Abstract
Objectives
To demonstrate how heart rate fragmentation gives novel insights into non-autonomic mechanisms of beat-to-beat variability in cycle length, and predicts survival of cardiology clinic patients, over and above traditional clinical risk factors and measures of heart rate variability.
Introduction
Precisely adaptive control of the heart’s beating rate and the resulting variability in beat-to-beat intervals is a hallmark of health. This variability is the integrated product of many factors, including extrinsic autonomic neurological control, factors intrinsic to cardiac tissue, and even variability in beating that occurs on a single sinus node cardiomyocyte level. 1 Changes in physical or emotional states, whether slow or abrupt, physiological or pathological, mandate a range of incessant small and large adjustments in the time of arrival of the next heartbeat, to optimize cardiac output to the body’s requirements at that exact point in time. Certain mechanisms underlying heart rate variability (HRV) are relatively easily detected, for example autonomic driven respiratory sinus arrhythmia can be revealed in the frequency spectrum of the heart rates or the times between beats, the RR intervals. Accordingly, quantification of HRV has traditionally been performed in the frequency domain, where the premise is that the amount of variance localized in discrete frequency bands can be ascribed to the action of the parasympathetic (high-frequency, HF, >0.4 Hz or 24/minute) and sympathetic (low-frequency, LF, 0.15 or 9/minute to 0.4 Hz) arms of the autonomic nervous system. 2
The heart rate frequency spectrum, though, reveals a great deal of variance outside of the respiratory frequency. While some of this represents changes in sympathetic nervous system activity, and some represents respiratory sinus arrhythmia (RSA) not well-localized because the respiratory rate is constantly changing, it is now reasonable to suppose that some of this variance is due to intrinsic variability at the level of sinoatrial node (SAN) tissue or indeed the individual SAN myocytes that comprise this remarkable structure. Contemporary evidence suggests substantial beat-to-beat variation in the functioning of the ensemble of sarcolemmal ion channels making up the membrane clock along with variation in the behavior of the ion channels, Ca2+ release channels and Ca2+ pumps comprising the Ca2+ clock, together comprising the coupled clock system that drives robust and flexible automaticity in SAN cells.1,3–5Beat-to-beat variation in the number of available channels and interaction with other regulatory molecules (e.g., in the case of SERCA, with phospholamban) is likely to underlie HRV in these denervated cardiac preparations. Since ion channel gating is stochastic, we might expect aperiodic characteristics of this very finely adaptive control, without a characteristic peak in the frequency spectrum.
In keeping with the idea that innate sinus node dynamics might be important, Costa and Goldberger recently introduced new analytics of short-term HRV that they call heart rate fragmentation,6,7 to quantify the previously described phenomenon of erratic sinus rhythm. 8 These measures quantify the phenomenon of small, aperiodic fluctuations in heart rate that are not easily explained by our understanding of extrinsic regulation of the SAN by the autonomic nervous system. Semi-quantitative analyses of erratic sinus rhythm have pointed to an adverse clinical effect of these fluctuations, and the metrics of Costa and Goldberger indeed are associated with worse prognosis in the MESA study. 9 The new measures number four: the percentage of inflection points (PIP), or points at which the first difference changes sign, the percentage of short segments (PSS), the inverse of the average segment length (IALS) and the percentage of alternating segments (PAS). For each, a larger value denotes a more fragmented heart rate structure.
The aims of our study were as follows. Firstly, to validate and characterize the novel heart rate fragmentation measures in a large, clinically replete dataset of 24-hour Holter recordings. Thus, we examined their relationship to age from 0 to 100 years, and correlated the fragmentation indices with both traditional and novel HRV measures. To examine mechanisms of fragmentation, we measured the effect of ectopy and atrial fibrillation, and tested the idea that fragmentation represents only undersampling of RSA by the heartbeat. Secondly, we tested how heart rate fragmentation and local dynamics score added independent information about survival in this cohort of cardiology patients.
Methods
Study population
2893 Holter recordings collected at the University of Virginia from patients of all ages were studied. In particular, we manually over read 1571 recordings of patients aged 40 and over because of the higher incidence of chronic heart disease and atrial fibrillation.10–12 Some patients had multiple Holter studies done, in which case only the first Holter was included in this study. Additionally, labels for AF, premature atrial contraction (PAC) or premature ventricular contraction (PVC) were acquired either from the Philips Holter software or our own manual corrections. 10
Patients were placed into one of three categories as follows: atrial fibrillation (when the arrhythmia occupied more than 5% of the record), sinus rhythm with ectopy (when ectopic beats occupied more than 10% of the record) or normal sinus rhythm.
In addition to the Holter recordings, other demographic data were collected for the subset of patients over 40, such as age, gender, and the presence of comorbidities including hypertension, hyperlipidemia, CHF, diabetes mellitus, history of tobacco use and the presence of cardiac pacemaker as recorded during visits to University of Virginia cardiologists. These demographics were described in detail for this group in previous work from our group – see Table 1 in Moss et al. 11 The (younger) group of patients were less exhaustively clinically characterized, but did have an average age of 23.0 years, and 546 (44.9%) were male.
