Abstract
Objectives
The aim of this study was to present a single-centre experience with EndoAnchors in patients who underwent endovascular repair for abdominal aortic aneurysms with challenging proximal neck, both in the prevention and treatment of endograft migration and type Ia endoleaks.
Methods
We retrospectively analysed 17 consecutive patients treated with EndoAnchors between June 2015 and May 2018 at our institution. EndoAnchors were applied during the initial endovascular aneurysm repair procedure (primary implant) to prevent proximal neck complications in difficult anatomies (nine patients), and in the follow-up after aneurysm exclusion (secondary implant) to correct type Ia endoleak and/or stent-graft migration (eight patients).
Results
Mean time for anchors implant was 23 min (range 12–41), with a mean of 5 EndoAnchors deployed per patient. Six patients in the secondary implant group required a proximal cuff due to stent-graft migration ≥10 mm. Technical success was achieved in all cases, with no complications related to deployment of the anchors. At a median follow-up of 13 months (range 4–39, interquartile range 9–20), there were no aneurysm-related deaths or aneurysm ruptures, and all patients were free from reinterventions. CT-scan surveillance showed no evidence of type Ia endoleak, anchors dislodgement or stent-graft migration, with a mean reduction of aneurysm diameter of 0.4 mm (range 0–19); there was no sac growth or aortic neck enlargement in any case.
Conclusions
EndoAnchors can be safely used in the prevention and treatment of type Ia endoleaks in patients with challenging aortic necks, with good results in terms of sac exclusion and diameter reduction in the mid-term follow-up.
Introduction
The proximal aortic neck still represents one of the challenges in endoluminal repair for abdominal aortic aneurysm (AAA): the risk of type Ia endoleak and endograft migration is increased in patients with short, large-diameter, conical or highly angulated necks, particularly in the presence of thrombus or calcium.1–7
Over the years, many different techniques have been used to manage a hostile proximal sealing site, including chimney procedures, fenestrated stent-grafts, and the use of standard stent-grafts with the adjunct of EndoAnchors.8–12
Mimicking the security of a hand-sewn aortic anastomosis, EndoAnchors are intended to provide fixation and sealing between endovascular grafts and the native aortic wall at the level of the proximal attachment site, with good assistance in the prevention and management of type Ia endoleaks and stent-graft migration in patients with challenging aortic necks.
The aim of this study is to present a single-centre experience with the use of the Heli-FX EndoAnchor System (Medtronic Vascular, Santa Rosa, CA, USA) in patients who underwent endovascular aneurysm repair (EVAR) for AAA, both in the prevention of proximal sealing site complications and for the treatment of type Ia endoleaks as well.
Materials and methods
This is a retrospective study that followed the principles outlined in the Declaration of Helsinki and used only information obtained from the review of medical charts and CT-scan images analysis. Patients gave written consent for the anonymous collection of their data on the standard consent sheet provided for by our institution. Data collection was carried out in conformity with the Italian laws on privacy (Art. 20–21, DL 196/2003) published in the Official Journal, volume 190, 14 August 2004, which explicitly exempts the need for ethical approval for the use of anonymous data.
We keep a database of all patients undergoing surgical interventions at our institution to observe their follow-up. We retrospectively analysed 17 consecutive patients treated with EndoAnchors in elective setting between June 2015 and May 2018. EndoAnchors were applied during the initial EVAR procedure (primary implant) to prevent proximal sealing site complications in nine patients with hostile proximal aortic neck anatomy (Figure 1), and in the follow-up after EVAR (secondary implant) to correct postprocedural type Ia endoleak and/or stent-graft migration (eight patients). Proximal aortic neck features for each patient in both groups are described in Table 1. The mean neck length and neck diameter were, respectively, 9 mm/29 mm in the primary implant group and 12 mm/30 mm in the secondary implant group, whereas thrombus and calcium were present in 44% of the primary implant patients and in 35% of the revision procedures. Significant angulation at the proximal neck was more frequent in the secondary implant group (25% of cases) compared to prophylactic procedures (11%). Conical shape neck was present in 22% of the primary implant cases and in 25% of the revisions. Technical success was defined as correct deployment of the anchors at the target site for both primary and secondary implant, and endoleak resolution in cases of secondary implant. Considering the small number of patients, no subgroups were analysed, and statistical analysis was not performed.
