Abstract
Aims
Prevention of heart failure relies on the early identification and control of risk factors. We aimed to identify the frequency and characteristics of individuals at risk of heart failure in the general population.
Methods and Results
We report cross-sectional data from the prospective Characteristics and Course of Heart Failure Stages A–B and Determinants of Progression (STAAB) cohort study investigating a representative sample of residents of Würzburg, Germany. Sampling was stratified 1:1 for sex and 10:27:27:27:10 for age groups of 30–39/40–49/50–59/60–69/70–79 years. Heart failure precursor stages were defined according to American College of Cardiology/American Heart Association: stage A (risk factors for heart failure), stage B (asymptomatic cardiac dysfunction). The main results were internally validated in the second half of the participants. The derivation sample comprised 2473 participants (51% women) with a distribution of 10%/28%/25%/27%/10% in respective age groups. Stages A and B were prevalent in 42% and 17% of subjects, respectively. Of stage B subjects, 31% had no risk factor qualifying for stage A (group ‘B-not-A’). Compared to individuals in stage B with A criteria, B-not-A were younger, more often women, and had left ventricular dilation as the predominant B qualifying criterion (all P < 0.001). These results were confirmed in the validation sample (n = 2492).
Conclusion
We identified a hitherto undescribed group of asymptomatic individuals with cardiac dysfunction predisposing to heart failure, who lacked established heart failure risk factors and therefore would have been missed by conventional primary prevention. Further studies need to replicate this finding in independent cohorts and characterise their genetic and -omic profile and the inception of clinically overt heart failure in subjects of group B-not-A.
Introduction
Chronic heart failure (HF) is considered a progressive disorder1–4 covering precursor stages that are characterised by risk factor patterns (HF stage A) or asymptomatic cardiac dysfunction (HF stage B), and overt symptomatic HF (HF stages C–D) characterised by typical signs and symptoms as well as cardiac dysfunction.1–3 This scheme as initially proposed by the American Heart Association (AHA)/American College of Cardiology (ACC) in 20013 is accompanied by progressively higher natriuretic peptide levels 2 and successively worse prognosis as evidenced by the development of symptomatic HF2,5–7 and death. 2
Prevention of HF relies on the early identification and control of risk factors. The current classification criteria of stage A comprise common cardiovascular risk factors known to contribute to the development of coronary heart disease and HF with reduced left ventricular ejection fraction (LVEF). However, a substantial proportion of adults with HF, mainly those with preserved LVEF, had no prior vascular disease but similarly dismal prognosis. 6 HF of non-cardiovascular origin, by contrast, is less well understood. In particular, its determinants as a potential source for prevention targets are objectives of intense research.
The prevalence of the precursor stages of HF has mainly been assessed in populations of advanced age,2,5,8–10 and little is known about the frequency of these stages in study samples also including younger adults. The present analysis intended to assess the prevalence and determinants of precursor stages of HF (stages A and B) in a representative sample of the general population over a wide age range including younger adults.
Methods
The Characteristics and Course of Heart Failure Stages A–B and Determinants of Progression (STAAB) cohort study is a joint project of the Comprehensive Heart Failure Center (CHFC) and the Institute of Clinical Epidemiology and Biometry (ICE-B) of the University Hospital and the University of Würzburg, Germany. The STAAB cohort study aims to investigate the prevalence and course of the precursor stages of HF in a representative sample of 5000 residents of the City of Würzburg, Germany, aged 30–79 years, who have no medical history of HF. A detailed description of the methodology has been published. 11 All procedures within the STAAB programme are subject to rigorous quality control, follow predefined standard operating procedures, and comply with the Declaration of Helsinki. The study protocol and procedures received positive votes from the ethics committee of the medical faculty of the University of Würzburg (vote #98/13) as well as from the data protection officer of the University of Würzburg (#J-117.605-09/13). All participants provided written informed consent prior to any study examination.
Population and sampling strategy
A random sample of residents of the city of Würzburg (source population 124,297 inhabitants as of 2011 census) was drawn in November 2013 from the local registration office with predefined age and sex strata with ratios 1:1 for sex, and 10:27:27:27:10 for age groups of 30–39, 40–49, 50–59, 60–69 and 70–79 years, respectively. Send out of invitations occurred in batches that were iteratively re-adjusted for sex and age groups according to the respective response rates in order to ascertain a continuously balanced recruitment. Figure 1 details this process for the first half of the STAAB study sample (derivation sample) consisting of all participants who were contacted within batches one to 16 and had their baseline examination between 12 December 2013 and 2 September 2016. The second half of the study sample (internal validation sample) consisted of all other participants; their baseline examinations were carried out between 2 January 2016 and 27 October 2017.
