Abstract
Invasive fungal infections are an important cause of morbidity and mortality in hematopoietic stem cell transplant and solid organ transplant recipients. Evolving transplant modalities and techniques, complex and extensive immunosuppressant strategies, and the increased use of broad spectrum antifungal prophylaxis has greatly impacted the epidemiology and temporal pattern of invasive fungal infections in the transplant population. The goal of this article is to provide an up-to-date review of the most commonly encountered invasive fungal infections seen in transplant recipients, including epidemiology, risk factors, clinical features, diagnostic dilemmas, management and their overall influence on outcomes.
Introduction
Recent advances and improvements in medical therapeutics, chemotherapy, and organ transplantation methodology have substantially reduced the overall morbidity and mortality associated with transplantation. However, along with these improvements, a variety of opportunistic infections frequently caused by relatively avirulent organisms have emerged. Critically ill, immunocompromised patients, especially those who have undergone transplants, are the prime targets for these opportunistic fungal infections, primarily due to
Candidiasis
Epidemiology

HIV, human immunodeficiency virus.
Clinical manifestation
Systemic candidiasis may be divided into two different categories: candidemia without organ involvement and disseminated candidiasis (organ infection by
Diagnosis
Unfortunately, findings from laboratory studies are either negative or nonspecific [Vazquez and Sobel, 2011; Pappas, 2006]. Clinicians are required to act definitively and early based on a high index of suspicion. Patients who remain febrile despite broad-spectrum antibiotic therapy, with either neutropenia or other risk factors and persistent leukocytosis, should be suspected of having systemic candidiasis. Cultures of nonsterile sites, although not useful for establishing a diagnosis, frequently demonstrate a high degree of candidal colonization. However, these positive cultures may be useful for initiating antifungal therapy in patients who are febrile and are unresponsive to broad-spectrum antimicrobials. It is important to always consider positive results from these sites significant and definitive evidence of infection. To be effective, appropriate antifungal therapy should be provided early and empirically in such high-risk patients [Morrell et al. 2005].
Characteristic features of biomarkers currently used for the diagnosis of invasive fungal infections.

Odabassi
BAL, bronchoalveolar lavage; FDA, US Food and Drugs Administration; GI, gastrointestinal; GVHD, graft
Management
The treatment of
Antifungal agents
Summary of systemic antifungal agents currently available.

GFR, glomerular filtration rate; IV, intravenous.
Polyenes include AmB-d and the LFAmB (AmB lipid complex, liposomal amphotericin B, and amphotericin B colloidal dispersion formulations) [Moen et al. 2009]. AmB was considered the gold standard of antifungal treatment for over 50 years. Unfortunately, its use is limited due to significant adverse events such as infusion-related reactions and nephrotoxicity. Lipid formulations of AmB were developed to overcome the limitations associated with the use of AmB-d. In general, LFAmB are better tolerated due to their different molecular structures. In fact, patients can be treated with larger doses of AmB without experiencing the typical side effects of AmB-d [Miceli and Chandrasekar, 2012]. The antifungal spectrum of activity, common drug–drug interactions, and side effects are shown in Table 4.
General patterns of susceptibility of

Vazquez and Sobel [2011]; S, susceptible; S-DD, susceptible dose-dependent; R, resistant; I, intermediately susceptible.
Until recently, the use of AmB and fluconazole was the standard therapy for all forms of candidiasis [Charlier et al. 2006]. The primary difference between the newer guidelines and the prior guidelines has to do with the upfront use of echinocandins in patients with candidemia and suspected candidiasis who have moderate to severe infections, patients with infections due to
In the non-neutropenic adult patient with candidemia or invasive candidiasis, most infections are due to the presence of an intravascular catheter in up to 70% of patients [Pappas et al. 2003, 2009]. Removal of all intravascular catheters appears to shorten the duration of candidemia and has been associated with reduced mortality [Pappas et al. 2009; Andes et al. 2012].
