Abstract
Introduction
In this study, we aimed to investigate the relationship between coronary slow flow (CSF) and carotid-femoral pulse wave velocity (CFPWV).
Methods
78 (27 women, mean age 43.95 ± 7.28) patients with CSF, and 70 (22 women, mean age 44.34 ± 7.08) healthy individuals were included in the study. Arterial stiffness measurement was performed to both groups via CFPWV, which is considered the gold standard. Aortic elastic properties (ASI-β and aortic distensibility) were evaluated in both groups.
Results
The CSF group had significantly higher CFPWV and aortic distensibility values and significantly lower ASI-β values compared to the control group. There was a positive correlation between TIMI frame count (TFC) obtained in all coronary arteries and CFPWV and aortic distensibility, and a negative correlation between TFC and ASI-β. It was determined that CFPWV predicted CSF with 97% specificity and 98% sensitivity at a 7.68 cut-off value (ROC area = 994, p < 0.001). ASI-β was determined to predict CSF with 64% specificity and 47% sensitivity at a 2.98 cut-off value (ROC area = 047, p < 0.001). Aortic distensibility was determined to predict CSF with 76% specificity and 79% sensitivity at a 3.94 cut-off value (ROC area = 706, p < 0.001).
Conclusion
Arterial stiffness increases in CSF patients, suggesting that CSF is a systemic pathology rather than a local disease and that a systemic cause such as atherosclerosis plays a role in etiology.
Keywords
Introduction
Coronary slow flow (CSF) is the case of the late removal of opaque material from non-atherosclerotic coronary arteries. The etiology of CSF is not clearly known. However, inflammation, increased microvascular resistance, and microvascular endothelial dysfunction have been accused as the primary causes. 1
Arterial stiffness shows the flexibility and distensibility of the arteries. 2 The deformation of especially the media layer of the arteries causes an increase in vascular stiffness and a decrease in vascular distensibility. Arterial stiffness has been shown to be impaired in many diseases such as atherosclerosis, aging, diabetes mellitus (DM), rheumatoid arthritis, and hypertension.3–5
Among the different methods for evaluating arterial stiffness, carotid-femoral pulse wave velocity (CFPWV) is the gold standard method. CFPWV is an easy-to-perform and reliable test that can detect cardiovascular events independent of conventional risk factors. 6
Most patients with CSF have been shown to have atheroma plaques that do not cause intimal thickening, diffuse calcification, or lumen irregularity in the coronary arteries.7–8 In light of this information, it is necessary to evaluate coronary slow flow phenomenon (CSFP) as a coronary artery disease. 1
Atherosclerosis is a disease that affects all vessels. Since it is possibly a common disease, atherosclerotic changes at any level of the arterial system can provide information about atherosclerosis in other arteries.
Since the peripheral arterial system may be affected in patients with CSFP, we aimed to investigate the relationship between CSFP and arterial stiffness using the CFPWV and aortic elastic properties.
Methods
The study was conducted prospectively between December 2018 and December 2019 in Cardiology Department of Niğde Ömer Halisdemir University Medicine Faculty Education and Research Hospital. 78 patients diagnosed with CSF on coronary angiography were included in the study and named as the CSF group. Age and gender matched 70 subjects with normal coronary angiographies were included in the control group. Routine biochemical tests were performed before coronary angiography and anamnesis was taken for the drugs used.
Patients who had abnormal sinus rhythm, left or right bundle branch block, peripheral arterial disease, LVEF <50%, cardiomyopathy (restrictive, hypertrophic, dilated cardiomyopathy), moderate-severe valve disease, arterial blood pressure >140/90 despite antihypertensive therapy, connective tissue diseases, active infectious diseases, chronic obstructive pulmonary disease, malignancy and systemic diseases affecting the aorta (such as Marfan, or Ehler-Danlos), hematological, interstitial pulmonary disease, or glomerular filtration rate <30 ml/min 1.73m2 were excluded.
The study was conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent was obtained from each participant. The study protocol was approved by the local Clinical Research Ethics Committee.
