Abstract
Background:
Prescribing opioids upon discharge after surgery is common practice; however, there are many inherent risks including dependency, diversion, and medical complications. Our prospective pre- and post-intervention study investigates the effect of a standardized analgesic prescription on the quantity of opioids prescribed and patients' level of pain and satisfaction with pain control in the early post-operative period.
Methods:
With the implementation of an electronic medical record, a standardized prescription was built employing multimodal analgesia and a stepwise approach to analgesics based on level of pain. Patients received an education handout pre-operatively explaining the prescription. Consecutive patients over a three-month period undergoing elective spine surgery as day or overnight stay cases who received usual care were compared to a similar cohort who received the standardized prescription and education. Patient satisfaction with post-operative pain control, post-operative pain scores, number of refills required, and opioids prescribed in oral morphine equivalents (OMEs) were compared before and after implementation of the standardized analgesic prescription.
Results:
Twenty-six patients received usual care (Control group) and 26 patients received the standardized prescription and education handout (Intervention group). There were significantly fewer OMEs prescribed in the Intervention group compared to the Control group. There was no difference between groups in: patient post-operative pain intensity score, post-operative satisfaction score, or number of refills required.
Conclusions:
This study demonstrates that a standardized prescription consisting of an appropriate amount of opioid and non-opioid analgesics is effective in reducing the OMEs prescribed post-operatively in elective spine surgery procedures, without compromising patient pain control or satisfaction or increasing the number of refills required.
Background
Over the last decade, the rise of opioid-related morbidity and mortality has prompted the development of an international multidisciplinary consensus statement in peri-operative harm reduction strategies to guide clinicians’ prescription habits. 1 However, despite recommendations by professional bodies intended to guide prescription opioid analgesia (POA) within the surgical specialties,1–11 the paucity of evidence-based guidelines for POA and patient satisfaction in surgical patients has led to wide variability in discharge prescriptions after spine surgeries. In 2016, Ontario (Canada) surgeons were responsible for 60% of the total POAs for individuals who had no POA in the previous 6 months. 14 Eight percent of these POAs were for high doses, defined as >90mg oral morphine equivalents (OME) per day, while 25% were for medium doses, defined as 51 mg–90 mg OME per day, and 11.5% of these scripts were given for a duration longer than seven days. 14 Several studies have described a positive correlation between the amount and duration of opioids prescribed upon discharge after surgery and the risk of chronic use.4,6–11
However, surgeons must also balance the risks of insufficient peri-operative analgesia including increased hospital length of stay (LOS), patient dissatisfaction, the development of chronic pain, increased time to mobilization, and possibly worse functional outcomes.5,6–15 These factors, and the bolstering of prescriptions to avoid the need for refills, affect how POAs are prescribed. 11
Reconciling adequate pain control peri-operatively, while mitigating the risks related to POAs, is pivotal to successful surgical outcomes and has been an area of active research for patients undergoing surgery.3,13,16–18 A strategy shown to be effective in a population of general surgery patients undergoing day surgery procedures is to standardize the post-operative analgesic prescription and provide patient education detailing when to fill the opioid and non-opioid components. 19 The aim of this study was to determine if the use of a standardized prescription and patient education could reduce the prescribed OME for patients undergoing outpatient spinal surgery without an adverse effect on post-operative pain control or patient satisfaction, or an increase in refills required.
Methods
Setting
At a single, tertiary-care academic health center, the orthopedic spine group consisting of four surgeons in collaboration with anesthesia colleagues developed a post-surgical multimodal analgesia protocol for all patients undergoing routine elective lumbar spine procedures without admission to hospital. Spine surgery volume is approximately 1000 cases per year at this center, including elective and urgent procedures. The institutional Research Ethics Board (REB) reviewed the study and granted REB review exemption due to the study qualifying as a continuous quality improvement (CQI) project.
The Control group had surgery between March and May 2019 and was prescribed analgesics post-operatively according to the treating surgeon’s discretion and individual practice patterns. A new electronic medical record (EMR) was implemented 1 June 2019, which facilitated the incorporation of standardized prescriptions through the creation of standardized order panels. The Intervention group had surgery between October 2019 and February 2020.