Candidemia requires treatment in all patient populations. In most situations, either fluconazole or an echinocandin are the drug of choice in the management of candidemia and disseminated candidiasis. The options listed should be considered depending on the history of a prior exposure to antifungals, the probability of fluconazole resistance, the presence of comorbid conditions, and the clinical status of the patient. Fluconazole (loading dose of 800 mg, then 400 mg daily) or an echinocandin (caspofungin: loading dose of 70 mg, then 50 mg daily; micafungin: 100 mg daily; anidulafungin: loading dose of 200 mg, then 100 mg daily) are recommended as initial therapy for most adult patients [Pappas et al. 2009]. However, an echinocandin is preferred in patients with moderate to severe illness, in patients who have a recent azole exposure, and in patients infected with a non-
Management of invasive candidiasis in patients with neutropenia may include an echinocandin, LFAmB 3–5 mg/kg/day or voriconazole (6 mg/kg administered intravenously twice daily for two doses, then 3 mg/kg twice daily) [Pappas et al. 2009]. Fluconazole 400 mg/day may also be an alternative.
Successful therapy for serious systemic
Antifungal prophylaxis of invasive candidiasis in patients who are in the high-risk group is currently recommended in several situations, which include patients with chemotherapy-induced neutropenia: fluconazole 400 mg daily, posaconazole 200 mg three times per day, or caspofungin 50 mg daily is recommended during induction chemotherapy for the duration of neutropenia [Viscoli et al. 1999; Husain et al. 2006; Ullmann and Cornely, 2006; van Burik et al. 2004]. In HSCT recipients, primarily those with allogeneic transplants, fluconazole 400 mg daily, or posaconazole 200 mg three times daily, or micafungin 50 mg daily is recommended during the period of neutropenia. In SOT recipients, fluconazole 200–400 mg daily or LFAmB 1–2 mg/kg daily for at least 7–14 days is recommended as postoperative prophylaxis for high-risk liver, pancreas, and small bowel transplant recipients.
Posaconazole has been shown to be effective prophylaxis against IFIs in high-risk patients with neutropenia and HSCT recipients, but its role as empirical therapy for candidiasis has not been established.
Empiric therapy
Empiric use of antifungal agents in patients who are febrile is widespread without much supporting data [Pappas, 2006, Leleu et al. 2002]. A major pitfall has been in establishing the definitive diagnosis of invasive candidiasis in the setting of negative blood cultures. It appears reasonable to initiate empiric antifungal therapy in selected patients with known risk factors. Echinocandins with their broad spectrum of activity and improved efficacy may be preferable, although less expensive fluconazole may also be an alternative. Some criteria for initiating empiric antifungal therapy include patients with known risk factors for candidiasis, patients who are febrile and on broad-spectrum antibiotics for over 96 h, and patients with multifocal
Invasive mold infections
Invasive mold infections (IMIs) have become an important cause of morbidity and mortality in HSCT and SOT recipients. Evolving transplant modalities and techniques, immunosuppressive strategies, and the use of antifungal prophylaxis has impacted the epidemiology and temporal pattern of IFIs in this population [Neofytos et al. 2009; Kontoyiannis et al. 2010; Pappas et al. 2010].
Epidemiology
Distribution of fungal pathogens causing invasive fungal infections in transplant recipients.

Neofytos
IFI, invasive fungal infection; HSCT, hematopoietic stem cell transplantation.
Non-
In the SOT population, the highest rate of IFIs was seen in small bowel transplants, followed by heart–lung, liver, pancreas, heart, and kidney transplants [Kontoyiannis et al. 2010]. Allogeneic HSCT recipients, especially unrelated or mismatched transplants, had a fivefold greater risk for IFIs compared with autologous HSCT recipients [Pappas et al. 2010]. Despite a slight increase in the incidence of all IFIs during 2002–2005, there was no significant increase in the incidence of mold infections in either the SOT or HSCT populations over the past decade. In contrast, there appears to be a comparative increase in the incidence of mucormycosis [Park et al. 2011; Kontoyiannis and Lewis, 2006; Petrikkos et al. 2012]. This increase may be a consequence of a greater number of at-risk patients undergoing HSCT or SOT, the use of more aggressive immunosuppressive treatments for GVHD and rejection, and possibly the increased use of voriconazole for antifungal prophylaxis or for empiric therapy [Kontoyiannis et al. 2006; Petrikkos et al. 2012; Trifilio et al. 2007; Spellberg et al. 2012; Xhaard et al. 2012, Lanternier et al. 2012].