Echocardiographic examination
Echocardiographic evaluations were performed by the same cardiologist using a 3.5-MHz transducer (EPIQ 7, Philips Healthcare, Andover, Mass., USA). The cardiologist was blind to the clinical diagnosis and status of the patients. Participants were evaluated after 10 minutes of rest. Systolic and diastolic blood pressures and pulses were evaluated before measurements. All measurements were based on American Echocardiography Society standards. 9 Left ventricular ejection fraction (LVEF) was calculated according to the Simpson’s method. Pulse wave mitral flow velocities were obtained from the apical four chamber view after the sample volume was placed on the mitral leaflet tips. Mitral early diastolic velocity (E, cm/s), late diastolic velocity (A, cm/s), E/A ratio, E deceleration time (DT, ms), and isovolumetric relaxation time (IVRT, ms) were calculated from the apical view. E’ velocities were taken by tissue Doppler imaging from the lateral and septal aspects of the mitral annulus, and their mean value was evaluated as E' mean (E’m).
To assess aortic stiffness, systolic and diastolic diameters of the ascending aorta were measured by M-mode echocardiography up to 3 cm above the aortic valve in parasternal long-axis images. Aortic diameters were calculated by taking the distances between the anterior and posterior wall inner edges of the aorta in systole and diastole. Aortic systolic diameter (AoSD) was obtained with the aortic valve fully open. The diastolic diameter of the aorta (AoDD) was taken simultaneously with the peak of the QRS on electrocardiography.
Calculation of aortic elasticity values
Aortic stiffness index-β (ASI-β) was used as a marker of aortic elasticity in all assessments. Relative diameter change in pressure increase was evaluated as aortic distensibility.
10
Aortic strain = (AoSD-AoDD)/AoDD ASI-β = log (Systolic blood pressure/Diastolic blood pressure)/Aortic strain Aortic distensibility= (2×Aortic strain)/(Systolic blood pressure/Diastolic blood pressure)
Coronary angiography
Selective coronary angiography (SCAG) was performed using 6 French (F) catheters with a femoral or radial artery approach. Coronary arteries were visualized in left and right oblique planes with cranial and caudal angles in all patients. A mean of 6–8 ml of contrast medium was injected manually for each exposure and coronary arteries were imaged at a rate of 25 fps.
TIMI frame count
TIMI frame count (TFC) method was used to detect and grade CSF. TFC was evaluated by two different, experienced cardiologists. If there is a difference in the TIMI frame numbers obtained, these values were averaged. The anterograde flow of the arterial lumen was accepted as the first frame when the first branch was almost completely filled and the last frame was accepted as the opaque at the distal endpoint of the artery. The distal bifurcation point in the apex was used to evaluate LAD, the onset of the first major branch of the crux or posterolateral extension was used to evaluate RCA and the distal separation point of the trunk or large obtuse marginal was used to evaluate CX. 11 The corrected LAD TFC was found to be divided by a coefficient of 1.7. Normal TFC was accepted as 36 for LAD, 20 for RCA, and 22 for CX. If these values were more than 2 standard deviations, CSF was accepted. 11
Arterial stiffness measurement
Arterial stiffness was performed by the same cardiologist using the SphymoCor® system (AtCor Medical, Sydney, Australia). Blood pressure, height, and weight values were measured after resting for 10 minutes. Patients were connected to the electrocardiography (ECG) leads of the SphymoCor® system. In addition to patient information for device measurement settings, the distance between the carotid and femoral measurement sites was measured by tape measure. The distance between the carotid and femoral sites to be measured was recorded in mm. Pulse wave records were made sequentially from the carotid artery and then from the femoral arteries. The device averaged at 10 cardiac cycles. The time between the R wave on ECG and the beginning of the pulse wave was calculated as the transit time. The transit time was divided by the distance between the carotid and femoral regions to obtain the CFPWV value (m/s).
Statistical analysis
Statistical analysis was performed using the SPSS 23.0 software. All values are given as mean ± SD. The Independent Samples t-test was used to examine the mean differences between CSF and NCA groups. The Chi-Squared test was used to examine the relationships between categorical variables. The ROC analysis was used to determine the diagnostic value of ASI-β, CFPWV, and aortic distensibility in patients with CSF. Pearson’s Correlation analysis was performed to evaluate the relation between ASI-β, CFPWV, and aortic distensibility in patients with CSF. p < 0.05 was considered significant for all analyses.
