Abstract
Background:
Although the value of fast diagnostic protocols in suspected acute coronary syndrome has been validated, there is insufficient real world evidence including patients with lower pre-test probability, atypical symptoms and confounding comorbidities. The feasibility, efficacy and safety of European Society of Cardiology (ESC) 0/1 and 0/3-hour algorithms using high-sensitivity troponin T were evaluated in a consecutive cohort with suspected acute coronary syndrome.
Methods:
During 12 months, 2525 eligible patients were enrolled. In a pre-implementation period of 6 months, the prevalence of protocols, disposition, lengths of emergency department stay and treatments were registered. Implementation of the 0/1-hour protocol was monitored for another 6 months. Primary endpoints comprised the change of diagnostic protocols and 30-day mortality after direct discharge from the emergency department.
Results:
Use of the ESC 0/1-hour algorithm increased by 270% at the cost of the standard 0/3-hour protocol. After rule-out (1588 patients), 1309 patients (76.1%) were discharged directly from the emergency department, with an all-cause mortality of 0.08% at 30 days (one death due to lung cancer). Median lengths of stay were 2.9 (1.9–3.8) and 3.2 (2.7–4.4) hours using a single high-sensitivity troponin T below the limit of detection (5 ng/L) at presentation and the ESC 0/1-hour algorithm, respectively, as compared to 5.3 (4.7–6.5) hours using the ESC 0/3-hour rule-out protocol (P<0.001). Discharge rates increased from 53.9% to 62.8% (P<0.001), without excessive use of diagnostic resources within 30 days.
Conclusion:
Implementation of the ESC 0/1-hour algorithm is feasible and safe, is associated with shorter emergency department stay than the ESC 0/3-hour protocol, and an increase in discharge rates.
Trial registration:
ClinicalTrials.gov, Unique identifier: NCT03111862.
Introduction
Accelerated 0/1 or 0/2-hour diagnostic protocols for the diagnosis of non-ST-segment elevation acute myocardial infarction (NSTEMI) are being recommended by the 2015 European Society of Cardiology (ESC) guidelines 1 as an alternative to the established ESC 0/3-hour protocol. Although accelerated protocols have been validated in numerous observational cohorts,2–11 implementation is very low worldwide. 12 Reasons for the limited use of fast protocols are multifactorial and include fear of litigation after missed myocardial infarction (MI) in early presenters, the presence of comorbidities that were excluded or were under-represented in observational studies and the limited clinical experience in patients presenting with symptoms other than chest pain or angina.13–16
Another issue is the lack of a clinically validated acceptable event rate after discharge. All-cause mortality rates between 0.1% and 2% have been suggested to be acceptable based on a survey among physicians who were asked to give their expectation on 30-day mortality rates. 17 In observational studies physicians were usually unaware of investigational biomarker results and protocols, and patients were discharged at the discretion of the attending physician. Adding to this dilemma, there are only few randomised biomarker-based trials evaluating the safety of discharge in low-risk patients, either using high-sensitivity troponin (hsTn) assays in combination with validated clinical scores,18,19 a dual biomarker strategy combining copeptin with cardiac troponin, 8 or an accelerated diagnostic protocol using hsTn I measurements 2 hours apart, together with electrocardiography (ECG) and either the thrombolysis in myocardial infarction (TIMI)9,20 or the emergency department assessment of chest pain (EDACS) score. 20 Moreover, incremental evidence comes from a large pre and post-implementation study on 31,332 patients providing findings on the lower length of emergency department (ED) stay and increased rates of discharge within 6 hours, without an adverse event when clinical pathways were correctly used. 21
In the light of sparse real world evidence, our prospective pre/post-implementation study sought to evaluate the feasibility, efficacy and safety of ESC recommended fast diagnostic protocols using hsTnT in a consecutive all-comer cohort with suspected acute coronary syndrome (ACS) based on a broad spectrum of symptoms.
Methods
In this prospective single centre study at Heidelberg University Hospital, we screened all consecutive patients with suspected ACS between 1 July 2016 and 30 June 2017. In this period, 7668 patients presented to the ED. Patients were managed in a chest pain unit (CPU), which represents a specialised ED that is led by a cardiologist and requires certification by the German Cardiac Society (Deutsche Gesellschaft für Kardiologie; DGK). In Germany, more than 320 certified CPUs are distributed across the country and represent the preferred facilities for the evaluation of patients with suspected ACS. The median number of patients per day (25th percentile–75th percentile) was 20 (17–24) patients. The ED of the department of cardiology is part of the internal medicine ED and follows the CPU quality criteria that are audited by the German Cardiac Society. The team consists of experienced resident physicians in training for cardiology working a three-shift schedule on weekdays (day shift 2, swing shift 1 and night shift 1 physician) and a two-shift schedule during weekends (day shift 1 and night shift 1 physician). The nursing team consists of experienced nurses working in a three-shift system (day shift 3, swing shift 3, night shift 2 nurses), with a ratio of one nurse per five patients. The ED is under the permanent supervision of a senior cardiologist who is responsible for the decision to admit or discharge, and for the indication and timing of an invasive strategy. There is unlimited access to coronary angiography or other diagnostic resources as per the required criteria for certification of a CPU. 22 Patient disposition, times and treatments were collected in a 6-month pre-implementation period followed by the implementation on 1 January 2017 that encouraged the use of the ESC 0/1-hour algorithm as the primary diagnostic strategy, and subsequently a post-implementation period of another 6 months to demonstrate changes.
Exclusion criteria comprised the following: (a) repeated presentations beyond the index admission (‘frequent flyer’); (b) patients referred from other hospitals for early or primary percutaneous coronary intervention (PCI) without receiving a standard diagnostic work-up; (c) diagnostic set of hsTnT samples not available (e.g. missing initial or consecutive blood sample); (d) patients with ST-segment elevation myocardial infarction (STEMI) were registered but were excluded for this analysis. A consort diagram illustrates the screening process (Figure 1).