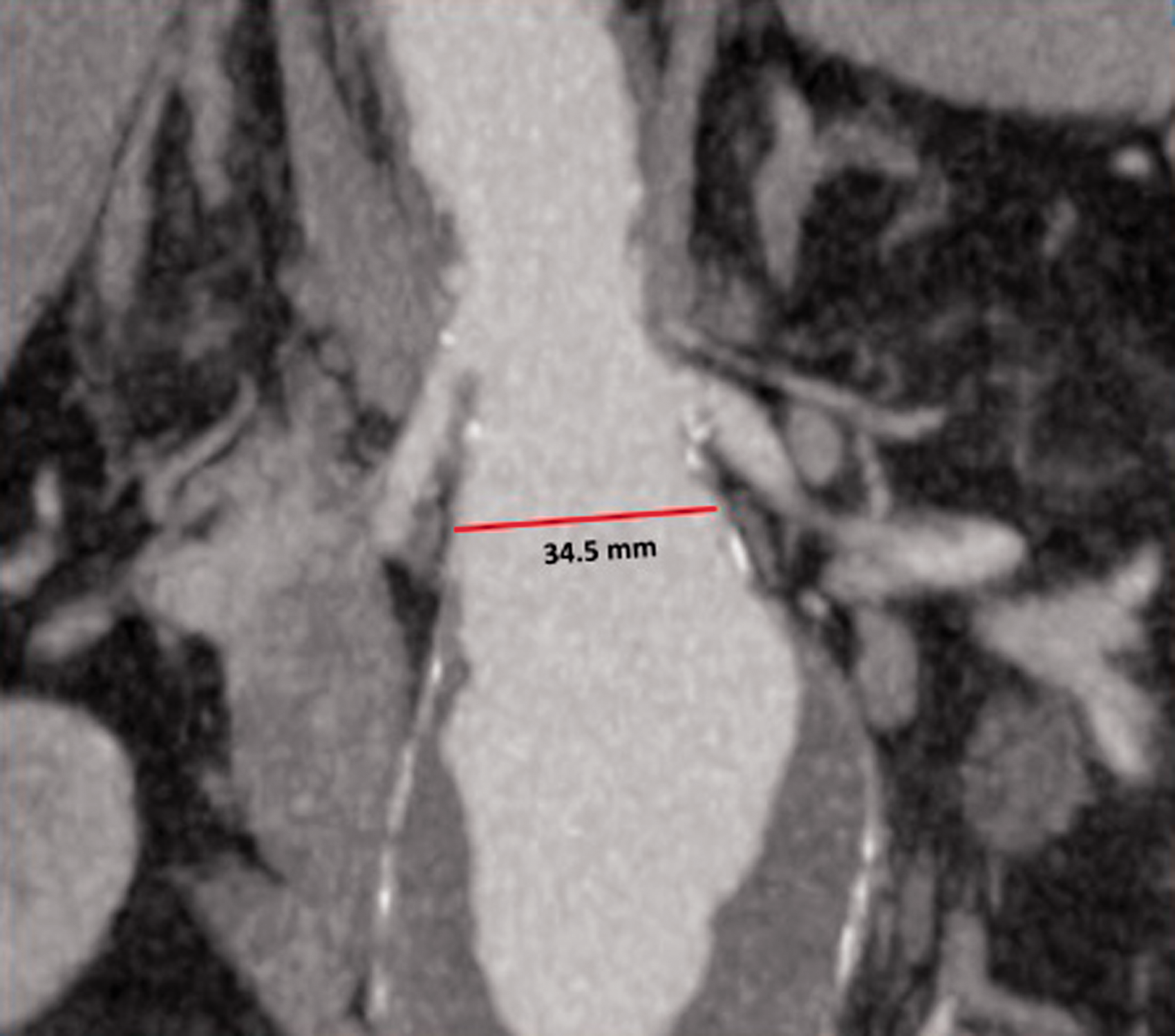
A challenging proximal aortic neck: short, wide, and reverse-tapered.
Proximal aortic neck features.

−: absent; +: present, less than half the circumference; ++: present, more than half the circumference.