Results
The clinical and demographic characteristics of the study groups are given in Table 1. Age, sex, body mass index (BMI), presence of diabetes mellitus (Type 1 or 2), dyslipidemia and HT, smoking status, systolic, diastolic and mean blood pressure, and heart rate showed no difference between the two groups (Table 1).
Comparison of clinical and demographic characteristics of CSF and control groups.
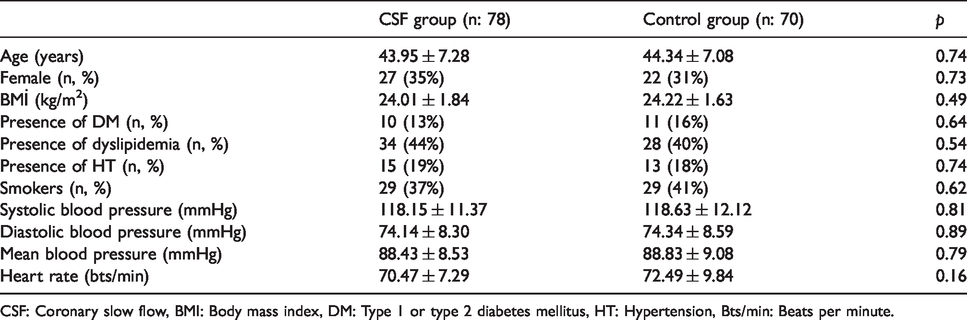
CSF: Coronary slow flow, BMI: Body mass index, DM: Type 1 or type 2 diabetes mellitus, HT: Hypertension, Bts/min: Beats per minute.
In laboratory analyses, there was no significant difference between the two groups in terms of biochemical parameters such as fasting glucose, urea, creatinine, total cholesterol, triglyceride, HDL cholesterol, and LDL cholesterol levels (Table 2).
Comparison of laboratory findings of CSF and control groups.

CSF: Coronary slow flow, HDLc: High-density lipoprotein cholesterol, LDLc: Low-density lipoprotein cholesterol.
There was no significant difference between the groups in terms of the use of drugs such as β-blocker, statin, nitrate, calcium channel blocker (CCB), angiotensin receptor blocker (ARB), and angiotensin converting enzyme inhibitor (ACEI) (Table 3).
Comparison of drug treatments of CSF and control groups.

CSF: Coronary slow flow, CCB: Calcium channel blocker, ARB: Angiotensin receptor blocker, ACEI: Angiotensin converting enzyme inhibitor.
There was no significant difference between the groups in terms of left ventricular ejection fraction (LVEF), left ventricular end-systolic (LVESD) and end-diastolic diameters (LVEDD), interventricular septum diameter (IVSD), peak early diastolic velocity (E), deceleration time (DT), isovolumetric relaxation time (IVRT), and E/E’m. Peak late diastolic velocity (A) was higher in the CSF group than the control group and the ratio of peak early to peak late diastolic velocities (E/A) was lower in the CSF group (Table 4).
Echocardiographic parameters of CSF and control groups.

CSF: Coronary slow flow, LVEF: Left ventricular ejection fraction, LVEDD: Left ventricular end-diastolic dimension, LVESD: Left ventricular end-systolic dimension, IVSD: Interventricular septum diameter, E: Peak early diastolic velocity, A: Peak late diastolic velocity, E/A: Ratio of peak early to peak late diastolic velocities, DT: Deceleration time, IVRT: Isovolumetric relaxation time, E/E’m: Septal and lateral E/E’ means.
As expected, TFC obtained by evaluating coronary angiographies was significantly higher in the CSF group than in the control group. In the CSF group, Corrected LAD TFC was 41.09 ± 3.96, CX TFC was 35.36 ± 6.72 and RCA TFC was 24.20 ± 5.30 (Table 5).
Comparison of TIMI frame counts (TFC) of CSF and control groups.

CSF: Coronary slow flow, LAD: Left anterior descending artery, TFC: TIMI frame count, CX: Circumflex artery, RCA: Right coronary artery.
