC visit and the late follow-up visit (days 24–32), the difference was 11.6 (95% CI: 2.7–20.3) and 16.6 (95% CI: 7.0–25.7), respectively, both in favor of plazomicin. In addition, sub-group analysis of composite cure numerically favored plazomicin in every group tested. The frequency of AEs was similar between groups, with 19.5% and 21.6% of patients reporting any event in the plazomicin and meropenem groups, respectively. The most common AEs reported for plazomicin were similar to Study P2-01. Similar numbers of patients experienced a ⩾0.5 mg/dL increase in serum creatinine in the plazomicin (3.7%) and meropenem (3.0%) groups while receiving IV therapy, with full renal recovery occurring in 81.8% and 100% of patients receiving plazomicin and meropenem, respectively. 44
Plazomicin achieved non-inferiority for all primary efficacy endpoints in both trials when compared with standard of care therapy for cUTI. Moreover, it exhibited excellent activity against numerous resistance phenotypes between the two studies, including aminoglycoside and fluoroquinolone resistance, extended-spectrum beta-lactamase and CRE, and MDR isolates (resistant to at least one agent in three different antibiotic classes). Of the nine isolates in the plazomicin group that were CRE, 77.8% were eradicated at the TOC visit in the EPIC trial.
Serious CRE infection
The CARE trial was a phase III, multicenter, randomized, open-label trial. Patients were randomized 1:1 to receive either plazomicin 15 mg/kg IV once daily or colistin 5 mg/kg IV loading dose (300 mg maximum) colistin base activity followed by 5 mg/kg per day IV maintenance dose q 8–12 h for 7–14 days. Plazomicin was adjusted based on TDM in patients with renal impairment to target an AUC0–24h of 262 mg*h/L. Both agents were administered in combination with either meropenem 2 g IV (3-h extended infusion) every 8 h or tigecycline 100–200 mg IV loading dose followed by 50–100 mg IV maintenance dose q 12 h. Patients enrolled in the study were between 18 and 85 years of age, had an Acute Physiology and Chronic Health Evaluation II score between 15 and 30, and had either a BSI, HAP, or VAP suspected/confirmed to be caused by a CRE. The primary efficacy endpoint was a composite of all-cause mortality at 28 days or clinically significant disease-related complications in the microbiologic MITT (MMITT) population (confirmed CRE infection who received ⩾1 dose of trial drug).
Unfortunately, the trial was ended prematurely due to slow enrollment. The difference in percent occurrence of a primary endpoint event in the MMITT population was 26 (95% CI: −55 to 6) in favor of plazomicin. Sub-group analysis by infection type revealed this difference to be 39 (95% CI: −69 to −4) in favor of plazomicin in the BSI group and 27 (95% CI: −48 to 82) in favor of colistin in the HAP or VAP group. Kaplan–Meier estimates in the MMITT groups showed the hazard ratio for all-cause mortality to day 28 [0.25 (95% CI: 0.05–1.19)] and day 60 [0.47 (95% CI: 0.19–1.19)] both favored plazomicin combinations. Plazomicin combinations also had a more favorable AE profile, with fewer serious adverse events (50%) and ⩾0.5 mg/dL serum creatinine increases (16.7%) occurring in the safety population than in the colistin group; 81% and 50%, respectively. 45
Due to the small sample size of the study, the FDA did not grant the CRE indication to plazomicin. Regardless, given the current lack of data in treating CRE infections in randomized controlled trials, these data, taken with
Clinical resistance
Pathogens demonstrating resistance to plazomicin were rarely encountered across these clinical trials. Due to concerns for balancing the intervention groups, isolates having baseline MICs considered to be non-susceptible to either meropenem or plazomicin were not included in the primary analysis of the EPIC trial; however, six isolates cultured in the CARE trial (two from the plazomicin arm and four from the colistin arm) had baseline MICs resistant to plazomicin. All of these isolates had MICs >128 µg/mL and were confirmed to express 16S rRNA methyltransferases.45,46
Treatment emergent resistance to plazomicin was also noted in phase III clinical trials, though this too was a rare occurrence. In total, seven isolates cultured from six patients met the criteria for resistance emergence (an isolate having a ⩾4-fold increase in MIC and whose baseline MIC changed from ⩽4 µg/mL to > 4 µg/mL after treatment). All of these patients were enrolled in the EPIC trial. Of these six patients, four achieved clinical cure at the TOC visit regardless, and only one of the other two patients required additional antimicrobial therapy following the initial administration of study drug. 46