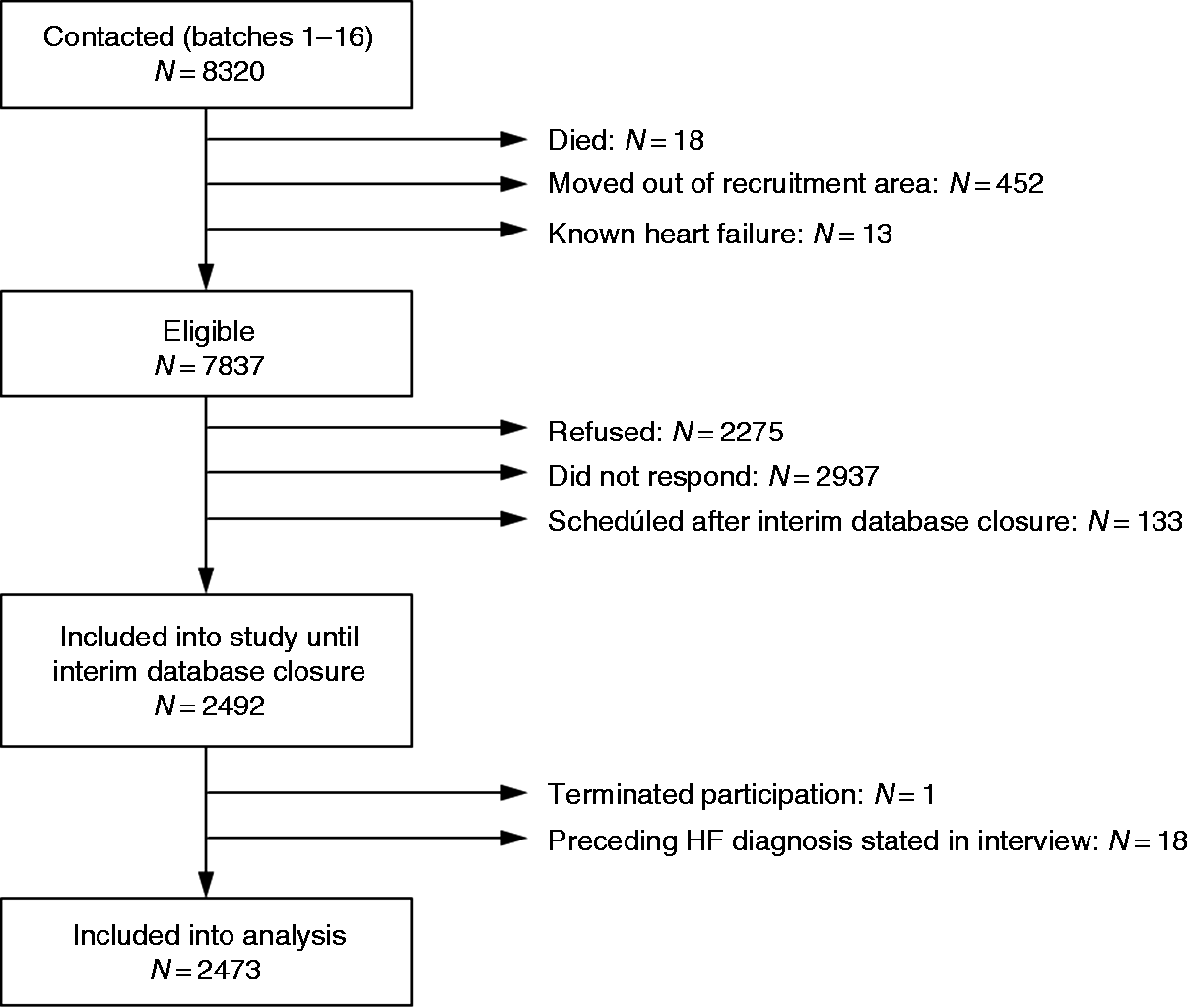
Study flow.
Baseline examination
Participants were evaluated at the Joint Survey Unit of the CHFC and the ICE-B. Blood pressure and anthropometry were assessed according to standard operating procedures. 11 A trained physician assessed medical history and the current pharmacotherapy and performed the physical examination. 11 Alcohol consumption and smoking status were assessed in a structured interview, symptoms of heart failure were self-reported in a questionnaire. 11
Routine laboratory measurements were performed at the Central Laboratory of the University Hospital Würzburg. Fasting and 2-hour venous whole-blood glucose for the oral glucose tolerance test were measured on-site (HemoCue, Hitado, Germany). 11 N-terminal pro B-type natriuretic peptide (NT-proBNP) was measured from stored serum samples (Cobas E411/Cobas Integra Roche, range 5 to 35,000 pg/mL, functional sensitivity 50 pg/mL).
Echocardiography
Echocardiography was carried out by dedicated and internally certified technicians 11 , 12 using a Vivid S6 ultrasound scanner with a M4S Sector Array Transducer (1.5–4.3 MHz; GE Healthcare, Horten, Norway) and a Vivid E95 ultrasound scanner with a M5Sc-D Transducer (1.5–4.6 MHz; GE Healthcare, Horten, Norway). A minimum of three cardiac cycles was recorded for analysis and stored digitally. The prespecified protocol adhered to recommendations formulated in current national and international guidelines. 13 A detailed methodology is given in the Supplementary methods file.