Univariate and multivariate predictors of mortality in Cox proportional hazards models of the 1321 patients of age 40 or greater who were in normal sinus rhythm during the Holter recording.
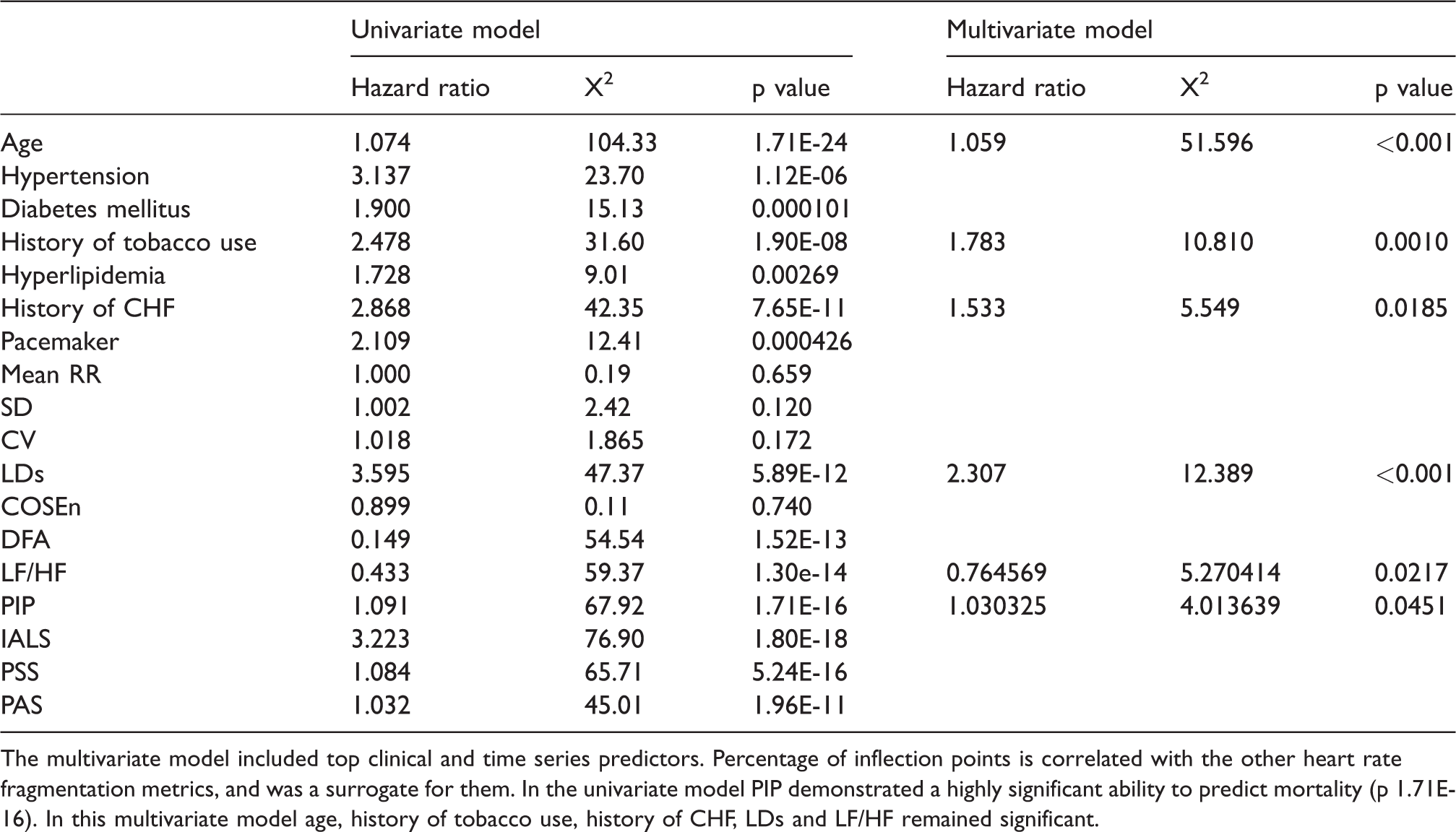
The multivariate model included top clinical and time series predictors. Percentage of inflection points is correlated with the other heart rate fragmentation metrics, and was a surrogate for them. In the univariate model PIP demonstrated a highly significant ability to predict mortality (p 1.71E-16). In this multivariate model age, history of tobacco use, history of CHF, LDs and LF/HF remained significant.
Heart rate variability metrics
Descriptive statistics
We computed the mean and standard deviation (SD) for 30 s RR interval time series, averaged results for each 10 min segment and then averaged over the entire 24 hours. The coefficient of variation (CV) was calculated every 10 minutes and then averaged over the 24 hours.
