Abstract
We report the case of a 33-year-old man with a testicular tumor and multiple lung metastases. After high orchiectomy, chemotherapy was administered. Subsequently, fluoroscopy-assisted thoracoscopic resection was performed for pulmonary lesions that had decreased in size after the chemotherapy but had persisted. Because four pulmonary lesions were too small to detect by fluoroscopy during the operation, Lipiodol marking was done before thoracoscopic resection. Sixteen days after the surgery, pneumonia after Lipiodol injection occurred on the lung contralateral to that injected with Lipiodol. Pneumonia improved rapidly after intravenous infusion of antibiotics.
Introduction
In the domain of pulmonary surgery, advances have been made in thoracoscopic surgical techniques for diagnostic excisional biopsies of pulmonary nodules as well as for therapeutic resection of peripheral lung malignancies (1). For small and deeply situated pulmonary nodules, however, a major factor limiting success of thoracoscopic resection is the difficulty in locating the target nodule because it cannot be palpated digitally. Fluoroscopy-assisted thoracoscopic resection of a small lung nodule marked with Lipiodol, which is generally used as a contrast medium for lymphatic vessels, has been reported to be useful in these cases (2,3).
Recently, we experienced a patient who underwent fluoroscopy-assisted thoracoscopic resection after marking of nodules with Lipiodol and thereafter developed pneumonia.
Case report
A 33-year-old man with multiple metastases to both lungs from a testicular tumor was
referred to our hospital. After a high orchiedectomy, he was given chemotherapy.
Even though the pulmonary lesions decreased in size, complete remission was not
achieved. Therefore, fluoroscopy-assisted thoracoscopic resection was attempted for
12 pulmonary lesions in the left lung, of which four were very small
Under CT fluoroscopic guidance (X Vigor Laudator; Toshiba Medical System, Tokyo,
Japan), a 21-gauge needle (PEIT needle; Hakko Medical, Chikuma, Nagano, Japan) was
advanced to the area adjacent to each lesion (4). When the needle tip was confirmed in a
position adjacent to a lesion, 0.3 mL of Lipiodol (Laboratoire Guerbet, Roissy,
France) was injected (Fig.
1a). For three of the lesions, the procedure was performed with the
patient in the prone position. For the remaining lesion in the upper lobe, the
patient was placed in the right lateral recumbent position. No complication related
to marking with Lipiodol occurred. A 33-year-old man with a testicular tumor
and multiple lung metastases. (a) CT fluoroscopy image obtained while
the patient was in the prone position shows Lipiodol (arrow) marking
near the pulmonary lesion in the lower lobe of the left lung
(arrowhead). (b) CT image obtained 16 days after surgery shows GGO
lesions. Dense areas corresponding to migrated Lipiodol are evident in
some of the GGOs (arrows). Such lesions are seen in the entire right
lung. (c) CT image obtained after treatment with antibiotics shows
almost complete disappearance of GGO lesions. There is still a high
density spot that is a focus of residual Lipiodol
(arrow).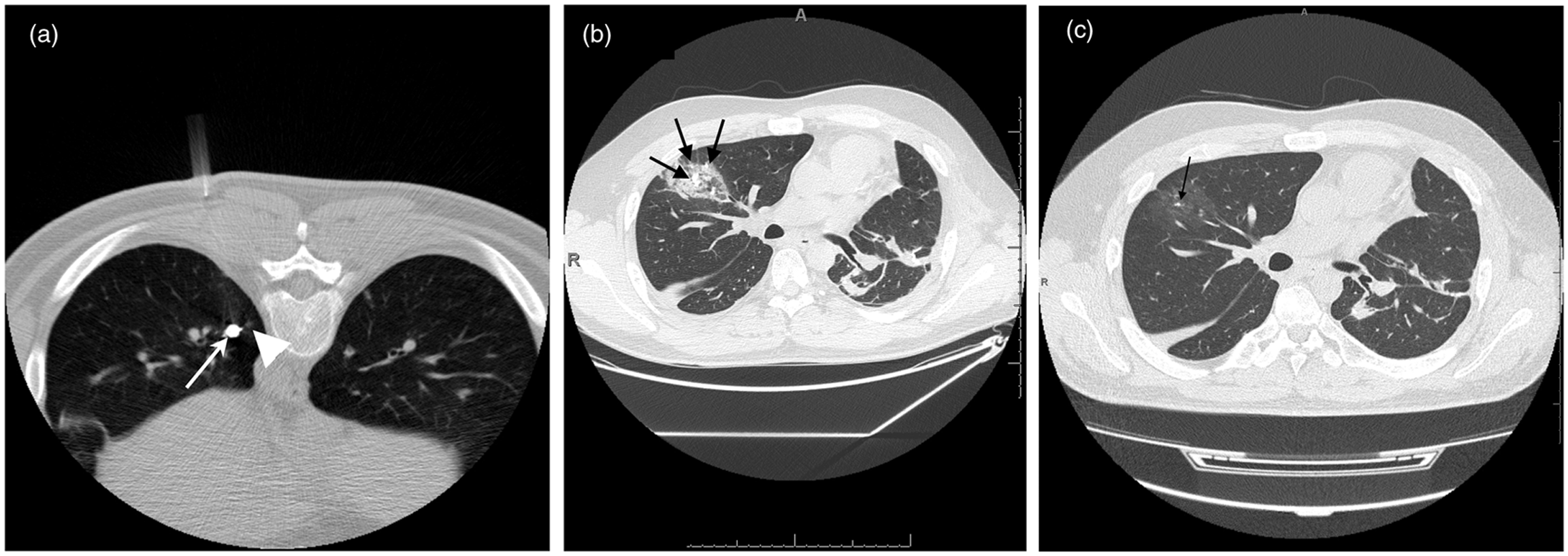
After the marking procedure the patient was taken to the operating room, and thoracoscopy was performed under general anesthesia with the patient in the right lateral recumbent position. A fluoroscopic unit with a C-shaped arm was used to identify the radiopaque nodules, which were grasped with a ring-shaped forceps during fluoroscopy using multiple projections. Successful resection was confirmed by viewing each radiopaque nodule within the resected specimens under the C-arm fluoroscope.