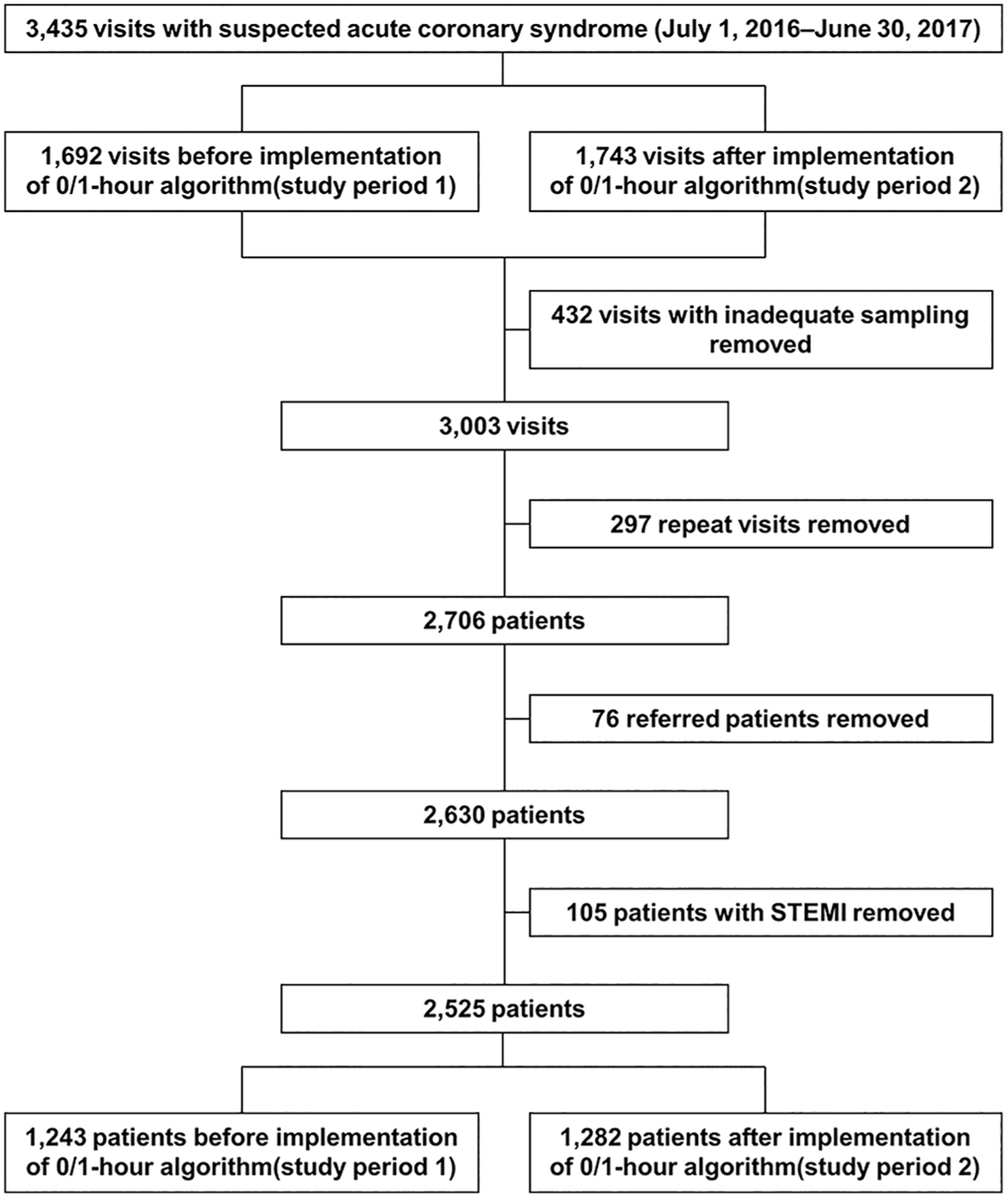
Standards for reporting diagnostic accuracy studies statement (STARD) patient inclusion flow diagram.
Patients qualified for enrolment with initial presentation of clinically suspected ACS, based on a broad spectrum of symptoms including atypical symptoms and dyspnoea. Patients on chronic haemodialysis were not included. Previously published findings 23 in symptomatic patients with atrioventricular nodal re-entrant tachycardia demonstrate ST-segment depressions and relevant troponin kinetics rendering the unequivocal differentiation between type 1 MI, type 2 MI, MI with non-obstructive coronary arteries, or acute myocardial injury impossible. Therefore, we decided not to include these patients. In the present study population, there are no patients with the following initial presentation: acute heart failure due to already known non-coronary heart disease without suspected ACS; confirmed primary pulmonary disease without suspected ACS; traumatic chest pain with preceded thorax injury without suspected ACS. Patients were not excluded for severe chronic kidney disease, older age, chronic heart failure, or atrial fibrillation. Patients were not included in the case of inappropriate command of the English/German language or permanent residence in a foreign country.
Reliable electronic time stamps were available for the time of arrival, referral and discharge, blood draws (between specimen intervals) and reporting of blood test results (‘turn-around-times’), and all diagnostic and therapeutic interventions. Acute MI was diagnosed in-hospital by treating clinicians based on all clinical information, using the diagnostic criteria of the 3rd universal MI definition. 24 Patients were categorised using the validated biomarker criteria for classification into rule-out, observe or rule-in as proposed by the 2015 ESC guidelines on non-ST elevation (NSTE)-ACS. 1 Levels of cardiac troponin were measured at presentation, after 1 or 3 hours and thereafter as long as clinically indicated. A small proportion of patients were categorised into ruled-out using hsTnT and copeptin at presentation, with biomarker cut-offs based on the BIC-8 trial. 8 HsTnT was measured on Cobas E 411 (Roche Diagnostics Ltd., Rotkreuz, Switzerland) and copeptin was measured on Kryptor (Thermo Fisher Scientific, BRAHMS GmbH, Hennigsdorf, Germany).
On 1 January 2017, the ESC 0/1-hour algorithm was officially implemented. Prior to this date, every staff member in the ED (nurses and physicians) had received training for the use and interpretation of the ESC 0/1-hour algorithm using hsTnT from 1 January 2017. This included formal education, posters and bedside cards based on the algorithm shown in Figure 3 of the 2015 ESC guidelines on NSTE-ACS. 1 The training was also implemented in the initial training for newly rotating personnel.
All patients underwent a clinical assessment that included medical history, physical examination, 12-lead ECG, continuous ECG monitoring, pulse oximetry and standard blood tests. Results were reported on the electronic patient record and were communicated to the clinicians responsible for patient care. Patients received treatment at the discretion of the attending physician, and all decisions to admit or discharge, or on the need and timing for invasive coronary angiography were made based on available information during the ED stay. The standard 12-lead ECG included routinely precordial leads V7–V9. The decision to discharge comprised clinical judgement from individual risk variables, or the GRACE score 25 that was generated by an electronic calculator embedded into the electronic file, and was thus accessible for all physicians.
Adjudication of final diagnoses in the ED was made prospectively in clinical routine by attending physicians and responsible cardiologists on duty, while confirmation of ED diagnoses for research purposes was done retrospectively by two cardiologists and a third cardiologist in case of discordance.
Definition of fast protocols
According to the ESC 0/1-hour protocol, patients with a 0-hour value below the limit of detection (LoD) (hsTnT <5 ng/L) and interval from the last chest pain episode exceeding 3 hours were classified as rule-out. Patients with a 0-hour value of hsTnT of 5 ng/L or greater and less than 12 ng/L and difference between the 0-hour and 1-hour value of hsTnT (Δ0–1 h) less than 3 ng/L were classified as rule-out. Patients with a 0-hour value of hsTnT of 52 ng/L or greater or difference between the 0-hour and 1-hour value of hsTnT (Δ0–1 h) of 5 ng/L or greater were classified as rule-in. Patients qualified for the ESC 0/1-hour protocol if the time window between the first and the second blood draw was between 30 and 90 minutes. According to the ESC 0/3-hour protocol, patients with a 0-hour value at or below the upper limit of normal (ULN) (hsTnT ≤14 ng/L) and interval from the last chest pain episode exceeding 6 hours were classified as rule-out. Patients with a 0-hour value at or below the ULN (hsTnT ≤14 ng/L) and absolute concentration change between the 0-hour and 3-hour value of hsTnT (Δ0–3 h) of 7 ng/L or less (defined as 50% of the ULN) were classified as rule-out. If the 0-hour value exceeded the ULN, patients with a relative concentration change between the 0-hour and 3-hour value of hsTnT (Δ0–3 h) of less than 20% of the 0-hour value were classified as rule-out. Patients qualified for the ESC 0/3-hour protocol if the second blood draw was between 150 and 210 minutes after the initial blood sample. Per protocol, blood samples for the baseline hsTnT value had to be obtained within 45 minutes according to laboratory time stamps and the follow-up specimens had to be obtained 1 or 3 hours ± 30 minutes after the initial specimen. Time sampling intervals between 90 and 150 minutes and blood draws beyond 210 minutes were summarised as ‘other’ protocols. These patients were commonly diagnosed using the criteria of either the ESC 0/1 or the ESC 0/3-hour protocol, whatever protocol came closer.
