Abstract
Background
Intravascular ultrasonography (IVUS) has become an important complementary tool in interventional cardiology, both for preprocedural planning and for optimizing the performance of percutaneous coronary intervention (PCI). However, this tool is not free of potential complications, because of that it is essential to be aware of them and their management. Over time, new uses of IVUS have emerged, and it is currently a potential tool for predicting the risk of coronary perforation.
Case report
We present the clinical case of a 51-year-old male patient who was admitted in the context of post-infarction angina. During coronary angiography, the patient presented with two acute complications, one of them associated with IVUS and the other associated with severe coronary calcification that predisposed to coronary perforation. Both complications were successfully treated.
Conclusion
IVUS, although a very useful imaging tool before and during PCI, is not without risk. The overall rate of complications with certain or possible relation to IVUS is 3.9%. Vascular spasm is the most frequent complication and acute vascular occlusion, with the need for emergency coronary artery by-pass grafting, the least frequent. On the other hand, IVUS can predict the risk of developing other complications, such as coronary perforation, by means of the C-CAT sign. Knowledge of the possible complications during PCI and the rapid procedure of the hemodynamic team allows adequate management of these potentially fatal complications.
Keywords
Background
Intracardiac ultrasonography for the study of cardiac valves and chambers was first described in 1971, and it was not until 1989 that the first in vivo images of the coronary arteries were obtained. 1 Since then, the use of intravascular ultrasonography (IVUS) has progressively increased, becoming an important complementary tool in interventional cardiology, providing a better characterization of vessel morphology, atherosclerotic plaque and optimizing the performance of percutaneous coronary intervention (PCI). 2 However, this tool is not free of potential complications, even with experienced operators, so it is essential to be aware of these complications and their management.3–5 Over time, new uses of IVUS have emerged, and it is currently a potential tool for predicting the risk of complications not directly associated with IVUS, such as coronary perforation (CP). 6 This is a rare but potentially preventable complication by knowing the coronary anatomy and high-risk characteristics. 7 We present the case of a patient who presented two acute, potentially fatal complications during the same interventional procedure, which were managed in a timely and successful manner.
Case report
History of presentation
A 64-year-old man with a past medical history of type I obesity and dyslipidemia showed up at the emergency room. Five days before his admission, the patient reported non-radiating oppressive chest pain of moderate intensity, associated with nausea, so he self-medicated with non-steroidal anti-inflammatory drugs with a gradual decrease in pain. On the day of his admission, he presented a recrudescence of a more intense chest pain so the patient looks for medical attention. On physical examination, blood pressure was 130/89 mmHg, heart rate was 92 bpm and oxygen saturation was 98% (without supplemental oxygen). No jugular ingurgitation or lower limb oedema was identified. Apical impulse, S1 and S2 were normal. On pulmonary examination, scarce crackles were auscultated in the bases of both lung fields.
Investigations
The electrocardiogram showed sinus rhythm with QS complexes and ST-segment elevation (1 mm) in V1-V2 leads associated with biphasic T waves in V1-V4 leads (Figure 1). Ancillary tests showed a high pro-brain natriuretic peptide (786 pg/mL) and elevated troponins (3.5 ng/mL). Chest X-ray showed pulmonary interstitial oedema. Transthoracic echocardiography (TTE) exhibited preserved biventricular function (left ventricle ejection fraction (LVEF) 55%), apical hypokinesia and preserved LV end-diastolic pressure. Therefore, the diagnosis of a 5-day evolving anteroseptal ST-segment elevation infarction (Killip Kimball II) associated with post-infarction angina was made and the patient entered to the cardiac catheterization laboratory.
