Abstract
Objective
To evaluate the efficacy of conventional and novel non-pharmacologic, non-invasive therapeutic interventions in physiotherapy for the treatment of phantom limb pain (PLP) in post-amputee patients.
Methods
A systematic search for the articles was conducted in multiple electronic databases such as PUBMED, Google Scholar, EMBASE, Cochrane library and Physiotherapy Evidence Database (PEDro), following the PRISMA method and only published articles from the last 12 years (2010–2022) evaluating the efficacy of different physiotherapy interventions for the treatment of PLP in post-amputee patients were included. The methodological quality and risk of bias of the articles were assessed and evaluated by two independent reviewers using the PEDro scale, Methodological index for non-randomized studies scale (MINORS), and Cochrane collaboration’s assessment tool.
Result
A total of 1840 articles were identified, out of which 17 articles (11 RCTs and 6 pilot studies) were ultimately chosen after the full-text screening. After reviewing the articles, evidence identified in RCTs and pilot studies indicates towards significant improvement in reducing the severity of PLP in post-amputee patients by using different physiotherapy interventions.
Conclusion and discussion
Physiotherapy interventions with advance modalities and exercises can be used to increase the overall effectiveness of the treatment and to reduce the severity of phantom limb pain in post-amputee patients. However, due to the lack of consistent evidence for a given intervention, it becomes even more difficult to reach a majority consensus as to which intervention better assess all the mechanism of PLP thereby alleviating the problem of PLP in post-amputee patients. Therefore, more rigorous randomized controlled trials will be required in the future to reach a conclusion.
Keywords
Introduction
The instance of post-amputation phenomenon, which occurs after partial or complete amputation of a limb, later called ‘Phantom Limb Pain’ was first documented by a French military surgeon Ambroise Pare during the 16th Century. The phrase ‘Phantom Limb Pain’ was coined by Silas Weir Mitchell (Neurologist) who was a military battlefield surgeon during the American Civil War (1871–72), who also provided the very first clinical description of ‘Phantom Limb’ in American soldiers who had undergone major or minor amputation surgery. 1
It is generally seen that 90 to 98% of patients almost immediately after amputation surgery experience a vivid ‘Phantom Limb’ sensation as soon as the anaesthesia wears off, which includes the perception of kinetic (voluntary or involuntary movement of the phantom limb), kinesthetics (awareness of phantom’s shape, size and position) and exteroceptive sensations (pressure, temperature, etc.) and the patients often describe the sensation of tingling, numbness, itching or as if their amputated limb is asleep or paralyzed occupying a ‘habitual posture’ which may or may not elicit pain. 2
A majority (70%–80%) of the amputee patients describe the ‘Phantom Limb Pain’ as a vivid sensation that the amputated limb is still present with intense episodes of pain sensation originating from the amputated limb. PLP are often characterized by the symptoms of dysesthesia. Patients often describe PLP as sharp, electric shock-like, stabbing, burning, crushing and aching pain. However, the timing of the onset of PLP vary from patient to patient. For Instance, in a study it is seen that the PLP is developed in 50% of patients within the first 24 h post-amputation surgery and 85% of the patients within the 1st week.3,4
It is generally seen that the majority of patients complain of excruciating painful ‘abnormal positioning’ of their phantom limb in the morning as they wake up or severe clenching spasms in their phantom hand accompanied by a sensation of the nail digging into their palm which resolves on its own within a few minutes, or sometimes even takes several hours only to return to their ‘Habitual Posture’. 5
PLP is often identified as a neuropathic pain condition in the majority of studies. For instance, fMRI studies of amputee patients’ brains have shown that there is an increased activity in the somatosensory and premotor cortical areas of the brain that are responsible for pain processing, indicating that the intensity or severity of PLP perceived by the patients is genuine and can even be compared to pain elicited by actual tissue damage. 6
According to an estimate in 1983, in India, the total population of amputee patients was estimated to be around half a million, and about 23,500 patients were added every year. A majority of which are males from rural areas of the working age group in their 20s or 30s belonging to the below the poverty line, 7 which poses a major clinical problem as 70%–80% of phantom limb remains painful even after 25 years of amputation surgery, 3 which imparts a significant impact on the quality of life of the patients and on to the society as a whole.
An epidemiological study conducted on 155 amputee patients in Kolkata, India, describes that the most common cause of amputation was due to trauma which accounts for 70.3%, followed by peripheral vascular disease. Diabetic foot condition is one of the most common causes of below-the-knee amputation in diabetic patients in India. In India, below-the-knee amputation accounts for about 94.8% of all amputations. 8
Earlier, it was believed that the phenomenon of PLP is usually seen in patients after major or minor amputation surgery. But in a study conducted by Wilkins et al. (1998) 9 to determine the prevalence of PLP in children born with a missing limb, it was found that the phenomenon of PLP is not only limited to the amputee patients (i.e., surgical group) but also in congenital amputee patients (7.2%), which indicates not only the prevalence of PLP in congenital amputee population but also its complex mechanism and nature. Therefore, a better understanding of the mechanism and aetiology of phantom limb pain is essential in order to develop novel treatment approaches and modalities that will not only help to alleviate the suffering of patients but also help millions of patients worldwide.
A recent meta-analysis conducted by Limakatso and colleagues
10
reviewed the available evidences from 15 studies (cross-sectional study) with a total of 4102 amputee patients and identified 25 potential candidate factors related to the prevalence of PLP. However, only three potential factors with the highest degree of evidence were seen closely related to the prevalence of PLP in amputee patients across the studies – residual limb pain (RLP), pre-amputation pain and non-painful phantom sensation. a. Residual Limb Pain (RLP): It can be described as the pain originating from the remaining portion of the limb after amputation, generally closer and localized to the site of amputation or Stump; therefore, it is often referred to as Stump pain. Patients typically describe RLP distinct from PLP by it's site of origin and severity. For example, in a study, it was seen that 33% of amputee patients describe RLP as more problematic as compared to PLP (24%).
11
It is generally seen that the RLP and PLP often occur together in amputee patients, where RLP tends to resolve on its own, most likely due to the resolution of the post-operative surgical wounds. In some patients, it can persist for decades either due to infection, pressure ischemia due to improper fitting of the prosthetic, etc. PLP remains consistent even after the complete healing of the surgical wounds, indicating a close relationship between peripheral factors (i.e. neuroma formation, infection, etc.) and initiation of RLP and thereby its progression into PLP. b. Pre-amputation pain: It is generally seen that patients who complain of intense pain in their limb prior to amputation surgery are more prone to develop PLP.12,13 In addition, it is seen that if the patients have any deformity in their limb, they are more likely to complain of the deformity being carried over into the ‘Phantom Limb’ after the amputation surgery indicating a mechanism of re-emergence of repressed memories of the limb before amputation. For example, many patients report that they can feel the wedding ring or watch band on their phantom limb. c. Non-Painful Phantom Sensation: Studies have shown that the prevalence of PLP is more common in patients with non-painful phantom sensation as compared to those without non-painful phantom sensation. For instance, an fMRI study conducted by Andoh found a similar pattern of somatosensory and premotor cortical activation upon inducing non-painful phantom sensation and phantom limb pain in amputee patients indicating a shared neural mechanism and common neural pathway.
14
One of the major challenges associated with the treatment and prevention of PLP in amputee patients is that as a painful condition, it is highly subjective and has multi-dimensional aspects to it, and is easily influenced by a variety of factors like the cause of amputation. It is generally seen that the prevalence of PLP is more common in amputation due to trauma, patient's past experiences with pain, their psychological or emotional state, and genetics such as single nucleotide polymorphisms in the genes such as GCH1 and KCNS1 15 ; which were found to be closely linked with the increased susceptibility of the patient to develop chronic pain, etc. In addition, PLP also projects several additional intrinsic challenges, such as the pain is perceived to originate from the body part that is no longer physically present, which not only complicates the process of quantifying the pain perceived by the patient but also the sensory, emotional and cognitive association of pain with the actual or potential tissue damage, thereby influencing or dominating the clinical picture of the pain perceived by the patient. Even though multiple theories and hypotheses have been proposed to explain the aetiology and the mechanism of PLP, there is still no clear treatment approach to prevent or to cure PLP in amputee patients.