According to the 2015 ESC guidelines for the management of NSTE-ACS, 1 repeat blood sampling was performed at the discretion of the treating physician, based on clinical assessment. The final diagnosis before admission or discharge was based on the complete clinical information and all hsTnT measurements.
Endpoints
The primary endpoints were: (a) the temporal change of implementation rate of the 0/1-hour algorithm and (b) 30-day all-cause mortality among patients discharged from the ED after rule-out. Secondary endpoints included: (a) the safety of discharge after 12 months; (b) the temporal trends of lengths of stay in the ED; and (c) the change of discharge rates before and after implementation. Additional endpoints included the prognostic role of baseline hsTnT concentrations on outcomes, as well as the impact of the severity of ED crowding on the length of ED stay and outcomes.
Follow-up was accomplished using telephone, questionnaire, patient’s hospital notes, the family physician’s records and the municipal registry on vital status. The study was approved by the ethics committee of the University of Heidelberg, and performed in accordance with the Declaration of Helsinki. Informed consent of the individual patients was not required.
Statistical analysis
Continuous variables were tested for normal distribution and were presented either as means with 95% confidence intervals, or as medians with 25th/75th percentiles (interquartile range). The normality of data distribution was assessed by the Kolmogorov–Smirnov test. Groups were compared using the χ2 test for categorical variables and the Mann–Whitney U-test for continuous variables. Absolute changes between baseline and follow-up samples were calculated by Ct2−Ct1 (where C is troponin concentration and t1 and t2 represents the time-point of blood draw, respectively) and relative changes with the formula (Ct2−Ct1/Ct1) × 100 (expressed as a percentage). Kaplan–Meier curves and the log-rank test were used to assess differences in outcomes between groups. A multivariate Cox proportional hazards regression was performed to determine predictors for discharge. All hypothesis testing was two-tailed and P values less than 0.05 were considered statistically significant. All statistical analyses were performed using MedCalc 11.1, R 3.5.1 (The R Foundation for Statistical Computing) and SAS 9.3 (SAS Institute Inc.).
Results
During the 12-month study period, 2525 patients met inclusion criteria, while 910 patients were excluded due to presentation as STEMI, repeated presentations, or inappropriate set of troponin (Figure 1). Patients were followed up for all-cause mortality for a median of 400 (316–459) days, and follow-up was complete for 98.7% (missing follow-up in 34 of 2525 cases). Discordance between the final diagnosis in the ED and the retrospective adjudication occurred in only eight NSTEMI cases (2.4%) and in 11 unstable angina cases (3.9%). Demographic characteristics for the entire study cohort, split by inclusion period are shown in Table 1.
Demographic characteristics for the entire study cohort by study period.
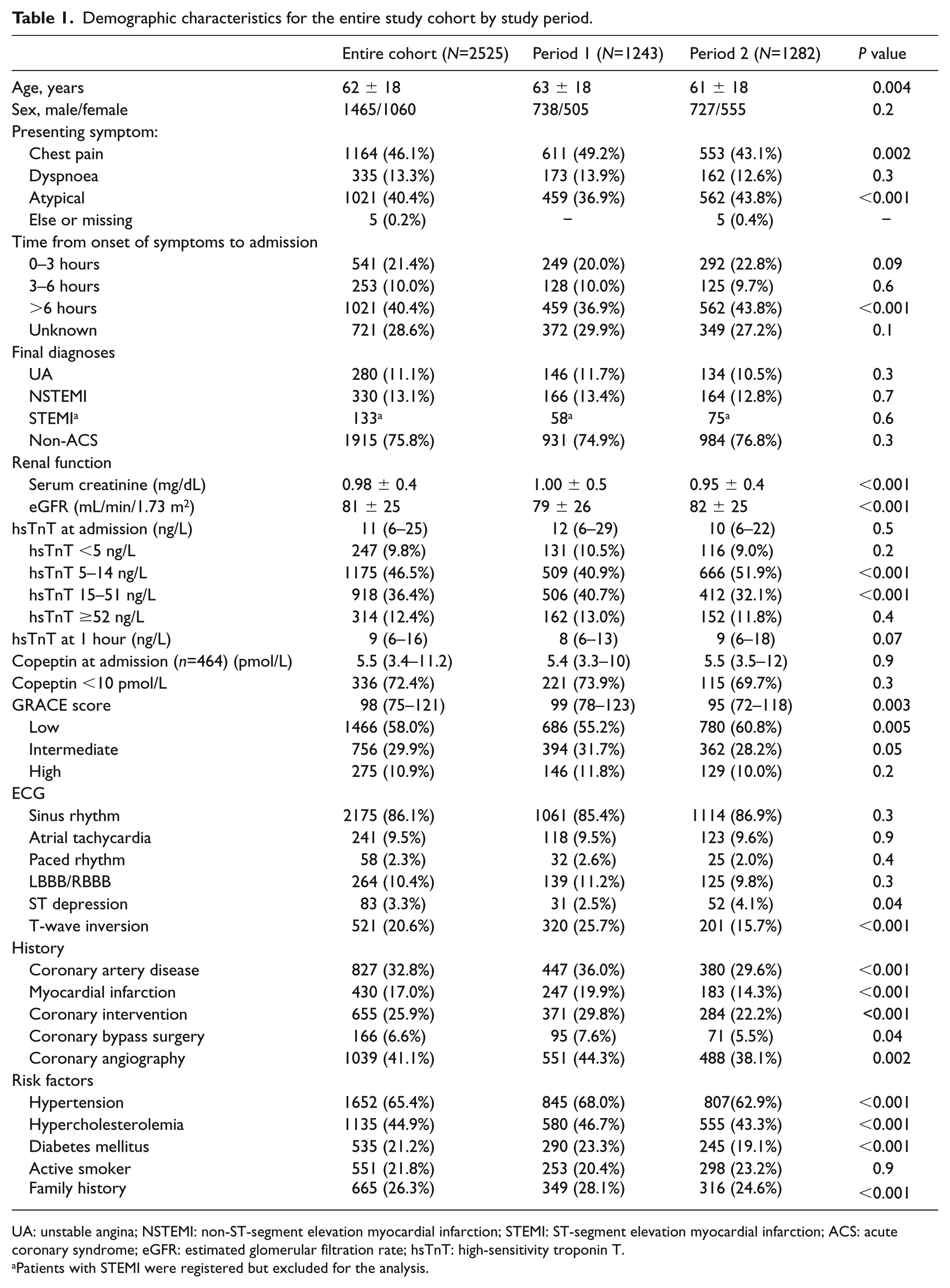
UA: unstable angina; NSTEMI: non-ST-segment elevation myocardial infarction; STEMI: ST-segment elevation myocardial infarction; ACS: acute coronary syndrome; eGFR: estimated glomerular filtration rate; hsTnT: high-sensitivity troponin T.
Patients with STEMI were registered but excluded for the analysis.