Categorisation of early heart failure stages
Patients were grouped into stages of HF according to AHA/ACC guidelines: 3 0 = no risk factor and no structural heart disease; A = at least one risk factor, but no structural heart disease and no clinical HF symptoms; B = structural heart disease, but no clinical HF symptoms; C = structural heart disease and clinical HF symptoms. Risk factors for the development of HF and respectively applied definitions were: hypertension, 14 atherosclerotic disease, diabetes mellitus, 15 obesity 16 and metabolic syndrome. 17 Based on current recommendations, structural heart disease was defined as the presence of at least one of the following: reduced LVEF, 13 left ventricular (LV) dilation, 13 LV hypertrophy, 13 any stenosis of mitral or aortic valve, 18 moderate or severe regurgitation at mitral 19 or aortic valve, 20 or diastolic dysfunction as an indirect measure of elevated left atrial pressure. 21 In addition, prior myocardial infarction was also considered to represent structural heart disease. In analogy to the Framingham criteria for congestive HF,22,23 clinical HF symptoms were defined as the presence of a combination of criteria suggestive of chronic HF integrating current guidelines and recommendations. 24 For detailed definitions, see Supplementary Table e1.
Quality control and plausibility
Data collection within the STAAB cohort study followed standardised operating procedures for all modules. Each new staff member was trained and certified according to standardised criteria. Ongoing quality control of the various modules was performed regularly. 11 Echocardiography of all participants was performed according to a prespecified protocol. Technicians were trained in the echocardiography laboratory of the CHFC and in regular iterative education sessions, and were regularly internally (re-)certified. 11 In weekly echo team meetings pathological findings were iteratively discussed and verified. Detailed results of the quality assurance programme have been published previously.11,12
Data handling and analysis
Crude frequencies of the HF stages are reported for the total sample and in strata defined by age groups and sex. The standardised prevalence of HF stage and its 95% confidence interval (CI) was computed as an estimate for the population of Würzburg aged 30–79 years with the sizes of demographic strata from 30 December 2016. Clinical characteristics of the entire cohort and the subgroups by HF stage are presented as frequencies and percentages, means and standard deviations, or medians and quartiles, as appropriate. As self-reported HF was an exclusion criterion, the subgroup of individuals in stage C was very small (n = 7) and, therefore, was excluded from further analyses. Comparisons between stages were performed by the chi-squared test, analysis of variance, or the Kruskal–Wallis test, as appropriate. P values were not provided for variables contained in the stage definition. Additional comparisons were done between subjects in stage B who exhibited risk factors that would qualify for stage A (referred to as B&A) and those who had no risk factor (referred to as B-not-A), and between stage 0 and the B-not-A subgroup. These analyses were adjusted for sex, if appropriate. In case of missing information on criteria defining stage A or B, it was assumed that respective criteria were not fulfilled. For all other variables, the presented data (in particular, percentages), refer to the amount of non-missing values. The supplementary materials contain expanded tables detailing the numbers of cases available for each analysis (see Supplementary Tables e2, e3, e5, and e6).
Validation
Results regarding the prevalence and determinants of the early stages of HF from the planned interim analysis after inclusion of the first half of the participants were validated internally in the second half of the participants, a sample of similar size from the same population with the same stratification criteria for sex and age groups.
Results
Derivation
In the derivation sample, we analysed 2473 participants (51% women; see Supplementary Table e2). The participants were distributed across the five age categories according to the stratification procedure as intended, that is, 10%/28%/25%/27%/10%. Stage A was apparent in 42% of participants (50%/34% of men/women, respectively). The prevalence of stage A was 18%/30%/45%/55%/57% in the respective age groups. Stage B was found in 17% of participants (13%/21% of men/women, respectively). Respective distribution across age groups was 13%/13%/16%/20%/33%. The standardised prevalence was 39.2% (95% CI 37.3–41.2%) for stage A, and 17.7% (16.0–19.3%) for stage B. Relationships with age and sex are displayed in Figure 2.
