Abstract
Aims
Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors consistently reduce low-density lipoprotein cholesterol (LDL-C) by 50–60% and lipoprotein(a) (Lp(a)) by 20–30%, but the mechanism of Lp(a) lowering remains unclear. If Lp(a) is cleared by the LDL receptor, similar to LDL-C, then one would expect PCSK9 inhibition to induce a concordant LDL-C/Lp(a) response in an approximately 2:1 ratio. We aim to determine the prevalence of discordant plasma LDL-C/Lp(a) response to the PCSK9 inhibitor alirocumab.
Methods
This is a post hoc, pooled analysis of 10 randomized controlled trials from the ODYSSEY Phase 3 clinical trial program for alirocumab. Patients enrolled in the trials were high cardiovascular risk and/or with heterozygous familial hypercholesterolemia. The primary end point was prevalence of discordant LDL-C/Lp(a) response to alirocumab at 24 weeks. Discordant response was defined as LDL-C reduction >35% and Lp(a) reduction ≤10%, or LDL-C reduction ≤35% and Lp(a) reduction >10%.
Results
Of the 1709 patients in the pooled study cohort, 62.4% were male, and the mean age was 59.2 (SD: 11.0) years. Baseline mean LDL-C was 126.5 (SD: 46.3) mg/dL and baseline median Lp(a) was 46.9 (interquartile range: 21.8-89.0) mg/dL. Total prevalence of discordant LDL-C/Lp(a) response was 21.5% (12.6% with LDL-C >35% reduction and Lp(a) ≤10% reduction; 8.9% with LDL-C ≤35% reduction and Lp(a) >10% reduction). Baseline Lp(a) and familial hypercholesterolemia status did not affect discordance.
Conclusion
A high prevalence of discordant LDL-C/Lp(a) response was observed with alirocumab, further suggesting that PCSK9 inhibitor therapy with alirocumab reduces plasma Lp(a) through alternative pathways to LDL receptor clearance.
Introduction
Proprotein convertase subtilisin/kexin type 9 (PCSK9) acts as a central regulator of lipoprotein metabolism by targeting the low-density lipoprotein receptor (LDLR) for degradation within hepatocytes. 1 Alirocumab and evolocumab are therapeutic monoclonal antibodies that target and neutralize plasma PCSK9, leading to drastic reductions in plasma concentrations of low-density lipoprotein cholesterol (LDL-C). In 2015, the U.S. Food and Drug Administration approved both of these PCSK9 inhibitors to treat adults with heterozygous familial hypercholesterolemia and/or patients with atherosclerotic cardiovascular disease (ASCVD) who require additional LDL-C lowering. More recently, two large trials demonstrated improved cardiovascular outcomes with the use of PCSK9 inhibitors in patients with a history of stable ASCVD (evolocumab) or recent acute coronary syndrome (alirocumab).2,3
Lipoprotein(a) (Lp(a)) is another circulating atherogenic lipoprotein4–8 whose plasma levels are affected by PCSK9 inhibition. Analyses of multiple Phase 2 and 3 trials for both PCSK9 inhibitors demonstrate 20–30% reductions in Lp(a).7,9–12 Clinical trial populations repeatedly demonstrate mean LDL-C reduction to be approximately twice that of mean Lp(a) reduction secondary to PCSK9 inhibition.13–21 While the mechanism of PCSK9 inhibitor-mediated LDL-C lowering is well-established, the mechanism by which PCSK9 inhibition reduces plasma Lp(a) remains poorly understood. Though Lp(a) may be cleared partly through the LDLR, several lines of evidence point toward alternative mechanisms.22–25 If Lp(a) clearance was fully accounted for by the LDLR pathway, one would anticipate PCSK9 inhibitor therapy to consistently reduce LDL-C and Lp(a) in a 2:1 ratio (a concordant reduction) in the same individual. We previously defined PCSK9 inhibitor-induced discordance as expected LDL-C reduction without significant Lp(a) reduction (LDL-C reduction >35% and Lp(a) reduction ≤10%), and recently published an analysis of four Phase 3 clinical trials to determine the prevalence of discordance with evolocumab therapy.22,23 Overall, the prevalence of expected LDL-C reduction in the absence of significant Lp(a) reduction (discordance) after evolocumab therapy was 18.4%. Furthermore, the prevalence of discordance was influenced by baseline Lp(a), being more common in patients with higher baseline Lp(a). These findings suggest that PCSK9 inhibitor therapy may reduce plasma Lp(a) through mechanisms independent of LDLR-mediated catabolism, particularly in patients with elevated Lp(a). Given potential differences between the two therapeutic PCSK9 monoclonal antibodies, we sought to determine the prevalence of discordance in response to treatment with alirocumab using the same definitions of concordance/discordance.22,23 Furthermore, we used a database including a significant proportion of patients with familial hypercholesterolemia, since it is important to determine whether discordant PCSK9 inhibitor response between these two lipoproteins is diminished or accentuated by a defective LDLR pathway.