Device description and technique
The Heli-FX EndoAnchor system comprises an electronically controlled applier (Heli-FX applier) with 10 helical anchors provided in a cassette and a deflectable 16F outer profile sheath (Heli-FX guide) designed for the delivery of the applier to the target location for anchors implantation. Each EndoAnchor is manufactured from MP35N-LT (nickel-cobalt-chromium alloy) and is 4.5 mm long with a diameter of 3 mm. In our standard technique, a 16F introducer sheath is inserted over a stiff guidewire from the femoral artery and advanced up to the infrarenal aorta, to give support and stability. The Heli-FX guide sheath is then positioned through the introducer just below the renal arteries and the Heli-FX applier is inserted inside it. By rotating and deflecting the guide sheath with the appropriate orientation under fluoroscopic guidance, four to six EndoAnchors are usually deployed along the aortic circumference just at the proximal edge of the stent-graft fabric, with the possibility of applying a second row below the first one. Each anchor is implanted in a two-stage process that allows retraction of the anchor and possible repositioning before final deployment.
This system has been tested with selected endografts: Zenith (Cook, Bloomington, IN, USA), Excluder (W.L. Gore, Flagstaff, AZ, USA), E-vita (Cryolife, Kennesaw, GA, United States) and AneuRx, Talent, Endurant (Medtronic Vascular, Santa Rosa, CA, USA) devices.
Operative details
Aortic anatomy and morphology were preoperatively assessed by thin-cut (0.5 mm) contrast-enhanced computed tomography (CT). Procedural planning was carried out using Aycan OsiriX PRO Workstation (Aycan Inc., Wuerzburg, Germany) to post-process CT images. The angles for the correct C-arm orientation to be used during the intervention were identified on the preoperative CT-scan in order to achieve a precise and effective clock-face positioning of the anchors along the stent-graft circumference (Figure 2).

Preoperative CT-scan images analysis, with identification of the angles of C-arm orientation to be used for fluoroscopic guided clock-face positioning of the anchors.
The interventions were carried out in the operative room preferably under local anaesthesia, according to patients’ clinical features and compliance, with the aid of a latest generation C-arm fluoroscopy/angiography equipment (Ziehm Vision RFD Hybrid, Ziehm Imaging, Nuremberg, Germany). The EndoAnchors procedure was done immediately after the stent-graft deployment in the nine patients with primary implant. Only one femoral access was used for the Heli-FX guide sheath insertion, deploying the anchors along the entire neck circumference with homogeneous distribution. In cases of stent-graft migration with the need of a proximal cuff, first the EndoAnchors were applied to the old stent-graft, when possible, to prevent further slipping, then the aortic cuff was deployed from the contralateral femoral access and secured to the aortic wall with additional anchors just below the renal arteries.
Completion angiography was performed at the end of the procedure in each patient (Figure 3). Technical success was defined as the deployment of the desired number of EndoAnchors at the target site with an adequate penetration into the vessel wall, and by the absence of persistent contrast filling of the aneurysmal sac at the end of the intervention in case of indication for endoleak correction.

Intraoperative angiography showing a type Ia endoleak (a and b, arrows) that is completely fixed after EndoAnchors deployment (c).
Prior to discharge, patients were evaluated with clinical examination and Duplex ultrasound. Contrast-enhanced CT-scan was performed one month after the procedure in all cases and thereafter on a yearly basis, according to individual patient’s needs (Figure 4).