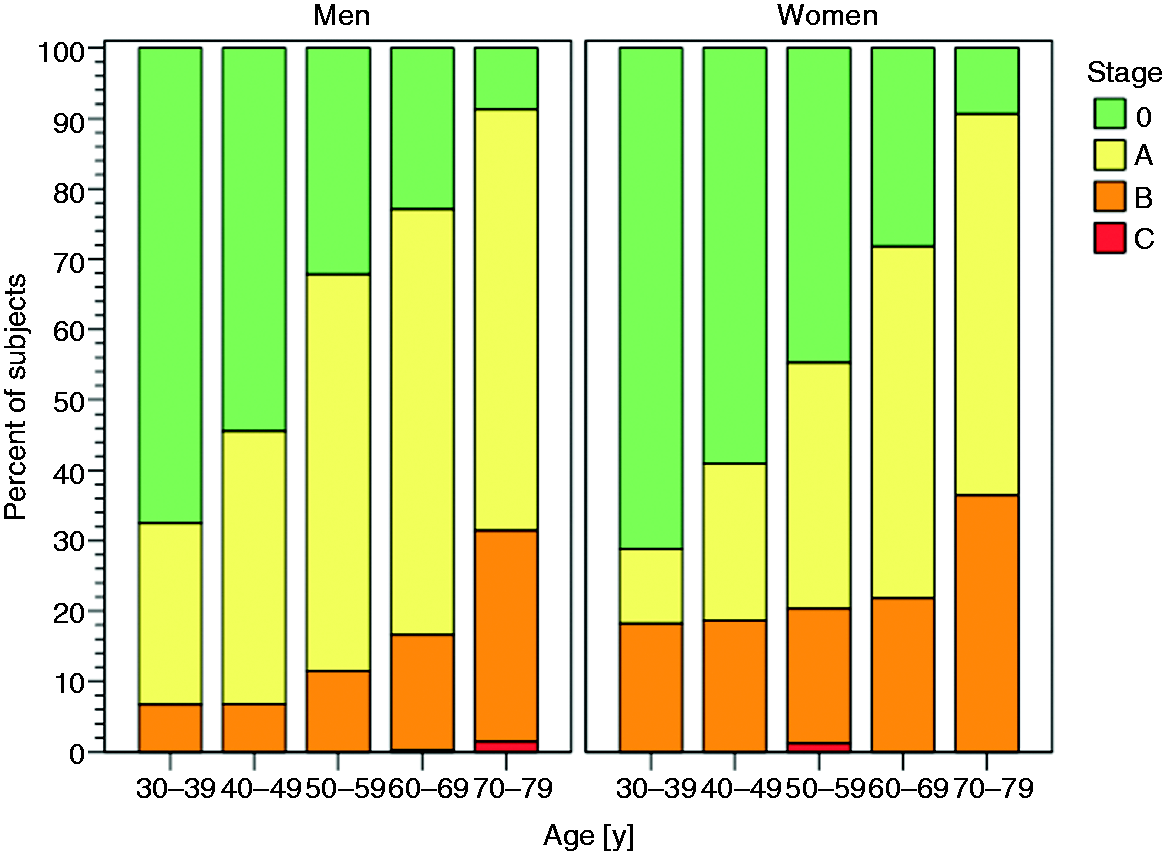
Crude distribution of heart failure stages 0, A, B, and C in men and women, by age decades.
Among individuals exhibiting risk factors for the development of HF, those with structural heart disease (stage B&A; n = 299) were older (58 vs. 63 years, P < 0.001), more often women (42% vs. 57%, P < 0.001), and revealed a higher total number of risk factors (1.8 vs. 2.1 risk factors, P < 0.001; see Supplementary Table e3) when compared to individuals without structural heart disease (stage A; n = 1039). However, potential additional risk factors such as smoking, alcohol consumption and comorbidities were not different between these groups. Renal function and levels of albumin, low-density lipoprotein (LDL) cholesterol, and haemoglobin were lower in B&A, and levels of haemoglobin A1c (HbA1c), fasting glucose, and NT-proBNP were higher (see Supplementary Table e3).
Within stage B (n = 430), 299 individuals exhibited risk factors qualifying for stage A (70%; in the following labelled ‘B&A’), but 131 did not (30%; in the following labelled ‘B-not-A’). Compared to B&A, B-not-A individuals were younger, more often women, more often in sinus rhythm, and less often had a family history of cardiovascular disease, less alcohol consumption, and better renal function, lower leukocytes, lower sex-adjusted haemoglobin, and lower levels of NT-proBNP. No differences were apparent regarding heart rate, smoking status and the prevalence of anaemia. Regarding B-qualifying markers, B-not-A exhibited less LV hypertrophy, diastolic dysfunction, and significant valve regurgitation, but more often showed LV dilation than individuals in group B&A. Although mean LVEF was higher in B-not-A, there was no statistically significant difference in the prevalence of reduced LVEF (Table 1; Supplementary Tables e2 and e3). Cumulative distribution functions of LVEF and left ventricular end diastolic volume index (LVEDVi) are displayed in Supplementary Figure e1. Differences regarding LVEDVi, LVEF, and left atrial volume index (LAVi) remained similar after adjusting for the presence of atrial fibrillation and diabetes mellitus (see Supplementary Table e4).
Baseline characteristics and prevalence of HF stage defining criteria in the total derivation sample and according to HF stages.29

Data are mean (SD), median (quartiles), or n (%), as appropriate.
P(0AB) refers to the null hypothesis that the values for groups 0, A, and B are not different. P values only reported for variables not included in definitions of HF stages. Individuals with previously diagnosed heart failure had been excluded from the study. Individuals with signs/symptoms of heart failure AND structural heart disease are displayed in this table as ‘stage C’ but did not enter the analysis due to low sample size.
P values for comparisons versus stage 0 are superscripted within cells by lowercase: a P < 0.05, b P < 0.01, c P < 0.001.