Methods
Study cohort
This study is a pooled analysis of participants from 10 randomized, double-blinded, placebo or ezetimibe-controlled trials of 24–78 weeks in duration within the ODYSSEY Phase 3 clinical trial program (Supplementary Material Table 1). All trial protocols were approved by the appropriate review body, and all patients provided informed written consent. These studies include ALTERNATIVE, COMBO I, COMBO II, FH I, FH II, HIGH FH, LONG TERM, MONO, OPTIONS I, and OPTIONS II. Study design and outcomes have previously been published.13–21 Two of these studies, ODYSSEY LONG TERM and HIGH FH, assessed the efficacy of alirocumab 150 mg every two weeks (Q2W). The other eight studies initiated alirocumab at a dose of 75 mg Q2W and the dose was increased to 150 mg Q2W at week 12 depending on achieved LDL-C at week 8. Alirocumab treatment was added on to maximally tolerated statin doses in six studies except for ODYSSEY ALTERNATIVE, MONO, and OPTIONS I and II. Patients included in these Phase 3 studies were at high cardiovascular risk, including both heterozygous familial hypercholesterolemia and non-familial hypercholesterolemia patients. Familial hypercholesterolemia status was evaluated either genetically or by clinical criteria (i.e. Simon Broome or World Health Organization (WHO)/Dutch Lipid Network criteria). Patients with known homozygous familial hypercholesterolemia were excluded. Both calculated LDL-C and Lp(a) were collected at baseline, and weeks 12 and 24, with week 24 as the primary time point for efficacy evaluation.
LDL-C and Lp(a) measurement
LDL-C was calculated using the Friedewald formula. 26 For patients whose triglycerides exceeded 4.5 mmol/L, the central laboratory measured LDL-C using the beta-quantification method. All studies used Medpace Reference Laboratories (Cincinnati, Ohio, USA; Leuven, Belgium; Singapore), except for LONG TERM, where Covance Central Laboratory Services were used. Serum Lp(a) was analyzed by Medpace Reference Laboratories in all studies, except for LONG TERM, where Covance Central Laboratory Services were used. However, all measurements were performed with an established immunoturbidimetric assay on a Siemens BNII (Siemens, Erlangen, Germany) validated against the International Federation of Clinical Chemistry and WHO standards.
Study end points
The primary end point for this post hoc analysis was the prevalence of concordant/discordant responses of LDL-C and Lp(a) to alirocumab after 24 weeks of therapy. Concordant response was defined as LDL-C reduction >35% and Lp(a) reduction >10% (and LDL-C reduction ≤35%; Lp(a) reduction ≤10%), as used in two prior studies.22,23 A discordant response was defined as LDL-C reduction >35% and Lp(a) reduction ≤10%, or LDL-C reduction ≤35% and Lp(a) reduction >10%.
There were initially 3141 patients in the combined alirocumab arms in the 10 Phase 3 trials for this pooled analysis. To qualify for this pooled analysis, patients needed to meet the following inclusion criteria: (a) alirocumab treatment for at least 24 weeks, (b) measurements of LDL-C and Lp(a) at both baseline and week 24, and (c) baseline Lp(a) ≥10 mg/dL. A baseline Lp(a) level of ≥10 mg/dL was chosen because percent reductions in Lp(a) would be exaggerated with lower baseline values. In total, 1709 patients met all inclusion criteria (Supplementary Material Table 2). Supplementary Material Table 3 shows the number of patients from each trial that met inclusion criteria for the pooled analysis.
Statistical analysis
For this post hoc analysis, the percentages of concordance and discordance were calculated in the overall population and in subgroups (familial hypercholesterolemia versus non-familial hypercholesterolemia; cutoffs of minimum baseline Lp(a) levels). The overlapping Lp(a) subgroups were: baseline Lp(a) ≥10 mg/dL, ≥30 mg/dL, and ≥50 mg/dL.
Continuous variables are summarized with means and standard deviations (SDs) or medians and interquartile ranges (IQRs). Categorical variables are summarized with counts and percentage. The differences in the proportions of concordance between the familial hypercholesterolemia and non-familial hypercholesterolemia groups were tested using Chi-square test. Clopper and Pearson methods were used to calculate 95% confidence intervals of binomial proportions and percentages. Spearman’s R2 coefficient was calculated for the correlation between LDL-C and Lp(a) values at week 24. All analyses were performed with SAS/STAT v9.4 software (SAS Institute).