The effective treatment approach to prevent or to cure phantom limb pain (PLP) in amputee patients is still a major issue that researchers and clinicians across the world continues to confront.
Pharmacological Therapies
The use of pharmacologic agents is still considered to be the first line of management of pain and symptoms of PLP in amputee patients, yet the condition remains poorly managed across the world. For instance, in a recent survey conducted by Corbett, 37 clinicians and neurosurgeons across 30 different UK pain clinics were interviewed to determine the effective treatment options available for PLP. 12 different pharmacological and non-pharmacological therapies were found to be used to treat PLP, with pharmacological treatment approach being the most popular among the clinician and patients. 16 As shown in Figure 1. The most commonly used Pharmacological agents for the treatment of PLP are anti-convulsant, tricyclic antidepressants, other N-methyl-D-aspartate receptor antagonists/epidural anaesthesia (such as gabapentin, pregabalin, calcitonin and ketamine), lidocaine and opioids, which are seen to be effective not only in reducing the symptoms, frequency and the severity of neuropathic pain but also to avoid or to reverse maladaptive cortical re-organization in some cases. 17 Despite their effectiveness in relieving the symptoms and pain of PLP in amputee patients, the use of opioids, oral analgesic drugs (i.e. vicodin), epidural anaesthesia, etc. are frequently associated with side effects, such as dizziness, nausea, sedation, loss of appetite, fatigue etc., coupled with a high rate of chances of addiction. Dependence and prolonged use of them may cause secondary complications in some patients such as liver damage, kidney damage, depression, mood swings, etc. 18
Surgical intervention
Surgical interventions are generally used as the last resort in patients with severe PLP or when all the other interventions fail to be effective and are specifically used to avoid or to reverse maladaptive central neuroplastic changes in the brain. Targeted muscle reinnervation, deep brain stimulation and spinal cord stimulation techniques are evaluated to be potentially promising options for the treatment of PLP. However, no surgical intervention has thus far been determined to be superior in the management of PLP. Robust research studies are still lacking to prove their efficacy in the prevention of PLP in patients, and it is generally seen that even after surgical intervention, PLP reappears in some patients. a. Targeted muscle reinnervation (TRM): It was originally developed in order to improve the functional utility and control myoelectric prosthetics limbs of amputee patients.
19
In the traditional amputation surgical technique, the nerves of the limb to be amputated are severed, due to which the neurons of the amputated limb undergo retrograde degeneration. The residual nerve fibres and axons attempt to re-establish the previous connection. However, the reconnection is impossible in case of amputation, which often results in the development of the disorganized mass of nerve fibres at the stump, known as neuroma, which subsequently leads to central sensitization, which later progresses or triggers PLP in patients. In the targeted muscle reinnervation technique, the residual nerves from the amputated limb are transferred to reinnervate new target muscles at the stump, which acts as the biological amplifier, not only amplifying motor signals from the residual nerve fibres at the stump but also prevent the misdirection nerve growth, thereby inhibiting the neuroma formation and development of PLP in amputee patients. For instance, a randomized controlled trial conducted by Dumanian et al. compared the effectiveness of conventional neuroma excision to TMR in 28 patients with chronic RLP and PLP. They found a significant reduction in the severity of both RLP and PLP in patients at 1-year follow-up.
20
b. Deep Brain Stimulation (DBS): It is a neurosurgical procedure that involves electrical stimulation of certain parts of the brain via electrodes implants (i.e. peri-aqua ductal grey matter stimulation and lateral thalamic stimulation). The amount of electrical stimulation of the brain is generally controlled by a small pacemaker-like device called a neurostimulator, which generally alters the electrical signals in the brain that are responsible for pain. Neuropathic pain is seen to be more likely to respond to thalamic stimulation as compared to peri-aqua ductal grey matter stimulation. For instance, in a study conducted by Bittar et al. (2005) to determine the efficacy of DBS in three patients with severe PLP, refractory to multiple pharmacological and non-pharmacological interventions were studied. They found a significant reduction in the severity of PLP by 62% (SD 7.4%, range 55%–70%) in patients indicating its efficacy and its potential use in the management of PLP.
21
However, due to its cost, associated dangers and secondary complications in patients (i.e., seizures), its potential side effects are generally seen to outweigh its benefits in amputee patients, due to which there are only a few studies advocating its use in the treatment of PLP in amputee patients. Therefore, both robust randomized clinical trials and studies must be needed. c. Spinal cord stimulation (SCS): It is a surgical procedure that involves the implantation of electrodes in between the spinal cord and vertebrae in epidural space connected to a small pacemaker-like device placed under the skin, usually near the abdomen delivering low-frequency current directly to the spinal cord. It generally works upon the principle of ‘Gate control theory of Pain’ which states that the electrical stimulation of inhibitory interneurons of Lamina I and Lamina II of the dorsal horn of the spinal cord via A-Beta fibres not only inhibits the transmission of pain to the brain but also activates the peri-aqua ductal grey matter and rostral ventromedial medulla which activates endogenous opioid pain suppression mechanism which further inhibits the transmission of pain to the brain by releasing endogenous opioids like enkephalin and dynorphin. For instance, in a trial conducted by Katayama et al. to determine the efficacy of SCS in 19 patients with PLP, they found a significant reduction in the severity of PLP in patients 6/19 (32%) for the long term.
22
However, just as deep brain stimulation technique, its cost and potential side effects and dangers such as epidural and intrathecal hemaorrhage, spinal cord and nerve trauma, paralysis, etc. outweigh its benefits in amputee patients. For example, Mekhail et al. reviewed over 707 cases of SCS and its associated complication – hardware related problems (38%), lead migration (22.6%), lead connection failure (9.5%), lead breakage (6%), biological related complications including pain at the site of application (12%), clinical infection (4.5%) and other complications (7.4%).
23
Physiotherapy interventions
In a recent survey conducted by Limakatso and Parker, 27 clinicians and researchers across 16 different countries were asked which treatment approach is viewed to be effective in treating PLP in amputee patients. Out of a total of 37 different treatment approaches for PLP management, seven treatment approaches were determined to be effective in reducing the symptoms of PLP in amputee patients by majority consensus (>50%), six of which were found to be non-pharmacological interventions and four were specifically designed to target the central contributor of PLP to restore the original cortical representation of the amputated limb by restoring the positive visual feedback of the amputated limb to the brain. 24
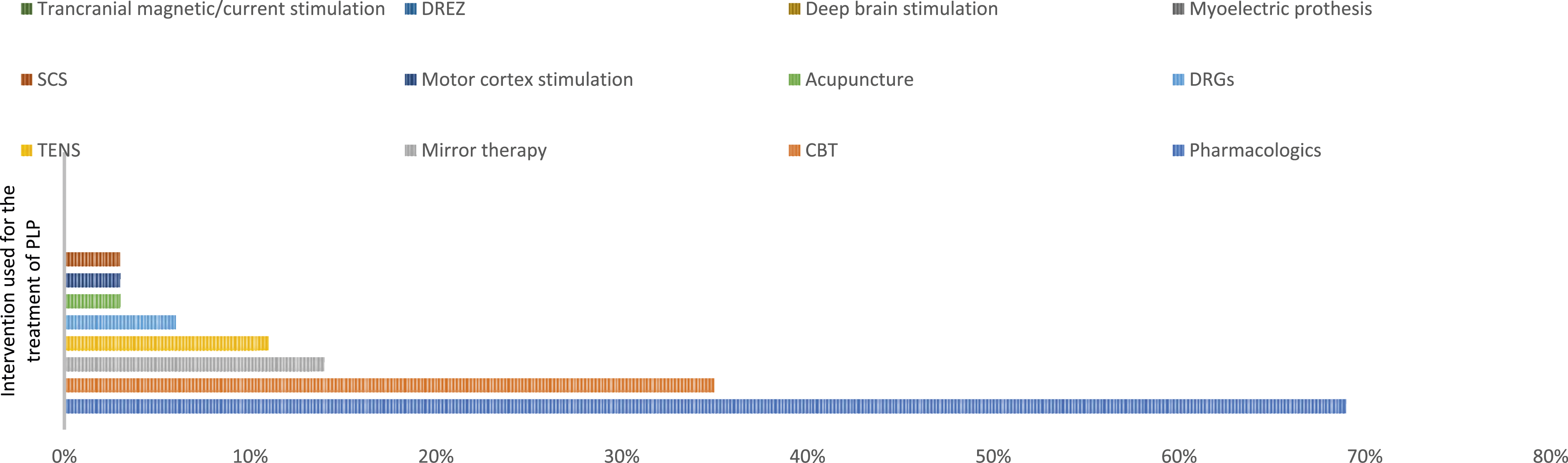
Prevalence of intervention used for the treatment of PLP. 16
Mirror therapy
Mirror therapy is a non-invasive and perhaps one of the least expensive and most effective non-pharmacological therapeutic interventions with no significant side effects used for the treatment of phantom limb pain in patients.