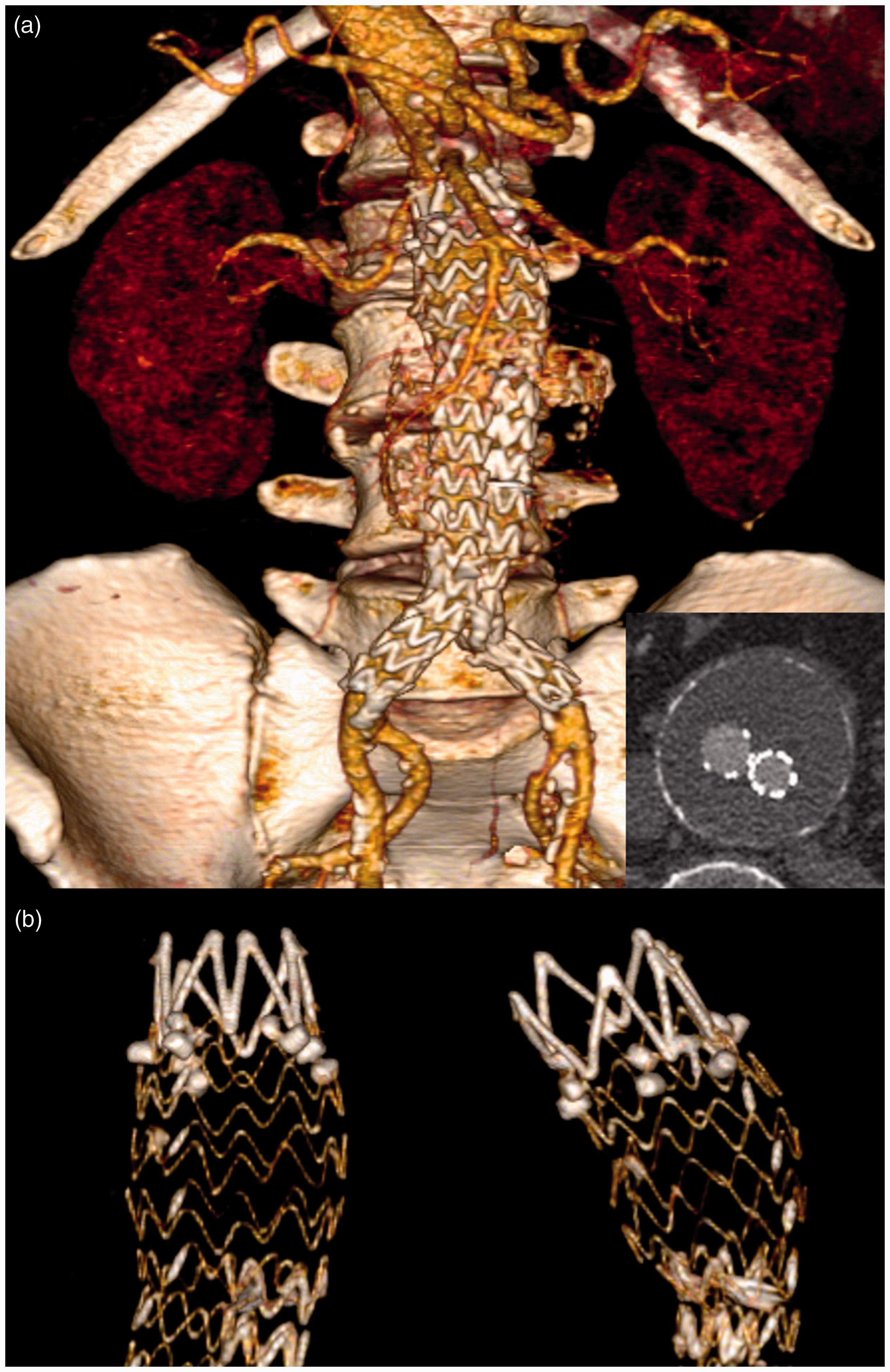
Post-operative CT-scan, confirming good stent-graft position with aneurysm exclusion (a) and effective anchors implant (b).
Results
Mean AAA diameter at the time of operation was 60 mm (range 43–88 mm). A bimodular endograft system (Medtronic Endurant II) was used in all the nine cases of primary implant, whereas a previous bimodular or trimodular stent-graft system was present in the eight cases of secondary implant (three Medtronic Endurant, two Medtronic Talent, three Cook Zenith).
Interventions were performed in 13 cases under local anaesthesia and in 4 cases under general anaesthesia. No conversion from local to general anaesthesia was required in any case.