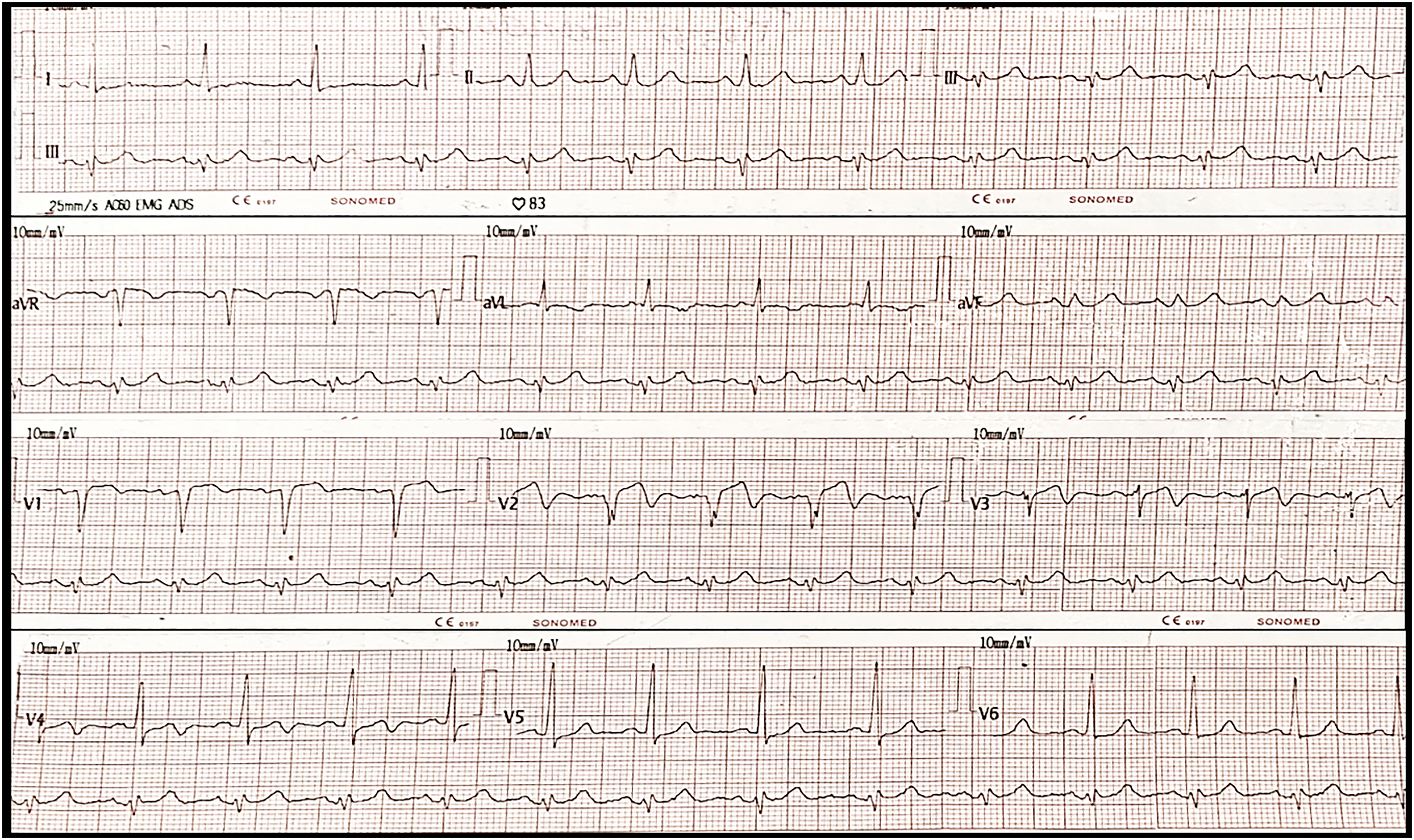
Admission electrocardiogram: sinus rhythm / HR 80 bpm / QRS axis 20° / PR 160 ms / QRS 80 ms / QT 320 ms / QS V1-V2 / ST-segment elevation (1 mm) V1-V2 / biphasic T waves V1-V4.
Coronary angiography (Video 1; Figure 2) showed severe ostial stenosis of the left anterior descending artery (LAD) and in its middle segment at the bifurcation with the 3rd diagonal (DG) (Medina 1-1-1), subocclusive ostial stenosis of the 2nd DG, mild stenosis in the proximal segment and moderate stenosis in the distal segment of the circumflex artery (Cx) (after the birth of the 1st marginal; Medina 0-1-0) and an anomalous birth of right coronary artery that did not present angiographically significant lesions. Pre-procedural IVUS corroborated the severe stenosis in the middle segment of the LAD (posterior to 3rd DG birth – lesion lumen area of 2.93 mm2) associated with significant calcification along its entire length (Figure 3(a)–(c)). IVUS ruled out the presence of significant lesions in the circumflex artery.

(a) Moderate stenosis in distal segment of Cx (white thin arrow), after the 1st marginal artery's birth. (b, d) Subocclusive ostial stenosis of the 2nd DG and severe stenosis in mid-segment of LAD at the bifurcation with 3rd DG (Medina 1-1-1) (blue thick arrow). (c) Severe ostial stenosis of LAD (blue arrowhead). (e, f) Anomalous birth of RCA (blue asterisk) without angiographically significant lesions.

Pre-PCI LAD IVUS shows severe stenosis in the middle segment of the LAD, posterior to 3rd DG birth, with lesion lumen area of 2.93 mm2 (c) associated with severe calcification throughout the proximal and mid segments (a, b) of LAD. C-CAT sign (b) describe a C typed eccentric lesion, approximately 270-degree, calcified plaque and contralateral thin plaque.