V S Ramachandran and Rogers-Ramachandran (1996) were the first researchers to successfully report the efficacy of mirror therapy in relieving both phantom limb pain and sensation in amputee patients by resolving the visual-proprioceptive dissociation in the brain. 5 For example, following limb amputation, an individual's cortical and peripheral body representations remain intact but the motor signals send by the motor cortex to the amputated limb via the parietal lobe, which monitors the command and simultaneously receives feedback from the limb, gets altered as command send to the limb are no longer physically present. This mismatch is exacerbated by the lack of visual feedback from the amputated limb, resulting in excessive pain despite the lack of sensory stimulus. 2 For instance, this phenomenon can easily be comprehended by a simple experiment. An individual is asked to place one of his/her hand on the table beside a rubber hand positioned in front of him/her on the table at a distance and the other hand just under the table beneath the rubber hand such that one of the individual’s own hand remains hidden from his/her own view. Both the actual and rubber hands are simultaneously stroked with the brush, causing the person to perceive the rubber hand as his/her own. To test the visual feedback to the brain and the participant response, the researchers then strike the rubber hand with a hammer. The participants flinch in fear of pain even though the actual body part remains unharmed.
A majority of the patients following the limb amputation surgery experience a vivid sensation of movement in their phantom limb (kinetic). For example, many patients describe that they can voluntarily move their phantom limb and sometimes their phantom limb might attempt to hold or to lift the glass on the table, wave goodbye or to even shake hands.
Generally, the motor commands sent from the motor and premotor cortex to the limb via the parietal lobe which monitors the commands and simultaneously receives feedback from the limb, that is, to clench the hand or to move the limb (repressed memories), are normally damped by error feedback signals send by the proprioceptors present in the limb. However, since such dumping of the motor signals is not possible in amputee patients, the motor output gets amplified even further. This overflow of signals thus produces the sensation of excruciating painful clenching spasms in their phantom hand accompanied by a sensation of the nail digging into their palm which resolves on its own within a few minutes or sometimes even takes several hours only to return to their ‘Habitual Posture’ by imagining voluntarily unclenching of the phantom hand. But with the passage of time, it is generally seen that the majority of the patients lose their ability to voluntarily move and control their phantom limb. One of the possibilities is that it is due to prolonged absence of confirmatory sensory and visual feedback of the amputated limb to the brain, that is every time when the motor signals send by the motor cortex to the amputated limb, the brain receives contradictory visual input that not only the limb is not moving but also missing, due to which the brain perceives that the phantom limb is fixed in a specific position or ‘paralyzed’. Therefore, the majority of patient’s complain that their phantom limb is frozen in a specific position and that they cannot produce voluntary movement in it, even with intense efforts. 2 For instance, a study conducted by J. Foell et al. (2013) indicates that mirror therapy causes a decrease in the activity of the inferior parietal lobe of amputee patients to return to the existing baseline configuration before the amputation surgery, thereby reducing the severity of PLP. 25
According to some randomized controlled trials and clinical studies, mirror therapy shows a significant result in reducing the severity of PLP and unpleasant phantom limb sensation by providing positive visual feedback of the amputated limb to the brain, thus creating the illusion that the amputated limb responds to the motor commands. To enable the amputee patient to perceive the real movement in their phantom limb, in mirror therapy, the therapist generally uses a ‘flat mirror’ placed parasagittally in front of the patient’s body with the reflective surface of the mirror towards the non-amputated limb. The reflection of the non-amputated limb mimics the amputated limb; thereby, the mirror provides an optical illusion that the phantom limb is moving simultaneously with the non-amputated limb. This positive visual feedback to the brain creates a vivid sensation of synchronous movement of the phantom limb, relieving the ‘paralyzes’ of the phantom limb and associated pain, thereby supporting mirror therapy as potentially promising for the treatment of PLP. However, mirror therapy has multiple limitations, for example, as a mirror image, the patient is required to focus on the reflection of their intact contralateral limb with no prominent functional disability. Even after numerous studies supporting mirror therapy, there is still no standardized treatment protocol. Therefore, more work is needed to elucidate the clinical efficiency of mirror therapy to alleviate PLP in amputee patients.
Augmented-virtual reality
The use of augmented-virtual reality is seen to be getting popular among surgeons, researchers, clinicians and physiotherapists around the world as it allows the users to interact with their surrounding in a 3-D virtual space, thereby creating a virtual world around the user. The user can not only interact with the virtual object but to also using certain devices and instruments can feel its temperature, dimensions and precise location and orientation of an object in the space just like the real object in the real world. For instance, in physiotherapy, it has emerged as a novel method of intervention for motor and cognitive rehabilitation of stroke,26,27 cerebral palsy, 28 Parkinson’s syndrome 29 and chronic neuropathic pain patients. 30
Augmented-virtual reality interventions have recently emerged as a novel treatment approach for the management of PLP in amputee patients over traditional non-pharmacological therapeutic interventions like mirror therapy. For instance, while mirror therapy has shown promising results as a potentially safe and effective therapy for the treatment of PLP, there are certain limitations that not only make its effective application in amputee patients challenging but also ineffective in many patients. For example, the patient is required to focus on the reflection of their intact contralateral limb with no prominent functional disability in order to successfully perceive the illusion that their phantom limb is responding to motor commands. However, if they lose focus or to even look at their intact limb, the visual illusion of their phantom limb would be lost, thereby having no effect. In addition, the patient must remain in a fixed position with their head orientated towards the mirror throughout the treatment session; it explains why mirror therapy is effective in some patients and completely ineffective in others. For instance, in a recent study conducted by Ortiz-Catalan et al. to determine the efficacy of virtual reality in 14 patients with refractory PLP, they found that after 12 sessions, the pain and the severity of PLP were significantly reduced in 12 out of 14 patients with no adverse side effects. 31
The use of augmented-virtual reality for clinical purposes and as a novel treatment approach for motor and cognitive rehabilitation of patients are commonly justified and encouraged by the assumption that it not only boosts neuromuscular rehabilitation but also encourages patients to use the muscle of the stump and the remaining part of the amputated limb, thereby decreasing the chances of neuroma formation. However, one of the major drawbacks of augmented-virtual reality is that it requires volitional voluntary musculature activity of the stump for the precise prediction of motor execution of the phantom limb. Thus, the use of augmented-virtual reality in patients with prominent nerve lesions of the muscle at around the stump affecting the muscular activity and prominent shoulder disarticulation is seen to be ineffective.
Transcutaneous electrical nerve stimulation
Transcutaneous electrical nerve stimulation is a non-invasive, non-pharmacological, safe and easy to use, adjunct analgesic technique which is used to deliver low-frequency pulsed electrical current to the skin (i.e. directly over or around the site of the highest intensity of pain) to activate/stimulate the underlying peripheral nerves via cutaneous electrodes connected to a portable battery-powered device to provide relief to pain. TENS is generally used as a stand-alone or in combination with other treatment techniques for the treatment of a wide variety of acute, subacute or chronic pains including phantom and stump pain.