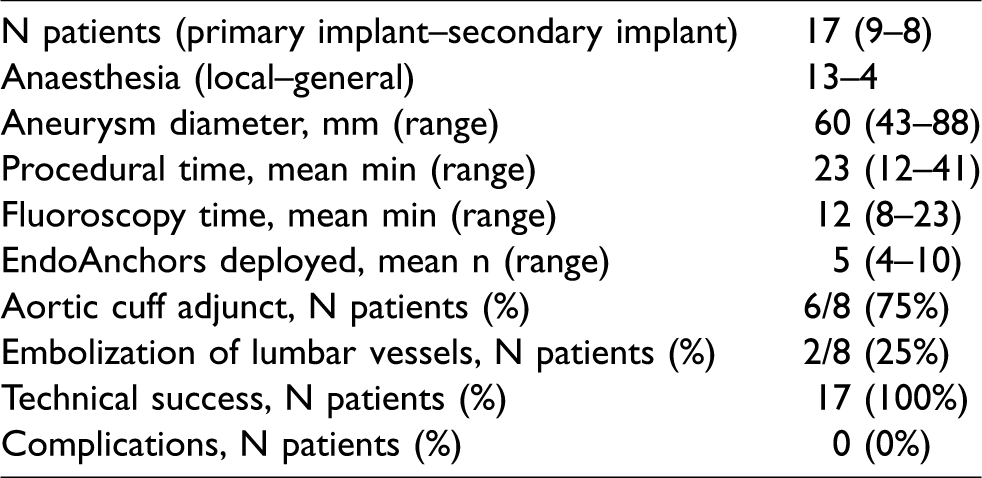
The mean procedural time for anchors implant was 23 min (range 12–41 min), with a mean fluoroscopy time of 12 min (range 8–23 min). We deployed a mean of 5 EndoAnchors per patient (range 4–10). Six patients (75%) in the secondary implant group required the addition of a proximal aortic cuff due to stent-graft migration ≥10 mm. In two cases of secondary implant, embolization of lumbar arteries was performed at the time of the anchors procedure. Technical success was achieved in all cases. We did not observe any intra-operative or post-operative complications related to anchors deployment (Table 2).
Procedural data.
At a median follow-up of 13 months (range 4–39, interquartile range 9–20), there were no aneurysm-related deaths or aneurysm ruptures, and all patients were free from reinterventions.
CT-scan surveillance showed no evidence of type Ia endoleak, anchors dislodgement or stent-graft migration. We observed a mean reduction of aneurysm diameter of 0.4 mm (range 0–19), with no evidence of sac growth or aortic neck enlargement in any case.
Discussion
One of the most relevant challenges of EVAR remains the proximal sealing zone below the renal arteries. Aortic neck diameter wider than 32 mm, neck length shorter than 10 mm, neck angulation of more than 60°, reverse-tapered shape, and the presence of thrombus and/or calcium are all factors associated with poor results in terms of aneurysm exclusion, both in the immediate and long-term follow-up.1–7 Indeed, the so-called ‘hostile aortic neck’ significantly increases the risk of ineffective proximal sealing, with possible consequent graft migration, early or late type Ia endoleak, and aortic rupture. 3
Over the last years, this risk has been managed by resorting to different techniques, in particular by chimney procedures or fenestrated stent-grafts, with the aim of extending the proximal neck length to obtain additional sealing zone.8–10
EndoAnchors have been introduced to stabilize the sealing at the proximal neck level without requiring its extension. By securing the endograft to the vessel wall, this approach imitates the efficacy and the safety of a hand-sewn aortic anastomosis, keeping in close contact the graft fabric with the aortic neck and increasing the resistance to distal migration.11,12 EndoAnchors are successfully used to fix type Ia endoleaks when endograft fabric is just below the renal artery with no migration and the use of an aortic cuff is not possible. In case of type Ia endoleaks associated with stent-graft migration, EndoAnchors are implanted to secure the primary endograft to the aortic wall preventing its further dislocation, before deployment of a proximal aortic cuff (that is usually secured with another row of anchors) to extend the sealing zone. 13
Several investigators have evaluated the EndoAnchors technique both in the prevention and treatment of endograft migration and type Ia endoleaks to achieve an effective proximal sealing.14–19 The ANCHOR prospective trial included 319 patients at 43 sites in the United States and Europe; 242 (75.9%) had EndoAnchors implanted at the time of the first operation (primary implant), whereas 77 patients were treated for proximal aortic neck complications after EVAR (secondary implant). The authors reported overall procedural success in 279 patients (87.5%), in particular in 217 of 240 (89.7%) in the primary implant arm and in 62 of 77 (80.5%) in the secondary arm. There were 29 residual type Ia endoleaks (9.1%) at the end of the procedure. At a mean follow-up of 9.3 months, there were no aneurysm-related deaths or aneurysm ruptures, with 301 patients (94.4%) free from secondary procedures. 20 In another paper, the same group published the one-year results of the trial: freedom from type Ia endoleak was 95% in the primary implant arm and 77% in the secondary arm. Aneurysm sacs regressed >5 mm within one year in 45% of the primary cases and in 25% of the secondary ones. 21
Data concerning the prophylactic use of anchors appear the most promising in terms of proximal endoleaks absence and sac size regression at follow-up. Comparing patients who received prophylactic EndoAnchors to patients treated with standard EVAR for aneurysms with challenging proximal necks, Muhs et al. found a higher rate of sac regression at two years’ follow-up in the anchors group (81.1% vs. 48.7%, p = 0.01), regardless of the presence of a wide neck or mural thrombus. 22
Recently, the combination of the Heli-FX EndoAnchor system and Endurant II/IIs stent graft has received the European CE Mark approval and U.S. Food and Drug Administration (FDA) approval to treat AAA in patients with neck lengths as small as 4 mm and ≤60° infrarenal angulation. This expanded indication enables the treatment of a wider range of patients with short, hostile aortic neck anatomies, independent of renal stenting that is required in chimney procedures or fenestrated stent-graft repair.