Heart rate fragmentation metrics
For every 24-hour RR interval time series, the differences between consecutive RR intervals, increments, were derived as follows: ΔRRi=RRi-RRi-1. When ΔRR is zero, the heart rate is constant and the ΔRR value remains 0. However, when ΔRR is smaller than zero, the increment is counted as an acceleration and the ΔRR value is replaced by −1. When ΔRR is larger than zero, the increment is counted as a deceleration and the ΔRR value is replaced by 1. From this ternary time series of values −1, 0 and 1, the four HR fragmentation metrics were calculated for the 24-hour record.
We excluded ΔRR values of >1 second or if they exceeded 4 SD of the ΔRR for the individual record. We excluded segments of <10 consecutive delta RR intervals. These steps resulted in excluding 1.8% of the overall data from all patients. We used the entire RR interval time series, not just the NN intervals, as this does not affect the impact of the fragmentation metrics. 6
Percentage of inflection points (PIP)
PIP is the fraction of RR intervals representing inflection points out of all RR intervals. An inflection is counted when there is any change in number in the ternary time series, for example from −1 to 0, −1 to 1 or 0 to 1.
Inverse of average segment length (IALS)
An acceleration/deceleration segment is a sequence of the same consecutive number; an acceleration segment is a sequence of −1’s and a deceleration segment is a sequence of 1’s. The length of a segment is the number of increments in that segment. IALS is the inverse of the average segment length.
Percentage of short segments (PSS)
PSS is the complement of the percentage of long segments. A long segment is an acceleration/deceleration segment with a length of at least three increments. The percentage of long segments is the number of increments in long segments divided by the length of the RR time series.
Percentage of alternation segments (PAS)
An alternation segment is a sequence of at least four increments where the ternary time series changes every step from −1 to 1 or vice versa, i.e. a sequence with the pattern “−1 1 –1 1”. The percentage of alternation segments (PAS) is the number of increments in these sequences divided by the length of the RR time series.
Coefficient of sample entropy (COSEn)
COSEn is a measure derived from the sample entropy (SampEn 13 ) optimized to detect AF in very short RR interval time series. 10 SampEn is the negative logarithm of the conditional probability that two sequences that match within tolerance r for m points will also match within r at the next point, where self matches are excluded in the calculation of the probability. Lake provided the quadratic entropy rate to correct for the value of the tolerance r, 14 and Lake and Moorman provided the coefficient of sample entropy for use in optimal estimation of entropy of human heartbeat time series. 10
Local dynamics score (LDS)
This measure of the dynamics of short RR time series is a function of the number of beats that are either very similar or very dissimilar to the surrounding beats. It is derived from a proportional hazards model relating these values to survival in this data set.11 We calculated LDS every 10 minutes, and then averaged over 24 hours.
Detrended fluctuation analysis
DFA exponent alpha quantifies the correlation properties of a time series. 15 Our group has shown that DFA is an efficient detector of ectopy, and was useful as part of a strategy for atrial fibrillation detection. 12 The DFA slope was calculated every 10 minutes over box sizes 4 to 12 (consistent with Costa and Goldberger6,7,9) and averaged over the 24-hour period.
Frequency domain analysis
We implemented the Lomb periodogram within the Physionet Cardiovascular Signal Toolbox, and report HRV quantified in the low-frequency (LF, 0.04 to 0.15 Hz), and high-frequency (HF, 0.15 to 0.4 Hz) domains of the power spectrum.
Statistical analysis
We calculated Pearson’s correlation coefficients of the heart rate fragmentation metrics among themselves as well as with the other nonlinear short-term dynamical metrics.
To test the statistical significance of age- and time-of-day-related changes in heart rate fragmentation metrics, we performed polynomial regression, a form of multiple linear regression where the terms are the zeroth to fourth powers of the dependent variable. We examined the confidence intervals of each coefficient to see if they included 0. If they did not, we interpreted that the trend was statistically significant.
In the survival analyses, only patients with age greater than 40 and sinus rhythm were included (n = 1321). To test association of various predictors with survival, univariate and multivariate Cox proportional hazards models were applied by using the coxphfit function in MATLAB R2018a, censoring patients not reaching an outcome at the last documented follow-up date.
To determine the features that resulted in a patient death within two years, a random forest model was implemented using the Random Forest Classifier module within Python’s scikit-learn library. In the analysis, 34 patients were excluded due to missing data in one or more feature variables. As a result 1284 patients were included in the random forest. The random forest model was trained on 70% of the data (n = 898) and constructed with 100 decision trees. Each tree was created by drawing a random bootstrap sample from the training set with replacement, and grown by selecting a random number of features at each node without replacement. Gini Impurity, defined as the total decrease in node impurity, was then used to calculate the feature importance of the random forest. Gini impurity is calculated for each feature by counting the number of times that feature is used to split a node across all trees, proportionally weighted by the total number of samples it splits.
Results
HR fragmentation decreases in early life, before increasing after the second decade of life
Figure 1 shows the four novel HR fragmentation metrics plotted as a function of patient age, with each dot representing a patient, with red dots for those patients who died within 2 years, and blue dots for those who survived the same time course. All fragmentation metrics followed the same trend with patient age, that is a decrease from birth continuing in early life before increasing once the patient reaches their 20’s, a trend that then continued throughout life. In all cases, 2 or more of the coefficients of a 4th order polynomial regression were significantly different from zero, indicating that the association of the fragmentation metrics with age is significant.