Management
In a first instance, surgical revascularization was considered, due to the absence of an appropriate landing zone in the LAD, so the procedure would involve lining the entire LAD up to the LMCA. After removal of the guidewire of the LAD (Figure 4(a)), complete occlusion of the LAD was observed (Figure 4(b); Video 2), that did not response to intracoronary nitroglycerin. Therefore, emergency angioplasty of the LMCA-LAD was decided (Video 2). The right radial approach was chosen and unfractionated heparin at 100 IU/kg was used to achieve an activated clotting time (ACT) > 250 s. Two 0.014 × 182 cm Floppy guidewires were passed into the distal bed of LAD and Cx. The mid and proximal segments of the LAD were predilated with a 2.75 × 15 mm Euphora NC balloon, targeting for a balloon/artery ratio of about 1. Two overlapping 3.0 × 32 mm and 4.0 × 29 mm Evermine drug-eluting stents (DES) were implanted in the mid and proximal segments of the LAD (Figure 4(c)–(e)), respectively. Then, the most proximal segment of the LAD and LMCA were predilated with a 4.0 × 15 mm NC Euphora balloon and, subsequently, a 4.5 × 22 mm Resolute Onyx DES was implanted at the LAD-LMCA (Figure 4(f) and (g)). Postdilatation was performed at the LMCA with a 5.0 × 12 mm NC Emerge balloon (Figure 4(h)). The Floppy guidewires were recrossed and kissing balloon (KB) was performed with a 3.0 × 12 mm NC Euphora and 4.0 × 20 mm NC Emerge balloons (Figure 4(i)). Angiographic control showed adequate DES expansion with TIMI III flow (Figure 4(j) and (k)). However, post-procedural IVUS showed distal DES underexpansion (Figure 5(a)–(c)) so postdilatation was chosen.

(a) Pre-angioplasty IVUS been performed. IVUS guidewire can be seen in the LAD (thin arrow). (b) Acute vessel occlusion seen after guidewire removal that did not response to intracoronary nitroglycerin, so emergency angioplasty was decided. (c)–(e) Predilatation of the mid and proximal segments of the LAD was performed and two overlapping Evermine 3.0 × 32 mm and Evermine 4.0 × 29 mm DES were implanted in these segments of the LAD. (f, g) Then, the proximal segment of the LAD and LMCA were predilatated and a 4.5 × 22 mm Resolute Onyx DES was implanted at the proximal LAD-LMCA. (h) LMCA's postdilatation with a 5.0 × 12 mm NC Emerge balloon was performed (asterisks). (i) Floppy guidewires were recrossed and KB was performed with a 3.0 × 12 mm NC Euphora and 4.0 × 20 mm NC Emerge balloons. (j, k) Angiographic control showed adequate DES expansion with TIMI III flow.

Post-PCI LAD IVUS showed improvement (red thin arrows) in the CSA in the proximal (a, b) and middle (c) segments of the LAD, however, there was persistent DES underexpansion (stent CSA 4.94 mm2) (c) so postdilatation was opted for.
Postdilatation, at nominal pressure, was performed with a 3.0 × 15 mm NC Euphora balloon in the mid and proximal segments of LAD (Figure 6(a)–(c)). On angiographic control, a contrast extravasation jet was seen through a > 1 mm orifice, so the diagnosis of an Ellis type III CP was made (Figure 6(d) and (e); Video 3). Immediately, the CP area was sealed with the postdilatation's balloon for 15 min at low pressure (5 atm). Concurrently, 50 mg of protamine was administered to achieve an ACT less than 150 s. Intraprocedural TTE revealed pericardial effusion, without right cavities collapse. After 15 min, the CP was complete sealed without residual leak (Figure 6(f); Video 4). Then, the patient was moved to the coronary care unit (CCU) where he presented tachycardia as the unique alarm sign, however there were no other intercurrences reported. Serial control TTE showed hypokinesia of segments in the territory of the LAD. LVEF was 55%; global longitudinal LV strain −18% (Figure 7). Right ventricle dimensions and function were preserved. There was no relevant valvulopathy. Inferior vena cava was dilated (25 mm) with less than 50% collapse. There was pericardial effusion with apical-lateral well, adjacent to the right atrium, anterior to the right ventricle and posterior to the left ventricle (Figure 8). Since there were no signs of cardiac tamponade, expectant management was chosen.
