Abstract
Syncope associated with bradycardia and ventricular arrhythmia is an indication of cardiac intervention. However, in adolescent patients with anorexia nervosa, the management of syncope and arrhythmia can be different. We present a case of a 17-year-old boy who was admitted to the hospital because of syncope during exercise. Electrocardiographic monitoring showed that his mean heart rate was 41 beats/minute, with many long pauses and frequent premature ventricular contractions. These results suggested that the syncope was probably caused by arrythmia. He had been on a diet and had lost 20 kg in the past 6 months, with a body mass index of only 15.3 kg/m2. He was diagnosed with anorexia nervosa. Pacemaker implantation or ablation was not performed. Refeeding therapy was performed with mirtazapine. A follow-up showed a stepwise increase in his heart rate and a stepwise decrease in premature ventricular contractions, with an increase in his body weight. The findings from this case show that vagal hyperactivity associated with anorexia nervosa might lead to multiple premature ventricular contractions and bradycardia.
Background
Syncope during exercise is highly indicative of the presence of heart disease, including myocardial ischemia, structural heart disease and arrhythmia. Severe bradycardia and ventricular arrhythmia can lead to syncope of all ages, and usually indicates the necessity of cardiac interventions, such as pacemaker/defibrillator implantation and radiofrequency ablation, even for adolescents.1,2 However, if a patient has anorexia nervosa, the management for arrhythmia might be different to that for other patients. We present a case of an adolescent who was diagnosed with anorexia nervosa and complex arrythmia, in whom the heart rhythm returned to normal after refeeding therapy.
Case presentation
A 17-year-old male student was sent to our hospital because of syncope. He had felt dizzy during running after class, and then passed out, fell on the ground and regained consciousness within 1 minute. No limb twitch, gatism, jaw chomping or foaming at the mouth was reported by his classmates. He claimed to have had no chest pain, dyspnea, palpitation, headache or other symptoms after the syncope and could move as usual. He was the only child of his family and grew up healthy with no history of chronic disease or hereditary disease, and behaved well in school with no obvious conflict with his classmates. He had been on a diet, frequently vomited after eating and lost for more than 20 kg in the past 6 months, with intermittent dizziness in the daytime during physical exercise or walking.
At admission, his heart rate was 43 beats/minute (bpm) and his blood pressure was 95/63 mmHg with no obvious postural hypotension. His body weight was 47 kg, height was 175 cm and body mass index was 15.3 kg/m2. A physical examination also showed that his heart beat was slow and irregular, but no other positive signs were identified.
At presentation, his initial electrocardiogram showed considerable sinus bradycardia with premature ventricular contractions (PVCs), and a normal P wave, PR and QT intervals (Figure 1). The size of the left ventricle was near the lower limit, and mild pericardial effusion was identified by an echocardiogram. Results of blood tests showed that the blood cell count, serum electrocyte, creatinine, cortisol and adrenocorticotrophic hormone concentrations at 8 am, and cardiac enzyme, cardiac troponin-T, brain natriuretic peptide and thyroid-stimulating hormone concentrations were normal. The free triiodothyronine concentration was 2.9 pmol/L, the total triiodothyronine concentration was 0.52 mmol/L, and the free thyroxine concentration was 7.8 mmol/L. The results of a tilt test were negative. The results of brain magnetic resonance imaging and chest computed tomography were normal.

Features of premature ventricular contractions on a 12-lead electrocardiogram.
A 24-hour ambulatory electrocardiogram was performed. Considerable sinus bradycardia with paroxysmal junctional and ventricular escape beats, a heart rate of 21 to 122 bpm and a mean heart rate of 41 bpm were observed. Additionally, 5900 long RR intervals (longer than 2 s) were recorded and the longest RR interval was 3.9 s, and 7426 PVCs (13.2% of all beats) were recorded with a unified form (Figure 2). A bedside electrocardiographic monitor showed that the longest RR interval was 5.9 s at approximately 1 am during sleep. No atrioventricular block or ventricular tachycardia was captured.