Results
Baseline characteristics of the pooled study population are described in Table 1. Of note, 1067 (62.4%) were male and mean age was 59.2 (SD: 11.0) years. Mean LDL-C and median Lp(a) in the overall population were 126.5 (SD: 46.3) mg/dL and 46.9 (IQR: 21.8–89.0) mg/dL, respectively. Baseline diagnosis of familial hypercholesterolemia was reported in 32.4% (n = 554) of patients. Baseline mean LDL-C by familial hypercholesterolemia and non-familial hypercholesterolemia status was 153.4 mg/dL and 113.6 mg/dL, respectively. Baseline median Lp(a) by familial hypercholesterolemia and non-familial hypercholesterolemia status was 45.0 mg/dL and 47.2 mg/dL, respectively.
Baseline characteristics of the pooled study population by familial hypercholesterolemia status.
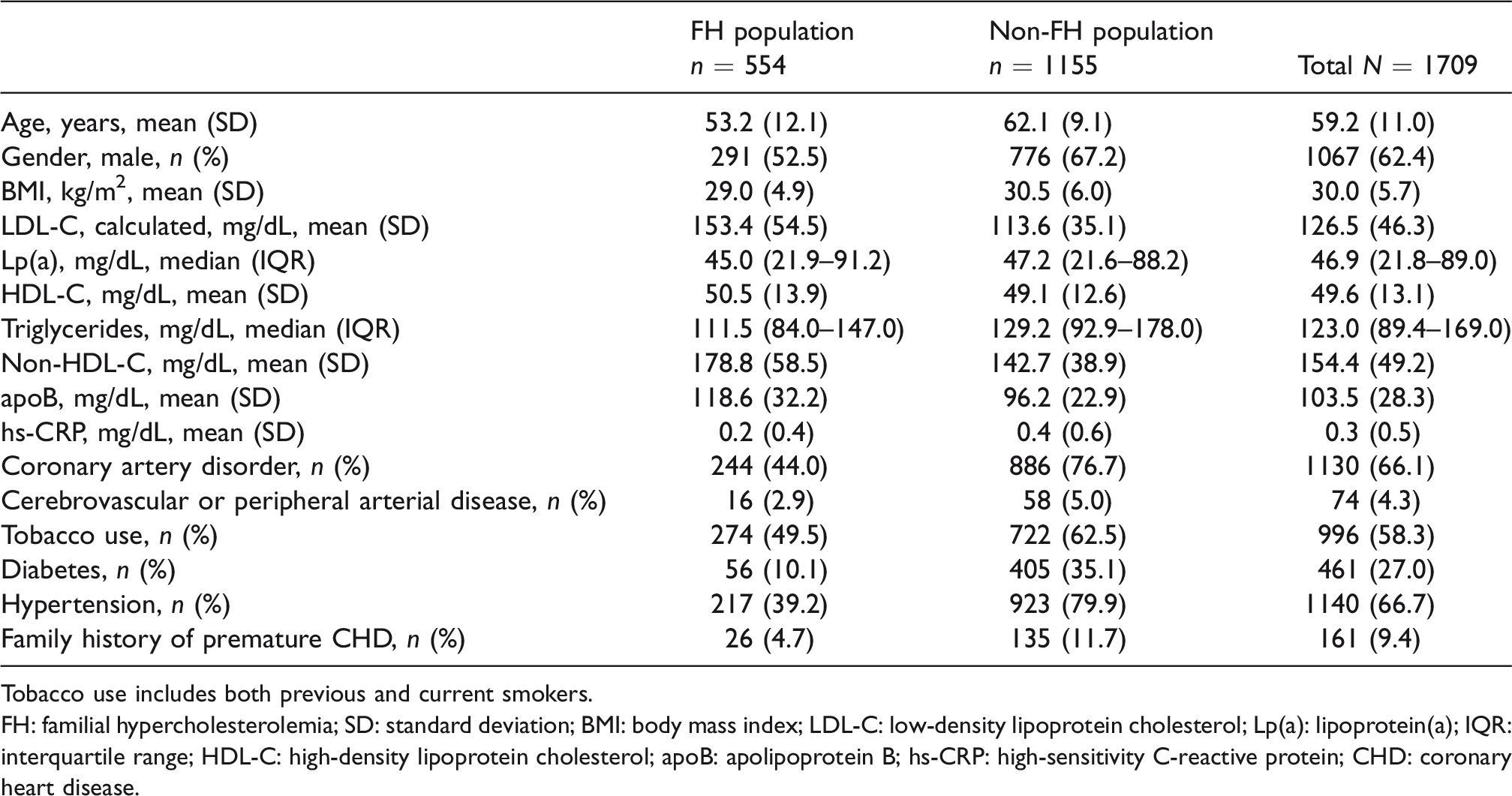
Tobacco use includes both previous and current smokers.
FH: familial hypercholesterolemia; SD: standard deviation; BMI: body mass index; LDL-C: low-density lipoprotein cholesterol; Lp(a): lipoprotein(a); IQR: interquartile range; HDL-C: high-density lipoprotein cholesterol; apoB: apolipoprotein B; hs-CRP: high-sensitivity C-reactive protein; CHD: coronary heart disease.