Patient inclusion
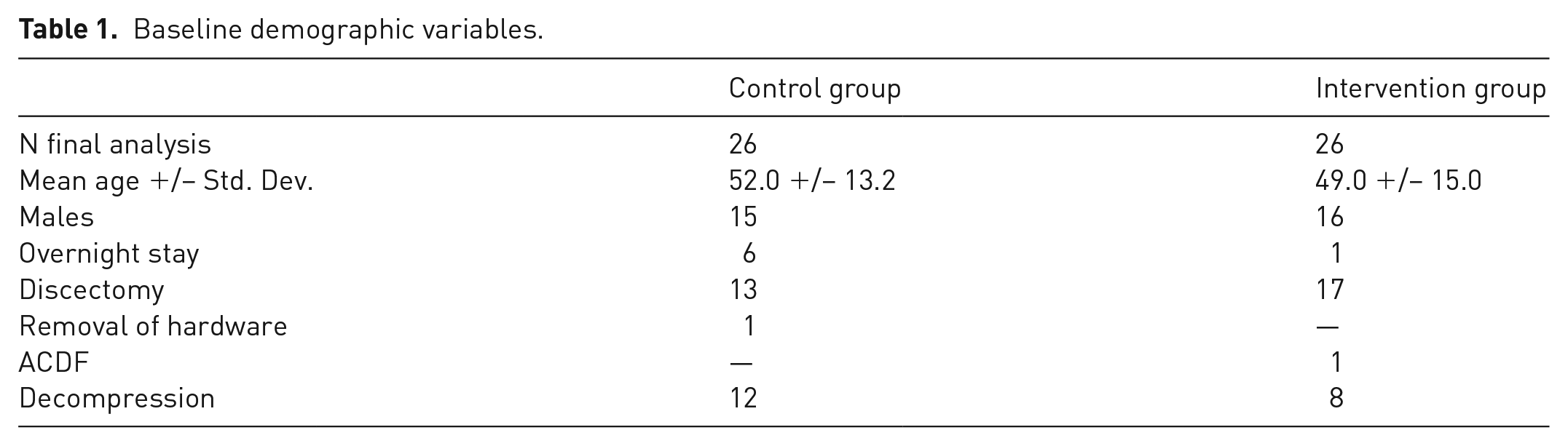
Consecutive patients over the age of 18 undergoing elective spine surgery identified as a day case or overnight stay (leaving hospital at 0630 h post-operative day one) by the treating surgeon were included. The types of surgical procedures were as follows and were routinely performed as day/overnight stay cases: lumbar discectomy, one- to two-level laminectomy and anterior cervical discectomy and fusion (ACDF), and removal of hardware (Table 1). Patients on high dose opioids (>90mg OME per day) prior to surgery were excluded.
Baseline demographic variables.
Intervention
A standardized prescription was developed through the consensus of a multidisciplinary peri-operative team, which included staff spine surgeons and anesthesiologists (Figure 1). It consisted of non-opioid analgesia, an adjuvant, a weak opioid (tramadol), a strong opioid (hydromorphone), and a stool softener. 19 On the protocol prescription, a standardized amount (12 tablets) and no refills were given for each of the weak and the strong opioids, for a total of 24 tablets. The prescriber chose the higher or lower dose for pregabalin and celecoxib based on age, renal function, and medical comorbidities. If a patient was on pregabalin or gabapentin pre-operatively, the patient was instructed to continue their previous dose of this medication and pregabalin was excluded from the post-operative prescription.
