Abstract
Objectives:
Cardiac implantable electronic device (CIED) infection has been a major clinical problem in addition to being a major financial burden. In spite of antimicrobial prophylaxis, CIED infection rates have been increasing disproportionately. We therefore conducted this meta-analysis to assess the role of TYRX antibiotic envelope for the prevention of CIED infection.
Methods:
Using extensive online search, we conducted a meta-analysis of studies reporting CIED infections with versus without the use of TYRX antibiotic envelope. A random-effect model was used, and between studies heterogeneity was estimated with I2. All analyses were performed with RevMan (version 5.0.20).
Results:
Five cohort studies were included in this meta-analysis. The pooled odds ratio (OR) of included studies was 0.29 [95% confidence interval (CI): 0.09–0.94; p < 0.004]. There was evidence of heterogeneity with I2 of 58%. There was also evidence of publication bias on funnel plot analysis. On sensitivity analysis, no statistically significant difference was noted when stratified by study design or duration of follow-up.
Conclusion:
The results of our study demonstrate a significant beneficial effect of TYRX antibiotic envelope for the prevention of CIED infections.
Introduction
Cardiac implantable electronic device (CIED) infections are a serious clinical problem associated with an increased morbidity, mortality, and healthcare costs.1–4 CIED infection rates have been estimated to be around 2–4%,5,6 which is much greater than ⩽1% of surgical site infections (SSIs) in a clean wound, as acknowledged by Centers for Disease Control. 7 Additionally, Centers for Medicare and Medicaid Services (CMS) in August 2012 published the Inpatient Prospective Payment System and Fiscal Year 2013 Rates – Final Rule, which added SSI after CIED implantation as a hospital-acquired condition; thus, hospitals will no longer be paid by CMS for treating these infections. 8 Multiple risk factors, including diabetes, prior history of infection, revision or upgrade procedure, renal failure, or congestive heart failure (CHF) have been described for CIED infections. 9 However, the risk of infection in an individual patient is mostly determined by the combination of risk factors rather than an absolute number. Although most CIED site infections manifest within the first few months, a delayed infection 6 months or more after implantation can also be seen. In a retrospective review of CIED infections CHF, corticosteroid therapy, and presentation with CIED-related infective endocarditis were shown to be associated with higher short-term mortality. 10 In addition to patient’s age, CHF, metastatic malignancy, corticosteroid therapy, renal failure, and CIED-related infective endocarditis were shown to be associated with higher long-term mortality. 10
Since local contamination with bacteria often occurs at the time of implantation, perioperative preventive measures are of critical importance. 11 Administration of perioperative parenteral antibiotics has been shown to reduce the risk of CIED infections. 12 American Heart Association and Heart Rhythm Society recommend prophylaxis with an antibiotic that has in vitro activity against staphylococci at the time of CIED implantation as a Class IA indication. 3 However, in spite of antimicrobial prophylaxis, CIED infection rates have increased more than what can just be explained by expanded implantation rates. 13 Possible reasons for rising CIED infections include the fact that more younger patients are receiving CIED, who survive long enough to require secondary interventions, and thus a higher infection rate.6,14
In 2008, the Food and Drug Administration approved the AIGISRx, now called TYRX, antibacterial envelope (Monmouth Junction, NJ now a subsidiary of Medtronic, Inc., Minneapolis, MN, USA). It is a polypropylene mesh that releases minocycline and rifampin in the generator pocket. There have been a few retrospective and prospective studies with the non-absorbable TYRX antibacterial envelope with conflicting results.15–19 Potential limitations to the use of TYRX antibacterial envelope have mainly stemmed from hospitals eating the cost up front for the device, lack of physician reimbursement for the use of device, and fear over intense scar or thickened capsule formation encasing the leads leading to device and patient to damage upon re-entry of the pocket. Recently, a second-generation, bio-absorbable version of the antibacterial envelope called TYRX-A was introduced. In a recent study, use of TYRX-A among high-risk subjects was shown to be associated with a very low prevalence of CIED infections, comparable to that seen with the older TYRX envelope.
Identification of simple yet effective strategies that can help prevent CIED infections is therefore needed to not only decrease overall morbidity and mortality, but also the associated healthcare costs and societal burden. We therefore conduct this meta-analysis of prospective and retrospective studies to assess the role of non-absorbable TYRX antibacterial envelope in the prevention of CIED infections.
