Abstract
Aims
A recent study suggested that women with heart failure and heart failure reduced ejection fraction might hypothetically need lower doses of angiotensin-converting enzyme inhibitors or angiotensin II receptor blockers ( = renin-angiotensin-system inhibitors) and β-blockers than men to achieve the best outcome. We assessed the current medical treatment of heart failure reduced ejection fraction in men and women in a large contemporary cohort and address the hypothetical impact of changing treatment levels in women.
Methods
This analysis is part of a large contemporary quality of heart failure care project which includes 5320 (64%) men and 3003 (36%) women with heart failure reduced ejection fraction. Detailed information on heart failure therapy prescription and dosage were collected.
Results
Women less often received renin-angiotensin-system inhibitors (79% vs 83%,
Conclusion
In this large contemporary heart failure registry, there were significant but relatively small differences in drug dose between men and women with heart failure reduced ejection fraction. Implementation of the hypothetical sex-specific target dosing schedule would lead to considerably more women adequately treated. In contrast, we identified a group of women who might have been relatively overdosed with increased risk of side-effects and intolerance.
Introduction
The overall enrolment of women in clinical trials investigating treatment and outcome in heart failure (HF) is generally low and, accordingly, women are underrepresented in these trials as compared to the real-world. 1 Studies on optimal dose in HF with reduced ejection fraction (HFrEF) are scarce and the number of included women was low.2,3 Likewise, the European Society of Cardiology (ESC) HF guidelines provide no sex-specific recommendations. 4 Recently, the hypothesis has been suggested that women with HFrEF might need lower dose of angiotensin-converting enzyme inhibitors (ACE-is), angiotensin II receptor blockers (ARBs) and β-blockers than men, which brings into question what the true optimal level of drug therapy is for women. 5 Whereas men obtained the maximal reduction of mortality and HF hospitalization at the guideline recommended target dose of β-blockers, ACE-Is and ARBs. In male HFrEF patients, not achieving target dose is equal to not achieving maximum treatment benefit.
In contrast, in women the largest treatment benefit was already observed at 50% of recommended target dose, achieving 30% lower overall cardiovascular risk (all-cause mortality or hospitalization for HF). At higher doses, no additional benefit was observed in women, therefore the hypothesis is that maximum benefit can be achieved at 50% of target dose at no further expense of intolerance of side-effects. 5 The clinical impact of this post-hoc analysis can be considerable but should be assessed in large contemporary HFrEF cohorts.
The Chronisch Hartfalen ESC-richtlijn Cardiologische praktijk Kwaliteitsproject HartFalen (CHECK-HF) registry is a large scale (
Methods
The design and methods of the CHECK-HF registry have been published in detail earlier.6,7 Briefly, the CHECK-HF study is a large contemporary cross-sectional observational cohort, including a total of 10,910 chronic HF patients from 34 participating Dutch centres between September 2013–September 2016. All patients were diagnosed and treated according to the 2012 ESC HF guidelines, 8 and almost all were seen at a dedicated outpatient HF clinic (96%). Detailed information on patient characteristics, echocardiographic parameters and HF therapy, including HF drug prescription, dose, contraindication and intolerance, as well as device therapy were recorded. This study was conducted according to the Helsinki Declaration, and was approved by the medical ethics committee in 2017 at the Maastricht University Medical Center (Maastricht, the Netherlands). Patients were not involved in the research process.
Left ventricular function, assessed during the most recent outpatient clinical visit, was used to categorise HF patients. Patients were categorised based on left ventricle ejection fraction (LVEF) or visual assessment of left ventricle (LV) function into HFrEF (LVEF<50% (
For a sub-analysis according to the newer 2016 ESC HF guidelines,
4
patients with an assessed LVEF<50% were categorised into HF with mid-range ejection fraction (HFmrEF) (LVEF 40–49% (
In order to investigate the impact of the hypothetical sex-specific dose schedule of β-blockers and renin-angiotensin-system inhibitors (RAS-is) (i.e. ACE-is or ARBs), we analysed the prescribed dosages expressed as a percentage of the recommended target dose and of the hypothetical target dosage (50% of the guideline recommend target dose) in women. Target doses of guideline-recommended HF therapy are presented in Supplementary Material Table 1 and in line with the ESC HF guidelines. 4
Patient characteristics in heart failure with reduced ejection fraction (HFrEF) patients.