P values for comparisons versus stage A are superscripted within cells by uppercase: A P < 0.05, B P < 0.01, C P < 0.001.
Percentages and mean values refer to subjects who had respective item recorded. Frequencies for each measurement are listed in Supplementary Table e1.
CVD: cardiovascular disease; dpw: drinks per week; COPD: chronic obstructive pulmonary disease; eGFR: estimated glomerular filtration rate; LDL: low-density lipoprotein; HDL: high-density lipoprotein; HbA1c: haemoglobin A1c; HF: heart failure; NT-proBNP: N-terminal pro B-natriuretic peptide; LV: left ventricle; LA: left atrium; TR: tricuspid regurgitation; LVEF: left ventricular ejection fraction; LVEDVi: left ventricular end diastolic volume index; LAVi: left atrial volume index; e′: early myocardial relaxation velocity; E: early mitral valve inflow velocity; TRmaxPG: tricuspid regurgitation maximal pressure gradient.
*Diabetes mellitus = insulin or antidiabetic drugs or fasting glucose ≥7.0 mmol/L or oral glucose tolerance test 2h glucose ≥11.1 mmol/L.
†Metabolic syndrome = ≥3 of the following: (a) waist circumference ≥102 cm (m)/≥88 cm (f); (b) triglycerides ≥1.7 mmol/L or on medication for high triglycerides; (c) HDL-cholesterol <40 mg/dL in males (m)/<50 mg/dL in women (f) or lipid modifying drug; (d) systolic BP ≥130 mmHg or diastolic blood-pressure ≥85 mmHg; (e) fasting blood glucose ≥6.1 mmol/L.
‡Hypertension = systolic blood pressure (BP) ≥140 mmHg or diastolic BP ≥90 mmHg or antihypertensive pharmacotherapy.
§Arteriosclerotic disease = coronary artery disease, stroke, or peripheral artery disease.
ǁObesity = body mass index ≥30 kg/m2.
#Family history of cardiovascular disease = mother and/or father had myocardial infarction, sudden death, and/or stroke.
**Anaemia = haemoglobin <12 mg/dL in women and <13 mg/dL in men.
††LV systolic dysfunction = LV ejection fraction <52% (m) and <54% (f).
‡‡LV dilation = LV end-diastolic volume index >74 mL/m2 (m) and >61 mL/m2 (f)
§§LV hypertrophy = LV mass index >115 g/m2 (m) and >95 g/m2 (f).
ǁǁLV diastolic dysfunction = ≥3 of the following in individuals with preserved LVEF (>50%): left atrial volume index >34 mL/m2, septal e’ <7 cm/s or lateral e’ <10 cm/s, average E/e′ >14, tricuspid regurgitation velocity >2.8 m/s.
##LA dilation = LA volume index >34 mL/m2.
Compared to individuals in stage 0 (n = 997), B-not-A were of similar age, but more often women (table 1; Supplementary Table e2). B-not-A individuals had lower creatinine, sex-adjusted haemoglobin, LDL-cholesterol and triglycerides. We further observed a trend towards less frequent chronic obstructive pulmonary disease (COPD), lower heart rate and higher HDL-cholesterol. There was no difference regarding blood pressure, frequency of sinus rhythm, body mass index, hip and waist circumference, alcohol consumption, smoking status, family history of cardiovascular disease and history of hypertension, malignancy, depression, thyroid disease and diabetes mellitus. Furthermore, no differences were apparent regarding white blood count, HbA1c, fasting glucose (Table 1; see Supplementary Table e2), change in body weight within the previous 5 years (P=0.33), and physical activity (P=0.15).
All markers classifying an individual as stage B were associated with stage A-defining risk factors, except for LV dilation (see Supplementary Table e5). Left ventricular dilation was associated with anaemia, but there was no association with diabetes mellitus, metabolic syndrome, hypertension, atherosclerotic disease, obesity, smoking, family history of cardiovascular disease, or renal dysfunction (see Supplementary Table e5).
All risk factors indicative of stage A were more prevalent in men, except for obesity (see Supplementary Table e6). Whereas a history of myocardial infarction was more frequent in men, LV dilation and LV dysfunction were more frequent in women. Relevant affection of the valvular apparatus was similarly frequent in both sexes. More women were affected by rheumatic diseases, anaemia and family history of cardiovascular disease, while significantly more men were smokers and consumed more than 14 dpw of alcohol. Heart rate greater than 80/minute, renal dysfunction and elevated levels of high-sensitivity C-reactive protein (hsCRP) or LDL-cholesterol showed no association with sex (see Supplementary Table e6).
Validation
In the validation sample (n = 2492; for characteristics compare Supplementary Table e7), the prevalence of HF stages was 43% for stage A, 22% for stage B and less than 1% for stage C. Again, among individuals with HF risk factors, those in stage B were older, more often women and had more HF risk factors. Of the subjects in stage B, 39% did not have a HF risk factor (B-not-A). As in the derivation sample, this subgroup was younger, more often female, and more often had LV dilation as the stage B qualifying criterion. Stage A-defining risk factors were more frequent in men, and were associated with higher frequencies of echocardiographic abnormalities qualifying for stage B except for LV dilation. LV dilation and LV dysfunction were more frequent in women, whereas myocardial infarction, LV valve stenosis and LV diastolic dysfunction were more frequent in men (see Supplementary Tables e8 and e9).