Transcutaneous electrical nerve stimulation generally works upon the principle of the Gate control theory of pain by Melzack and Wall (1965). 32 TENS can be used to selectively stimulate large diameter A-beta afferent fibres without activating small diameter A-delta and C fibres to elicit segmental analgesic effect (conventional TENS/High TENS) or to stimulate small diameter A-delta or C fibres to elicit extra-segmental analgesic effect (acupuncture TENS/Low TENS). According to multiple research studies, the use of TENS may increase blood flow, 33 reduces muscle spasms and inhibits 2nd order neurons, 34 thereby inhibiting the transmission of pain to the brain, indicating its plausible role as an adjunct analgesic technique for the treatment of PLP, stump pain or both. For instance, in a pilot study conducted by Mulvey et al., 2013 to determine the efficacy of TENS in alleviating PLP and stump pain in 10 amputee patients, they found a significant decrease in the intensity of pain score after a 60-min treatment session (p < .05). 35
Transcutaneous electrical nerve stimulation applied to the contralateral extremity has shown promising results in the management of PLP and stump pain in amputee patients. However, most of the evidence advocating the use of TENS in the treatment of PLP is limited to the case and pilot studies, 36 due to the concern that the application of TENS on the stump or remaining portion of the limb after amputation may exacerbate the pain, thereby increasing the severity of PLP.
How the application of TENS on the contralateral limb can help to alleviate PLP and stump pain in amputee patients can be explained by the neuromatrix theory of pain proposed by Melzack, 37 according to which the sensory experiences create a unique neuromatrix which gets imprinted in the brain. When the limb gets amputated, the lack of afferent signals from the amputated limb affects the previously set neuromatrix causing a maladaptive cortical re-organization triggering the abnormal generation of pain in spite of the lack of a sensory stimulus. TENS works by replacing or substituting the lack of afferent signal from the amputated limb. In case of contralateral limb stimulation, (i.e. stimulation of the area on the intact limb approximate to the pain felt in the phantom limb) the relevant afferent signals from the contralateral limb activate the cortical areas of the brain of the deafferented limb, thereby relieving the severity PLP in amputee patients.
Repetitive transcranial magnetic stimulation/transcranial direct current stimulation
Repetitive transcranial magnetic stimulation is a non-invasive, non-pharmacological, neuromodulation technique that delivers a brief, high-intensity magnetic field emitted by an r-TMS coil positioned tangentially to the scalp of the patient, approximately at an angle of 45° from the midline of the skull. It targets the somatosensory and motor cortex of the brain, which is proposed to create an electric field, which in turns excites the neurons, thereby activating the descending inhibitory pathway of pain to the thalamus, which not only helps in reducing the severity of PLP in amputee patients but also helps in preventing maladaptive cortical re-organization. Barker A and his colleagues created this technique in 1985 (A. Barker et al., 1985), and it emerged as a revolutionary therapy strategy for the management of numerous neurological conditions such as Parkinson’s disease, neuropathic pain and so on.
Similarly, transcranial direct current stimulation is a non-invasive, non-pharmacological, neuromodulation technique that delivers low-intensity current to different cortical areas of the brain via cutaneous electrodes attached to the scalp of the patient. It modulates the spontaneous neural activity of the somatosensory and motor cortex of the brain, which is likely to mediate the reversal of maladaptive cortical re-organization of the brain to the baseline before the amputation, thereby alleviating the problem of PLP in the amputee patients.
Both of these techniques generally work upon the same principle that after amputation of the limb, the cortical area that was once represented by the amputated limb gets re-occupied by adjacent zones of the primary somatosensory and motor cortex corresponding to the other body parts i.e., lower ipsilateral face, 38 in-line of amputation – deltoid muscles of the amputated limb 38 and contralateral hand. 5 This is known as maladaptive cortical re-organization. The spontaneous neural activity of the somatosensory and motor cortex of the brain can mediate the reversal of maladaptive cortical re-organization of the brain to the baseline before the amputation, thereby alleviating the problem of PLP in amputee patients.
The current evidence involving the use of r-TMS and t-DCS techniques for phantom limb pain and other neurological disorders is limited but emerging and promising in the future.
Considering the above, the primary aim of this review is to describe, examine and determine the efficacy of the available conventional and novel adjunct non-pharmacologic, non-invasive therapeutic interventions in physiotherapy for the management of PLP in post-amputee patients. The secondary aim is to determine the best choice of adjunct physiotherapy interventions for the management of PLP in amputee patients. The exploratory aim is to identify preventive measures that clinicians can take to not only reduce the severity of PLP but to reduce the prevalence of PLP in post-amputee patients.
Method
This systematic review study was designed in accordance with the recommended guidelines by Preferred Reporting Items of Systemic Reviews and Meta-Analysis (PRISMA)
39
and has been registered in PROSPERO (International Prospective Register of Systematic Reviews) under the registration number
Search strategy
On December 2022, a search for published articles from the last 12 years (i.e. 2010 to 2022) was conducted in electronic databases such as PUBMED, Google Scholar, EMBASE, Cochrane library and PEDro database using the keywords ‘phantom limb’, ‘pain’, ‘mirror therapy’, ‘TENS’, ‘augmented reality’, ’virtual reality, ‘Transcranial magnetic stimulation’, ‘Transcranial direct current stimulation’ and ‘physical therapy’, combined with the Boolean operators like ‘AND’ and ‘OR’.
Inclusion criteria
The inclusion criteria used for selecting the articles and studies were as follows: 1. Clinical trials, randomized controlled trials and pilot studies which were published in the indexed scientific electronic databases from the last 12 years (i.e. 2010 to 2022) were included in this article in order to focus on the recent development. 2. Articles and studies which were published in English. 3. We generally sought articles and studies which addressed the efficacy of traditional and novel adjunct non-pharmacological therapeutic interventions in physiotherapy. 4. To be included in this article, the articles and studies were needed to quantitatively measure the effect of adjunct non-pharmacologic, non-invasive therapeutic interventions in physiotherapy on the intensity of PLP perceived by the patient before the treatment and after the treatment, its treatment duration, frequency and follow-up. 5. The scope of search used in this article is designed to not only identify all the potentially relevant evidence supporting the efficacy of the intervention used but also try to identify the aetiology and mechanism of PLP in amputee patients.
Exclusion criteria
The exclusion criteria used for eliminating the articles and studies were as follows: 1. Duplicate studies, case reports, on-going trials, single-subject studies, review articles (i.e. literature review, scoping review and systematic review) and meta-analyses were excluded. 2. Which did not specifically address the PLP. 3. Which involves the use of pharmacological and surgical interventions. 4. Studies that assess the PLP in congenital amputee patients. 5. Studies that did not assess the intensity of PLP perceived by the patients instead focus on the associated symptoms such as depression, anxiety, etc. 6. Studies that involve research conducted on animals.
Selection and data collection process
After conducting the search in the electronic database, all the relevant articles and studies were preselected based upon their abstracts and title. The final decision to include them in this article was made after a full-text screening of articles to determine their relevance to this article. All the studies identified as relevant to this article were assessed by two independent reviewers (SG and AKS) and any disagreement was resolved via discussion.