Comparing the CFPWV, ASI-β, and aortic distensibility values of the CSF and control groups, there was a significant difference between the groups. According to these findings, CFPWV and aortic distensibility were higher and ASI-β was lower in the CSF group (Table 6).
Comparison of CFPWV, ASI-β and aortic distensibility of CSF and control groups.

CSF: Coronary slow flow, CFPWV: Carotid-femoral pulse wave velocity, ASI-β: Aortic stiffness index-β.
The relationship between TFC and CFPWV and aortic elasticity values (ASI-β, aortic distensibility) in the CSF group was also examined. There was a positive correlation between TFC obtained in all coronary arteries and CFPWV and aortic distensibility, and a negative correlation between TFC and ASI-β (Table 7).
Correlation between TIMI frame counts (TFC) and CFPWV, ASI-β, and Aortic distensibility in the CSF group.

CFPWV: Carotid-femoral pulse wave velocity, ASI-β: Aortic stiffness index-β, LAD: Left anterior descending artery, TFC: TIMI frame count, CX: Circumflex artery, RCA: Right coronary artery.
**p < 0.01.
The receiver operating characteristic curve (ROC) analysis was performed for the role of CFPWV, ASI-β, and aortic distensibility in predicting CSF. In this analysis, it was determined that CFPWV predicted CSF with 98% sensitivity and 97% specificity at a 7.68 cut-off value (ROC area = 994, p < 0.001). ASI-β was determined to predict CSF with 47% sensitivity and 64% specificity at a 2.98 cut-off value (ROC area = 047, p < 0.001). Aortic distensibility was determined to predict CSF with 79% sensitivity and 76% specificity at a 3.94 cut-off value (ROC area = 706, p < 0.001) (Figure 1).

Relationship between CSF and CFPWV, ASI-β, and Aortic distensibility (ROC analysis).
Discussion
Three main results were obtained as a result of the study. Firstly, patients with CSFP had higher CFPWV and ASI-β values than the control group. Secondly, in the CSF group, a statistically significant positive correlation was observed between the number of LAD corrected TFC, CX TFC, and RCA TFC and CFPWV. Finally, the CFPWV method is more sensitive and specific than ASI- β and aortic distensibility in predicting CSF.
The clinical course of atherosclerosis and its interaction with risk factors vary considerably from patient to patient, starting from the subclinical period. Endothelial dysfunction is an early sign of cardiovascular disease. Endothelial dysfunction is thought to play an important role in prognosis. It has long been observed that pathologies in large arteries cause cardiovascular events. Many studies have been conducted on the onset and progression of pathological changes in the arteries. Non-invasive evaluation of the great arteries and the increase in the information about the elastic and anatomical properties will help shed light on this issue.
CSFP is characterized by a delay in the flow of opaque material without any lesions causing stenosis of the epicardial coronary vessels during coronary angiography. Although CSFP has been described in many studies, it is still not a well-known clinical condition. The pathogenesis of the disease is responsible for increased vasoconstrictor response, microvascular dysfunction, endothelial dysfunction, increased endothelin-1, functional contraction caused by capillary edema, and tonus increase. The slowing of coronary blood flow can be expressed objectively and numerically by the TFC method. Because of the absence of atherosclerotic lesions on coronary angiography, coronary artery wall structures of CSFP patients were considered normal. The reason for CSFP was the increase in microvascular resistance. However, with the development and more frequent use of intravascular ultrasonography (IVUS), it was observed that calcifications and atheromatous changes were mostly present in the vascular wall of these patients. 7 As a result of these findings, the idea that CSFP should be considered as a subtype of coronary artery disease (CAD) was put forward. They evaluated these findings in the epicardial coronary arteries as part of diffuse atherosclerosis affecting the whole arterial system.
A relationship between atherosclerosis and arterial stiffness may have attracted the attention of researchers for a long time. 12 Increased PWV values have been shown to be associated with atherosclerosis and coronary artery disease. Patients with high aortic PWV values have been demonstrated to have more atherosclerotic lesions in the carotid arteries and lower extremity arteries and abdominal aorta compared to those with low PWV. 13 Contrary to these studies, we investigated the relationship between arterial stiffness and CSFP in non-atherosclerotic CSF patients.