Table 2 details the changes in plasma LDL-C and Lp(a) concentrations from baseline to week 24 of alirocumab therapy. Regardless of alirocumab dosing regimen, the majority of patients achieved an LDL-C reduction of >35% (n = 1446, 84.6%) with alirocumab therapy at 24 weeks. A similar number of patients achieved a Lp(a) reduction of >10% (n = 1383, 80.9%). Overall, the total prevalence of concordant response of LDL-C and Lp(a) to alirocumab was 78.5% (n = 1342). The prevalence of the two patterns of discordance was similar. There were 215 patients (12.6%) who achieved LDL-C >35% reduction and Lp(a) ≤10% reduction. The opposite pattern of discordance (LDL-C ≤35% reduction and Lp(a) >10% reduction) was slightly less common (n = 152, 8.9%). The total prevalence of discordant responses was 21.5% (n = 367).
Percent reduction in plasma LDL-C and Lp(a) amongst patients treated with alirocumab at 24 weeks by baseline plasma Lp(a) concentration.
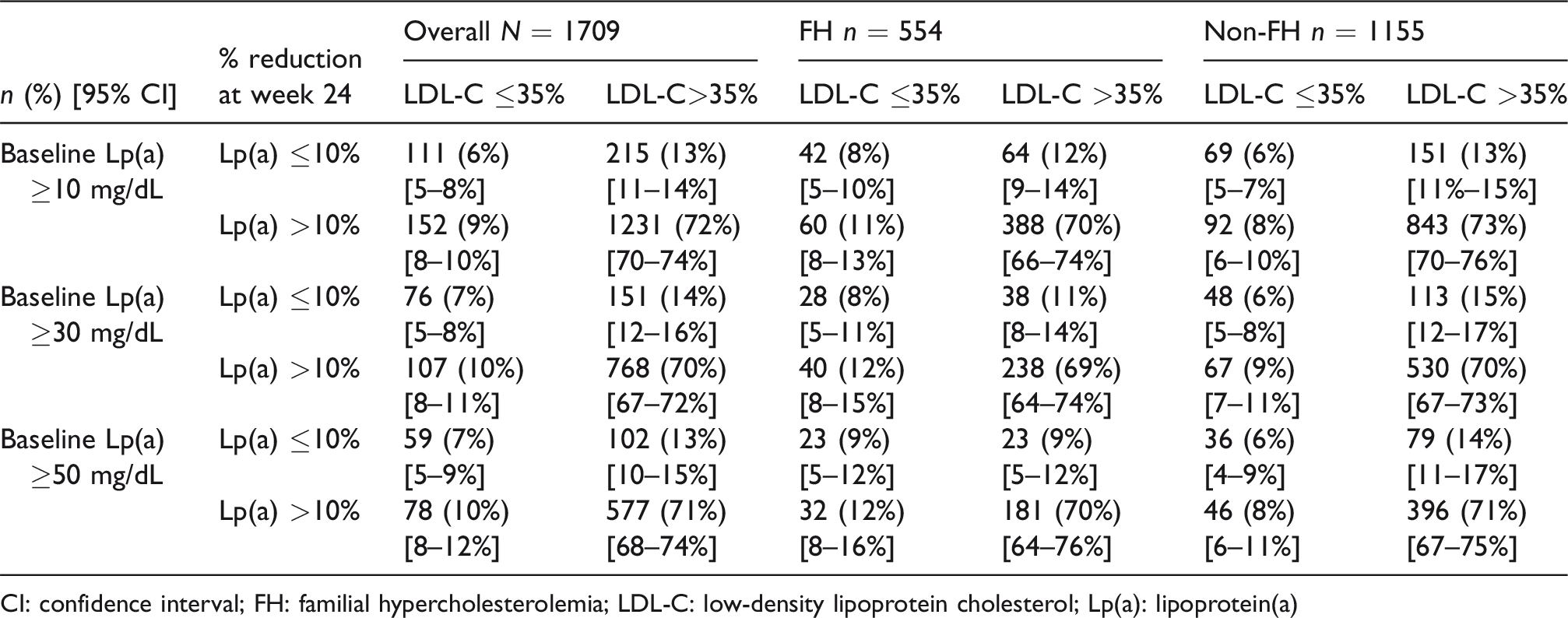
CI: confidence interval; FH: familial hypercholesterolemia; LDL-C: low-density lipoprotein cholesterol; Lp(a): lipoprotein(a)