When applying to the first half of our cohort, the cut-off values for Caucasians as recently reported by the World Alliance of Societies of Echocardiography Normal Values (WASE) study 25 only a minor shift in prevalence rates was observed: stages 0, 42%; stage A, 39%; stage B-not-A, 3%; stage B&A, 15%; stage C, less than 1%. Again, individuals in stage B-not-A were younger (–14 years, P < 0.001) and more often women (+23%, P < 0.001) when compared to B&A.
Discussion
The current analysis reports four major findings derived in a sample of 2473 individuals from the general population and internally validated in another 2492 subjects. First, the prevalence of the early HF stages A and B was high: about one-third of individuals in the youngest age decade (30–39 years) were in HF stage A or B, and the prevalence of both stages increased with advancing age; men predominated in stage A, women in stage B. Second, in the subgroup of individuals with risk factors for HF, individuals also revealing structural heart disease were significantly older, more often women, and their number of risk factors was higher. Third, about one-third of individuals in stage B showed no risk factors defining stage A; these individuals were younger, more often women, with LV dilation as the predominant criterion qualifying for stage B. Fourth, stage A-defining risk factors were more frequent in men and associated with all B-defining echocardiographic abnormalities, except for LV dilation; LV dilation and LV dysfunction were associated with female sex. All four major findings were confirmed in the validation sample of 2492 participants from the same source population with identical demographic stratification and assessment.
We report cross-sectional data from a representative sample of residents of a medium-sized town in Germany over a wide age range and including younger adults. Stratification goals regarding age and sex were exactly met, allowing us to provide standardised prevalence rates for early stages of HF. To our knowledge, this report is the first to evaluate the prevalence and determinants of the early stages of HF in a current population-based study over such a broad age spectrum. The high prevalence of stages A and B in the youngest age group (30–39 years) highlights the importance of extending early HF stage study populations also to younger adults in order to capture and characterise such individuals at risk, to monitor disease evolution and progression and, subsequently, to intervene at the earliest time point possible.
In individuals with prevalent risk factors for HF (stage A and group B&A), we found that age (and thus a potentially longer duration of risk factor exposure), female sex and the number of prevalent risk factors were the most important determinants of structural heart disease. These results endorse the concept of HF as progressive disorder which is triggered by risk factors1,3,4 and confirm the pathophysiological basis for current screening strategies regarding primary prevention of HF.
Unexpectedly, we found a sizeable number of individuals in stage B, who exhibited no risk factors qualifying for stage A. Compared to B&A, B-not-A individuals were younger, more often women, and showed a different phenotype of structural heart disease, with LV dilation as the most prominent qualification criterion for stage B. Prevalence and characteristics of this subgroup were exactly confirmed in the validation sample. Compared to B&A, individuals in stage B-not-A revealed a similar prevalence of reduced LVEF, but mean LVEF was significantly higher, prevalence of LV hypertrophy was significantly lower, and none of the individuals in stage B-not-A had aortic or mitral valve stenosis or more-than-mild diastolic dysfunction. To the best of our knowledge, this subgroup of individuals in stage B-not-A, who would be missed by current primary prevention screening strategies, has not been described before.
Our findings might potentially be influenced by incorrect cut-off values for the definition of structural heart disease. Cumulative distribution functions of the LVEF and the LVEDVi revealed that using the currently recommended thresholds, 13 women were more likely to be classified as abnormal compared to men. In line with this, recently published data from the multi-ethnic WASE study indicate that current cut-off values might overestimate LV dilation but, on the other hand, might underestimate LV hypertrophy. 25 Nevertheless, application of the proposed WASE cut-off values for Caucasians to our sample slightly changed proportions observed for respective HF stages, but consistently revealed a relevant subgroup of individuals with structural heart disease showing no risk factors for HF and affecting mainly women of younger age.
A potential explanation might be that stage A-defining factors causing structural heart disease in these individuals were not present any more at the time of our investigation due to the cross-sectional design of our study. However, the prevalence of self-reported history of diabetes mellitus and history of hypertension as well as of significant weight loss within the previous 5 years showed no statistically significant differences when compared to stage 0. In addition, individuals of B-not-A revealed a significantly different phenotype of structural heart disease when compared to B&A, which renders such a scenario unlikely.