The comparative distribution of IMIs varies among the type of organ transplanted (Table 6). Overall, invasive aspergillosis (IA) and other mold infections predominated among HSCT recipients. Among SOT recipients IA was most common in lung transplant recipients, accounting for 44% of all IFIs compared with 23%, 14%, 11%, and 5% in heart, kidney, liver, and pancreas transplant recipients respectively [Pappas et al. 2010].
Incidence, timing and outcomes of invasive fungal infection after transplantation.

Neofytos
IFI, invasive fungal infection; HSCT, hematopoietic stem cell transplantation; SOT, solid organ transplantation.
Risk factors
Risk factors for invasive mold infections in transplant recipients.

Park et al. [2011]; Petrikkos et al. [2012]; Trifilio et al. [2007], Spellberg et al. [2012]; Safdar et al. [2010]; Fortún et al. [2012]; Silveira and Husain [2007]; Singh et al. [2003]; Husain [2009]; Ibrahim et al. [2011].
CMV, cytomegalovirus; GVHD, graft
In SOT recipients, the risk factors for IMIs are strongly associated with end-organ failure, especially renal or hepatic insufficiency [Nucci, 2003; Fortún et al. 2012; Silveira and Husain, 2007]. A study from Spain reported a 29-fold higher risk of IA in liver transplant recipients who required retransplantation and a 24-fold higher risk in patients requiring dialysis after transplantation [Fortún et al. 2012]. Lung transplant recipients who have documented prior colonization with
Clinical features
The clinical features of IMIs are frequently nonspecific. Although most IMIs cause pulmonary infection, infections may also involve the paranasal sinuses, the central nervous system (CNS), the skin, the gastrointestinal tract or occasionally they can become disseminated (Figure 1). Table 6 summarizes the frequency of the organ sites involved in IMIs in transplant recipients.