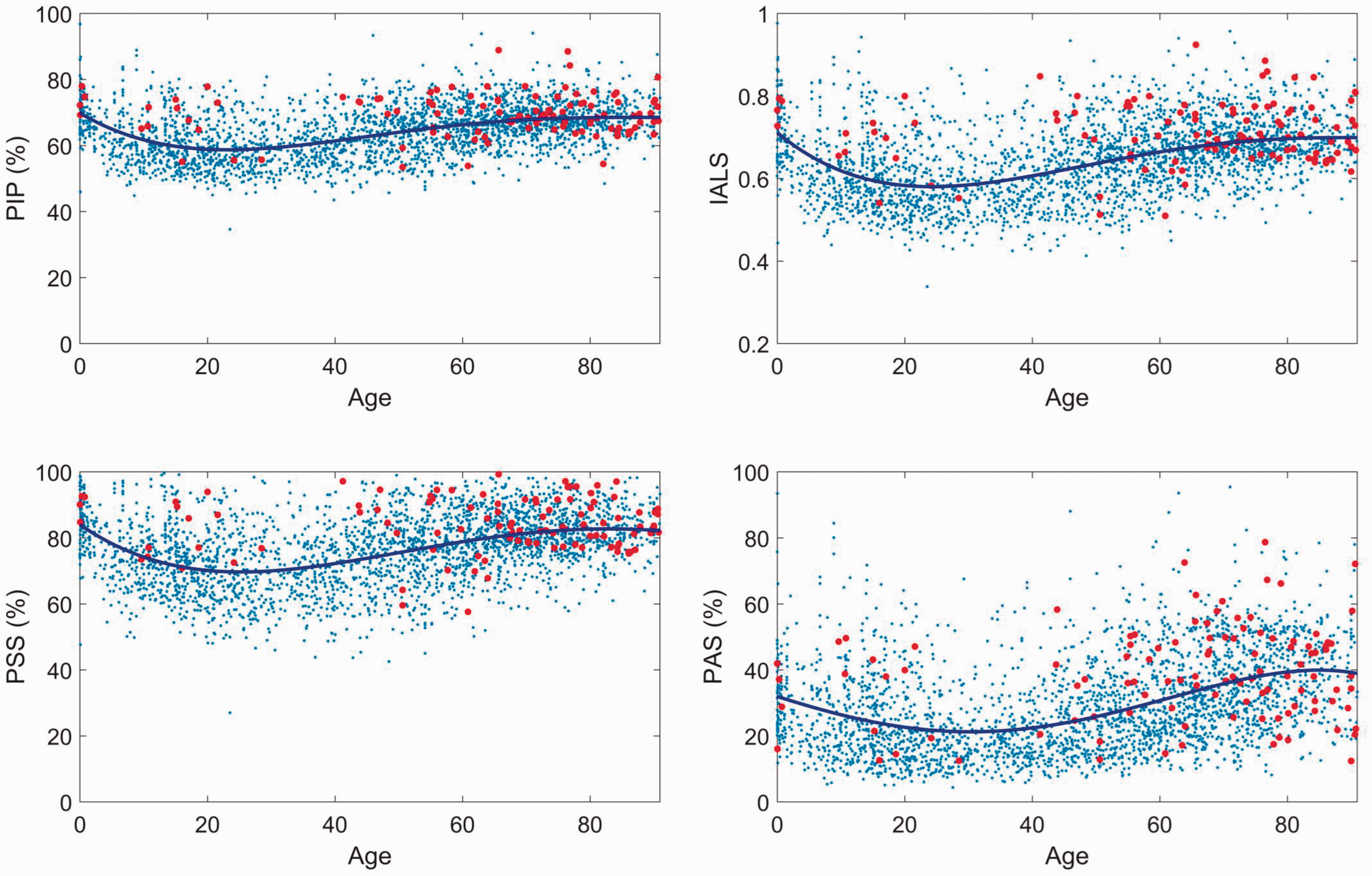
Heart rate fragmentation metrics plotted as a function of age for 2983 patients who had 24-hour ambulatory Holter EKG monitoring. Blue lines are fourth order polynomials fitted to the data. Red dots mark patients who died within 2 years of the Holter recording. PIP = percentage of inflection points, IALS = inverse of average segment length, PSS = percentage of short segments, PAS = percentage of alternating segments. Coefficients of polynomial fit were significantly different from 0 (p < 0.05), signifying a statistically significant association of the metrics with age.
Clinical ectopy burden does not markedly contribute to HR fragmentation metrics
Figure 2 shows HR fragmentation metrics plotted as a function of percentage of ectopic beats (regardless of whether the ectopy was ventricular or supraventricular) contained within the 24 hour recording. The metrics were calculated on 10-minute windows. In the typical clinical range of 0 to 20% ectopy, PIP, IALS and PSS increased only by 3.5, 0.040, and 1.9%, respectively. PAS increased more, by 14.1%. These relative changes in PIP, IALS and PSS are small, leading us to conclude that the effect of clinical levels of ectopy on these measures of heart rate fragmentation is negligible.