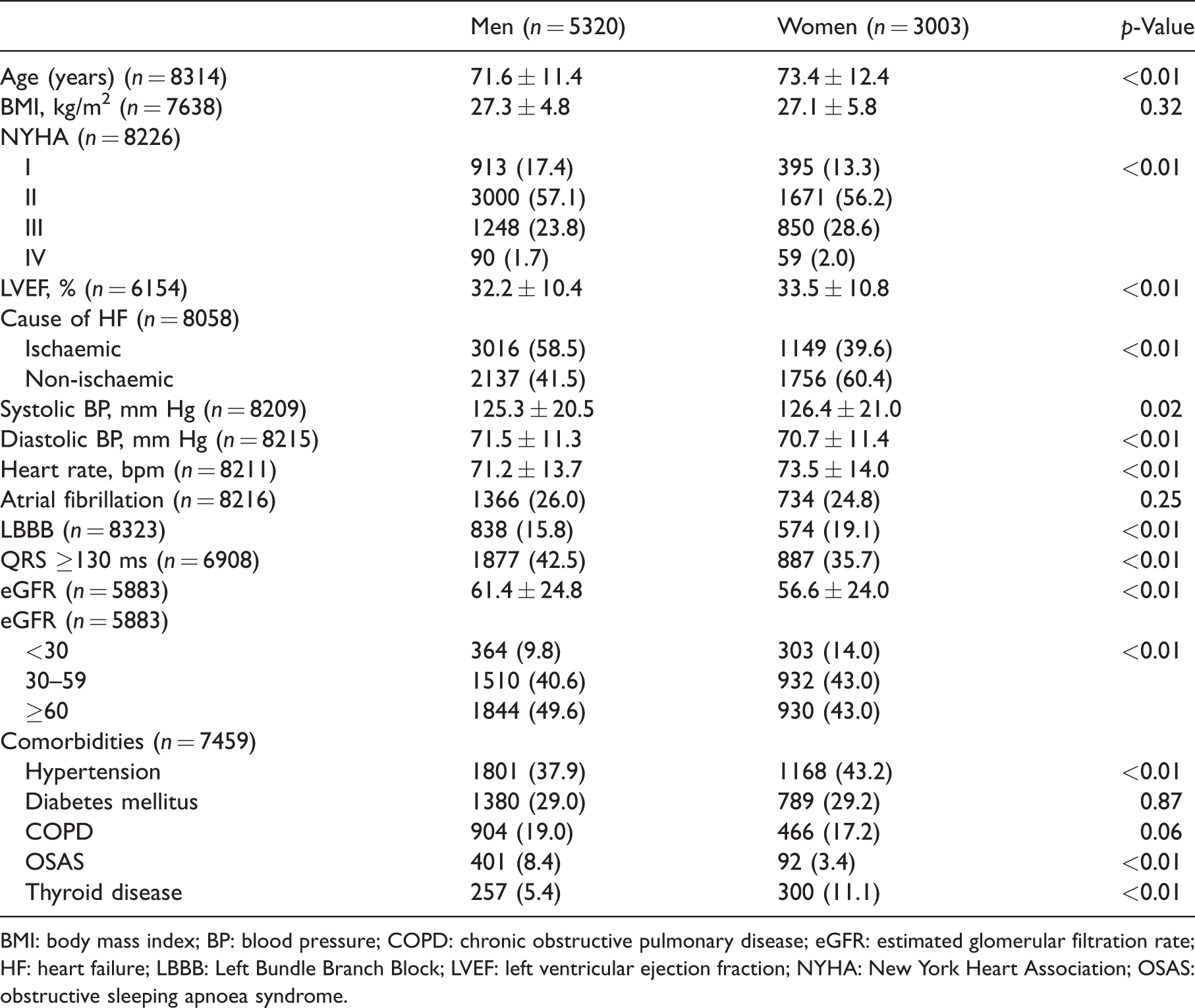
BMI: body mass index; BP: blood pressure; COPD: chronic obstructive pulmonary disease; eGFR: estimated glomerular filtration rate; HF: heart failure; LBBB: Left Bundle Branch Block; LVEF: left ventricular ejection fraction; NYHA: New York Heart Association; OSAS: obstructive sleeping apnoea syndrome.
Statistical analysis
Continuous data are expressed as mean value ± standard deviation (SD) or median and interquartile range, depending on the distribution of the data, and compared by one-way analyses of variance (ANOVAs) or Mann-Whitney U-test. Categorical data are expressed as counts and percentages, and compared by the Pearson Chi-square test. The prescribed dosages are expressed as a percentage of the recommended target dose. The differences between the recommended and newly suggested target dose were compared by the McNemar test.
Multivariable predictors of HF medication use were assessed using multivariable logistic regression analysis. All predictors of medication use in univariable analysis at a
For variables with missing data in the multivariable analysis, we used multiple imputation modelling. If the missing variables showed a monotone pattern of missing values, the monotone method was used, otherwise, an iterative Markov chain Monte Carlo method was used with a number of 10 iterations. A total of five imputations was performed, and the pooled data were analysed. All analyses were performed with SPSS Statistical Package version 25.0 (SPSS Inc., Chicago, Illinois, USA).