Primary endpoints
Utilisation of ESC 0/1-hour algorithm
Trends for utilisation of diagnostic algorithms changed significantly after the implementation of the ESC 0/1-hour protocol. In particular, there was an increase of the ESC 0/1-hour algorithm by 270% and a concomitant decrease of the ESC 0/3-hour algorithm by 62%. The algorithms based on a single baseline hsTnT of less than 5 ng/L (LoD) remained almost stable. In parallel, the median interval between the initial and the first follow-up hsTnT specimen shortened by a median of 45 minutes from 2.2 (1.48–3.08) to 1.45 (1.15–2.03) hours post implementation. The proportion of patients categorised into rule-out, observational zone and rule-in was 62.9%, 16.2% and 20.9%, respectively.
Safety of discharge after rule-out
The overall discharge rate was 58.5% (1476 of 2525 patients), and discharge rates increased from 53.9% to 62.8% post implementation (P<0.0001). Among the 1588 patients who were classified as rule-out, 76.1% (n=1209) were discharged directly from the ED. The baseline characteristics of patients admitted to hospital versus discharged from the ED are displayed in Table 2. Briefly, patients admitted to hospital were 15 years older (54 ± 17 vs. 69 ± 14, P<0.0001), more frequently had unstable angina (1.9% vs. 41.2%, P<0.0001), more often had hypertension, diabetes and hypercholesterolemia, a history of cardiovascular disease and higher GRACE scores (80 ± 27 vs. 106 ± 27, P<0.0001).
Baseline characteristics of patients admitted to hospital versus discharged from emergency department.
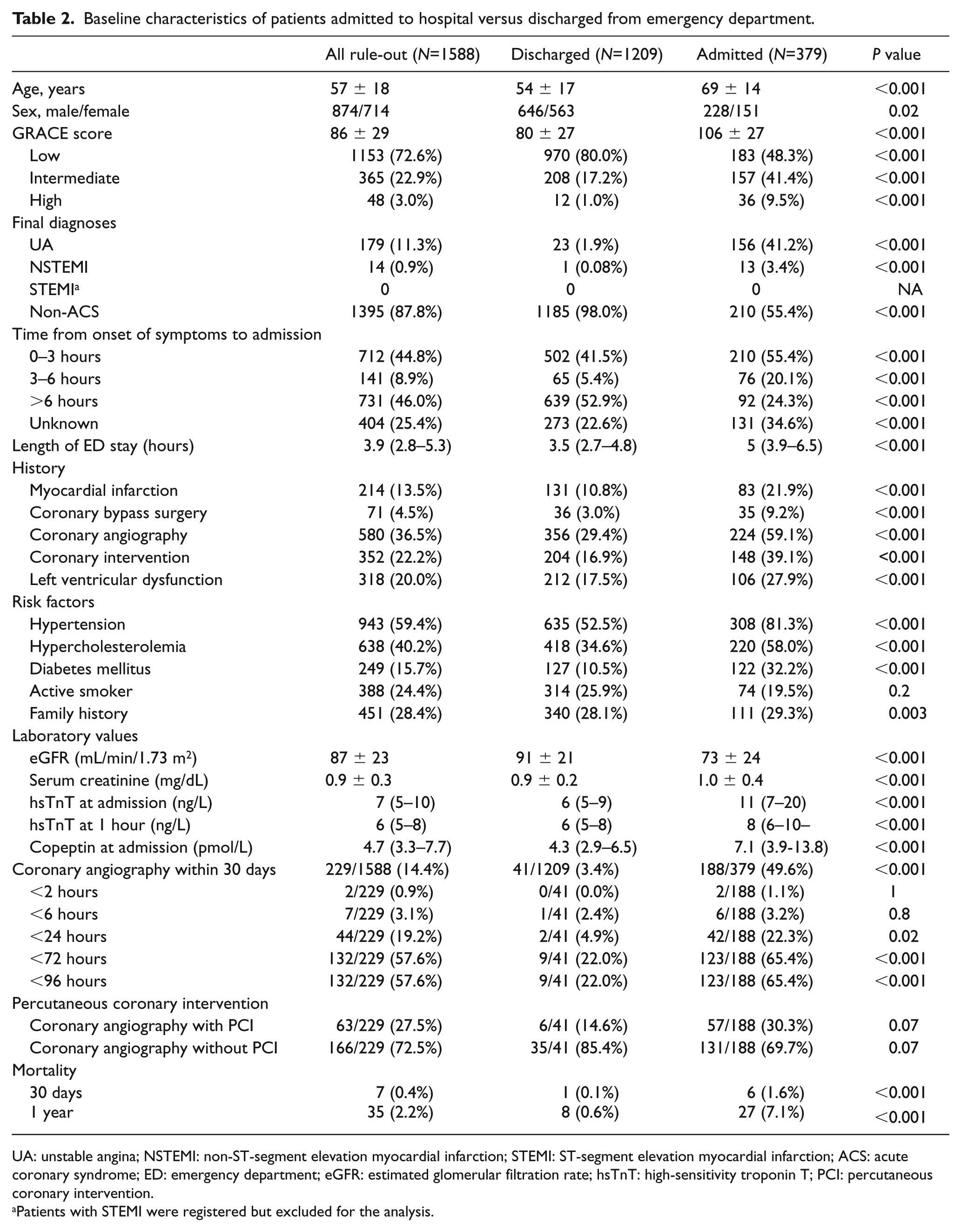
UA: unstable angina; NSTEMI: non-ST-segment elevation myocardial infarction; STEMI: ST-segment elevation myocardial infarction; ACS: acute coronary syndrome; ED: emergency department; eGFR: estimated glomerular filtration rate; hsTnT: high-sensitivity troponin T; PCI: percutaneous coronary intervention.
Patients with STEMI were registered but excluded for the analysis.
After discharge, only one patient died 13 days post-discharge, yielding an all-cause mortality rate of 0.08% (Figure 2). This 89-year-old woman presented with typical chest pain onset over 6 hours before presentation and had a GRACE score of 136 points. NSTEMI was ruled out based on two serial hsTnT values 2 hours apart (both 12 ng/L). Investigations identified urinary tract infection and mild hyponatremia. Work-up revealed chest radiography suspicious for lung cancer. The patient left hospital at her own request despite a recommendation for hospitalisation.