Cutaneous lesion of aspergillosis. Reproduced with permission from Dr Pranatharthi Chandrasekar.
Infections due to
Although IMIs caused by
Diagnosis and management of mold infection
EORCT/MSG definitions
Organ involvement in invasive mold infections among transplant recipients.
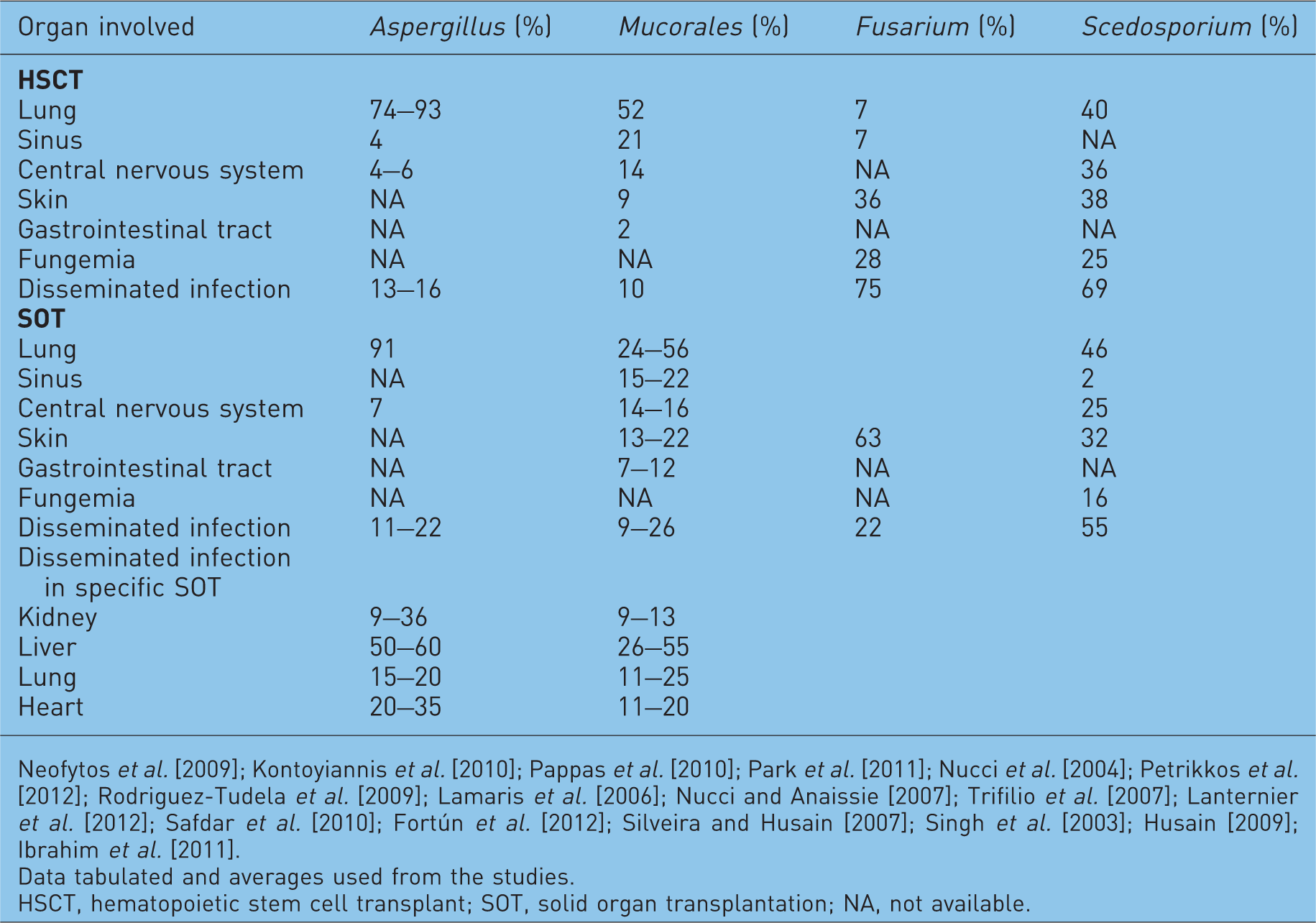
Neofytos et al. [2009]; Kontoyiannis et al. [2010]; Pappas et al. [2010]; Park et al. [2011]; Nucci et al. [2004]; Petrikkos et al. [2012]; Rodriguez-Tudela et al. [2009]; Lamaris et al. [2006]; Nucci and Anaissie [2007]; Trifilio et al. [2007]; Lanternier et al. [2012]; Safdar et al. [2010]; Fortún et al. [2012]; Silveira and Husain [2007]; Singh et al. [2003]; Husain [2009]; Ibrahim et al. [2011].
Data tabulated and averages used from the studies.
HSCT, hematopoietic stem cell transplant; SOT, solid organ transplantation; NA, not available.
Microbiologic criteria for the diagnosis of proven IFIs rely on direct tests (cytology, direct microscopy and culture) demonstrating the presence of fungal elements [De Pauw et al. 2008]. However, obtaining tissue samples or performing invasive procedures is not always feasible because of cytopenias or the poor clinical condition of these patients. Thus, the initiation of appropriate antifungal therapy is frequently delayed. The difficulty in establishing an early diagnosis is one of the primary reasons for the high mortality rates seen in IMIs [Chamilos et al. 2006; Rinaldi 1991; von Eiff et al. 1995].
Nonculture diagnostic assays such as the galactomannan (GM) and BG for the diagnosis of IFIs have been developed over the last two decades [Boudewijins et al. 2006; Mennik-Kersten and Verweij, 2006]. The advent of these indirect tests represents a major advance in the management of patients at risk for IFIs (Table 3).