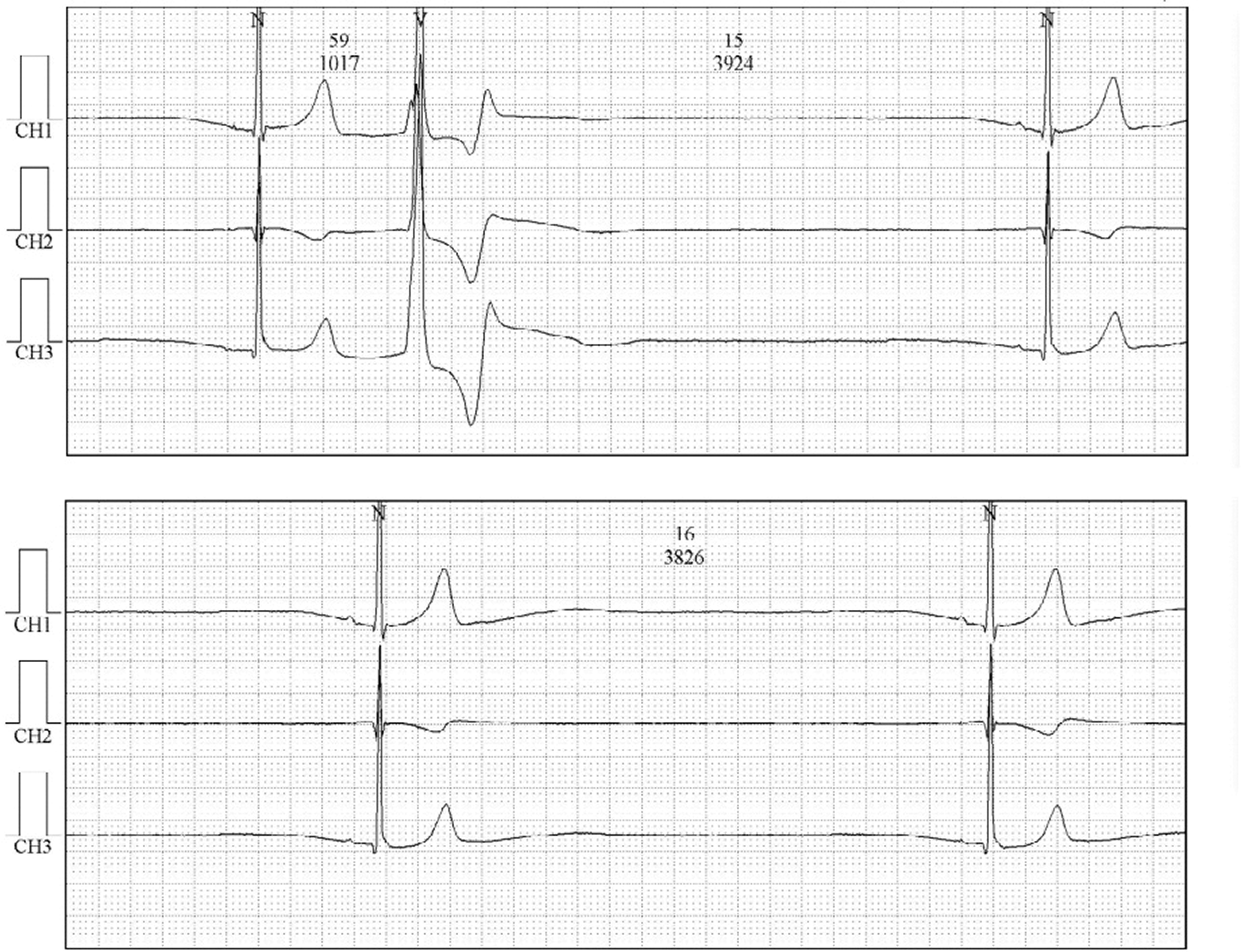
A 24-hour electrocardiogram shows a long pause and a premature ventricular contraction.