The prevalence of discordance stratified by familial hypercholesterolemia status and baseline Lp(a) level is depicted in Figure 1. Familial hypercholesterolemia status did not significantly modulate the degree of discordant responses of LDL-C and Lp(a) to alirocumab at 24 weeks. The prevalence of discordance (LDL-C >35% reduction, Lp(a)
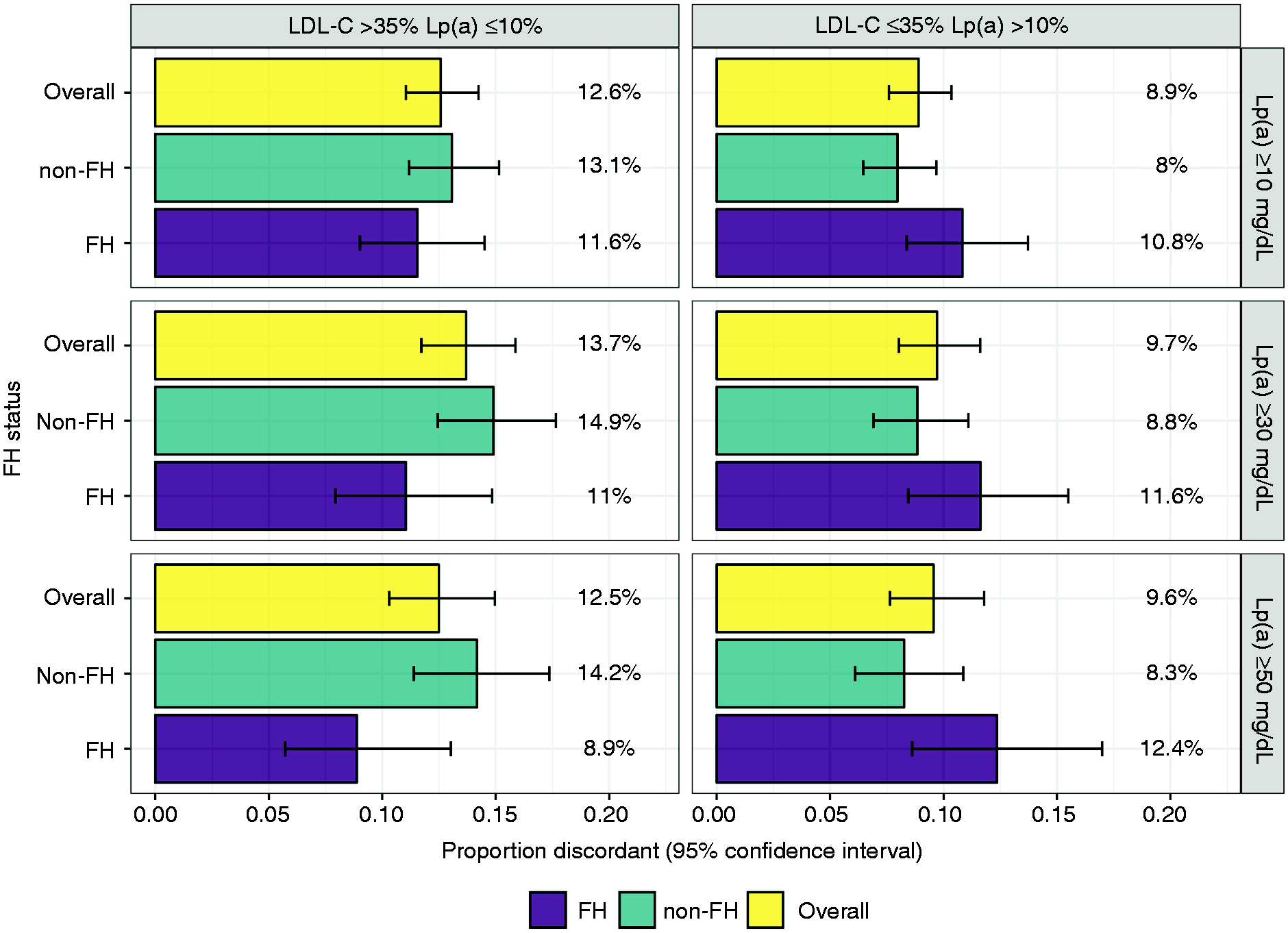
Percent LDL-C/Lp(a) discordant response to alirocumab stratified by baseline Lp(a) and FH status. No significant differences (p< 0.05) were observed in LDL-C/Lp(a) discordant responses to alirocumab by baseline Lp(a) or FH status. Data shown as percent of total number of all, FH, or non-FH patients in each of the concordance and discordance categories, with error bars representing 95% Clopper–Pearson binomial proportion confidence interval.
Reduction in Lp(a) at 24 weeks was assessed according to baseline LDL-C and baseline Lp(a) concentrations (Figures 2 and 3). No correlation was observed between baseline LDL-C and percent change in Lp(a) from baseline to 24 weeks (R2 = 0.002; p = 0.1075). Additionally, no correlation was observed between baseline Lp(a) and percent change in Lp(a) from baseline to 24 weeks as evidenced by a R2 value close to zero (R2 = 0.080; p <0.0001).