Quality assessment
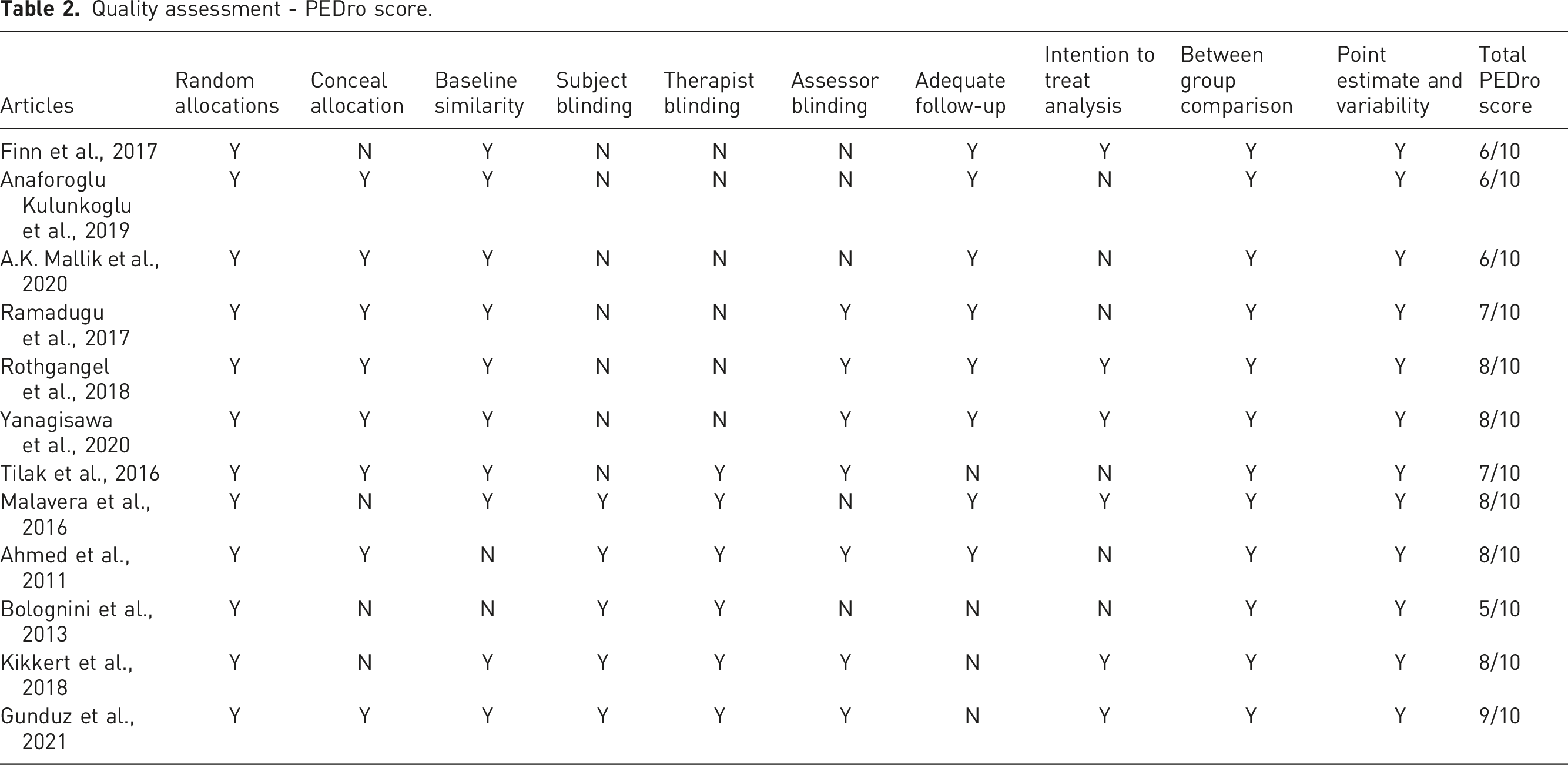
The quality of included RCT articles is evaluated and calculated using the PEDro scale, which mainly consists of 10 items scale which relates to the selection, performance, attribution bases and information. An article with a PEDro score of ≥6 out of 10 is considered good or excellent, and an article with a PEDro score of ≤4 out of 10 is considered poor. The PEDro scores for the included articles were obtained from the official PEDro website (https://www.pedro.org.au/). 40 In case the score for the included articles was not available on the website, two independent reviewers (SG and AKS) scored the quality of the included article across the 10 items scale of PEDro scale, and in case of any doubt or disagreement, the final decision was made through discussion.
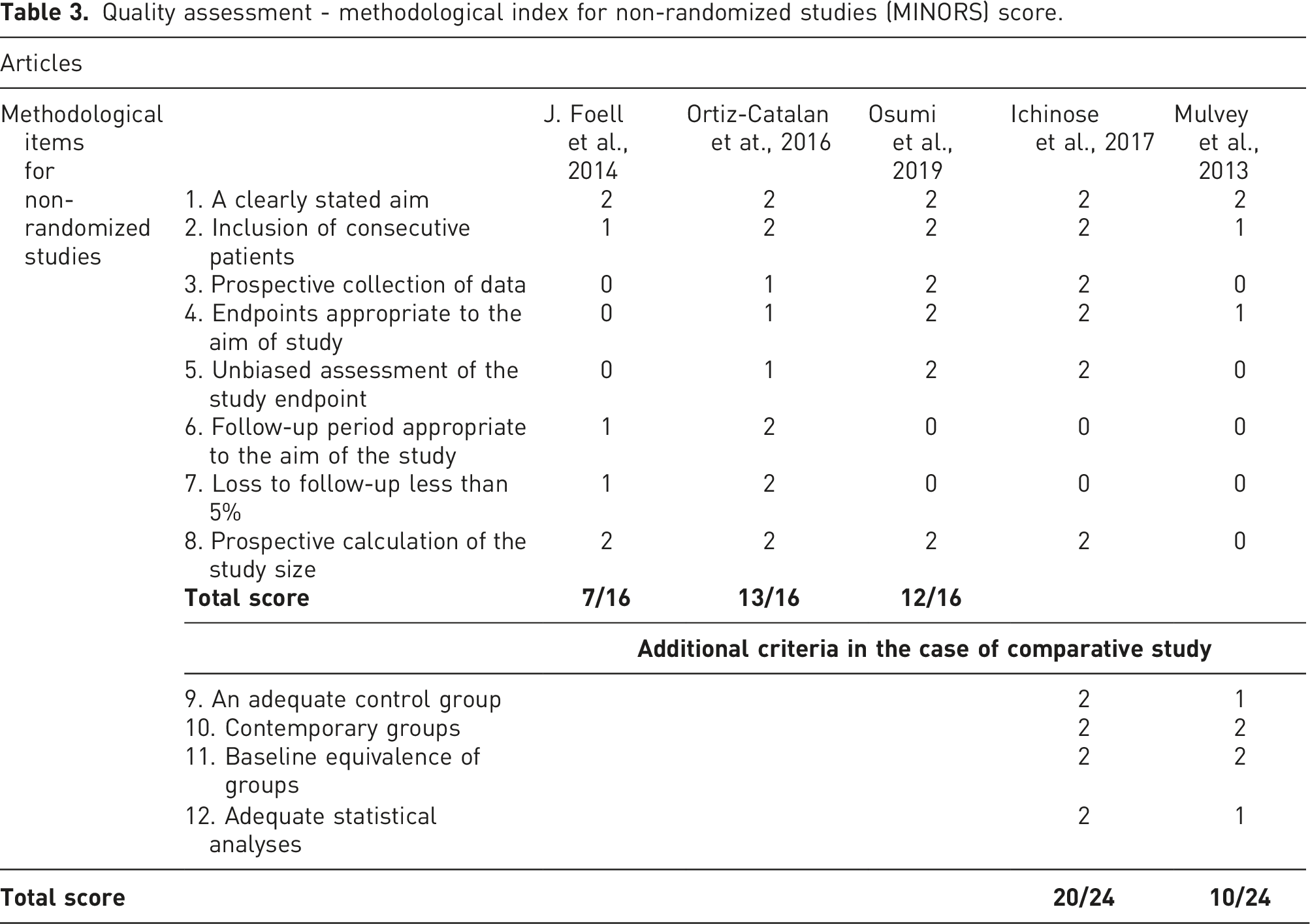
The quality of included non-RCT articles is evaluated and calculated by two independent reviewers (SG and AKS) using the Methodological Index for Non-Randomized Studies (MINORS) scale, which mainly consists of 8 items scale for non-comparative studies and 12 items scale for comparative studies, with a maximum score of 16 for non-comparative studies and 24 for comparative studies. Therefore, it can be used to evaluate both comparative and non-comparative studies. 41
Risk of bias assessment
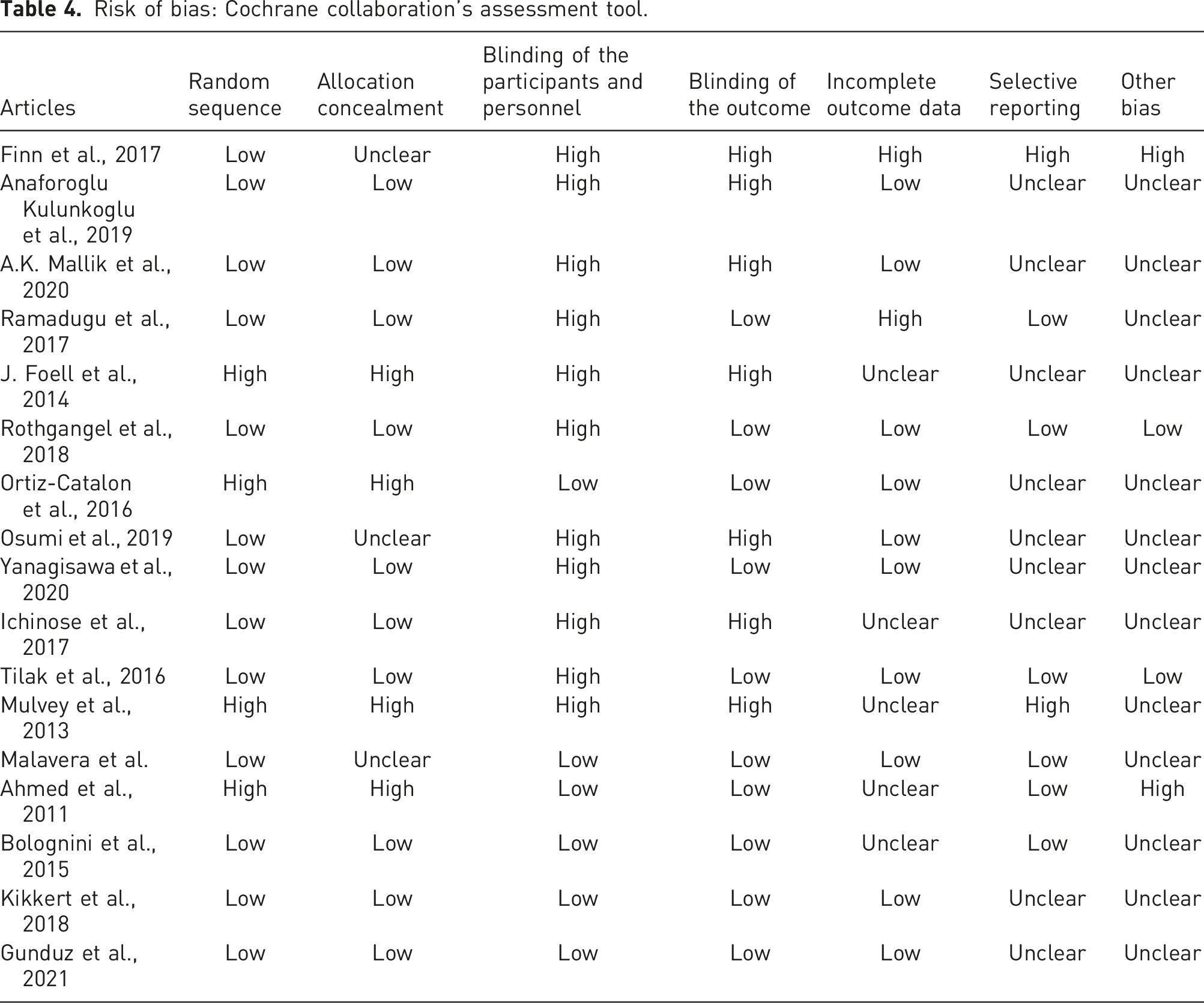
Two independent reviewers (SG and AKS) assessed all the identified relevant articles and studies to evaluate and calculate the risk of bias using the criterion given in the Cochrane Handbook for Systematic Reviews of Interventions: a. Selection bias – Random sequence generation, allocation concealment b. Performance bias – Blinding of the participants and personnel c. Detection bias – Blinding of the outcome assessment d. Attrition bias – Incomplete outcome data e. Reporting bias – Selective reporting f. Other bias
In case of any doubt or disagreement, the final decision was made through discussion.
Data analysis
Data extracted from each and every article and study were entered into a tabulated form on Microsoft Excel Spreadsheet for analysis. Studies were grouped in accordance to the type of intervention used. Differences between the studies, results and their subsequent potential impact on the severity of PLP in the follow-up period in amputee patients were compared with each other. The following data were extracted from the included articles: 1. Author and year of publication 2. Objective of the study 3. Study design 4. Intervention used 5. Sample size, amputation and gender of the patient 6. Treatment characteristics and duration 7. Pain assessment tool used 8. Follow-up 9. Results
Result
Search result