Ectopy increases heart rate fragmentation metrics. Results are plotted in bins of width 2%; lines are means, and ribbons are 95% CI. The effects are modest over the clinically observed burdens up to 20%.
HR fragmentation metrics are fixed for atrial fibrillation
We note that metrics measured in patients with AF were closely grouped between patients – each AF record had a PIP around 66%, IALS around 0.67, PSS around 80% and PAS around 50%. Gaussian random numbers had similar results (not shown).
HR fragmentation metrics are correlated among themselves and with other HRV metrics
Figure 3 shows a color-coded matrix of the correlations between the novel HR fragmentation parameters (first 4 rows and first 4 columns) and the other established HRV metrics included in this study, for 3 patient groups: those in atrial fibrillation (left column), those in normal sinus rhythm (middle), and those in normal sinus rhythm with more than 10% ectopy (right). The dark colors in the upper left corner represent very strong correlations among the heart rate fragmentation metrics themselves, r > 0.9, with the exception of PAS, which is only moderately strongly correlated (0.6<r < 0.8). HR fragmentation metrics are strongly positively correlated with LDs, moderately negatively correlated with LF/HF and DFA, and weakly negatively correlated with COSEn.

Correlation among the HRV metrics in this study. Top row shows the absolute values, and the bottom row gives the signed values; the gray scale and color scale are shown to the right of each table. Left, records with more than 5% atrial fibrillation; middle, normal sinus rhythm; right, normal sinus rhythm with more than 10% ectopy.
Generally, COSEn was least correlated with the other measures – it was not correlated at all with LF/HF, DFA and PAS, and only weakly negatively correlated with LDs, PIP, PSS and IALS. LF/HF was weakly correlated with LDs but strongly correlated with DFA.
HR fragmentation metrics have circadian rhythm in sinus rhythm but not atrial fibrillation
Figure 4 shows 24 hour plots of the HR fragmentation metrics measured every 10 minutes for one example patient in sinus rhythm, and a further example patient with persistent atrial fibrillation. In SR, there is more variability across the 24 hours, especially in nocturnal hours. There is virtually no circadian variation in HR fragmentation metrics in the patient with AF. Further, note that the HR fragmentation metrics of the AF patient are very close to the mean of the AF group, indicated by the red line, suggesting that the presence of AF renders HR fragmentation parameters void and nondiscriminatory with respect to time of day in the same patient, or indeed between patients.

Circadian rhythm of heart rate fragmentation metrics in a patient with normal sinus rhythm (left) and a patient with persistent atrial fibrillation (right). Holter recordings are affixed during the day, and the middle section of the plot represents the night. The red line is the group mean for patients with atrial fibrillation. While fragmentation of normal sinus rhythm varies, in atrial fibrillation it does not.
Figure 5 shows plots of the mean and SD of the HR fragmentation metrics, measured every 10 minutes over 24 hours, of all patients who had either NSR over 90% of the time (blue lines) or AF over 90% of the time (red lines). In NSR, 2 or more of the coefficients of polynomial regression were significantly different from zero, meaning that there was circadian rhythmicity in parameters of heart rate fragmentation. In AF, there was no discernible circadian rhythmicity. Accordingly, for AF patients, the variances were much less than for SR patients.