Results
Baseline characteristics of men and women with HFrEF are presented in Table 1.
Pharmacological therapy in HFrEF
Female HFrEF patients significantly less often received a RAS-i (78.9% vs 82.6%,
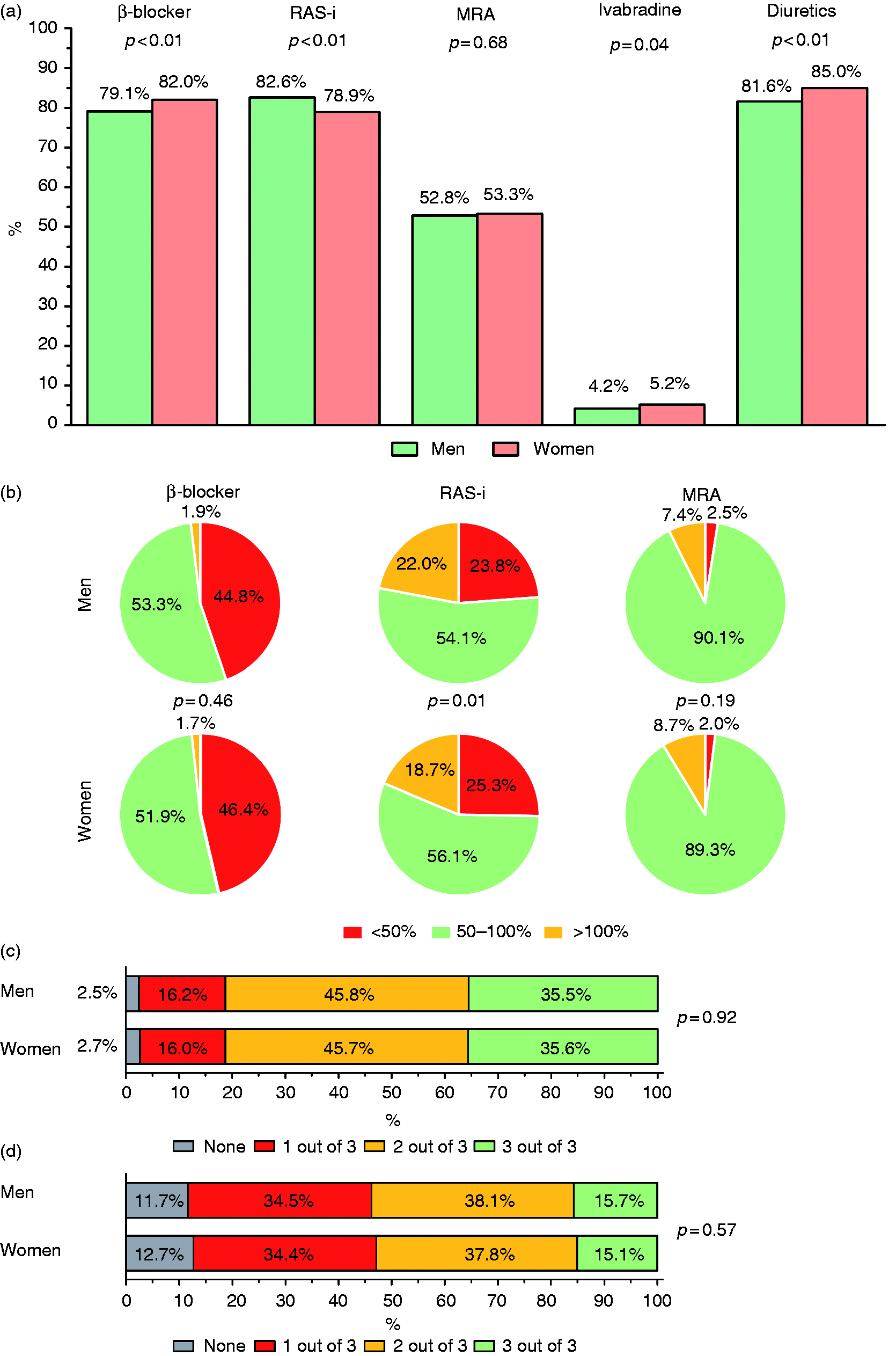
(a) Heart failure (HF) therapy usages; (b) prescribed dosages expressed as a percentage of recommended target dose; (c) triple therapy prescribed; and (d) triple therapy at ≥50% of the recommended target dose prescribed in men and women. RAS-i: renin-angiotensin-system inhibitor; MRA: mineralocorticoid receptor antagonist.
Multivariable predictors of the use of heart failure (HF) therapy in heart failure with reduced ejection fraction (HFrEF) patients in relation to gender.