Overall, a total of 1840 articles were retrieved from the electronic databases published between 2010 and 2022 using the keywords ‘phantom limb’, ‘pain’, ‘mirror therapy’, ‘TENS’, ‘augmented reality’, ’virtual reality, ‘transcranial magnetic stimulation’, ‘Transcranial direct current stimulation’ and ‘physical therapy’, combined with the Boolean operators like ‘AND’ and ‘OR’, of which only 976 remained after the removal of duplicates and articles which were not relevant to this article based upon their titles. Following the removal of articles which do not meet the inclusion criteria, only 90 relevant articles were preselected and screened for inclusion based upon their abstract. Following a screening of the full text, a total of 73 articles were excluded because they fail to meet the inclusion criteria or due to a high risk of bias in the articles. In total, 17 articles were included in this article as shown in the Figure 2. PRISMA flow chart: graphical representation of the process of search and selection of relevant articles from the electronic database.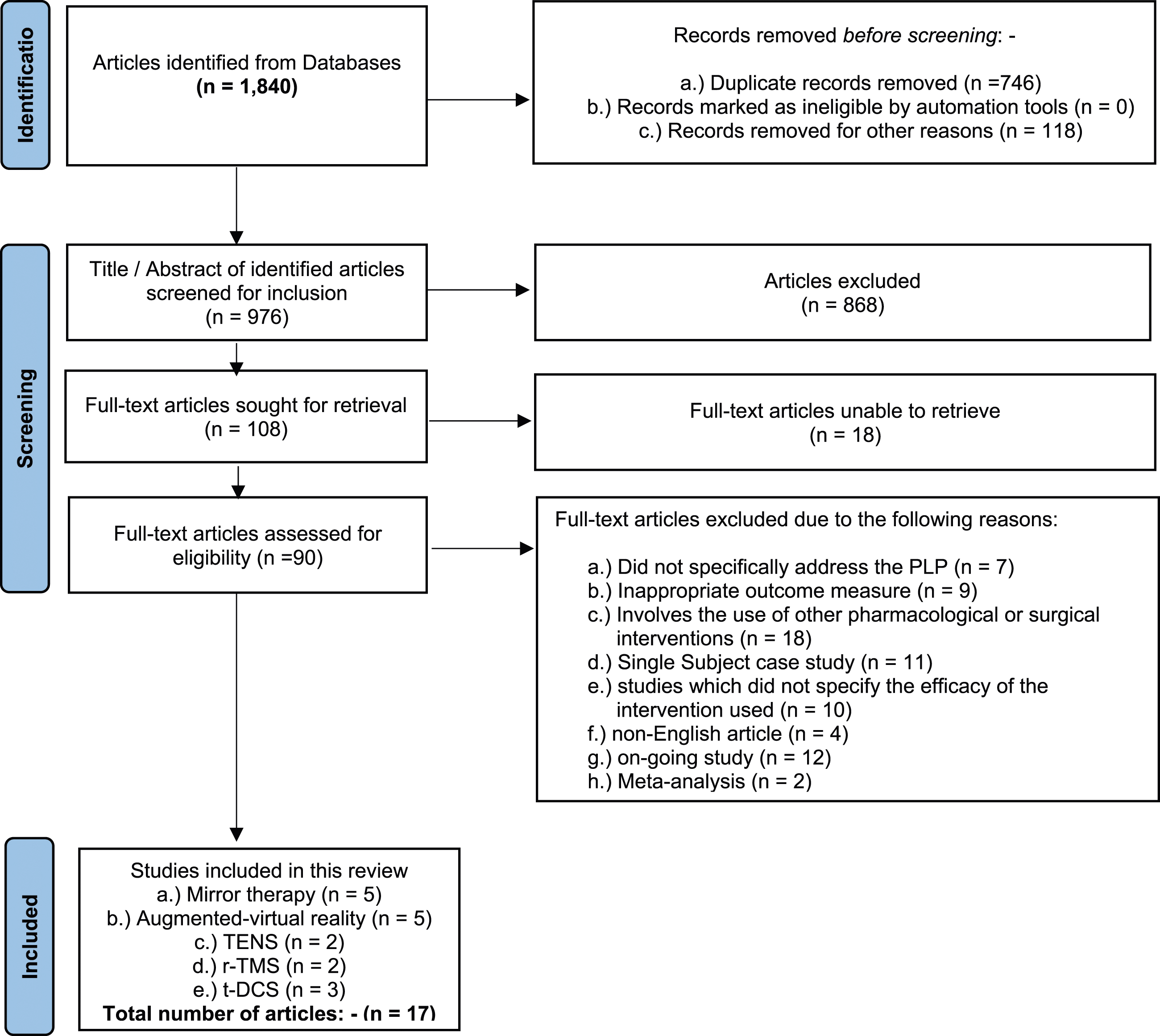
Effectiveness of non-pharmacologic, non-invasive therapeutic intervention in physiotherapy for the management of PLP
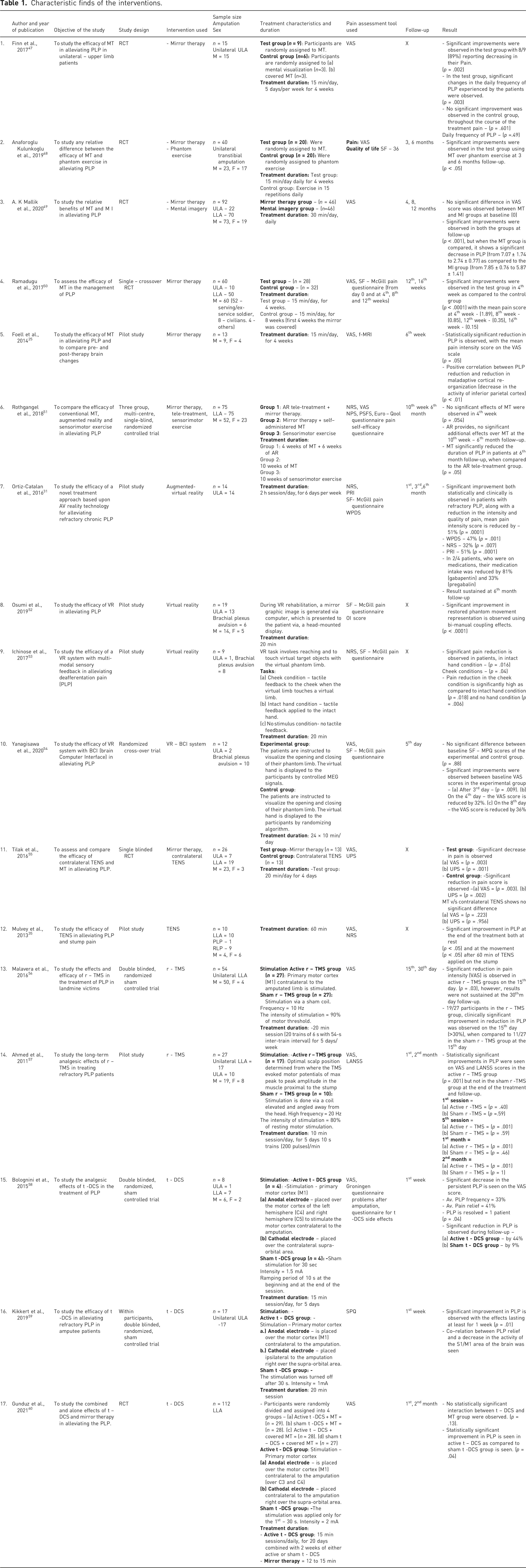
Characteristic finds of the interventions.
Mirror therapy
In our search, we yielded five different articles which investigated the efficacy of mirror therapy alone or in combination with other non-pharmacologic, non-invasive therapeutic interventions in physiotherapy as a potential treatment approach for the treatment of PLP in amputee patients, out of which 4 were RCTs and 1 was pilot study. Across all the studies included in this article, mirror therapy has shown a significant positive result in not only reducing the severity of PLP but to also in some studies, it is seen that it helps the patients to gain voluntary control of their phantom limb, thereby reducing the incidences of unpleasant phantom sensation such as the sensation of excruciating painful in-voluntary clenching spasms in their phantom hand accompanied by a sensation of the nail digging into their palm and so on.
Augmented-virtual reality
In our search, we yielded five different articles which investigated the efficacy of different treatment approaches utilizing augmented-virtual reality technology using various virtual reality implementations such as the creation of a virtual limb in the virtual space from the mirror image of the intact limb, controlling the movement of the virtual limb via electromagnetics sensors and myoelectric pattern recognition to create a more realistic immersive illusion of the phantom limb and so on for the treatment of PLP in amputee patients, out of which 2 were RCT and 3 were pilot studies. Results from these studies trends to indicate towards the use of augmented-virtual reality technology as a therapeutic intervention for the management of PLP over conventional non-pharmacologic, non-invasive therapeutic interventions such as mirror therapy with no adverse side effects. For instance, augmented-virtual reality has multiple advantages over traditional mirror therapy as it can create a more sophisticated and realistic immersive illusion of their amputated limb in the virtual world. Furthermore, it allows the user the direct control of motion and sense of agency of their phantom limb and using certain devices like myoelectric pattern recognition strapped around the stump, allowing accurate prediction of motor execution of the phantom limb as intended or desired by the patient. Thereby, it can be used in patients having bilateral amputation or even in patients having a prominent functional disability in the contralateral limb, in which the use of traditional mirror therapy for the treatment of PLP is proven to be ineffective and limited.