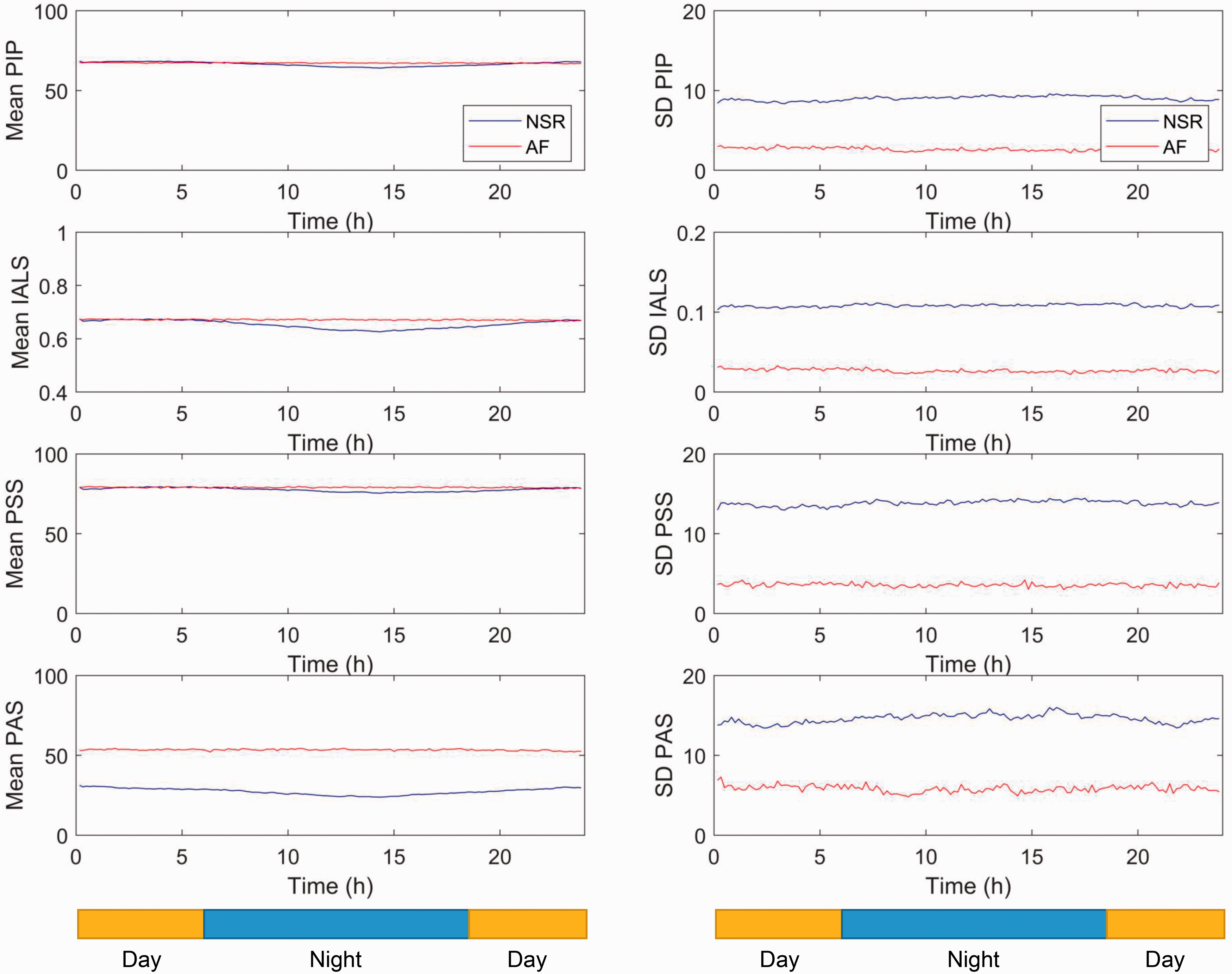
Means and standard deviations of heart rate fragmentation metrics. In atrial fibrillation (red), the heart rate fragmentation index values were not greatly different from normal sinus rhythm, but they showed no circadian rhythm, and had much lower variability.
Is heart rate fragmentation a result of under-sampling respiratory sinus arrhythmia?
Figure 6 shows results of numerical experiments on a sine wave of frequency 12 per minute, mimicking respiration. The sampling rate is shown on the x-axis, and is analogous to the heart rate. Because the breathing rate/sine wave has a frequency of 12/min, the Nyquist frequency is 24 beats per minute. The plot shows that the HR fragmentation metrics have a range of values that depend on the sampling rate (i.e. on the heart rate), and are much higher at lower heart rates (often peaking at sampling/heart rates of 30–40). However, at usual human resting heart rates of near 70 beats per minute the predicted fragmentation metrics are markedly lower than observed in the Holter data, suggesting that the observed human heart rate fragmentation represents more than just undersampling of periodic RSA, and must instead represent some other physiological phenomena influencing the heart’s rhythm on a beat-to-beat basis.

Heart rate fragmentation is not solely due to undersampling of RSA. Numerical experiments on a sine wave of frequency 12 per minute with varying heart rate. There is increased fragmentation for percentage of inflection points and inverse of average segment length at heart rates even exceeding 100 bpm. No increased fragmentation is present in percentage of short segments at rates above 75 bpm, nor in percentage of alternating segments at rates about 40 bpm.
HR fragmentation metrics are associated with survival
Figure 7 shows patient survival based on quintiles of HR fragmentation metrics. More fragmentation (higher quintile number) was associated with progressively poorer survival.

Survival as a function of quintile of heart rate fragmentation metrics. More fragmentation was associated with poorer survival.
Table 1 shows the univariate and multivariate predictors in a Cox proportional hazards model. In the univariate models (2nd thru 4th columns), heart rate, standard deviation of heart rate, and COSEn all failed to reach statistical significance affirming our previous assertion that COSEn has its main value in atrial fibrillation detection from short segments10,16 rather than as a predictor of mortality. In the univariate model, PIP (chosen to represent all other heart rate fragmentation measures) demonstrated a highly significant ability to predict mortality (p = 1.71E-16).
We selected the 3 most significant clinical predictors of death within 2 years – age, tobacco usage and congestive heart failure – and the 3 most significant time series analytics – local dynamics score, ratio of low to high frequency content of the heart rate time series, and HR fragmentation, represented in this instance by PIP. All were statistically significant in the multivariable model (Table 1).
To test the robustness of these results, we used a random forest modelling approach, and Figure 8 shows the relative importance of the predictor variables. This analytical approach shows near-equal importance of the HR fragmentation parameters with the established time series metrics, each of which exceeds the importance of all the clinical risk factors, except age.