The results of the examinations suggested that the syncope and dizziness were probably caused by considerable bradycardia and the long pauses. However, pacemaker implantation was not performed immediately. Radiofrequency ablation was also not performed because there was no proof of ventricular tachycardia. A psychiatrist confirmed the diagnosis of anorexia nervosa (AN), and encouraged the patient to eat and prescribed mirtazapine. One week later, he could eat food and took mirtazapine (30 mg each night) regularly without obvious vomiting. He was then discharged with a prescription of mirtazapine for 1 month.
One month after discharge, his body weight had increased to 55 kg. A 24-hour electrocardiogram showed that his mean heart rate was 54 bpm, 177 long RR intervals were recorded and the longest RR interval was 3.0 s, and 3247 PVCs were recorded. No syncope and less frequent dizziness were reported. The patient stated that he had a good appetite without vomiting and then administration of mirtazapine was ceased. Six months later, his body weight had increased to 60 kg. A 24-hour electrocardiogram showed that his mean heart rate was 61 bpm, 2 long RR intervals were recorded and 918 PVCs were recorded. One year later, his body weight was 64 kg. A 24-hour electrocardiogram showed that his mean heart rate was 64 bpm. No long RR interval was recorded and five PVCs were recorded. No syncope or dizziness was reported.
Discussion
AN is an eating disorder characterized by a fear of eating and gaining weight, voluntarily restriction of food intake and a low body weight. 3 This condition is relatively unfamiliar to cardiologists. AN might lead to a series of cardiac complications, including hypotension, arrythmia and structural heart disease.4,5 Sinus bradycardia is relatively common in patients with AN, as shown by a study of long-term cardiac monitoring, 6 and is sometimes symptomatic as found in our case. However, pacemaker implantation is not usually recommended for patients with AN, even though their heart rate can be less than 30 bpm with frequent long pauses. This lack of recommendation is because bradycardia might be caused by increased vagal tone, which is an adaptation to the hypometabolic state of AN and can be reversed after refeeding therapy. 7
The frequent PVCs observed in our patient were unusual. A ventricular escape beat is common in patients with severe bradycardia. However in this case, the dominant ventricular beats were PVCs, which is rare, as reported by a previous study of long-term cardiac monitoring. 6 The pathogenesis of the PVCs in this case is unclear. AN might lead to cardiomyopathy, which mainly presents as a decreased ventricular mass, and decreased cardiac output 4 and myocardial fibrosis.8,9 Therefore, there could be an association between AN and ventricular arrhythmia. However, in this case, the PVCs had a unified morphology, including large R wave on leads II, III and aVF, and precordial lead transition on lead V3. This finding indicated that the PVCs might have originated from the left ventricular outflow tract and were less likely to be caused by cardiomyopathy. However, the PVCs might have been induced by increased vagal tone. Although most PVCs are positively associated with sympathetic activity, a previous study showed that 10% of PVCs were more sensitive to parasympathetic activity. 10 In this case, a 24-hour electrocardiogram showed that PVCs in the night time were more frequent than those in the day time. Additionally, the number of PVCs decreased in a stepwise manner with the elevation in mean heart rate. We speculate that, with the improvement in nutrition in our patient, vagal hyperactivity decreased and this then led to the decrease in PVCs.
The findings in this case suggest that PVCs are indicative of vagal hyperactivity caused by AN together with considerable bradycardia.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
The data collection and publication of this case report were approved by the Ethics Committee of the Fourth Affiliated Hospital, Zhejiang University School of Medicine (reference number: 20190057). Written consent was obtained from the patient and his parents.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (No. 81600393, 81971688) and a grant from the Education Department of Zhejiang Province (No. Y201942034), which funded for the process of data collection, laboratory examinations and publication of the manuscript.