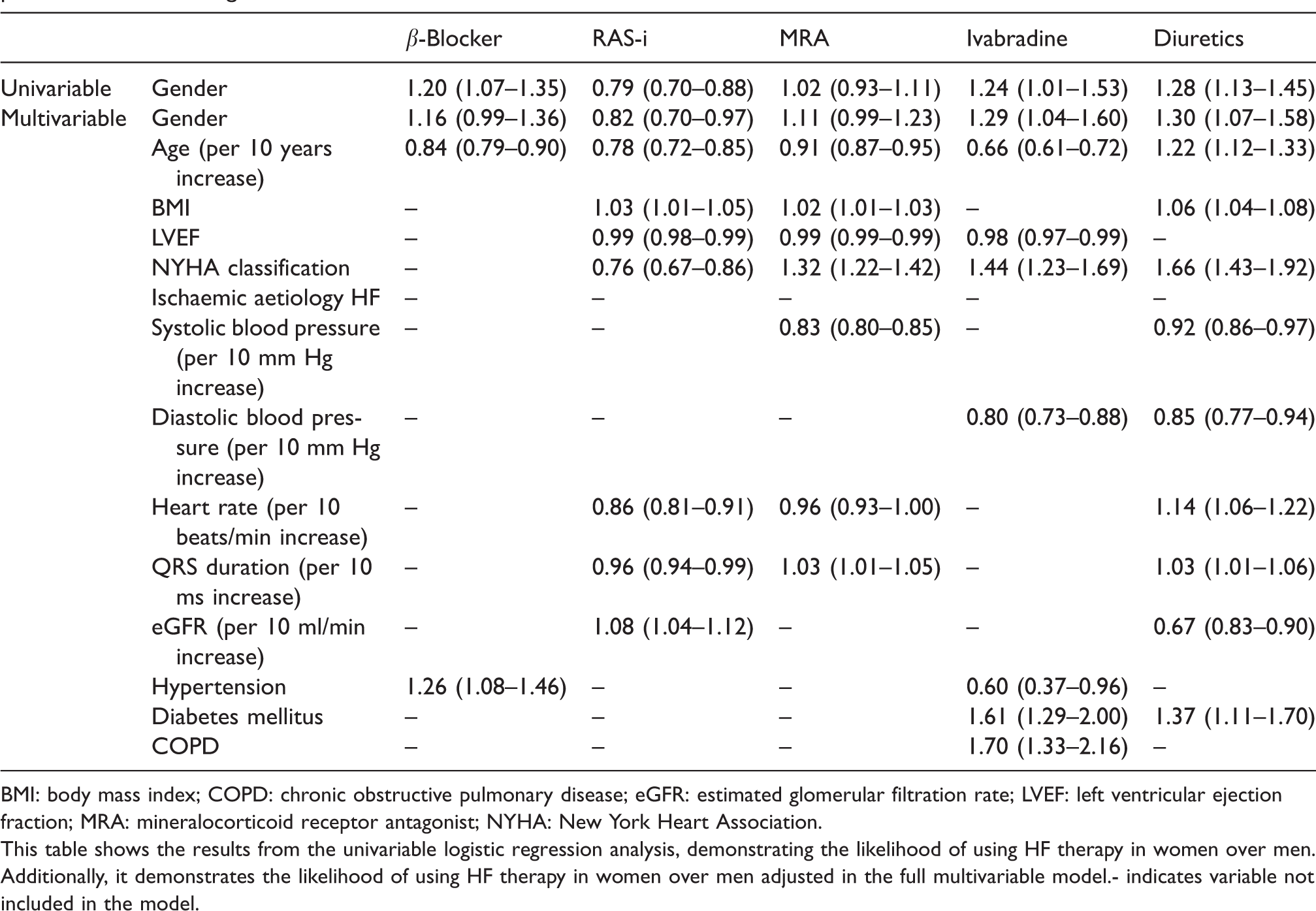
BMI: body mass index; COPD: chronic obstructive pulmonary disease; eGFR: estimated glomerular filtration rate; LVEF: left ventricular ejection fraction; MRA: mineralocorticoid receptor antagonist; NYHA: New York Heart Association.
This table shows the results from the univariable logistic regression analysis, demonstrating the likelihood of using HF therapy in women over men. Additionally, it demonstrates the likelihood of using HF therapy in women over men adjusted in the full multivariable model.- indicates variable not included in the model.
Predictors of prescription and target dose of HF therapy
In the multivariable regression analysis, lower age and the presence of hypertension were significant predictors of prescription of β-blockers; lower age, male gender and renal insufficiency were significant predictors of prescription of a RAS-i; and lower age, higher New York Heart Association (NYHA)-classification and lower systolic blood pressure were significant predictors of prescription of MRAs (Table 2). In multivariate analysis, the chance of receiving the guideline-recommended target dose of a RAS-i was independently related to male gender, while the chance of receiving the guideline-recommended target dose of MRA was independently related to female gender (Table 3). Multiple imputations did not change the results (data not shown).