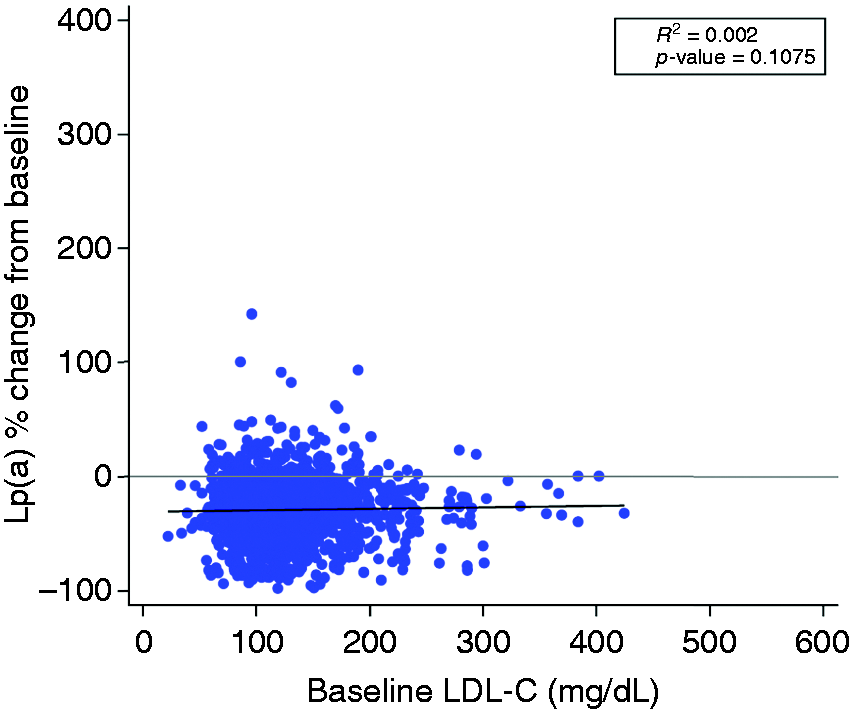
Relationship between percent change in Lp(a) at week 24 of alirocumab treatment and baseline LDL-C. Spearman’s correlation R2 of baseline LDL-C, % change in Lp(a), and p-value are shown with least-squares linear fit line.
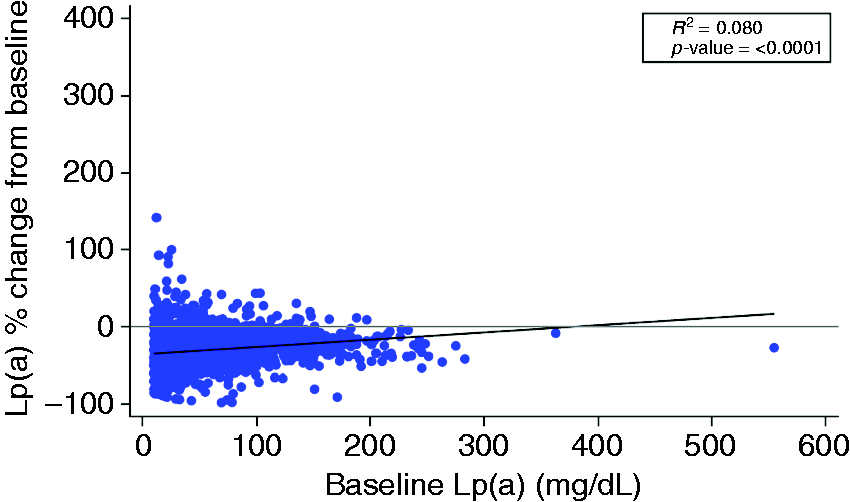
Relationship between percent change in Lp(a) at week 24 of alirocumab treatment and baseline Lp(a). Spearman’s correlation R2 of baseline LDL-C, % change in Lp(a), and p-value are shown with least-squares linear fit line.
Discussion
We analyzed the prevalence of discordant LDL/Lp(a) responses among 1709 trial participants who received alirocumab. In the 1446 patients who achieved LDL-C reduction >35% on alirocumab, Lp(a) decreased by ≤10% in 215 of them, which is 12.6% of the total study population. Interestingly, there were also many patients who achieved significant Lp(a) reduction without the anticipated LDL-C reduction, leading to a total prevalence of discordance of 21.5%. Baseline Lp(a) and familial hypercholesterolemia status did not significantly impact the observed prevalence of discordant lipoprotein responses.