Variable importance in the Random Forest model. With the exception of age, time series metrics including heart rate fragmentation were more important than clinical variables.
Discussion
We have studied novel parameters of heart rate fragmentation in a real world scenario, utilizing a large clinically well characterized set of patients seen by cardiologists in the out-patient setting with 24-hour ambulatory ECG monitoring. We have confirmed the prior finding of Costa et al.6,7,9 that heart rate fragmentation increases with age in patients 40 years and older. However, using our population that included young people we have added the novel finding that parameters of heart rate fragmentation actually decrease initially in early life, before beginning to increase in the third decade of life, a trend that continues throughout the rest of life, with an ultimate plateauing in old age. We have also confirmed their previous finding that heart rate fragmentation is a strong predictor of future mortality, with low levels of heart rate fragmentation conferring high survival rates, and the heart rate fragmentation parameters adding independent information to the more traditional clinical factors and frequency domain HRV measures.
Ectopic beats, whether coming from the atria, ventricles or the AV junction, have a profound effect on the analysis and interpretation of traditional HRV parameters, and yet are a fact of life, being almost ubiquitous in continuous 24 hour recordings from both young and old individuals. We have shown in this new work that ectopy up to a clinically relevant burden of 20% had only small effects on measures of heart rate fragmentation. This has important connotations for the usability and fidelity of heart rate fragmentation in real world clinical situations which will almost certainly contain ectopy.
Traditional HRV parameters are of little use in the context of atrial fibrillation. We investigated whether heart rate fragmentation parameters would be any more useful when the heart’s predominant rhythm was AF. We have shown however that heart rate fragmentation in atrial fibrillation is fixed and unvarying – it is extremely similar across individuals, and does not show any evident circadian rhythmicity within individuals, unlike in sinus rhythm where we have uniquely shown that all heart rate fragmentation parameters demonstrate significant circadian rhythmicity.
We found that HR fragmentation parameters are very strongly correlated with each other, and so it may be sufficient to only select a single heart rate fragmentation parameter to represent all the others, rather than calculating all the 4, simplifying workflow. Importantly, we have shown that the observed levels of HR fragmentation observed in our recordings is more than could be expected if the observed fragmentation were related solely or in large part to the undersampling of RSA, suggesting that some as yet uncharacterized physiological phenomenon/phenomena is responsible for heart rate fragmentation. This mandates further study.
External validation of HR fragmentation metrics
Costa and Goldberger developed HR fragmentation metrics to capture the phenotype of frequent small variations in heartbeat intervals, faster than the familiar RSA, originally described by Stein et al. as erratic sinus rhythm. In three seminal papers, Costa and Goldberger showed that increased fragmentation had clinical association with death in the CAST trial (Stein), with age and coronary disease in the THEW data set, and with cardiovascular death in the MESA study (Costa, Goldberger). Here, we add a new data set, and extend the findings to compare heart rate fragmentation to other non-linear measures as well as traditional time- and frequency-domain ones.
We tested the idea that heart rate fragmentation might arise from undersampling of RSA – it does not. In fact, a clinically unfeasible heart rate of over 200 beats per minute is required given a respiration rate of 12 per minute to reduce fragmentation to less than 10% (see Figure 6), but clinically usual heart rates of 70 to 90 beats per minute would be expected to yield heart rate fragmentation metrics much less than observed if the fragmentation were due solely to RSA. For example, the PIP would be 25–30% if heart rate fragmentation arose only from the RSA mechanism at such clinically normal resting heart rates. Instead, the average observed PIP exceeds 60%. From this analysis, we conclude that heart rate fragmentation does not uniquely arise from undersampling of the respiratory rate by the sinus node, and instead arises from some as yet incompletely characterized physiological process.
Heart rate fragmentation in atrial fibrillation
Heart beat interval time series in atrial fibrillation have numerical properties of nearly-Gaussian random numbers. We found fixed and unvarying values of heart rate fragmentation in the clinical records of patients with atrial fibrillation. Thus, as a prognostic indicator, heart rate fragmentation has no value in these patients. Moreover, the average fragmentation values in atrial fibrillation are not greatly different than in sinus rhythm, and they do not seem promising as targeted atrial fibrillation detectors, except perhaps that their very low variance with time might be diagnostic.
Impact of HR fragmentation and local dynamics on survival
The prognostic value of HRV measures in the time and frequency domains was established in the 1980s by Kleiger, Bigger and others in landmark studies of patients after myocardial infarction, and a large literature has followed. 17 We found that these new heart rate fragmentation measures add information independently to established time and frequency domain measures with respect to their ability to predict future mortality (Figure 8), suggesting that aperiodic dynamics operating across a range of heart rates and variances can inform on the future clinical status of patient.
What mechanisms underlie heart rate fragmentation parameters if they are not due to autonomic signaling to the heart?