In our study, we used the CFPWV method, which is accepted as the gold standard method for the measurement of arterial stiffness, and the aortic stiffness method measured by echocardiography. Previously, intravascular ultrasonography coronary artery intima-media thickness determination and TFC and intracoronary pressure measurement were detected in patients with CSF. 8 Endothelial dysfunction in coronary arterioles due to atherosclerosis in the early phase, decreased vasodilator reserve, and manifest atherosclerosis should be monitored in these patients.
In our study, we found that CFPWV values, which are the non-invasively gold standard indicator of arterial stiffness, were statistically significantly higher in the CSF patient group than in the control group. These findings suggest that CSF may be an early indicator of subclinical atherosclerosis and endothelial dysfunction. In addition, it can indicate the presence of a common pathology that affects both coronary arteries and systemic arteries in CSF patients. In fact, it has been shown that histopathological changes leading to increased arterial stiffness occur simultaneously in the coronary arteries with the arterial system. 14
Left ventricular relaxation and filling disorder is the earliest finding of myocardial ischemia. 15 It has been shown that left ventricular filling is impaired by the PW doppler method in patients with CFS and that E velocity and E/A ratio decrease, A velocity increases, and isovolumetric relaxation time (IVRT) increases. 16 In patients with CSF whose left ventricular diastolic function was evaluated with Tissue Doppler, diastolic velocities were taken only from the lateral and septal regions of the mitral annulus, and CSF was shown to lead to impaired left ventricular diastolic functions at the tissue level in these regions. 16 In our study, there was a statistically significant increase in A velocity and a decrease in the E/A ratio in the CSF group compared to the NCA group. There was no statistically significant difference between the groups in terms of parameters such as E velocity, IVRT, or deceleration time. Our findings show that similar to the results of previous studies, left ventricular diastolic function is impaired in patients with CSF.
Previous studies have shown that PWV is associated with diastolic dysfunction, in line with our study. 17 The relationship between arterial stiffness and left ventricular diastolic dysfunction is not clearly known. Microvascular ischemia, as previously demonstrated in CSF, is thought to play a role in the pathogenesis of systemic pathology that causes an increase in arterial stiffness.
It has been shown that there is a relationship between the presence of CAD and deterioration in the elastic parameters of the aorta. 18 A relationship has been found between the presence and severity of CAD and the aortic distensibility and strain. 19 Aortic pulse pressure and pulsatility index have been shown to be increased in patients with CSF. 20 They evaluated aortic stiffness by echocardiography in patients with CSF and showed that aortic stiffness increased in patients with CSF. 21 We also examined the relationship between aortic stiffness and CSF echocardiographic measurements besides the CFPWV method. We found ASI-β to be significantly higher in patients with CSF compared to the control group. We also found that CFPWV is a better determinant for CSF than ASI-β and aortic distensibility.
Conclusion
There is an increase in arterial stiffness in endothelial dysfunction, which is one of the early signs of atherosclerosis. Mortality and morbidity can be reduced by early detection of stiffness and appropriate treatment. Our findings show that CSFP should be considered as a CAD subgroup and that an increase in CFPWV is predictive. As a simple and inexpensive method, CFPWV can be used for this purpose.
Study limitations
In our study, the exclusion of pathologies such as pulmonary and renal diseases, connective tissue diseases, acute infectious and inflammatory cases, and peripheral arterial disease affecting arterial stiffness, as well as the small number of patients may have affected our findings. Therefore, more accurate results can be obtained by further studies with more patients to determine the role of PWV in predicting CSF.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study protocol was approved by the Niğde Ömer Halisdemir University Clinical Research Ethics Committee. Written informed consent was obtained from each study participant. The study was conducted in accordance with the principles of the Declaration of Helsinki.
Guarantor
HA is the guarantor and accepts full responsibility for the work and/or the conduct of the study, had access to the data, and controlled the decision to publish.
Contributorship
HA: study design, acquisition of data, writing and drafting the manuscript, and final approval; guarantor of the study, correspondence author. EEG: study design, acquisition of data, writing and drafting the manuscript, and final approval.