Materials and methods
We conducted a search in PubMed, CINAHL, and Cochrane databases for studies that reported CIED infections with/without the use of TYRX antibacterial envelope. We used the following keywords for our search: cardiac implantable electronic device infections and antibiotics, cardiac implantable electronic devices with antibiotic envelope, and implantable devices with antibiotic envelope. The search was performed for studies in English language and was limited to human subjects. Articles from the reference list relevant to the clinical question were also considered. Both full-text articles and published abstracts were included in the analysis. In case of multiple reports from the same study, we used the most complete and/or most recently reported data. We also conducted a manual search for abstracts presented at the scientific sessions of the American College of Cardiology, the American Heart Association, the European Society of Cardiology, and the Heart Rhythm Society over the past 5 years.
We included studies reporting the rate of CIED infections with/without the use of TYRX antibacterial envelope. Only studies comparing event rates between two or more groups with complete information available were included in this report. Data for each trial were abstracted by an investigator (S.A.) and were confirmed by a second investigator (O.K.).
The meta-analysis was performed by computing unadjusted odds ratio (OR) using random-effects model. OR for CIED infections was calculated along with the 95% confidence intervals (CIs). Between studies heterogeneity was analyzed by means of I2. I2 of more than 50% suggests heterogeneity. Publication bias was assessed graphically using a funnel plot. We also conducted sensitivity analysis stratified by study type (prospective versus all included studies) and duration of follow-up (⩽6 months versus all included studies). All analyses were performed with RevMan Analyses Version 5.0.20 (© Nordic Cochrane Centre, Ringshopitalet 2008).
Results
Overall, we found 234 reports on the primary search of which we excluded 7 studies due to duplication. From the remaining 227 reports, we excluded 222 studies after full-text review. Of the 5 included studies, all were cohort studies with 4 being retrospective and 1 prospective in nature. Basic characteristics of these studies are shown in Table 1.
Baseline characteristics of study included in meta-analysis.

ICD, implantable cardioverter-defibrillator.
The four retrospective studies included in the meta-analysis were those by Mittal et al. 15 Shariff et al., 16 Kolek et al., 17 and Hassoun et al., 19 while the only prospective study was that by Henrikson (Citadel/Centurion 18 ). Overall, there were 4779 people included in this meta-analysis of which 2214 (46%) were in the TYRX group versus 2565 (54%) in the non-TYRX group. Combining the control and interventions groups, Citadel/Centurion study 18 had the highest number of subjects − 1580 (33%), the longest duration of follow-up (12 months), and was financed by TYRX. On the other hand, study by Hassoun et al. 19 had the minimum number of subjects − 184 (3.9%) while studies by Mittal et al. 15 and Shariff et al. 16 had the shortest duration of follow-up (6 months). For our analysis, we only compared the individuals from control and intervention groups who had the most comparable baseline risk factor profile as determined by each study. In total, there were 14 cases of CIED infection identified in the TYRX/experimental group versus 60 cases of CIED infection in the non-TYRX/control group. The pooled OR was 0.29 [95% confidence interval (CI): 0.09–0.94; p < 0.004] (Figure 1).

Pooled odds ratio for cardiac implantable electronic device infection across cohort studies between patients with versus without TYRX antibiotic envelope.
Sensitivity analysis (stratification by study type and duration of follow-up) did not alter the main conclusion (Table 2). There was evidence of heterogeneity noted with I2 = 58% (Figure 1). The funnel plot analysis showed asymmetrical distribution of OR estimates suggesting potential publication bias (Figure 2).
Sensitivity analysis.