Galactomannan assay
GM is an
In the clinical setting, the detection of serum GM antigen has been shown to be a useful screening test for the early diagnosis of IA in patients at risk [Maertens et al. 2001; Pfeiffer et al. 2006; Sulahian et al. 2001]. Serum is the most frequently tested specimen and appears to provide the highest sensitivity (up to 95%, depending on the patient population and previous antifungal therapy) [Chamilos et al. 2006]. Galactomannan is water soluble and therefore can be detected in specimens other than serum, including bronchoalveolar lavage (BAL), cerebrospinal fluid, pleural fluid and urine [Klont et al. 2004]. Except for serum and BAL, the use of GM in other specimens remains investigational.
A recent meta-analysis study was conducted to determine the role of BAL-GM in the diagnosis of IA. In this study, BAL-GM sensitivity and specificity varied from 84% and 95%, respectively, depending on the population tested and the cut-off used [Zou et al. 2012]. BAL-GM may be used as an adjunctive tool in establishing the diagnosis of IA (see http://www.accessdata.fda.gov/cdrh_docs/pdf6/K060641.pdf and http://www.accessdata.fda.gov/cdrh_docs/pdf9/K093678.pdf). Typically, a serum GM value of at least 0.5 is considered positive. Using this suggested cutoff point, the reported sensitivity and specificity of the GM assay was 80.7% and 89.2% respectively [Chamilos et al. 2006]. Conversely, due to the lack of data, the threshold for positive BAL-GM remains under debate.
The use of serum GM is also an excellent tool for the early diagnosis of IA. Sulahian and colleagues showed that GM might be detected in serum as early as 5–8 days before the clinical manifestations of IA develop [Sulahian et al. 2001]. These results support the use of GM as a screening tool for patients at high risk of developing IA. In this setting, the detection of positive results, particularly in two consecutive serum samples, provides strong support for the diagnosis of IA [von Eiff et al. 1995; Mennick-Kersten and Verweij, 2006; Maertens et al. 2001]. Recently, some authors have also suggested that GM could serve as a surrogate marker of clinical response to treatment in patients with IA [Miceli et al. 2008; Park et al. 2011; Maertens et al. 2009; Boutboul et al. 2002]. Several studies showed that the titer of GM tends to decrease in cases that demonstrated a clinical response. Similarly, increasing GM titers were associated with poor outcomes [Park et al. 2011; Maertens et al. 2009; Boutboul et al. 2002; Woods et al. 2007; Segal et al. 2008]. False-positive reactions have also been reported in 1–18% of the tested samples and may be due to cross reactivity or false-positive GM (Table 3).
Radiographic imaging
Radiographs of the chest and sinuses have been used as a primary means of diagnostic assessment. However, they are frequently inadequate to establish a diagnosis. Initial chest computed tomography (CT) scan findings in IA are dominated by the nodule and its associated ‘halo sign’. The main finding in IA is generally a pulmonary nodule greater than 1 cm in diameter, that is, the mancronodule. It is defined as a localized, space-occupying, ovoid, soft-tissue opacity that displaces rather than conforms to the shape of the preexisting aerated lung [Georgiadou et al. 2011]. More than 90% of patients with mycologically proven IA have at least one pulmonary nodule. The halo sign apparent on the CT scan is a modifier of the macronodule. It is defined as a perimeter of ground-glass lung opacity surrounding a pulmonary nodule. On initial CT scan, a study of patients with mycologically proven IA, about 33% have one or more macronodules with a halo sign. The ‘air crescent sign’ generally follows the halo sign approximately 1 week later (Figure 2).
Computed tomography scan showing necrotic nodular infiltrate in a hematopoietic stem cell transplant recipient with pulmonary aspergillosis.
Management
Despite the advances in the field and the advent of newer technologies, identification of fungal pathogens continues to be difficult and early diagnosis is not always possible [Georgiadou et al. 2011; Miceli and Lee, 2011; Revankar and Sutton, 2010]. Because early treatment is crucial in the management of these patients, initiation of empiric antifungal therapy is not uncommon when IFI is suspected [Walsh et al. 2008].