A mechanistic understanding of Lp(a) catabolism remains unclear despite the discovery of this enigmatic lipoprotein almost 60 years ago. 27 Nonetheless, there is compelling epidemiologic and genetic evidence that consistently suggests that Lp(a) is an independent risk factor for ASCVD4,5 and calcified aortic stenosis.6,8 The atherogenicity of Lp(a) is likely multifactorial, including its physiochemical similarity to LDL, the presence of oxidized phospholipids on the apolipoprotein (a) (apo(a)) moiety, its homology to plasminogen, and the lack of apo(a) lipid domain allowing binding to exposed lysine-rich vascular endothelium. 28 Given that Lp(a) is associated with higher risk of ASCVD and the absence of an effective Lp(a)-lowering therapy other than lipid apheresis, 29 the observation that PCSK9 inhibition consistently lowers plasma Lp(a) concentrations is of great interest.7,9–12 It has been difficult to reconcile this unanticipated finding, as other therapies that lower LDL-C through upregulation of the LDLR, such as statins, do not modulate the plasma levels of Lp(a). 30 In fact, statins tend to increase plasma Lp(a) levels. 31 Experimental studies suggest that alternative mechanisms beyond the LDLR pathway may play a role in the observed Lp(a) reduction with PCSK9 inhibitor therapy, including effects on hepatocyte apo(a) secretion 24 and Lp(a) production, 25 though a recent kinetics study suggests alirocumab does not affect apo(a) production. 32 Additionally, Lp(a) is implicated in binding to a number of other receptors (e.g. lectin, plasminogen, scavenger, toll-like receptors), which may be involved in its clearance. 33 If any of these LDLR-independent mechanisms are modulated by PCSK9 inhibition, this may explain the discordant responses in LDL-C and Lp(a) reduction observed with alirocumab.
Based on previously defined cutoffs for concordant and discordant LDL-C and Lp(a) responses to PCSK9 inhibition,22,23 this pooled analysis from 10 ODYSSEY Phase 3 studies shows discordance in 21.5% of the study population, suggesting that discordant lipoprotein responses to PCSK9 inhibition are common and pointing towards LDLR-independent pathways for Lp(a) lowering. It is important to note that two discordance patterns are possible in response to PCSK9 inhibition: 1) expected LDL-C lowering without expected Lp(a) lowering, and 2) expected Lp(a) lowering without expected LDL-C lowering. The former scenario challenges the prevailing view of LDLR-mediated Lp(a) clearance. If in fact increased LDLR preservation secondary to PCSK9 inhibition played an exclusive role in Lp(a) clearance, then one would expect significant Lp(a) reduction in all cases where LDL-C was lowered to the anticipated extent. In this analysis, we observed the expected LDL-C lowering in the absence of significant Lp(a) lowering in 12.6% of the overall population. The prevalence of this discordant response did not vary by baseline Lp(a) level or familial hypercholesterolemia status.
The opposite discordance pattern (LDL-C reduction ≤35% and Lp(a) reduction >10%) in response to PCSK9 inhibition is perhaps even more challenging to the simple concept that Lp(a) is lowered because of activation of the LDLR by PCSK9 inhibition. In fact, when Lp(a) is reduced more than LDL-C by PCSK9 inhibition, the LDLR cannot be the driver of the effect (since LDLR has much lower affinity for Lp(a) than for LDL). An exaggerated lowering of Lp(a) by PCSK9 inhibition in the absence of expected LDL-C lowering is compatible with either specific catabolic effects (activation of a “Lp(a) receptor”) or modulation of synthesis or assembly. Interestingly, the two discordance patterns were observed to a similar extent in this study. Another interesting observation was a higher prevalence of the opposite discordance pattern in familial hypercholesterolemia compared with non-familial hypercholesterolemia patients. However, this latter finding did not achieve statistical significance.
The findings of the present analysis agree with our recent study to assess the prevalence of discordance in response to the other PCSK9 inhibitor, evolocumab. 23 Using the same definition of discordance, we found a similar prevalence of discordance amongst clinical trial participants receiving evolocumab (21.2% vs. 21.5% in this study). However, in the previous study, the discordance pattern predominantly manifested as significant LDL-C reduction in the absence of significant Lp(a) reduction. In fact, in the prior work, only 2.8% of the entire study population exhibited the opposite discordance pattern. Explanations for the differences in the prevalence of discordance patterns in the two studies are not obvious. The prior study compared lipid levels at baseline and post-evolocumab at 12 weeks. In the present analysis, lipid levels were evaluated at baseline and post-alirocumab at 24 weeks. However, the findings of our discordance analysis were similar when analyzing lipid data at weeks 12 and 52 (Supplementary Material Tables 4A and 4B). While direct comparisons between the two study populations cannot be made, baseline characteristics appear similar, including baseline LDL-C and Lp(a). The current study included a significant proportion of patients with familial hypercholesterolemia (32.4%); however, familial hypercholesterolemia status did not influence the prevalence of discordance. In this study, baseline Lp(a) did not modify the relationship between LDL-C and Lp(a) lowering in response to alirocumab. This finding differs from the prior analysis with evolocumab, which demonstrated a progressively increasing prevalence of discordance with higher baseline Lp(a). 23 Given the inverse relationship between Lp(a) mass and apo(a) isoform size, perhaps more patients in the prior analysis with evolocumab had smaller apo(a) isoforms compared with our current study, which may account for the discrepancy. However, this is only speculative as apo(a) isoform measurements were not reported for either analysis. Furthermore, there is evidence that alirocumab may reduce Lp(a) independent of apo(a) isoform size. 34 More patients in the evolocumab analysis achieved LDL-C reduction >35% compared with our study (93.7% vs. our 84.6%). This may be due to the fact that many patients in our analysis (i.e. all studies except HIGH FH and LONG TERM) remained on the initial alirocumab 75 mg Q2W dose. Fewer patients achieving the LDL-C reduction cutoff in the present analysis may explain why we observed a similar overall prevalence of total discordance but a higher prevalence of the opposite pattern of discordance. Differences in the antibodies themselves may have contributed to the differences in the findings, though it is not clear by what mechanism this would occur, as the two PCSK9 inhibitors operate on the same mechanism of action and have similar LDL-C lowering efficacy at maximum doses.