Transcutaneous electrical nerve stimulation
In our search, we yielded two different articles which investigated the efficacy of transcutaneous electrical nerve stimulation (TENS) alone or in combination with other non-pharmacologic, non-invasive therapeutic interventions in physiotherapy as a potential treatment approach for the treatment of PLP in amputee patients, out of which one is RCT and one pilot study. Across all the studies included in this article, TENS has shown a significant positive result in not only reducing the severity of PLP but also in reducing the severity of RLP. However, according to Cochrane reviews in 2010 and 2015, there was an insufficient and low level of evidence available to justify the efficacy of TENS in the management of PLP and stump pain. A need for a more randomized controlled trial with homogeneity in terms of devices used, the frequency of intervention, and the duration of treatment sessions is required in the future to confirm its positive outcome in patients.42,43
Repetitive transcranial magnetic stimulation/transcranial direct current stimulation
In our search, we yielded five different articles (RCTs) which investigated the efficacy of Repetitive Transcranial Magnetic Stimulation (r-TMS) and Transcranial Direct Current Stimulation (t-DCS) techniques. Results from these RCTs trend to indicate towards the use of r-TMS and t-DCS as therapeutic interventions in physiotherapy for the management of PLP in amputee patients and other neurological conditions in the future. However, due to the reason of lack of treatment protocol, being still new and in the research stage, these studies were underpowered, which not only reduces the significance of the finding but also its general applicability in treating PLP in post-amputee patients.
Quality assessment
Quality assessment - PEDro score.
Quality assessment - methodological index for non-randomized studies (MINORS) score.
Risk of bias
Risk of bias: Cochrane collaboration’s assessment tool.
Discussion
Studies involving available adjunct conventional and novel treatment techniques and approaches for the treatment of PLP in amputee patients and their efficacy are essential for any healthcare professionals and clinician, helping them to make a better decision regarding the use of adjunct therapeutic intervention to better serve their patients. Therefore, to obtain an estimate regarding the efficacy of available and novel adjunct physiotherapy interventions, we gathered and critically appraised all the available scientific literature, pilot studies, clinical trials, RCTs etc. on phantom limb, pain, mirror therapy, TENS, augmented and virtual reality, transcranial magnetic stimulation and transcranial direct current stimulation.