Our study describes a single-centre series with a median follow-up of 13 months and 100% technical success; results are favourable and comparable with published data from other authors. We found the implant of EndoAnchors satisfactory in terms of clinical outcome and aneurysm sac size regression both in case of proximal neck late complications treatment and in prophylactic use as well. All the nine patients with hostile proximal neck in the primary implant group were evaluated for different treatment options before the intervention, including open repair, fenestrated stent-graft, chimney technique, and standard endograft plus anchors. The main factors taken into account in the decision-making process were anatomy, age, and operative risk. We usually reserve open repair for patients with long life expectancy that are at low surgical risk. On the other hand, in patients unfit for open surgery and with short proximal neck (<15 mm in length) that is at least 7 mm long and up to 32 mm in diameter, our standard practice is to prefer the use of the Endurant stent-graft with the eventual adjunct of anchors, considering the relative simplicity of the procedure, the immediate availability of devices, and the satisfactory reported early- and mid-term results in terms of aneurysm exclusion and stability.15,16,20,21 For those cases with a proximal neck shorter than 7 mm and/or wider than 32 mm that are not eligible for open surgery, there is the need to extend the proximal sealing zone above the renal arteries to prevent late complications in the follow-up, so we prefer to resort to other endoluminal techniques such as fenestrated stent-grafts, or chimney procedures when there is no possibility to wait for a custom-made device.
From a technical point of view and according to our experience, EndoAnchors should be deployed within the first centimetre or less (taking into account the neck length) from the proximal margin of the endograft fabric, in order to achieve a secure sealing and maximize penetration into the aortic wall. In case of secondary implants, we did not experience any problem with anchors deployment related to the proximal bare metal stent struts of the previous endografts. We usually consider the presence of severe thrombus and calcium as a contraindication to anchors due to the risk of ineffective penetration or dislodgement. 23 Similarly, one should avoid EndoAnchors when the distance between the endograft and the aortic wall is more than 2 mm, because they will not be able to ensure effective fixation and sealing. For all these reasons, the aortic neck should be carefully evaluated using CT-scan prior to the intervention for an effective clock-face anchors positioning planning. CT image analysis is essential for correct C-arm orientation and accurate fluoroscopic guidance for anchor deployment at the proper location of the aortic neck. We believe this to be the most relevant point in the use of these devices to increase their effectiveness.
The main limitation of this study is the small number of patients, which is why no subgroups were analysed and statistical analysis was not performed.
Conclusions
The use of EndoAnchors is becoming widespread and an increasing number of papers demonstrate that these devices lead to excellent short- and mid-term results, representing a useful adjunct to EVAR in patients with challenging aortic neck anatomy. Our experience confirms that EndoAnchors are a suitable device to prevent and treat type Ia endoleaks with or without endograft migration in the presence of a short, wide or angulated proximal neck. Nevertheless, a larger patient cohort and longer-term follow-up data are needed for stronger conclusions.
Footnotes
Contributorship
RG and OB contributed equally to the conception of this work and the acquisition of data, and drafted the article. CC, GS, and CD researched literature and contributed to the acquisition of data. RG, ML, and GP critically revised the article for intellectual content. All authors approved the final version of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
RG.