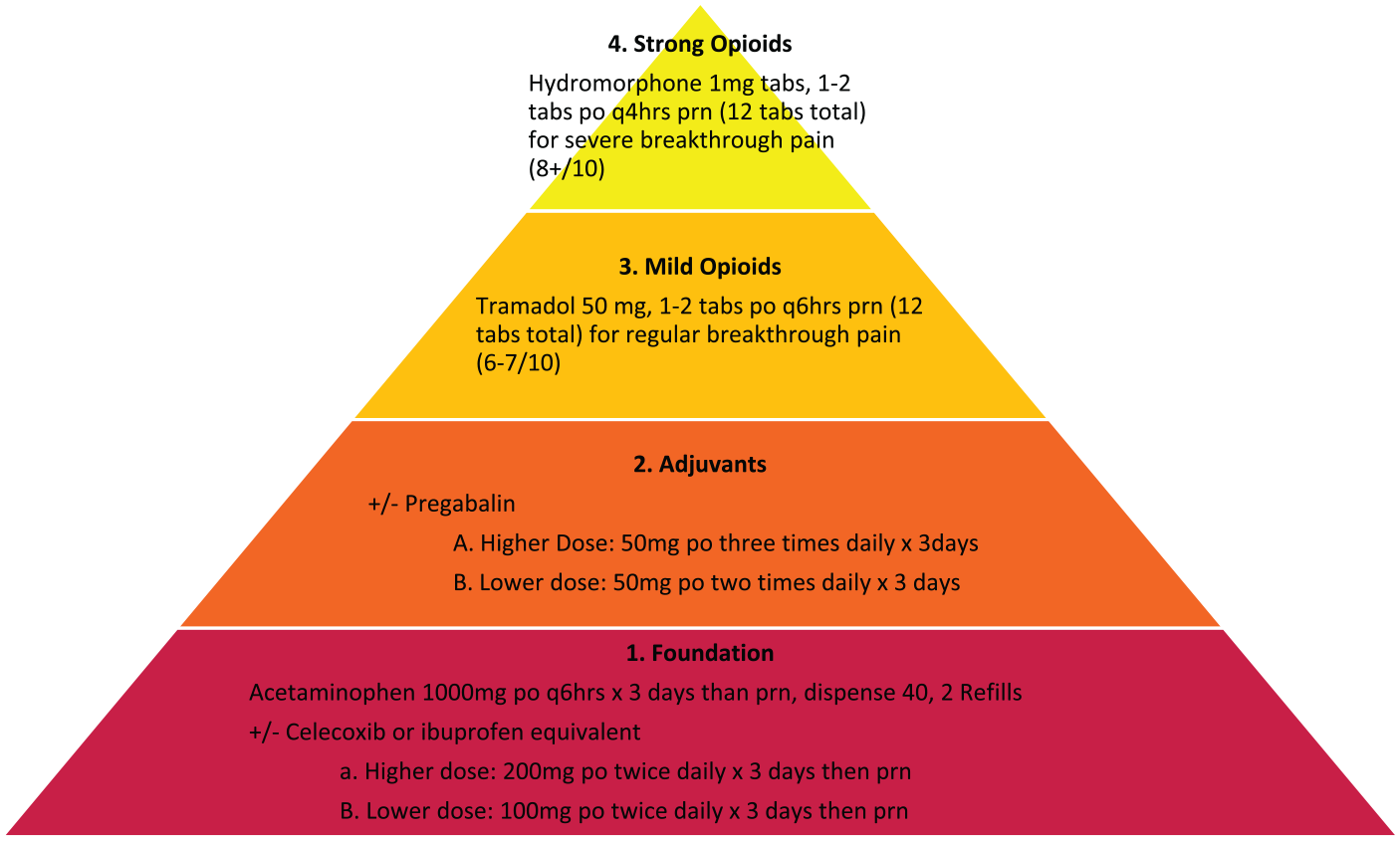
Multimodal analgesia pyramid used for the standardized prescription.
The patient education sheet included a pain intensity grading scale, based on the Numeric Rating Scale (NRS), to guide patients as to when it would be appropriate to use which components of their analgesia prescription (Appendix 1). The 11-point NRS was utilized for its high validity, reliability, and responsiveness for assessing pain in adults. 20 At their pre-operative appointment and again post-operatively, the education sheet and NRS were explained to patients as follows: zero represents no pain, while 10 represents the worst pain ever experienced.
Outcome measures
The primary outcome measure was OMEs prescribed. Secondary outcomes measures were patient-reported post-operative pain scores, satisfaction with pain control, and number of refills required in the early post-operative period.