Whole genome sequencing revealed that five of the seven isolates shared the genetic profile of the baseline pathogen with the addition of a plasmid-encoded 16S rRNA methyltransferase. No definitive resistance mechanism was determined for the other two isolates, but genetic changes consistent with increased aminoglycoside MICs (
Safety
Plazomicin was evaluated in six phase I clinical trials, one phase II clinical trial in patients with cUTI, and in two phase III clinical trials (one in patients diagnosed with severe CRE infections and one in patients diagnosed with cUTI). It should be noted that the FDA approved plazomicin with a Black Box Warning for aminoglycoside class effects (nephrotoxicity, ototoxicity, neuromuscular blockade, and pregnancy risk) as it has for other aminoglycosides; however, plazomicin demonstrated a safe AE profile across all clinical trials. In the EPIC trial, which enrolled the most patients of any other trial (303 received plazomicin), the most common AEs reported were decreased renal function (3.7%), diarrhea (2.3%), hypertension (2.3%), headache (1.3%), nausea (1.3%), vomiting (1.3%), and hypotension (1.0%). Given that plazomicin is an aminoglycoside, decreases in renal function are expected as a class effect; however, a similar frequency of clinically significant renal function decreases (increase ⩾0.5 mg/dL serum creatinine from baseline) occurred in patients receiving meropenem (3.0%). Furthermore, most patients in the plazomicin group had full renal recovery by the final follow-up visit (81.0%). 44 Patients experiencing any severe AE were similar between groups (1.7%). Although ototoxicity events were rare in clinical trials, patients should be monitored for these events especially if they have structural abnormalities or a history of otologic disease as these patients were excluded from participation. While the safety data provided here are promising, conditions in clinical trials often differ from clinical practice, especially in the duration of therapy. This should be considered when using plazomicin in practice as these percentages may not extend to patients being treated due to the differing contexts.
Relevance to patient care and clinical practice
Plazomicin has received an FDA indication for the treatment of cUTIs, including acute pyelonephritis, following positive results in a phase II and III clinical trial against the current standard of care. The majority of cUTIs are not caused by MDR organisms and can be effectively treated with more targeted therapy. Given its exceptional
Newer beta-lactam/beta-lactamase inhibitors have demonstrated excellent activity against most major carbapenem-resistant phenotypes; yet, emergence of resistance to ceftazidime/avibactam (the first novel beta-lactam/beta-lactamase combination commercially available) has already been reported, occurring both prior to exposure to the antibiotic and during active treatment.
47
Aminoglycosides have been utilized as add-on therapy with beta-lactams for serious infections for decades due to their synergistic mechanisms of action. However, recent spread of resistance determinants against aminoglycosides has threatened this antimicrobial class. This is especially true in CRE isolates, which have been shown to harbor numerous AME phenotypes. This could be another avenue for plazomicin to enter routine clinical use. It bears repeating that plazomicin did not receive an FDA indication for treating severe CRE infections; however, several data, both
Caution should be exercised when using plazomicin to treat New Delhi metallo-beta-lactamase (NDM)-producing
This trend has not yet been associated with any other CRE phenotype, including other MBLs. Numerous commercially available rapid detection tests can identify the presence of carbapenemases, including NDM, which can guide empiric administration of plazomicin.53,54 A rapid
Conclusion
Because of the persistence of bacterial evolution, it seems unlikely that a single agent will emerge as a panacea against infection; rather, an armamentarium seems necessary to keep pace in the fight against antimicrobial resistance. Plazomicin appears poised to help fill the need for new agents to treat infections caused by MDR
Footnotes
Author contributions
Both JAC and DSB contributed significantly towards the conception of the project, the interpretation of the data, drafting/editing process, and ultimate approval of the published draft.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