Strategies for the management of mold infections
Current strategies for the management of IFIs include prophylaxis, empiric, preemptive, and targeted therapy (Figure 3) [Ruhnke et al. 2012; Freifeld et al. 2011]. Antifungal prophylaxis involves the administration of an antifungal drug to high-risk patient populations before the onset of signs or symptoms of infection. In addition to neutropenia during the pre-engraftment period in HSCT recipients, these patients are at high-risk for mold infection as a consequence of severe cell-mediated immunodeficiency due to GVHD and its therapy (Table 8). Similarly, certain SOT recipients are also at high risk for mold infections (Table 8). Prophylaxis with anti-mold agents has been recommended in these select patient groups [Tomblyn et al. 2009; Singh et al. 2013]. In this setting, the antifungal agent is started despite the fact that adequate microbiological diagnosis of IFIs is unavailable [Freifeld et al. 2011]. Preemptive therapy is often initiated when nonspecific radiographic signs are present or laboratory tests are suggestive of IMIs, in the absence of microbiological or histopathological confirmation of IFIs. Although preemptive antifungal therapy has been used successfully in patients with neutropenia who are febrile, there are no standard recommendations that fully support its use [Pasqualotto and Colombo, 2010; Kontoyiannis and Lewis, 2011; Lortholary et al. 2010]. Targeted therapy relies on treating microbiologically and histologically documented cases of IFIs [Ruhnke et al. 2012; Freifeld et al. 2011].
Strategies for the Management of Neutropenic Patients at High Risk for Invasive Fungal Infections.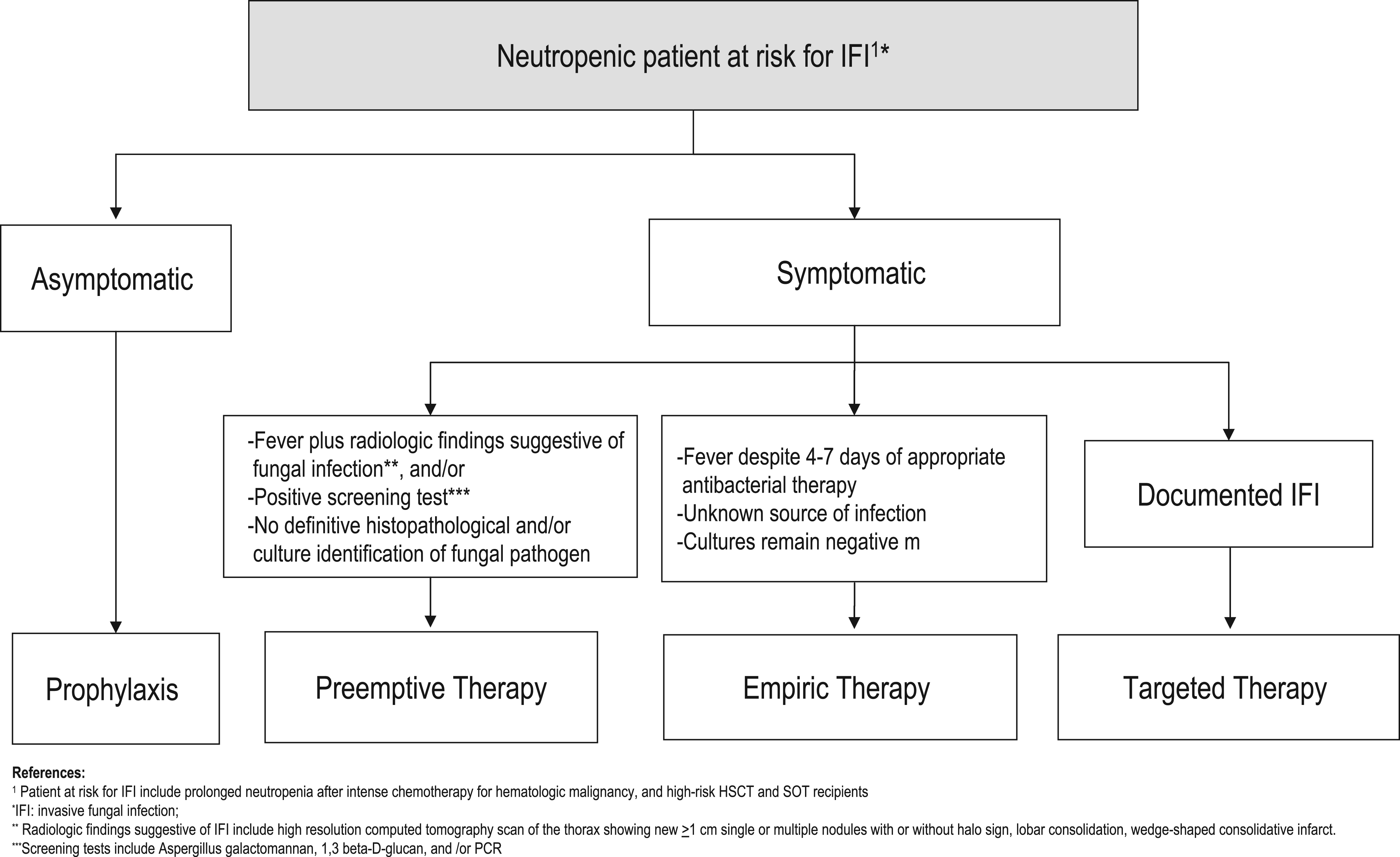
Specific management issues
Specific antifungals used for the treatment of IFI are summarized in Table 4. The management and prognosis of IA depends on the specific form of disease and the degree of immunosuppression. For over 50 years, AmB-d was the mainstay of antifungal therapy. Guidelines for the management of IA have been published by the Infectious Diseases Society of America [Walsh et al. 2008]. The current mainstay of therapy for IA is considered to be voriconazole. A randomized, multicenter study compared AMB-d with voriconazole as initial therapy for IA. This pivotal study demonstrated that initial therapy with voriconazole led to better responses and improved survival with fewer serious side effects, such as renal insufficiency and infusion-related toxicity [Pasqualotto and Colombo, 2010]. The appropriate dose of voriconazole is 6 mg/kg twice daily for 1 day, followed by 4 mg/kg twice daily.