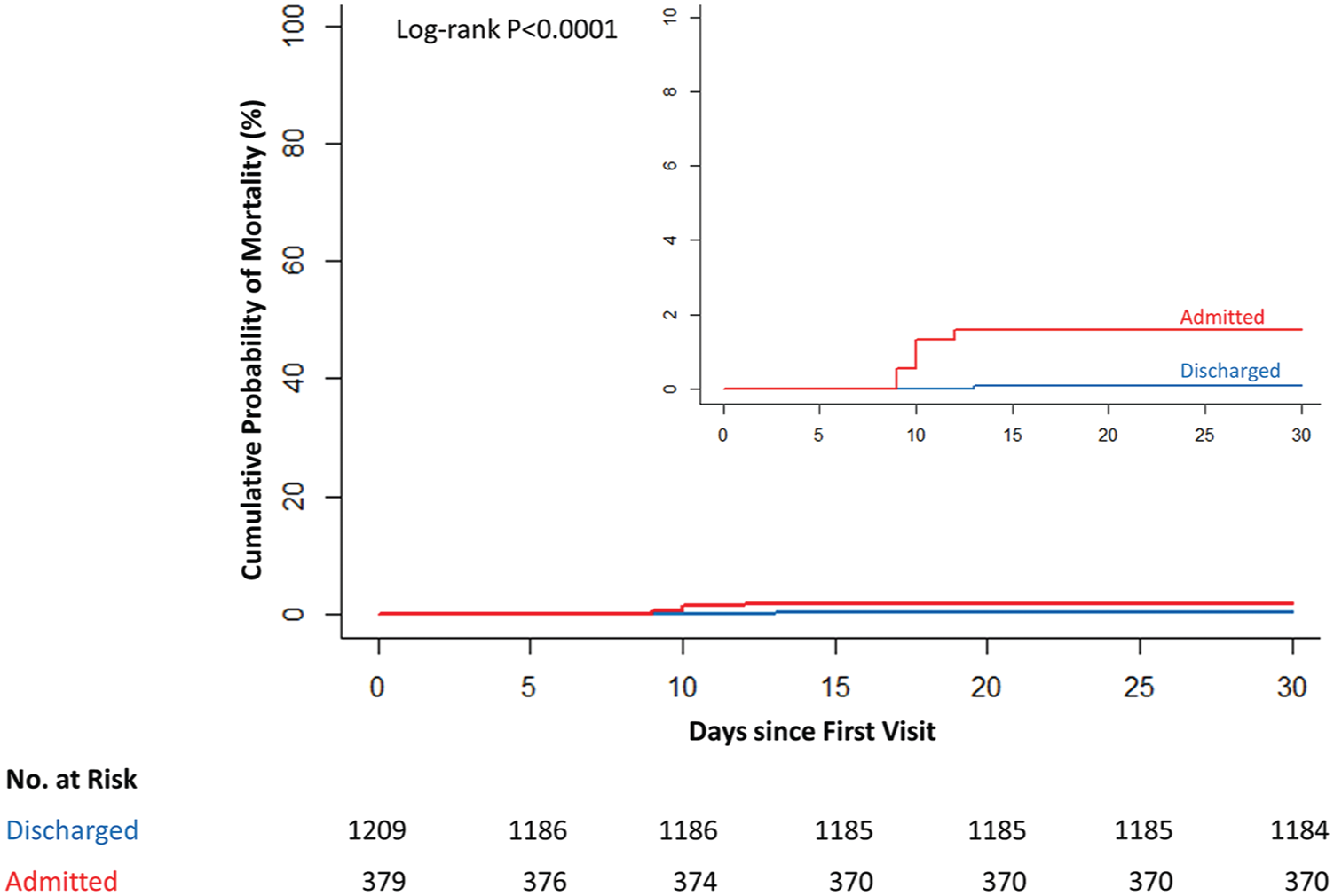
Kaplan–Meier estimates of 30-day mortality in patients with rule-out of acute myocardial infarction by hospital admission: discharged from emergency department (blue) and admitted to hospital (red).
In Cox regression analysis, several variables listed in Table 3 were independently associated with a lower probability of discharge. These variables included unstable angina, dyspnoea as the primary presenting symptom, history of diabetes, intermediate or high GRACE score, hsTnT concentration above the 99th percentile and history of left ventricular dysfunction.
Independent factors associated with a lower probability of discharge.
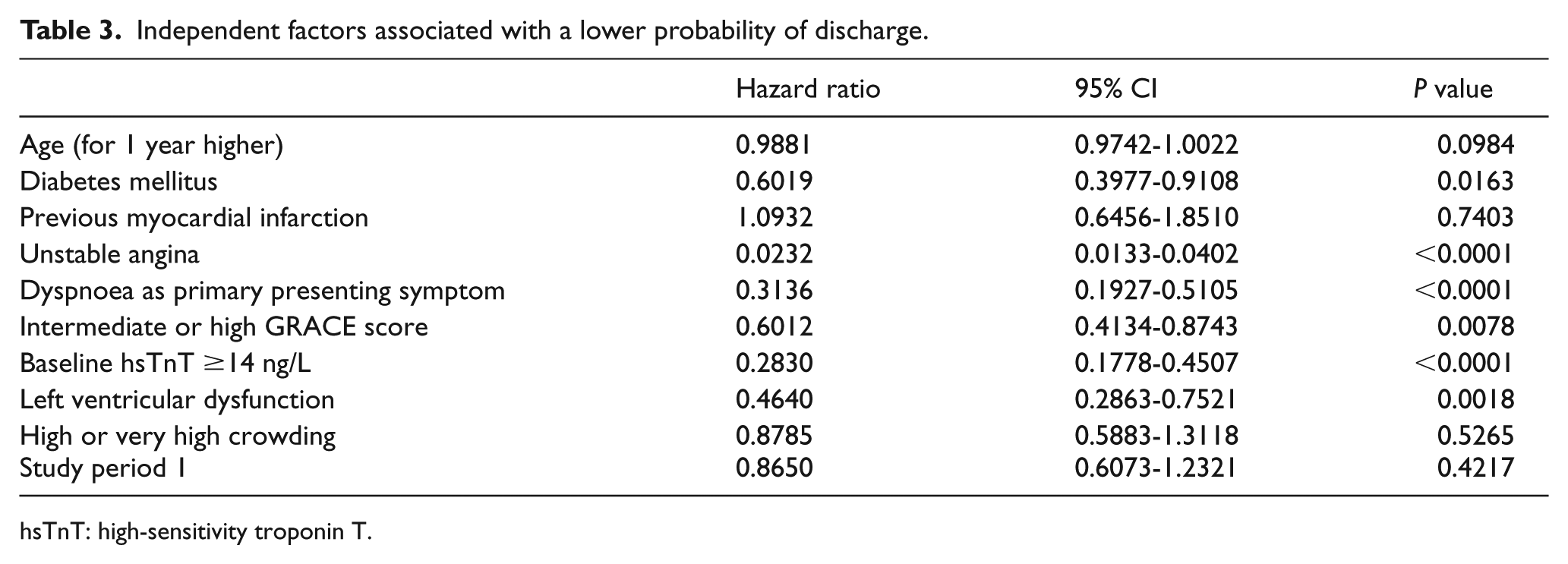
hsTnT: high-sensitivity troponin T.
Secondary endpoints
Length of stay in ED
The median length of stay (LoS) in the ED was 3.9 (2.8–5.3) hours for the entire study population, and significantly shorter for patients discharged from the ED vs. admitted to hospital (3.5 (2.7–4.8) vs. 5.0 (3.9–6.5) hours, P<0.001). The LoS grouped by different rule-out protocols is displayed in Figure 3. Median LoS was 2.9 hours with the single hsTnT less than LoD, 3.2 hours with the ESC 0/1-hour and 5.3 hours with the 0/3-hour protocol.