In light of the above-mentioned findings and evidence (Table 1), it became evident that the use of adjunct conventional and novel, non-pharmacologic, non-invasive therapeutic interventions such as mirror therapy, augmented-virtual reality, TENS, r-TMS and t-DCS can be implemented to increase the treatment outcomes. However, the RCTs conducted in the past 12 years has failed to demonstrate any consistent evidence for a given intervention, to come to a majority consensus as to which intervention better assess all the mechanism of PLP (peripheral and central) in amputee patients, thereby alleviating the problem of PLP in post-amputee patients.
Mirror therapy, since its discovery in 1996, has emerged as a revolutionary therapy strategy for the management of PLP in amputee patients. However, even though a large number of research has been conducted and published, there is still no standardized treatment protocol and no significant change in its methodology.
Augmented-virtual reality: In spite of the growing number of research surrounding the future use of augmented-virtual reality and artificial intelligence (AI) in the healthcare system, research, surgery and so on. The use of augmented-virtual reality has recently emerged as a prominent novel non-invasive, painless, simple, cost-friendly method in treatment of PLP in amputee patients, demonstrating encouraging and significant results in reducing the severity and frequency of PLP. However, being still new, there are multiple technological limitations and only a few numbers of randomized controlled trials and studies with high level of evidence were available advocating its use for the treatment of PLP in amputee patients. Therefore, more randomized double blinded controlled trials are needed using a large sample size and with homogeneity in terms of devices used, the frequency of intervention and duration of treatment sessions in the future in order to develop more novel treatment approaches and methods to validate the conventional methods of treatments.
Although, the main focus of this article is to determine the efficacy of adjunct physiotherapy interventions in reducing the severity of PLP, but due to the reason of so close association between RLP and PLP, it became inevitable to talk about PLP without considering RLP. It is seen that the use of mirror therapy and augmented-virtual reality has no significant impact in reducing the severity of RLP, despite reducing the PLP, which not only indicate towards the lack of underlying knowledge of the mechanism of PLP and its action but also towards the high risk of bias in the studies. For instance, studies involving the use of mirror therapy and augmented-virtual reality are particularly more susceptible to high risk of bias as they are often designed to be more engaging for the patients and due to the difficulties in the blinding of the participants and personnel associated with the trial, unlike the studies which involves pharmacological treatment or medication in the general population because they are specifically designed and controlled to reduce the risk of bias by conducting a placebo-controlled double blinded randomized controlled trials.
Transcutaneous Electrical Nerve Stimulation (TENS) is one of the most commonly used low-frequency modality (1 Hz to 100 Hz), a non-invasive, safe, easy to administer, analgesic technique used by physical therapists and clinicians around the world. Despite the overall evidence available about the efficacy of TENS in reducing the severity of chronic pain in patients, the evidence regarding the efficacy of TENS in alleviating the problem of PLP in amputee patients is extremely limited. For instance, in our search, we yielded only one RCT article about the efficacy of TENS in reducing PLP. One of the major challenges associated with the use of TENS in amputee patients is the safety issues and lack of standardized treatment protocol. For example, in many studies, it is seen that the use of TENS on the stump can not only intensify the pain intensity of RLP but also can cause damage to the integrity of the stump.
Repetitive Transcranial Magnetic Stimulation (r-TMS)/Transcranial Direct Current Stimulation(t-DCS): Both are neuromodulation techniques that generally work upon the principle of spontaneous neural activity of somatosensory and motor cortex of the brain which can mediate the reversal of maladaptive cortical re-organization of the brain to the baseline before the amputation, thereby alleviating the problem of PLP in the amputee patients. However, one of the major challenges associated with both r-TMS and t-DCS is that it requires specialized equipment and skilled technician to operate, and due to the lack of RCTs involving neuroimaging of the brain (i.e. f-MRI) and how r-TMS and t-DCS assess the mechanism to reduce PLP in amputee patients. Although both of these techniques have shown promising results across the studies, but due to the reason of lack of treatment protocol, being still new and in the research stage, further studies in the future may be required with homogeneity in terms of devices used, the frequency of intervention and duration of treatment sessions to confirm its positive outcome in patients.
Preventive measures
Use of prosthesis
The maladaptive cortical re-organization of the somatosensory and motor cortex of the brain in amputee patients was estimated to be a major contributor to PLP. According to a recent research, it is seen that the patients who use prostheses, especially myoelectric prosthetics with somatosensory feedback on a daily basis, have a lower cumulative level of PLP as well as a lesser degree of cortical re-organization than patients who have never used prosthesis, indicating a negative correlation between prosthesis usage and cortical re-organization and PLP perceived by the patients. For instance, in a comparative study conducted by Dietrich et al., 2018, in 14 amputee patients with below-the-knee amputation, to compare the efficacy of prosthesis with somatosensory feedback to reduce the severity of PLP, they found that the majority of the patients (9/14) report a significant decrease in their PLP, along with an overall increase in the functionality of the prosthetic. 44 Thus, the use of myoelectric or cosmetic prosthesis might be one way to influence or to prevent the prevalence of PLP in amputee patients. However, one of the major challenges associated with the prosthesis is its cost. In India, the majority of the prosthesis are imported from other countries, and each and every prosthetic is made especially for the patients to better suit their condition, functionality and amputation. These cause the price of the prosthesis to skyrocket; thereby, a majority of the patients are unable to buy prostheses. However, more rigorous randomized controlled trials will be required in the future using large sample sizes and with homogeneity in terms of devices used to confirm its positive outcomes.
Pre-amputation pain
In a study conducted by Katz and Melzack, in 1990, it is seen that patients who complain of intense pain in their limb prior to amputation surgery are more prone to develop PLP as compared to other amputee patients, indicating that there is some sort of mechanism that may revive the repressed somatosensory pain memories after amputation surgery in patients that leads to PLP. For instance, patients who have any deformity in their limb prior to amputation are more likely to complain of the deformity being carried over into their ‘Phantom Limb’ after the amputation. 45 This simple observation by Katz and Melzack can be used in the treatment of PLP in post-amputee patients by using the pre-emptive analgesia method (i.e. treatment or administration of analgesic medication prior to a surgical procedure). The pre-emptive analgesic method can be used to prevent PLP in amputees by early intervention by using epidural anaesthesia like ketamine and calcitonin which have shown promising results in reducing the intensity and prevalence of PLP in amputee patients. 46 However, the number of studies regarding the efficacy of the pre-emptive analgesia method to prevent the prevalence of PLP in amputee patients is still limited. Therefore, more rigorous randomized controlled trials will be required in the future using a large sample size to confirm its positive outcomes.
Conclusion
Physiotherapy interventions with advanced modalities and exercises can be used in conjunction with pharmacological and surgical treatment to improve the overall effectiveness of the treatment and to reduce the severity of phantom limb pain in post-amputee patients. However, the RCTs conducted in the past 12 years has failed to demonstrate any consistent evidence for a given intervention, which makes it even more difficult to reach to a majority consensus as to which intervention better assess all the mechanism of PLP (peripheral and central), thereby alleviating the problem of PLP in post-amputee patients. However, novel physiotherapy interventions like augmented-virtual reality, repetitive transcranial magnetic stimulation (r-TCS) and transcranial direct current stimulation (t-DCS) techniques do show promising results as a potential non-invasive, non-pharmacological, therapeutic intervention for the treatment of neurological and musculoskeletal disorders. In addition, as the prevalence of amputation is increasing exponentially, further research on phantom limb pain, its neural basis and its mechanism is becoming an inevitable task for scientists, researchers, neurologists, surgeons and clinicians around the world. Therefore, a more rigorous randomized controlled trials and research will be required in the future to reach a conclusion.
The results of this review do not support the efficacy or promote any single technique or approach to be superior over another.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
AKS: The guarantor is the person willing to take full responsibility for the article, including for the accuracy and appropriateness of the reference list. This will often be the most senior member of the research group and is commonly also the author for correspondence. Please use initials only: BJP operates a double-blind peer review process so full names of authors should not be listed on this form.
Contributorship
All the authors listed have contributed significantly, directly, and intellectually to the work of this review, whether that is in the analysis of the data, interpretation, or reviewing the article, and have given their final consent for it to be published.
Trial registration
PROSPERO (International Prospective Register of Systematic Reviews) under the registration number CRD42023404058.