There might also be gender differences regarding HF precursor stages. The current qualification criteria for stage A might more reliably detect HF-associated risk in men. It is also conceivable that risk factors predisposing to the development of HF but particularly affecting women are yet un-identified. In the Health ABC cohort, the annual incidence of HF could be predicted mainly by cardiovascular risk factors. 9 Nevertheless, an analysis from the same cohort regarding the incidence of HF in relation to vascular disease revealed that 27% of individuals with incident HF had no vascular and 37% no coronary disease. Moreover, 67% and 55% of these two subgroups, respectively, had HF with preserved ejection fraction. 6
As expected,2,9 we found traditional risk factors for HF more prevalent in men, but associated with structural heart disease independent of sex, with one important exception. LV dilation predominantly affected women and was not associated with traditional risk factors for HF. This implies that some risk factors for LV dilation may not be included in the current definition of stage A, for example, anaemia. Anaemia as iron deficiency relates to HF, for example, via an increase in catecholamine levels as well as a reduction in the concentration and activity of muscular oxidative enzymes and respiratory proteins, 26 , 27 has a high prevalence in patients with symptomatic HF, and predicts worse outcome. 28 Further research should investigate this relationship in more detail potentially to identify additional risk factors for HF.
Finally, the precursor phase stage A may be ‘skipped’ in a substantial number of individuals. Whereas such a sequence is well acknowledged for HF with reduced ejection fraction, for example, events such as myocarditis, myocardial infarction, or Tako-Tsubo cardiomyopathy might suddenly hit a previously considered healthy individual and shift it into symptomatic stage C, this is not established for HF with preserved ejection fraction. Rather, the precursor stages of HF typically predisposing to HF with preserved ejection fraction are considered as progressively building up over time.1–3
The concept of HF as a progressive disorder with asymptomatic precursor stages 3 has shown its prognostic relevance regarding the development of symptomatic HF and survival,2,7,8 and is the current basis for the primary prevention of HF. Yet, the size of the subgroup B-not-A, which was found in the derivation sample and could be confirmed in size and characteristics in the validation sample, was unexpectedly high and is unsatisfactorily addressed regarding the primary prevention of HF. In particular, other factors might exist causing accelerated impairment of a previously healthy heart, thus advancing the respective individuals from stage 0 into stage B without the precursor stage of preventable risk factors, stage A.
Limitations
We provide cross-sectional data of a representative sample of the general population of the city of Würzburg according to predefined age and sex strata. However, although stratification goals were achieved, the response rate of 33.5% might have introduced a selection bias hampering the generalisability of our findings. According to the study protocol, the oral glucose tolerance test was only performed in individuals attending the joint survey unit before 11:00 a.m. (62% of participants), thus reducing information for the whole sample. However, fasting glucose and HbA1c were assessed allowing for reliably diagnosing diabetes mellitus. 15 Potential further causes of structural heart disease and HF could not be evaluated due to the lack of detailed information on prior pregnancies or potentially cardiotoxic chemotherapy and remain subject to future studies.
Dedicated recruitment strategies with the achievement of stratification goals in the derivation and the validation sample made extensive internal validation possible. Nevertheless, the presence of the subgroup B-not-A has to be validated externally in cohorts from different source populations. Furthermore, a slight deviation from normal might not translate into an increased risk of developing symptomatic heart failure, and current reference values might not exactly be applicable to our study population.
Another important aspect regarding HF risk prediction might be exercise tolerance. Exercise chronotropic incompetence was more pronounced in individuals with higher numbers of cardiovascular risk factors. 30 Furthermore, cardiopulmonary exercise test performance predicted events in HF patients. 31 In our study, this aspect was not evaluated at baseline, but will be subject to follow-up investigations.
As participants of the STAAB cohort will be followed in 5-year intervals, the recording of the temporal changes of risk factors, cardiac dimensions and function, and incident cardiovascular events will provide insights into the natural course of individuals across the different stages of HF and might help to refine thresholds defining deviation from normal.
Conclusions
The prevalence of precursor HF stages A and B in the general population was found to be high across all age groups and increased with advancing age. Primary prevention programmes for HF should aim to identify and address individuals at high risk of developing HF already in younger adulthood. We identified a hitherto undescribed patient subgroup exhibiting structural heart disease but lacking risk factors for HF. This group, B-not-A, deserves further study either to detect novel risk factors for HF that mainly affect younger women or to identify adverse circumstances inducing structural heart disease in previously healthy individuals.
Supplemental Material
sj-pdf-1-cpr-10.1177_2047487320922636 - Supplemental material for Prevalence and determinants of the precursor stages of heart failure: results from the population-based STAAB cohort study
Supplemental material, sj-pdf-1-cpr-10.1177_2047487320922636 for Prevalence and determinants of the precursor stages of heart failure: results from the population-based STAAB cohort study by Caroline Morbach, Götz Gelbrich, Theresa Tiffe, Felizitas A Eichner, Martin Christa, Renate Mattern, Margret Breunig, Vladimir Cejka, Martin Wagner, Peter U Heuschmann and Stefan Störk in European Journal of Preventive Cardiology
Footnotes
Author contribution
CM, SS, PUH and GG contributed to the conception or design of the work. CM, GG, TT, FE, MC, RM, MB, VC, MW, PUH and SS contributed to the acquisition, analysis, or interpretation of data for the work. CM and GG drafted the manuscript. TT, FE, MC, RM, MB, VC, MW, PUH and SS critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work ensuring integrity and accuracy.