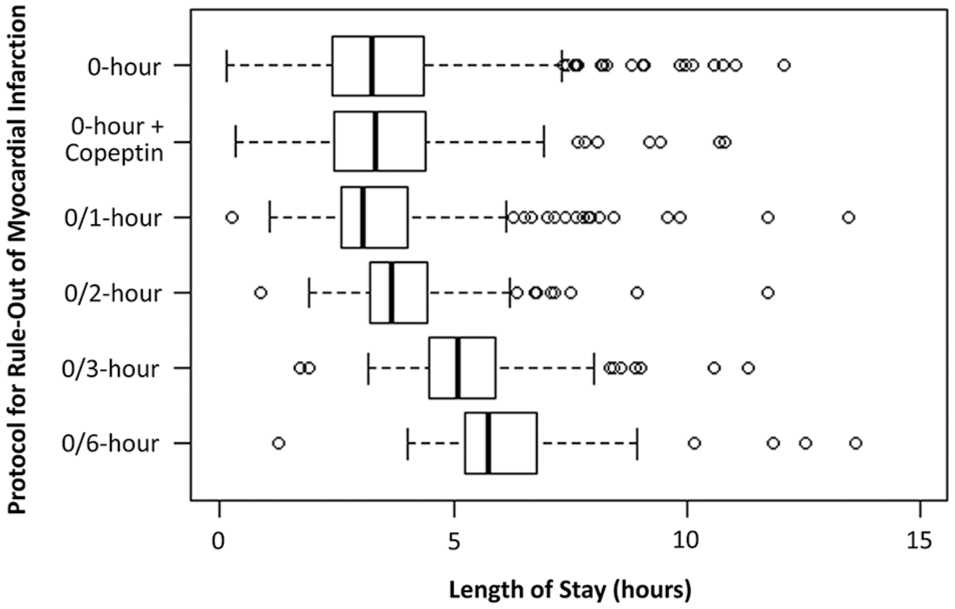
Length of stay in the emergency department by protocol for rule-out of acute myocardial infarction.
Safety of discharge within 30 days and one year by diagnostic categories
At 30 days, total all-cause mortality across all diagnostic categories at 30 days was 1.9%. Mortality rates were 0.4%, 1.2% and 6.3% for rule-out, observe and rule-in (Figure 4).
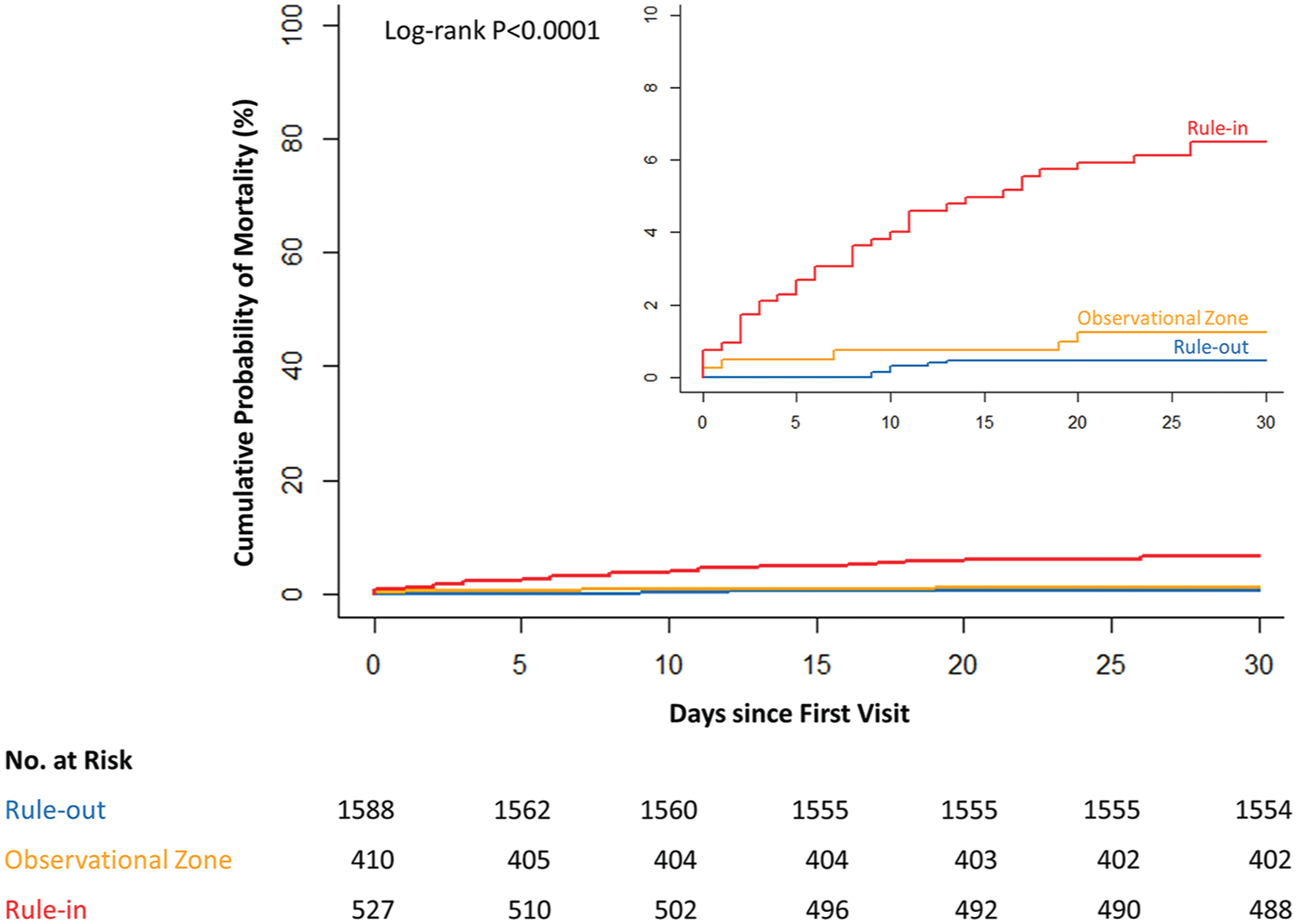
Kaplan–Meier estimates of 30-day mortality by diagnostic rule classification: rule-out (blue), observational zone (orange) and rule-in (red).
At one year, the total all-cause mortality rate across diagnostic rules was 5.7%, with corresponding rates of 2.2%, 6.1% and 16.1% for rule-out, observe and rule-in (Supplementary Figure 1).
Misclassifications
Among the 2525 patients, a total of 4872 hsTnT samples (median 2 (2–2), min-max 1–4) were collected. Beyond the minimally required diagnostic set of hsTnT measurement(s), 944 additional samples were collected (one additional sample in 846 cases and two additional samples in 49 cases), allowing a reclassification of patients from the rule-out category to the rule-in category, and from the observational zone to the rule-in zone. There were 19 reclassifications from rule-out to rule-in including five cases of missed NSTEMI and 12 reclassifications from the observational zone to rule-in including four missed NSTEMI. Selected clinical, laboratory, angiographic parameters and revascularisation procedures, disposition and outcomes are displayed in Supplementary Tables 1 and 2.
All-cause mortality by baseline hsTnT
As displayed in Supplementary Figure 2, all-cause mortality increased in proportion to the baseline hsTnT concentration demonstrating no death among patients presenting with a hsTnT of less than 5 ng/L and the highest all-cause mortality among those presenting with a hsTnT of 52 ng/L or greater.
Additional findings
Kaplan–Meier survival plots for all-cause death at 30 days and one year according to different diagnostic rules for rule-in are illustrated in Supplementary Figures 3 and 4. After rule-out of MI (n=1588 patients), a total of 379 patients (23.9%) were admitted to hospital. The use of diagnostic work-up during index admission and subsequent 30 days following discharge is displayed in Supplementary Table 3, split by study period.