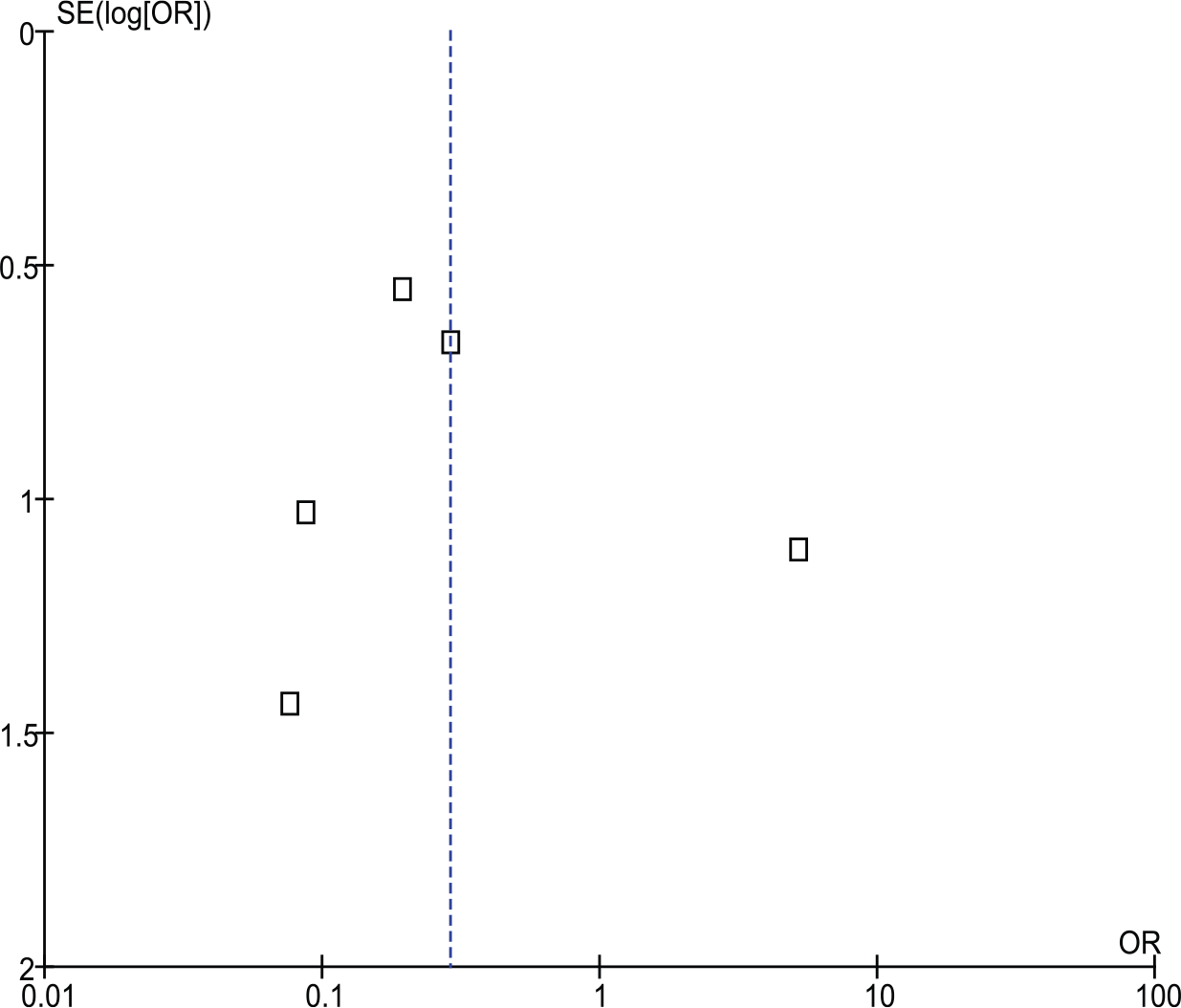
Funnel plot for pooled analysis for cohort studies. There is evidence of publication bias seen.
Discussion
Based on the findings of this meta-analysis, TYRX antibacterial envelope use is associated with a significant reduction in CIED-related infections. In addition, sensitivity analysis (stratification by study type or duration of follow-up) did not alter the results.
Our results are consistent with prior animal studies. Hansen et al. 20 in their study on rabbit model implanted pacing devices with or without the use of TYRX antibacterial envelope in implant pockets. These pockets were then inoculated with infection from various bacterial strains, including Staphylococcus epidermidis, Staphylococcus capitis, Escherichia coli, and Acinetobacter baummannii. After approximately 7 days, there was no evidence of infection in implant pockets among TYRX group versus the non-TYRX group which did become infected.
However, data on the beneficial effects of TYRX antibacterial envelope have been inconsistent in human studies. Bloom et al. 21 conducted a study on 624 human subjects undergoing CIED procedures utilizing TYRX antibacterial envelope at 10 US academic, community, and Veterans Affairs medical centers. Approximately, 50% of patients had at least three predefined risk factor for CIED infection. However, they demonstrated >99% success rate for CIED implantation with only 3 infections in 1.9 ± 2.4 months of mean follow-up. These infections were seen only among those undergoing replacement or revision procedures. This study did not have a comparison group and was therefore not included in our meta-analysis.