Chart review was performed to identify and record every post-operative opioid prescription provided. The total prescribed was then converted into total OME as it is the most international opioid standard against which opioids can be compared in terms of potency and toxicity. 21 The Center of Medicaire and Medicaire service and the 2018 CDC Opioid NDC and Oral MME Conversion was utilized for tramadol (0.1ME) and hydromorphone (4ME).23–25
Secondary outcome measures were obtained through a survey administered at patients’ first post-operative visit, which occurred on post-operative day 10–14. The survey captured the following self-reported information: pre-surgical use of non-opioid and opioid analgesia, average NRS pain intensity scale in the week following surgery, satisfaction with post-operative pain control (scale 0–10, 0 = not at all satisfied, and 10 = extremely satisfied), if the patient filled the opioid prescription (yes/no), and the need for refill in opioids (yes/no). 18
Statistics
Data was entered into an Excel spreadsheet and analyzed using SAS 9.4 Statistical Package. Descriptive statistics were used to describe the group. Average doses of medications were calculated only for those that were prescribed the medication. Skewness (<1.0) and Shapiro-Wilk (p > 0.05) tests were used to examine for normal distribution. Between group comparisons was made using Student’s t-test for continuous parametric data, Wilcoxon for non-parametric (non-normally distributed) data and Pearson chi-square for dichotomous data. For dichotomous variables with less than 5 outcomes in one group, a Fisher exact test was performed. Ad hoc power analyses were not done because this was a pilot study conducted to assess feasibility. Intent-to-treat analysis was used, and all 52 patients were enrolled and included in this analysis.
Results
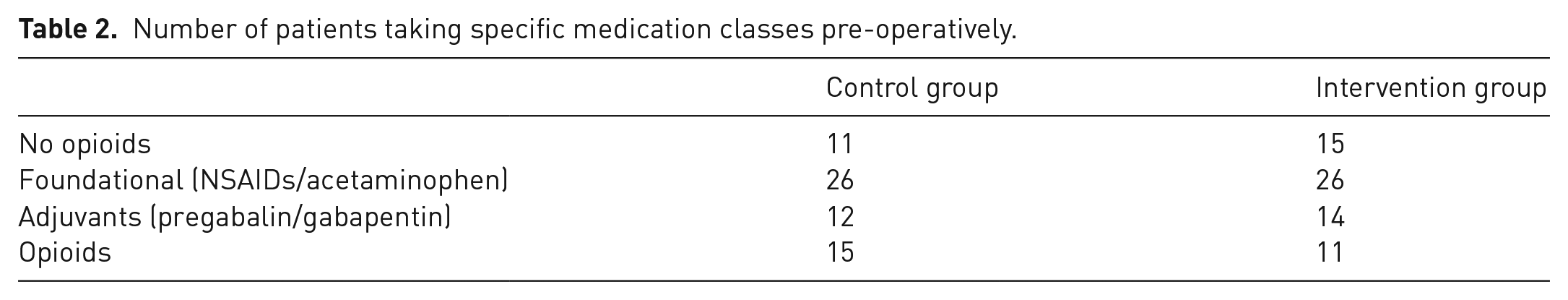
Fifty-two patients were enrolled and included in the analysis; their baseline demographics and pre-operative analgesic use are summarized in Tables 1 and 2, respectively.
Number of patients taking specific medication classes pre-operatively.
The Intervention group had a significantly lower amount of total OMEs prescribed than the Control group (Table 3). Compliance in using the standardized prescription by the prescribers was 92.3% (24/26 patients). The two individuals in the Intervention group who were not given the standardized prescription were prescribed 240 and 580 OMEs instead of a total of 108 OMEs (the amount prescribed with the standardized prescription). Post-operatively, there were no significant differences in any of the following outcomes: pain intensity, satisfaction with pain control, or need for a prescription refill (Table 4).
Oral morphine equivalents (OME) prescribed post-operatively.

Post-operative patient outcomes related to opioid prescription.