Sixteen days after surgery, the patient experienced general fatigue and fever of 39℃. White blood cell count was 7230 and C-reactive protein was elevated to 19.55 mg/dl. Chest CT showed multiple ground-glass opacities (GGOs) distributed at the upper, middle, and lower lobes of the right lung (Fig. 1b). Within the GGOs in the upper and middle lobes, high density spots corresponding to Lipiodol were seen. The patient was then treated for pneumonia, although information from a sputum culture, procalcitonin testing, and polymerase chain reaction testing was not obtained. After intravenous infusion of antibiotics, the symptoms disappeared. On CT 1 month later, the GGOs had markedly disappeared (Fig. 1c). Fifty-two days after the fluoroscopy-assisted thoracoscopic resection in the left lung, the pulmonary lesions in the right lung were resected by the same procedure.
Discussion
Several methods are used for localizing small pulmonary nodules before thoracoscopic resection. These include percutaneous injection of water soluble dye (5) or Lipiodol (2,3,6), percutaneous insertion of a hook wire (7,8), and barium marking via bronchoscopy or percutaneous injection (6). Because of rapid diffusion of dye in lung tissue after injection, the general dye method has drawbacks. Marking must be performed within 3 h before thoracoscopy to enable dye detection and diffusion sometimes blurs the injection site (2,6). With water soluble dye, dye-marked lesions cannot be localized after formalin fixation (3). Barium appears as a lesion in hematoxylin and eosin-stained sections and can cause inflammatory changes in lung tissue, possibly making a pathologic diagnosis difficult (2,3). With the hook-wire technique, dislocation of the hook wire has caused varying degrees of failure and was reported to cause massive air embolism (8).
Lipiodol, as used in the present case, offers the following advantages: (i) over-resection of normal lung tissue can be avoided because Lipiodol marks nodules as clear spots; (ii) Lipiodol can remain for up to 3 months after marking; (iii) Lipiodol does not affect pathologic findings; and (iv) because Lipiodol diffuses only to a small extent, leaving a clear spot, even deeply situated nodules can be easily localized (2). A 100% success rate for the use of Lipiodol for marking prior to thoracoscopic resection was reported (2,3).
Interstitial pneumonia induced by 131I-labeled Lipiodol administered from the hepatic artery as treatment of hepatocellular carcinoma was reported (9). Earlier, pneumonia following chemoembolization for liver tumors in which Lipiodol was used was described (10). Suggested mechanisms for such pneumonia were Lipiodol embolism by arteriovenous shunting or hepatic vein invasion, an immunoallergic phenomenon, and/or radioactive-induced lesions in patients after 131I-labeled Lipiodol infusion (9). To our knowledge, however, pneumonia occurring after Lipiodol marking has not been reported. We hypothesize the following to explain the occurrence of pneumonia in this patient. Part of the Lipiodol injected for marking the pulmonary nodules in the left lung distributed in the alveoli and bronchioles. The Lipiodol then moved to the contralateral lung transtracheally by gravity during the thoracoscopic resection procedure during which the patient was placed in the right lateral recumbent position. Thereafter, Lipiodol distributing in the entire right lung was involved in causing pneumonia. The occurrence of this complication might be avoided by minimizing the time that a patient spends with the opposite lung dependent.
In conclusion, care must be taken to avoid the occurrence of pneumonia after Lipiodol injection to localize pulmonary nodules before fluoroscopy-aided thoracoscopic resection.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