Supplementary Figure 5 shows the steep uptake of the ESC 0/1-hour algorithm at the cost of the ESC 0/3-hour algorithm starting immediately after the official implementation date (1 January 2017 – 6 months after the start of the study). Small changes of implementation rates before the official implementation were presumably motivated by the 2015 ESC guideline recommendation on the usefulness of the ESC 0/1-hour algorithm as an alternative to the ESC 0/3-hour algorithm. 1
Additional blood sampling beyond the minimal diagnostic set was obtained in 30% of the patients and reflects clinical practice, in which decisions to extend observation or continue blood sampling is left at the discretion of the physician, rather than on the protocol of a clinical trial. The additional measurements allowed us to obtain information on potential misclassifications, e.g. transition from rule-out to rule-in. Such reclassifications – from rule-out to rule-in – occurred in 19 patients, of whom five patients would have a missed NSTEMI diagnosis. In these five cases, maximal hsTnT values were low (maximal hsTnT in the ED ranging from 21 to 29 ng/L) suggesting a very small infarct size that would have been missed by a conventional sensitive and probably also by a contemporary sensitive cardiac troponin assay. Consistently, only two of the five patients with theoretically missed MI underwent PCI and there were no mortality events at 30 days among these five patients.
Additional information on outcomes, diagnostic rules, timing of coronary angiography, rates of revascularisation are provided in Supplementary Table 4.
Discussion
There is a striking contrast between the excellent diagnostic performance of fast protocols,2–11 the consistent findings for lower observation times in the ED,8,19,21 relevant cost savings26–29 and the limited adoption of fast diagnostic protocols despite an urgent need to decongest crowded EDs.12,30 Uncertainties that reduce the enthusiasm of physicians to implement single biomarker or accelerated protocols are mostly driven by the fear of litigation in case of missed MI or death. 13 Previously, warnings were expressed that clinicians should apply the 1-hour algorithm with caution and only in low-risk patients,14,15 and that decisions should rather be based on rising or falling patterns of troponin than on single cut-off values. 14 In support of the former, 2014 American College of Cardiology (ACC)/American Heart Association (AHA) guidelines 16 cautioned against early troponin testing given that some values may not become abnormal for up to 12 hours. However, at the time of publication, high sensitivity tests were not available in the USA. This fear is further fuelled by the paucity of evidence from randomised trials evaluating the safety of discharge using accelerated protocols and hsTn assays, as well as issues to extrapolate findings derived from observational studies that enrolled patients with higher pre-test probabilities for an ACS. Currently, most evidence from randomised trials has focused on the implementation of hsTn in combination with validated clinical scores,18,19 a dual biomarker strategy combining copeptin with cardiac troponin, 8 or a discharge of low-risk patients based on a normal hsTnI measurement 2 hours apart, together with a normal ECG and either a TIMI score of 1 point or less,9,10,21 or a low EDACS score. 20 A large pre and post-implementation study on 31,332 patients with suspected ACS demonstrated a reduced LoS and increased proportions of patients discharged from the ED within 6 hours, without an adverse event when clinical pathways were correctly applied. 21
The data from this large study confirm previous observations and add information on feasibility, efficacy and safety of discharge using the ESC 0/3-hour but most importantly the ESC 0/1-hour rule-out protocols in an all-comers registry with broad inclusion criteria. The study provides several important findings.
First, our findings suggest that the ESC recommended 0/1-hour algorithm can be implemented as the predominant diagnostic algorithm, and is clinically feasible. Second, discharge after rule-out is safe with or without the use of the GRACE score, with 30-day all-cause mortality rates less than 0.1%. In this cohort, mortality was defined as all-cause death, and the only fatality occurred 13 days after discharge due to lung cancer. There was no statistically significant difference regarding the safety of the ESC 0/1-hour algorithm compared to the standard ESC 0/3-hour protocol. Even the rule-out strategy based on a single low baseline hsTnT below the LoD (<5 ng/L), or a normal hsTnT (≥5 but ≤14 ng/L) together with a normal copeptin (<10 pmol/L) at presentation were not associated with a higher risk of all-cause mortality after discharge among 1309 patients primarily discharged from the ED. Third, lengths of stay in the ED were closely related to the specific protocols and were as low as a median of 2.9 hours using a single rule-out hsTnT, 3.2 hours using the 0/1-hour protocol and 5.3 hours with the 0/3-hour protocol. Likewise, the interval between the first and the second blood draw shortened by 45 minutes after implementation of the ESC 0/1-hour protocol. Fourth, there is no overuse of resources for diagnostic work-up in order to facilitate earlier discharge, which is important as others14,15 have warned about an increase of unnecessary and costly diagnostic work-up before discharge. Conversely, utilisation rates of stress testing, imaging and coronary angiography were in the range of 10–20% among patients discharged, and thus very similar to rates of investigations performed during the index visit or 30-day follow-up reported by Mokhtari et al. 7 Among 1038 patients with suspected ACS, exercise ECG, echocardiography, computed tomography coronary angiography and coronary angiography were performed in 13.8%, 18.3%, 0.6% and 13.3% of patients, respectively. Interestingly, rates of utilisation of stress testing and imaging before discharge vary considerably between studies showing low rates19,26,31 or very high utilisation rates of investigations 31 suggesting different adherence to guideline recommendations 1 and local practice across institutions and continents. 32
The consistencies and differences of our study compared to other observational studies require detailed discussion. Our study is distinct to other observational studies regarding several aspects. First, we enrolled a consecutive cohort of all-comers with a broad spectrum of presenting symptoms without exclusion for older age, heart failure or chronic kidney disease, with the consequence of a further lowering of clinical specificity. Accordingly, rates of NSTEMI within the rule-in and observational zone were 57.5% and 3.2%, and thus considerably lower than reported in other observational trials.2–4,6,7 Second, LoS in the ED in our study are very short, ranging from 2.9 hours with rule-out based on a hsTnT less than LoD to 5.3 hours with the 0/3-hour diagnostic algorithm. In the literature, reported LoS vary between 4 hours,8,26 5.3 hours, 38 5.5 hours, 39 6.4 hours 19 to 26.3 hours, 31 with the shortest observation times (median LoS 4 hours in both studies) using the dual biomarker strategy, 8 or the 0/1-hour rule-out in the TRAPID study. 26 Conversely, the longest LoS have been reported in an Australasian cohort, 31 in which LoS was associated with high utilisation rates of investigations before discharge. As compared to most other observational studies in which treating physicians were blinded to the investigational hsTn results and patients were not managed in accordance with these results, physicians in the present study reacted on the fast protocols thus accelerating the disposition of patients.
Third, post-implementation discharge rates after rule-out were 62.8% and thus at the upper end of the reported range. Discharge rates in other observational or randomised studies are heterogeneous with low discharge rates among low-risk patients between 18.4% and 26%,9,18,21 intermediate discharge rates between 42.3% and 55%19,39,40 and high discharge rates between 67.8% and 72%.8,38,41 Only one study 42 reported significantly longer hospitalisation stays by 35%, fewer early discharges after a negative result (7% vs. 22%, P=0.0001), more coronary angiograms (77% vs. 55%, P=0.0001) and revascularisations (45% vs. 31%, P=0.0001) after implementation of hsTn assays by 35%.