The mechanisms underlying fragmentation in the heart rate time series reflect a smoothly adaptive system that changes heart rate on a beat-to-beat basis in the young and healthy, but that becomes dysfunctional and fragmented with age. The time-scale of such fragmentation is faster than that which could be accounted for by the functioning of the autonomic nervous system. The mechanisms underlying the development of this fragmentation are not known, but the aperiodic nature of the time series, and the unvarying PQRST waveform suggests that they arise from altered microregulation of the SA node. The question remains, what mechanism or mechanisms might be predominantly responsible for this microregulation (see Figure 9)? Detailed consideration of potential contributors are beyond the scope of this paper, but would include extracellular, sarcolemmal and intracellular mechanisms.

schematic figure of the factors influencing beat to beat variation in behavior of the SAN. The figure depicts important modifiers of SAN function at the level of the whole heart, the level of the intact SAN, and at the level of individual SAN myocytes. See text for details.
Extracellular mechanisms impacting beta-to-beat variation in SAN functioning include the renin-angiotensin-aldosterone system, 18 nitric oxide signaling 19 and reactive oxygen species, 20 the latter also representing an intracellular mechanism of beat-to-beat SAN regulation.
Sarcolemmal mechanisms that impact beat-to-beat fluctuation in SAN beating rate include variation in behavior/availability/number of autonomic receptors on the cell surface leading to beat to beat variability, 21 along with variation in the so-called membrane clock.1,22 This latter group would include variation in number/availability/gating/conductance through ion channels on a beat to beat basis.23,24 SAN cells are known to exhibit marked heterogeneity in their electrophysiological characteristics; 25 the cells of the SAN that lead pacemaking vary on a beat-to-beat basis; 22 and there is a putative loss of SAN cells with age. 26 Thus, there are multiple ways in which heart rate fragmentation may arise from the cellular electrophysiology and biology of the SAN cell membrane.
Finally, intracellular mechanisms exhibit variable behavior on a beat-to-beat basis including variation in the calcium clock i.e. variation in number/availability/probability of opening of L- and T-type Ca2+ channels, RyRs, and SERCA.3,5,27,28 There will also be beat-to-beat variation in modulators of both membrane and calcium clocks - protein kinase A (PKA), Ca2+-calmodulin kinase II (CAMKII) and protein kinase C (PKC), along with beat-to-beat variation in dephosphorylation mechanisms (protein phosphatases and phosphodiesterases 29 ) There will also be variation in the behavior of other regulatory molecules such as phospholamban24,30 and variation within the energy state of the cell, including variation in mitochondrial content and behavior. 31
We have shown in previous work 32 that the variability of increasingly reductionist components of the SAN behave with increasing degrees of variability, for example SAN cells demonstrate markedly more variability in beating than does the whole isolated spontaneously beating SAN in vitro. This may be a manifestation uncoupling of the cells, not only from the autonomic nervous system, but also from each other. Could then HR fragmentation be reflective of progressive uncoupling of the cells of the SAN with age, or even uncoupling of individual components of the coupled clock system from each other, leading to random threshold crossings, and thus fragmented interbeat intervals?
Is all the information contained in the heart rate?
A recent Journal of Physiology CrossTalk series pitted two extreme points of view about HRV analysis. Malik et al. hold that the order of heart beat intervals – and, in particular, the periodic nature of their order, as measured in the frequency domain – has important information about the autonomic nervous system. 33 Boyett et al., on the other hand, hold that HRV is merely an innocent bystander to heart rate, which holds all the information. 34
We have another point of view – fine control of the heart rate results from clinically important sinus node physiology and pathophysiology that may or may not be controlled by the autonomic nervous system, but is most certainly not well-quantified by the mean or the frequency spectrum. We justify this point of view in part by the fact that the only clinical application of HRV analysis that improves outcomes is heart rate characteristic monitoring for early detection of sepsis in premature infants. There, the abnormal finding is reduced variability and transient decelerations, which alters neither the mean 35 nor the frequency spectrum. 36
Limitations
We studied Holter recordings of patients who visited cardiologists, and thus the landscape of heart disease and cardiac drugs will not match that of the general population. We used only the basic metrics of heart rate fragmentation – percentage of inflection points, inverse of average segment length, percentage of short segments, and percentage of alternating segments – but have not tested the refinements - symbolic dynamics, and “hard” VS “soft” inflection points – that followed (Costa 2017 b).
Conclusion
Heart rate fragmentation is a useful addition to the battery of techniques currently available for the characterization of complexities in the heart’s rhythm that are not visible to the naked eye, and that reveal important information concerning mechanisms of heart rate control on a beat to beat basis. The mechanisms giving rise to heart rate fragmentation cannot be easily accounted for by autonomic signaling to the heart, and are likely reflective of fine tuning of the heart’s rhythm by the many non-autonomic mechanisms illustrated in Figure 9. Many of these are intrinsic to the SAN and its individual cells, though the magnitude to which these factors contribute to HR fragmentation requires further study. Other critical characteristics of HR fragmentation, including its relationship to age, time of day and morbidity/mortality are described. Heart rate fragmentation parameters are robust compared to many other HRV parameters, which are markedly affected by the presence of ectopy and prevailing heart rate.
Footnotes
Acknowledgements
None.
Contributorship
All authors contributed equally to the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
None.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
Randall Moorman MD.