Multivariable predictors of receiving guideline-recommended target dose of HF medication in HFrEF patients in relation to gender.
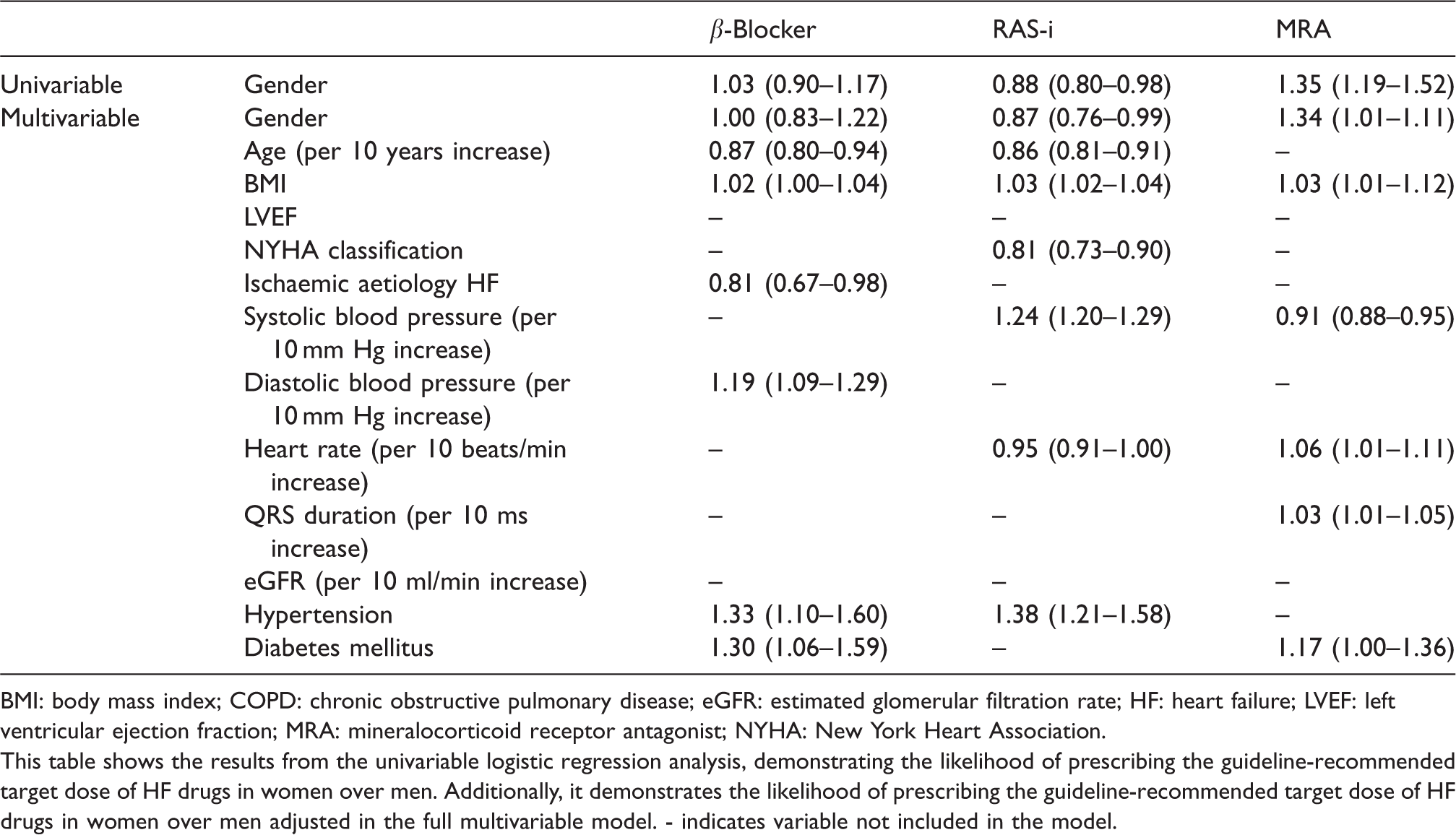
BMI: body mass index; COPD: chronic obstructive pulmonary disease; eGFR: estimated glomerular filtration rate; HF: heart failure; LVEF: left ventricular ejection fraction; MRA: mineralocorticoid receptor antagonist; NYHA: New York Heart Association.