Our study findings are consistent with other observational studies with regard to important aspects. First, the prevalence of NSTEMI was 13.1% in our study and was thus similar to reported rates between 7.0% and 23.3%,33,34 with an overall prevalence in the pooled population of 9241 patients of 15.4%. 33 Second, total mortality rates at 30 days and one year across all diagnostic categories were 1.9% and 5.7%, respectively, indicating enrolment of a risk that is at least as high as in other observational studies,2–5,21 and the need for risk stratification before discharge, even among patients ruled-out for MI. Along with others, total mortality rates in the TRAPID acute MI study, 4 a study that used a similar protocol, were lower than in our cohort, highlighting the broader inclusion of consecutive patients. At 30 days, total mortality rates were 0.2%, ranging between 0.1%, 0.4% and 0.5% for those ruled-out, in the observational zone, and ruled-in. At one year, total mortality rates were 4.1%, ranging between 0.7%, 9.7% and 9.3% for those ruled-out, in the observational zone and ruled-in, respectively. Consistently, a recent meta-analysis on 9241 patients in 11 cohort studies noted no death occurring within 30 days among low-risk patients defined as hsTnT of less than 5 ng/L without ECG changes. 33 Thus it appears that fast rule-out protocols allow safe classification and risk stratification of patients with suspected ACS, with or without the use of clinical scores, enabling the safe discharge of a low-risk cohort, with an all-cause mortality rate below 0.01%. Third, rates of missed MI following fast rule-out were 0.3% (five of 1588 patients), which is at the lower end of reported rates between 0.2% and 1.2% using hsTn and fast protocols.4,5,35,36 There were no deaths or MI, all underwent coronary angiography, but only two patients (10.6%) received a PCI within 30 days, supporting the claim of safe discharge after rule-out using hsTn and accelerated protocols. Finally, rates for the diagnostic categories rule-out, rule-in and observe were 62.9%, 20.9% and 16.2%, which is very consistent with other observational studies on the 0/1-hour protocol using hsTnT or hsTnI. 37 Of interest, the inclusion of patients with a broad spectrum of symptoms with higher prevalence of myocardial injury unrelated to ischaemia did not augment the observational zone but – as expected – fewer patients received a diagnosis of NSTEMI within the rule-in and observational zone.2–4 The yield of NSTEMI was similar to Mokhtari et al. 7 reporting 50% NSTEMI among rule-in but only 1.9% in the observational zone.
These findings extend previous studies on the safety of discharge of low-risk patients after rule-out of MI using hsTnT in general and the ESC 0/1-hour protocol in particular. Our findings might increase the confidence of physicians to apply accelerated diagnostic protocols and are likely to speed up the slow implementation of accelerated diagnostic algorithms in crowded EDs.
Limitations
Our findings suggest that the use of a fast rule-out using hsTnT in a 0/1-hour protocol allows safe discharge without the need for extensive pre or post-discharge work-up, provided the residual risk is estimated appropriately. We identified variables that were independently associated with a hospital admission despite rule-out. These variables included individual risk factors, hsTnT level and a high GRACE score. However, an extremely low all-cause mortality rate after discharge prohibits any meaningful speculations on the utility of clinical scores in the era of hsTn assays. Chapman et al. 43 recently reported that when clinical risk scores were applied in the High-STEACS pathway at thresholds to rule out MI that were considerably lower than the 99th percentile value, the proportion of patients ruled out halved without improving safety. In our institution, the use of the GRACE score, which represents a validated and objective tool for estimation of risk for death or MI, 25 was not mandatory for the decision to discharge, highlighting the importance of the physicians’ experience to estimate future risk. Accordingly, our liberal discharge policy might not be transferrable to other EDs with lower levels of physicians’ experience.
Second, we cannot exclude under-reporting of diagnostic work-up within 30 days that might have escaped our follow-up. Third, as a consequence of early discharge after rule-out without a gold standard diagnosis based on a 6-hour algorithm, we cannot exclude the later development of rule-in or NSTEMI. In order to reduce numbers of false negatives, we strictly followed ESC guideline recommendations and refrained from rule-out and discharge of patients based on a single hsTnT less than LoD, if the interval from the last pain episode was unclear, equivocal, or shorter than 3 hours. Moreover, physicians were instructed to continue hsTnT measurement in patients with refractory or recurrent symptoms, a persistent high level of clinical suspicion for evolving MI. Additional blood draws beyond the adequate diagnostic set were collected in 30% of patients and allowed us to identify 19 of 1588 patients (1.2%) who changed the diagnostic category from rule-out to rule-in. Among these, five patients had a missed NSTEMI (0.3%), characterised by small hsTnT between 17 and 23 ng/L, none died within 30 days, all received coronary angiography but only two patients required PCI. The numbers of missed MI wrongly classified as rule-out are very similar in other observational studies ranging from 7/813 (0.9%) in the TRAPID study, 4 to 5/2488 (0.2%) in the pooled cohorts of APACE and BACC trials, 36 20 of 2533 (0.8%) in the APACE study, 35 and between four of 342 (1.2%) and 12 of 2160 (0.6%) in two High-STEACS study reports.5,35
Finally, the present study was performed in a single centre – the ED of the department of cardiology, which is certified as a CPU. Our ED is constantly supervised by a cardiologist and has a volume of patients that is representable compared to other CPUs. The number inclines to more than 40 patients per day when patients admitted to the entire internal medicine ED are also considered. While we cannot fully exclude a referral bias, patients with suspected ACS are almost exclusively referred to the CPU. This has the advantage to evaluate consecutive patients across almost the entire spectrum of risk. Our study findings are consistent regarding the prevalence of ACS and rates of death or MI during follow-up as compared with other European observational studies.3,6,7 Given that our findings were obtained in a German CPU, led by a cardiologist and certified by the German Cardiac Society only after operating criteria were met, our results cannot be automatically generalised to other EDs outside Germany, unless they provide similar infrastructural characteristics or tight interdisciplinary collaboration with cardiologists.
Conclusions
Implementation of the ESC 0/1-hour algorithm is feasible and is associated with very low mortality of discharged patients after rule-out. Furthermore, the ESC 0/1-hour algorithm is associated with a significantly shorter length of ED stay than the ESC 0/3-hour protocol. After implementation of the ESC 0/1-hour algorithm, discharge rates increased significantly, without excessive use of diagnostic resources.
Supplemental Material
Supplemental_Material – Supplemental material for RAPID-CPU: a prospective study on implementation of the ESC 0/1-hour algorithm and safety of discharge after rule-out of myocardial infarction
Supplemental material, Supplemental_Material for RAPID-CPU: a prospective study on implementation of the ESC 0/1-hour algorithm and safety of discharge after rule-out of myocardial infarction by Kiril M Stoyanov, Hauke Hund, Moritz Biener, Jochen Gandowitz, Christoph Riedle, Julia Löhr, Matthias Mueller-Hennessen, Mehrshad Vafaie, Hugo A Katus and Evangelos Giannitsis in European Heart Journal: Acute Cardiovascular Care
Footnotes
Acknowledgements
The authors would like to thank the patients who participated in the study and the staff of the ED. They also express their gratitude to the study nurses Heidi Deigentasch, Melanie Hütter and Elisabeth Mertz.
Conflict of interest
MB reports grants and non-financial support from AstraZeneca, non-financial support from Thermo Fisher. MMH reports grants and speaker honoraria from Roche Diagnostics; grants and non-financial support from BRAHMS Thermo Scientific. HAK received honoraria for lecturers from Roche Diagnostics, AstraZeneca, Bayer Vital, Daiichi-Sankyo, and held a patent on cTnT that has expired. EG received honoraria for lectures from Roche Diagnostics, AstraZeneca, Bayer Vital, Daiichi-Sankyo, Eli Lilly Deutschland. He serves as a consultant for Roche Diagnostics, BRAHMS Thermo Fisher, Boehringer Ingelheim, and has received research funding from BRAHMS Thermo Fisher, Roche Diagnostics, Bayer Vital and Daiichi Sankyo. All other authors have no conflicts of interest to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by a research grant from Roche Diagnostics International Ltd.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