A crucial factor in optimizing therapy in any patient with IA is the decrease or elimination of the immunosuppressant whenever possible. The recent literature suggests that if patients are diagnosed and treated early with appropriate antifungal therapy, the response rates may reach 50% or greater [Ruhnke et al. 2012].
Successful treatment of mucormycosis requires a high index of clinical suspicion for an early diagnosis [Kontoyiannis and Lewis, 2006; Ibrahim et al. 2011; Freifeld et al. 2011]. Mortality rates as high as 85% have been documented. Treatment requires reversal of the underlying condition, when possible; wide and extensive surgical removal of the affected tissue; and early antifungal therapy. Unfortunately, prospective randomized clinical trials have not been performed. Current recommendations include high-dose LFAmB at doses of 7–10 mg/kg/day [Kontoyiannis and Lewis, 2006]. The optimal duration of therapy is unknown, but a total dose of 2–6 g has been used in some cases.
In addition, posaconazole has demonstrated
The overall prognosis of the infection depends on several factors, including the site of infection, the rapidity of diagnosis, and the type and severity of immunosuppression. Although the overall mortality rate for mucormycosis is approximately 50%, the mortality rate for the rhinocerebral form is approximately 85%.
Patient survival in patients with infections due to
The successful management of invasive scedosporiosis also depends on the early diagnosis and the early initiation of appropriate antifungal therapy, as well as the correction of the host’s immune status [Musk et al. 2006; Husain et al. 2005]. If possible, immunosuppression should be either discontinued or reduced and the neutropenia reversed. Although clinical studies have not yet been performed, voriconazole is the drug of choice in the treatment of infections due to
Conclusion
Invasive fungal diseases have become an infection of increasing importance in the transplant recipient. Recent advances in antifungal therapy, such as the echinocandins, voriconazole and posaconazole have made a significant impact on the selection of antifungals due to their broader spectrum of activity, their excellent safety profile, and their ease of use in these critically ill, severely immunosuppressed patients. Additionally, the earlier recognition of the high-risk patient and the known difficulty in establishing a definitive diagnosis warrant the use of early antifungal therapy in an attempt to decrease the exceedingly high morbidity and mortality associated with these infections.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.