Similar beneficial effects of TYRX antibacterial envelop were noted in all the studies included in our meta-analysis other than that by Hassoun et al. 19 Among the included studies, Henrikson 18 and Kolek et al. 17 only included high-risk subjects as defined by subjects undergoing replacement procedure and those with ⩾2 risk factors for CIED infection, respectively. However, all subjects undergoing CIED implantation were included in the studies by Mittal et al. 15 and Shariff et al. 16
In the study by Hassoun et al., 19 there was a higher incidence of major infection among the TYRX versus the control group (5.4% versus 1.1%). Also, noted among the TYRX group were longer hospitalizations (6.8 ± 10.7 versus 3.1 ± 5.2 days), higher chronic corticosteroid use, higher rates of replacement or revision (51.1% versus 8.7%), and a greater proportion of devices with >2 intracardiac leads (42.4% versus 29.3%) as compared to the control group, thereby increasing the susceptibility to infection. This likely explains the differential results seen in this when compared to the other included studies where the experimental/control groups were more closely matched, with persistence of favorable effects in spite of a higher proportion of participants requiring ⩾2 leads implantation, early re-intervention, generator change out, or device upgrade in the TYRX group.
Mittal et al., 15 in their study, created a logistic regression model that identified independent risk factors for CIED infection [C index of 0.72 (95% confidence interval 0.61–0.83)]. The risk factors identified included early pocket re-exploration, male sex, diabetes, need for an upgrade procedure, history of CHF, hypertension, and a glomerular filtration rate (GFR) of <60 ml/min. Thus, a scoring system based on the baseline comorbidities to determine eligibility for TYRX antibacterial envelope merits careful consideration.
Prior studies looking into the risk factors for CIED infection have also shown device revision or upgrade, 22 use of >2 pacing leads or the need for early pocket re-exploration to be associated with a higher risk of CIED infection.6,23 Also, the presence of multiple leads has been shown to increase the risk of central venous thrombosis which can then serve as a potential site of secondary seeding of bacteria. 24 Additional procedure-related factors shown to be associated with an increased CIED infection risk include procedure time, temporary pacemaker use prior to implantation, and postoperative hematoma at the pocket site. 25 Importantly, ICD replacement has been associated with a 2.5× greater incidence of pocket-related events with the need for re-intervention increasing with every consecutive replacement. 14
Pathophysiologically studies suggest an important role played by biofilm formation in the pathogenesis of CIED infections. 26 The bacteria housed in the biofilm are much more resistant to antibiotics owing to the limited host immune cell response and antibiotic penetrance as a result of the adherent biofilm. 27 Therefore, strategies aimed at preventing biofilm formation in the first place can prove to be highly successful. TYRX antibacterial envelope, a novel modality, has been shown to effectively prevent the biofilm formation in vitro model. 28 The antibiotic coating is active within 2 h of the device implantation and continues to elute the medication over the next 7–10 days. 29
CIED infections have been associated with a significant health care cost resulting from prolonged hospital stays, longer duration of antibiotic therapy, management of sepsis and complications, device extraction and reimplantation. 2 These infections typically cost around at least $52,000, but the cost may even exceed $100,000.1,30 Shariff et al. 16 in their study also analyzed the financial impact of using TYRX antibacterial envelope. Based on the rate of infection and cost incurred in the non-TYRX group, they estimated 6.2 additional infections costing approximately $340,000 in the TYRX group, had it not been used. This was noted to be almost similar to the actual cost of the devices used in the TYRX group. This underscores the economic feasibility of TYRX antibacterial envelope.
Our meta-analysis has several strengths. A large sample size which improves the statistical power to detect smaller effect size. There was similarity in pooled effect estimate between prospective and retrospective cohort studies suggesting minimal unmeasured confounding. However, there are several limitations to this meta-analysis. All included studies were observational and most retrospective in nature. The intervention and control groups were from different time points. There was heterogeneity in the study design, number of participants, and duration of follow-up across the different studies included in this meta-analysis. In addition, lack of long-term follow-up limits the information on safety of TYRX antibacterial envelope in the long run. Finally, the type of TYRX antibacterial envelope utilized in these studies has now been replaced by a second-generation TYRX-A device which is fully absorbed within several weeks of implantation.
Conclusion
Overall, this meta-analysis shows a significant reduction in CIED infections with the use of TYRX antibacterial envelope. Additional long-term, randomized controlled trials with hard clinical endpoints and overall economical impact are needed for further insight. However, for now only few data exist on this topic while awaiting the results of the randomized, clinical controlled trial, World-wide Randomized Antibiotic Envelope Infection Prevention Trial (WRAP-IT). WRAP-IT is a large randomized clinical trial that will assess the efficacy of TYRX-A antibacterial envelope in reducing CIED infections and define its cost effectiveness.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