Acknowledgements
The authors greatly appreciate the time of all STAAB participants and their willingness to provide data to the study. They also thank the mayor of the city of Würzburg and the local registration office for their sustained support of our study. They also wish to thank Jasmin Simon, Yvonne Memmel, Frances Knobeloch, Heike Hergenröder, Melanie Roos, Sabine Cäsar, Timo Ludwig, and the entire study team, study nurses, technicians, data managers and students for their efforts on the STAAB study. The authors also thank M Ertl, G Fette and F Puppe from the CHFC DataWarehouse, Institute of Informatics VI, University of Würzburg, as well as T Ludwig, ICE-B, for diligent data management.
The authors do hereby declare that all illustrations and figures in the manuscript are original and do not require reprint permission.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Caroline Morbach reports a research cooperation with the University of Würzburg and Tomtec Imaging Systems funded by a research grant from the Bavarian Ministry of Economic Affairs, Regional Development, Amgen, and Energy, Germany, speakers honorarium from Amgen and Tomtec, a travel grant from Orion Pharma and Alnylam, and participation in Advisory and Patient Eligibility Boards sponsored by AKCEA, Alnylam, Pfizer and EBR Systems outside the submitted work. Götz Gelbrich reports a research cooperation with the University Hospital Würzburg and TomTec Imaging Systems funded by a research grant from the Bavarian Ministry of Economic Affairs, Regional Development and Energy, Germany, and a grant from the German Research Council (DFG) as the senior biometrician of the FIND-AF II trial outside the submitted work. Theresa Tiffe, Felizitas Eichner, Martin Christa, Renate Mattern, Margret Breunig, Vladimir Cejka and Martin Wagner have nothing to disclose. Peter Heuschmann reports grants from the German Ministry of Research and Education, German Research Foundation, European Union, Charité – Universitätsmedizin Berlin, Berlin Chamber of Physicians, German Parkinson Society, University Hospital Würzburg, Robert Koch Institute, German Heart Foundation, Federal Joint Committee (G-BA) within the Innovationfond, University Hospital Heidelberg (within RASUNOA-prime; RASUNOA-prime is supported by an unrestricted research grant to the University Hospital Heidelberg from Bayer, BMS, Boehringer-Ingelheim, Daiichi Sankyo), grants from Charité – Universitätsmedizin Berlin (within Mondafis; Mondafis is supported by an unrestricted research grant to the Charité from Bayer), from University Göttingen (within FIND-AF randomized; FIND-AF randomized is supported by an unrestricted research grant to the University Göttingen from Boehringer-Ingelheim), outside the submitted work. Stefan Störk reports research grants from the German Ministry of Education and Research, European Union, University Hospital Würzburg; participation in Data Safety Monitoring or Event Adjudication in studies sponsored by Roche, Medtronic; participation in Advisory Boards for Novartis, Bayer, Boehringer Ingelheim, Thermo-Fisher, Boston Scientific; principal investigator in trials (co-)sponsored by Boehringer-Ingelheim, Novartis, Bayer, Lundbeck; speaker honoraria by Boehringer-Ingelheim, Servier, Novartis, Astra-Zeneca, Pfizer, Bayer, Thermo-Fisher, outside the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article. This work was supported by the German Ministry of Research and Education within the Comprehensive Heart Failure Centre Würzburg (BMBF 01EO1004 and 01EO1504). This publication was funded by the German Research Foundation (DFG) and the University of Würzburg in the funding program Open Access Publishing. The funding source did not have any role in the collection, analysis, and interpretation of the data, in the writing of the report, nor in the decision to submit the paper.
Collection of biomaterials was supported by the Interdisciplinary Bank of Biomaterials and Data of the University Hospital of Würzburg and the Julius Maximilian University of Würzburg (ibdw). The implementation of the IBDW has been supported by a grant of the Federal Ministry for Education and Research (grant number FKZ: 01EY1102).
The STAAB Consortium consists of: S Frantz (Dept. of Medicine I, Div. of Cardiology, University Hospital Würzburg); C Maack (Comprehensive Heart Failure Center, University Hospital and University of Würzburg); G Ertl (University Hospital Würzburg); M Fassnacht (Dept. of Medicine I, Div. of Endocrinology, University Hospital Würzburg); C Wanner (Dept. of Medicine I, Div. of Nephrology, University Hospital Würzburg); R Leyh (Dept. of Cardiovascular Surgery, University Hospital Würzburg); J Volkmann (Dept. of Neurology, University Hospital Würzburg); J Deckert (Dept. of Psychiatry, Psychosomatics and Psychotherapy, Center of Mental Health, University Hospital Würzburg); H Faller (Dept. of Medical Psychology, University of Würzburg); R Jahns (Interdisciplinary Bank of Biomaterials and Data Würzburg, University Hospital Würzburg).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