This table shows the results from the univariable logistic regression analysis, demonstrating the likelihood of prescribing the guideline-recommended target dose of HF drugs in women over men. Additionally, it demonstrates the likelihood of prescribing the guideline-recommended target dose of HF drugs in women over men adjusted in the full multivariable model. - indicates variable not included in the model.
Clinical impact of new hypothetical sex-specific target dose schedule
Using the hypothetical sex-specific target doses (at 50% of the current recommended dose in the ESC guidelines in women only), leads to an considerable increase in the number of women who received the target dose for β-blockers (87.2% vs 53.6%,
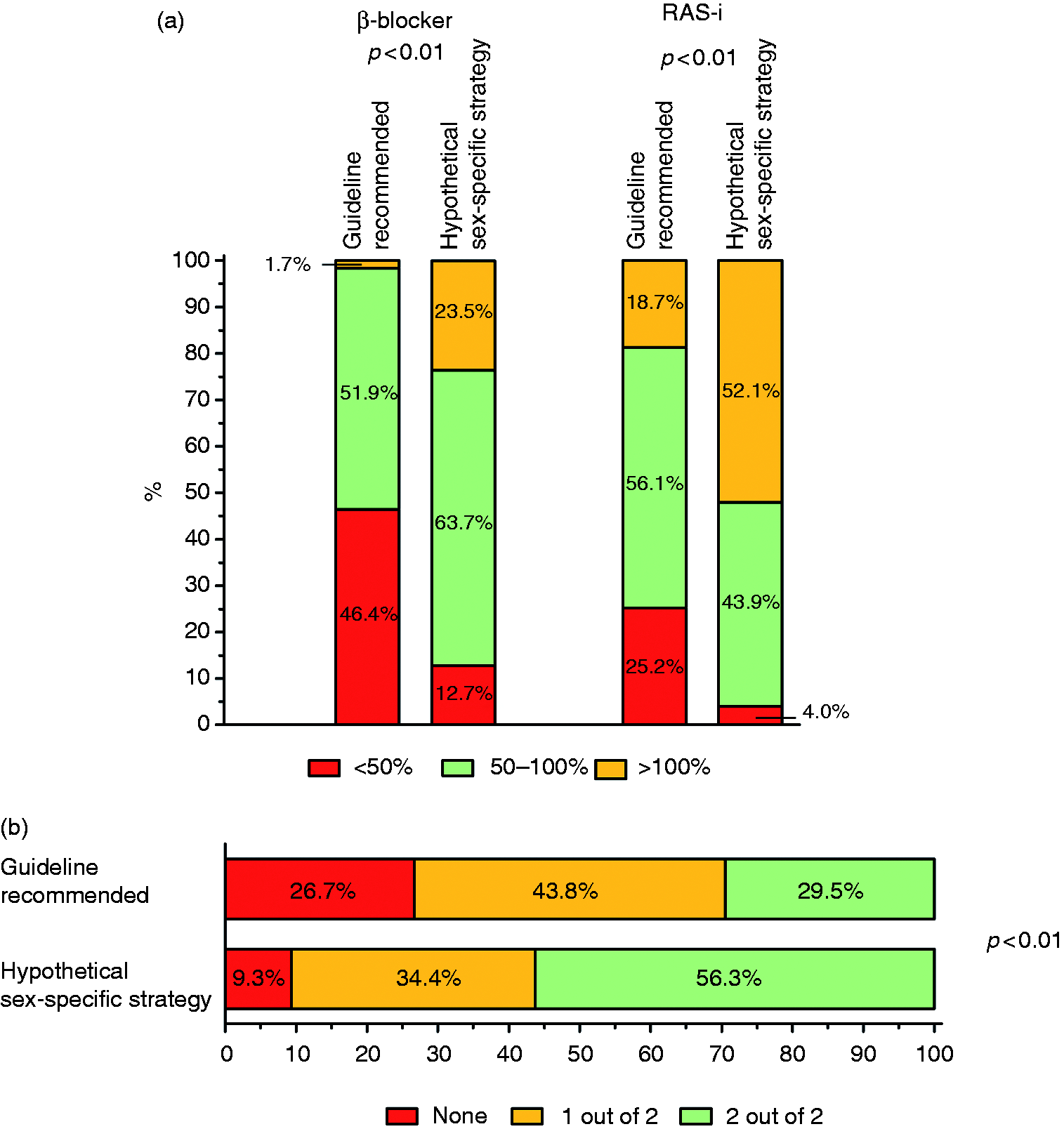
Impact of the newly proposed sex-specific target dose strategy (a) prescribed dosages expressed as a percentage of the guideline and newly proposed target dose of β-blockers and renin-angiotensin-system inhibitors (RAS-is), and (b) dual therapy at ≥50% of the guideline and newly proposed target dose prescribed in women.