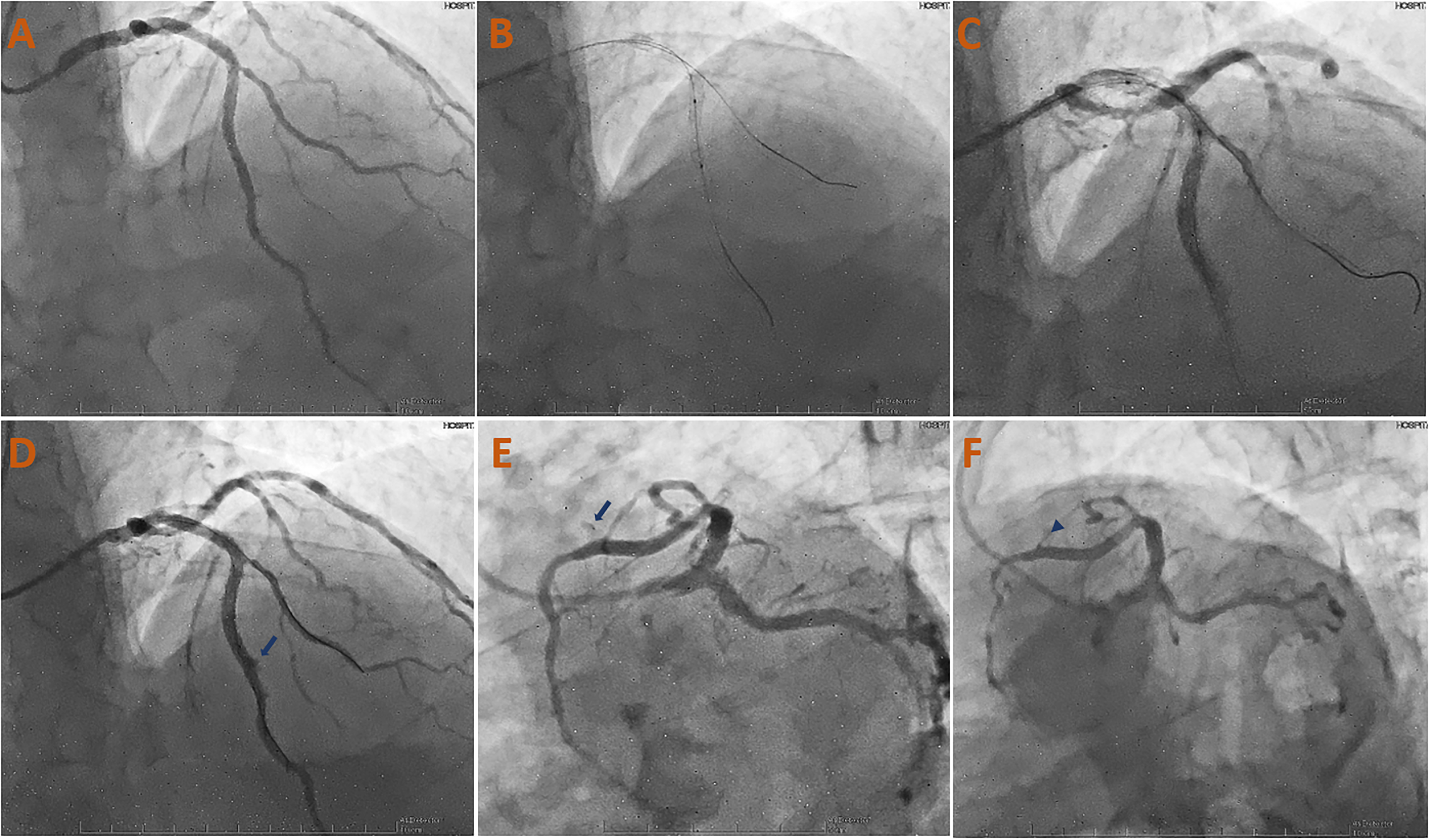
(a) Angiographic control with a TIMI III flow, however, IVUS showed DES underexpantion, so postdilatation was decided. (b, c) Postdilatation was performed with a 3.0 × 15 mm NC Euphora balloon in the mid and proximal segments of LAD. (d, e) On angiographic control, a contrast extravasation jet (blue arrow) was seen through a > 1 mm orifice, making the diagnosis of an Ellis type III CP. Immediately, the CP was sealed by prolonged low-pressure insufflation and protamine was administered. (F) After awaiting 15 min, angiographic control showed complete sealing of the CP and absence of residual leak (blue arrowhead).

Strain curves in 4-chamber, 3-chamber and 2-chamber views. Show differences in systolic and post-systolic strain of segments from different coronary territories. Global longitudinal left ventricular strain: −18%.