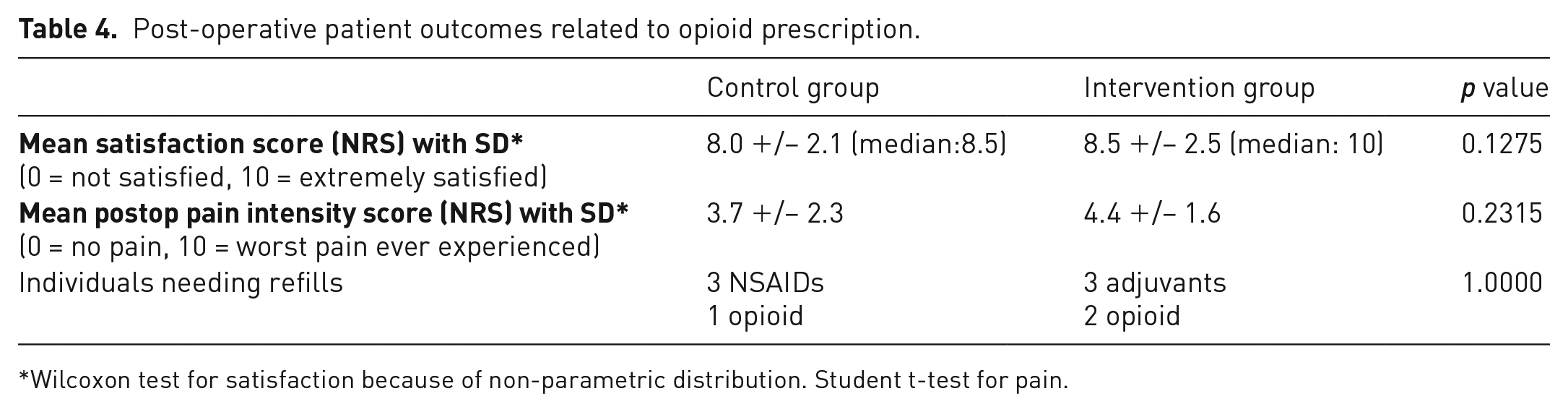
Wilcoxon test for satisfaction because of non-parametric distribution. Student t-test for pain.
Discussion
Our study demonstrated that the implementation of a standardized prescription in combination with patient education reduced the OME prescribed after routine spine surgery cases and provided similar patient satisfaction score as the pre-intervention usual care. The prescription provided to patients along with an explanation regarding how to use the prescription may have increased the use of non-opioid analgesia. Our findings are in keeping with other studies that demonstrate patient education in combination with multimodal analgesia leads to a significant reduction in filling and taking opioids without a decrease in patient pain management satisfaction.7,16–19
Furthermore, surgeons’ concern of increased requests for analgesic prescription refills in the Intervention group was not seen in this study (p = 1.0). These results support the literature which has failed to show any correlation with prescription strength and number of dispensed tablets with refill requests. In fact, the greater the amount of OMEs prescribed and the lack of instruction for disposal drive opioid diversion.13,18
Our study did not demonstrate perfect compliance with the standardized prescription as two patients in the Intervention group failed to receive the standardized prescription. This was due to two visiting fellow surgeons who were not familiar with our intervention. Therefore, the intent-to-treat analysis effectively increased the heterogeneity of OMEs prescribed in the Intervention group in the study. Another limitation of the study is that the number of patients included in this study was small and the sample size was one of convenience as we sought to capitalize on the new EMR go-live date in implementing the new standardized prescription. Similarly, the actual number of OME taken by patients in both study arms is unknown, and prescribed OME was used as a proxy as it did not rely on patient self-report. Another limitation is that post-operative pain scores have limited value in the absence of pre-operative pain scores to use as a reference, which were not available in this study. Furthermore, non-specific post-operative pain scores may be less useful than those associated with movement and rest. Future research should include disease-specific patient-reported outcome measures and focus on analgesia as it relates to function due to the limitations in, and risks of, unidimensional pain assessment tools such as the NRS.26,27
This prospective cohort study is limited in its external validity as the intervention was only studied in a small subset of patients seen by spine surgeons. However, the results are comparable with a similar protocol in general surgery procedures, such as laparoscopic cholecystectomy and inguinal hernia repair, 22 and as such it is reasonable to assume it may be applicable to other relatively minor surgical procedures in other specialties.
Conclusions
Although utilized in other surgical specialties, the application of a standardized multimodal pain analgesia and patient education protocol to minimize the quantity of opioids prescribed is a novel concept for elective same-day or overnight stay spine surgical patients. Our study demonstrated this intervention led to a decrease in OMEs prescribed post-operatively, the same patient satisfaction scores, and no increase in analgesic prescription refill requests. Opportunities exist to further this research with standardized prescription protocols in other day surgical procedures, and the integration of validated disease-specific functional outcome scores, which may lead to a significant decrease in opioid prescription rates within the surgical community.
Footnotes
Appendix 1
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