Analysis in patients with HFmrEF according to 2016 ESC guidelines
A sub-analysis studying the different cut-off of HFmrEF and HFrEF according to the ESC 2016 guidelines does not change the inferences of this analysis (Supplementary Material Table 3, Supplementary Material Figure 1). Likewise, the subgroup of semi-quantitative LV function showed similar differences (Supplementary Material Figure 1).
Discussion
The current analysis shows that HF treatment between men and women differs in this large real-world contemporary cohort of HFrEF patients. Women received lower doses of HF drugs compared to men. The level of the target dose of HF drugs or maximally tolerated levels has been frequently discussed and recently gained more attention from a study suggesting that the optimal dose level might be 50% lower in women compared with men at maximum sex-specific treatment benefit. 5 This hypothesis has major implications for HF treatments in general, and we assessed the impact of this new hypothetical dose schedule in women.
In this patient sample, doctors were urged to titrate to guideline-recommended dosages, and this was successful in some, but not all, patients.
In male patients with HFrEF, the post-hoc analysis from BIOlogy Study to TAilored Treatment in Chronic Heart Failure (BIOSTAT-CHF) shows that male patients only achieve maximum treatment benefit at the full recommended target dose of HF medication. In women, the maximum treatment benefit was observed at 50% of the target dose with no further gain in benefit, with only futile risk of intolerance. Naturally, if lower dosages were accepted as ‘optimal’ in females, a much larger proportion of the female patients would be regarded as being treated optimally when 50% of the recommended dose would be regarded as optimal. Notably, this approach identifies a potential subgroup of women who are relatively overdosed (>100% dose in females) with an increased risk of side effects and intolerance at no incremental benefit of treatment.
Guideline adherence and sex
Women with a (non-ST-segment elevation) myocardial infarction receive the guideline-recommended therapy less often.9,10 Additionally, sex-specific treatment strategies for these conditions have been proposed previously.10,11 Similarly, multiple registries have demonstrated sex-related differences in guideline adherence, with women less often receiving β-blockers, 12 ACE-is,12–14 MRAs 12 and more often ARBs 15 and diuretics.1,12 The current results of our analysis are in line with these previous registries, although sex-specific differences particularly regarding guideline-recommended target doses were relatively small in CHECK-HF. Differences in patient characteristics could influence clinicians in their decision-making, but these differences do not fully explain the sex-related differences in HF drug usage and dosages in our dataset.
Optimal doses of HF therapies in men versus women
Women are underrepresented in clinical trials investigating the efficacy of HF drugs, as only 10–40% of the patients included in these trials were women. 16 Furthermore, only one trial investigated efficacy prospectively stratified by sex, 17 while all other studies analysed sex-related effects retrospectively and in post-hoc analyses, limiting these results. In women, the use of ACE-is leads to a non-significant reduction in all-cause mortality and hospitalizations compared to placebo. 18 The use of ARBs reduced all-cause mortality and hospitalizations in women compared to placebo. 19 Women using β-blockers had a better clinical outcome compared to women receiving placebo therapy,20–22 and similar favourable treatment effects were seen in women using MRAs.23,24 Studies investigating the ideal target dose in HFrEF are scarce, especially in women,2,3 therefore a one-size-fits-all strategy is recommended in the ESC HF guidelines. 4
Several sex-related pharmacological differences can cause differences in the efficacy of HF drugs between men and women. So differences in body weight, medication clearance rate and the effect of sex hormones contribute to higher plasma concentrations, and stronger effects of HF drugs in women.25,26 Additionally, it has been suggested that HF drugs might have a larger effect in women compared to men, even if the plasma concentrations are similar. 27
HF therapy dose and sex
Data on the ideal dosages in women are scarce. Two post-hoc analyses from the Heart failure Endpoint evaluation of Angiotensin II Antagonist Losartan (HEAAL) and Assessment of Treatment with Lisinopril and Survival (ATLAS) trials demonstrated that a lower RAS-i dosage in women was equally effective, or even more effective, compared to higher dosages.2,28 In contrast, higher dosages of RAS-is were more effective in men. These results suggest that using a one-size-fits-all target dose could lead to overdosing in women. A sub-analysis from these trials investigating the potential overdosing and its effect would be of great interest, especially since a post-hoc analysis from the Digitalis Investigation Group (DIG) trial demonstrated that women had a higher serum concentration compared to men, although they used a slightly lower dose adjusted for body-mass index.29,30 A similar effect has been seen in the use of β-blockers, with women having a higher serum concentration while using a similar dosage. 25
New hypothetical target dose levels in women
Recently, a post-hoc analysis from the BIOSTAT-CHF study investigated whether sex-related differences in the optimal dose of β-blockers and RAS-is for preventing all-cause mortality and HF-related hospitalization exists in HFrEF patients, and validated the results in the Asian Sudden Cardiac Death in Heart Failure (ASIAN-HF) dataset. 5 This post-hoc analysis demonstrated that in women a 30% risk reduction in all-cause mortality and HF-related hospitalizations can be obtained with approximately 50% of the recommended target dose of β-blockers and RAS-is, with no further decrease in risk at higher dose levels in BIOSTAT-CHF and ASIAN-HF validation cohorts. In contrast, in men the largest reduction was observed if 100% of the recommended target dose was reached. These results suggest that women with HFrEF might have similar clinical outcomes with lower doses of β-blockers and RAS-is than recommended in the ESC HF guidelines. 4 Naturally, if lower target doses were accepted as optimal in women, a larger proportion of women would be regarded as treated optimally, in our registry. Additionally, we identified a large group of women who were potentially overdosed, possibly without an incremental benefit. It is generally believed that women are more often affected by drug-related adverse effects31–33 and differences in target doses could be an explanation for this. Unfortunately, we do not have adequate data on side-effects to support this statement from our study. In these women, the doses might be lowered, improving patient compliance and lowering intolerance rates.