(a) TTE, 4-chamber view. Evidence of pericardial effusion with apical-lateral well (12 mm) and adjacent to the right atrium (9 mm). (b) Mitral respiratory variability less than 25%. (c) TTE, subcostal view. Pericardial effusion with well anterior to the right ventricle (7 mm) and posterior to the left ventricle (10 mm). (d) Dilated inferior vena cava with collapsibility less than 50%.
After percutaneous revascularization and management of the CP, the patient remained hospitalized in the CCU for ten days and was discharged with dual antiplatelet therapy, beta-blocker and statins with outpatient follow-up. Six months after discharge, the patient is asymptomatic (functional class NYHA I, CCS I) with preserved LV systolic function (LVEF 60%).
Discussion
IVUS is an imaging modality used in interventional cardiology, the use of which has been increasing in recent years. It allows a better characterization of the vascular anatomy, provides fundamental data to guide PCI, such as the morphology and content of the lesion, plaque burden, guides the size of the stent, its adequate apposition and expansion, and allows us to identify post-intervention complications. However, currently, the highest level of recommendation in the guidelines regarding the use of IVUS is IIA. 8 Likewise, despite the potential usefulness of IVUS, this imaging modality is not free of risks, which have been poorly studied (Figure 9 – central illustration).

Central illustration. IVUS general information.
The exact incidence of complications due to IVUS is difficult to assess, as it is often performed in conjunction with other interventional procedures. In the European registry 3 of 718 patients, the acute complication rate associated with IVUS was 1.1% with no permanent clinical consequences. On the other hand, in the US registry, 4 the overall rate of complications with certain or possible relation to IVUS was 3.9% (spasm 2.9%, dissection, acute occlusion or thrombus 0.7%, non-fatal myocardial infarction or emergency coronary by-pass grafting 0.3%).
The risk of developing these IVUS-associated complications (other than spasm) was significantly increased in patients admitted in the context of an acute coronary syndrome and in those who underwent post-procedural IVUS. 5 However, it is difficult to determine whether this complication attributed to IVUS is actually a complication due to the ultrasonographic study or to the interventional procedure per se. On the other hand, the rate of complications associated with IVUS performed solely for diagnostic or preprocedural purposes are even rarer. 5
What is more, IVUS also allows us to identify risk factors for the development of complications not associated with IVUS, such as CP. Anatomical and procedural predictors of CP have been defined in other studies, 7 while imaging predictors include the C-CAT sign (C typed eccentric, approximately 270-degree, calcified plaque and contralateral thin plaque). 6 It should be noted that this sign has been reported in only case series, and has to be validated in larger registries to assess their predictive impact, yet. 6 However, we can affirm that our patient fulfilled these high risk's characteristics (Figure 3). Likewise, despite the fact that IVUS is associated with the use of larger, longer and more stents, 9 the SCAAR Registry has shown that IVUS is used in the same percentage of CP as in non-CP procedures without any increased risk of CP, contrary to prior publications.10,11
CP, depending on its severity, can be complicated by pericardial tamponade, cardiogenic shock and even death if not managed promptly. The Ellis classification allows stratification of the severity of CP, which, together with its location, determines management. 7 In general, Ellis type I and II perforations can be treated conservatively with transient limitation of blood flow by prolonged intracoronary balloon insufflation. 12 While type III and IV perforations usually require occlusion of the perforated area with covered stents or permanent occlusion of the affected vessel with coils, microspheres, autologous subcutaneous fat or even surgical management. 12
In the case presented, the patient developed an Ellis III type CP associated to moderate pericardial effusion but without significant collapse of the right cardiac cavities neither echocardiographic nor clinical signs of cardiac tamponade. Despite of being a severe perforation, it was managed in a conservative manner with reversal of anticoagulation, prolonged and low-pressure balloon inflation with adequate control of the complication.
Conclusion
IVUS is an important adjunctive tool in the cardiac catheterization laboratory that can be useful to determine plaque morphology and guide PCIs. However, it is not without risk, with vascular spasm being the most frequent complication and acute vascular occlusion with the need for emergency coronary by-pass grafting the least frequent. Likewise, there are clinical scenarios that increase the probability of complications related to IVUS. On the other hand, there are novel imaging predictors of CP such as the C-CAT sign, which, although it still needs to be validated in larger studies, alerts the interventional cardiologist to the risk of CP. The severity of the perforation, according to the Ellis classification, and the clinical status will determine patient management. Finally, knowledge of the protocol for the management of an intraprocedural complication together with teamwork significantly reduces patient mortality.
Supplemental Material
Supplemental Material
Supplemental Material
Supplemental Material
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