Similar to these findings, specific guideline recommendations or target dosages might be warranted for different subgroups as well, for example specific guidelines for races or body mass index (BMI) category. However, these should be evaluated by additional research.
Limitations and strengths
Our study has some limitations. CHECK-HF has a cross-sectional design with no follow-up data on patient outcomes. Other prospective studies integrating dose findings of HF therapy in women and outcome are needed. Still, our analysis shows the potentially large impact of the newly proposed target levels. In addition, for some variables a limited number of data were missing, however, after using multiple imputation this did not impact the results. Additionally, with changing HF categories based on LVEF in the newer guidelines, 4 our analysis was limited by a small number of patients where LV function was semi-quantitatively analysed with echocardiography. Strengths of the CHECK-HF registry include the large scale, contemporary (2016), and a reflection of the real-world practice of outpatient HF management in the Netherlands, representative of Western European countries. Furthermore, the availability of a large number of women with detailed information on medication prescription and dosage is important due to the lack of data in this subgroup, as previously noted.
Conclusion
In this large contemporary registry, drug dose significantly differed between men and women with HFrEF, although the differences where relatively small. As the first large HF study, we demonstrate the clinical impact of a hypothetical adjustment to a lower target dose schedule in women, by which more women would be considered adequately treated. On top of better adherence, this identifies a considerable large subgroup of women who are relatively overdosed in HF medication at no further reduction in CV risk but, rather, at higher risk of intolerance when the dose could have been further reduced.
Supplemental Material
sj-pdf-1-cpr-10.1177_2047487320923185 - Supplemental material for Impact of sex-specific target dose in chronic heart failure patients with reduced ejection fraction
Supplemental material, sj-pdf-1-cpr-10.1177_2047487320923185 for Impact of sex-specific target dose in chronic heart failure patients with reduced ejection fraction by Jesse F Veenis, Hans-Peter Brunner-La Rocca, Gerard CM Linssen, Ayten Erol-Yilmaz, Arjen CB Pronk, Domien JM Engelen, Rob M van Tooren, Hetty JJ Koornstra-Wortel, Rudolf A de Boer, Peter van der Meer, Arno W Hoes, Jasper J Brugts and for the CHECK-HF investigators in European Journal of Preventive Cardiology
Footnotes
Acknowledgments
The author(s) greatly acknowledge the participation of HF nurses and cardiologists of all participating sites for including patients and imputing patient data.
Author contribution
HPBLR, GCML, AWH and JJB contributed to the conception or design of the work. JFV, HPBLR, GCML, AEY, ACBP, DJME, RMVT, HJJKW, RADB, PVDM and JJB contributed to the acquisition, analysis or interpretation of data for the work. JFV and JJB drafted the manuscript. HPBLR, GCML, AEY, ACBP, DJME, RMVT, HJJKW, AWH, RADB and PVDM critically revised the manuscript. All gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HBRLR reports grants and personal fees from Novartis, grants and personal fees from Vifor, grants and personal fees from Roche Diagnostics, grants and personal fees from Boehringer-Ingelheim, outside the submitted work. RADB is a minority shareholder of scPharmaceuticals, Inc. RADB has received personal fees from Abbott, AstraZeneca, MandalMed Inc. and Novartis. All other authors report no conflict of interest. All authors had joint responsibility for the decision to submit for publication.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Servier, the Netherlands, funded the inclusion of data and software programme. The steering committee (HBRLR, GCML, AWH and JJB) received no funding for this project. This analysis was initiated by the authors and designed, conducted, interpreted and reported independently of the sponsor. The current study had no other funding source or any with a participating role in outcome assessment, or writing of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