While this study makes use of prospectively collected clinical trial data, it is a post hoc analysis and should be considered exploratory in nature. Nonetheless, the data analyzed are from multiple randomized, double-blinded, controlled clinical trials. The study population of the present analysis is larger than in prior reports and the overall effect size is similar. However, a more precise estimate of the prevalence of discordance would require a larger number of patients, ideally derived from one study population.22,23 The interval between baseline and post-alirocumab LDL-C and Lp(a) measurements was relatively short (24 weeks). It is conceivable that longer exposure to alirocumab would yield different results; however, an exploratory analysis with data collected at 52 weeks yielded similar results (Supplementary Material Table 4B). Perhaps most important is the operative definition of concordance/discordance. Currently, there is no widely accepted definition of discordant response between LDL-C and Lp(a) secondary to PCSK9 inhibition. We chose the previously used definition of discordance22,23 to remain consistent with prior studies and to facilitate meaningful comparisons between prior analyses. Moreover, this definition was conservatively formulated to avoid misclassification (overprediction) of discordance and in fact likely yields an underestimate of the prevalence of meaningfully discordant lipoprotein responses to PCSK9 inhibition. Finally, although LDL-C/Lp(a) discordant response to PCSK9 inhibition suggests alternative pathways to LDLR-mediated clearance of Lp(a), dedicated mechanistic studies, including evaluation of production and other clearance pathways, still need to be performed to further understand this observation.
Conclusion
The aim of this study was to define the prevalence and explore the biologic implications of alirocumab-induced discordance of plasma LDL-C and Lp(a) responses. We confirm a high prevalence of discordance in the degree to which these two lipoprotein fractions respond to alirocumab when the cutoffs for response to therapy were LDL-C reduction >35% and Lp(a) reduction >10%. Importantly, the two patterns of discordance were equally prevalent and suggest that Lp(a) does not always or exclusively utilize the LDLR as a clearance receptor. Alternative pathways of Lp(a) reduction affecting both clearance and production rates may be modulated by PCSK9 inhibition with alirocumab.
Supplemental Material
CPR915803 Supplemental Material - Supplemental material for Discordant responses of plasma low-density lipoprotein cholesterol and lipoprotein(a) to alirocumab: A pooled analysis from 10 ODYSSEY Phase 3 studies
Supplemental material, CPR915803 Supplemental Material for Discordant responses of plasma low-density lipoprotein cholesterol and lipoprotein(a) to alirocumab: A pooled analysis from 10 ODYSSEY Phase 3 studies by Tahir Mahmood, Jessica Minnier, Matthew K Ito, Qian H Li, Andrew Koren, Ivy W Kam, Sergio Fazio and Michael D Shapiro in European Journal of Preventive Cardiology
Footnotes
Author contribution
TM, SF, and MDS contributed to the conception or design of the work. All authors contributed to the acquisition, analysis, or interpretation of data for the work. TM and MDS drafted the manuscript. All authors critically revised the manuscript. All gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Acknowledgement
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MDS has received compensation for advisory activities from Esperion and Amarin. SF has received compensation for advisory activities from Amarin, Amgen, Astra-Zeneca, Esperion, and Novartis. TM and JM declare no conflict of interest. MKI and AK are employees/stockholders of Sanofi. QHL and IWK are employees/stockholders of Regeneron Pharmaceuticals Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MDS was partially supported by NIH K12HD043488. SF was partially supported by NIH 5R01HL132985. This study was approved by Sanofi/Regeneron Pharmaceuticals, Inc., but no direct or indirect funding was provided to either Oregon Health & Science University (OHSU) or the co-authors from OHSU. The sponsor provided funding for the parent studies and via employee coauthors provided input into the design and conduct of the study as well as collection, management, and analysis of the data review. The sponsor reviewed the manuscript prior to submission, but the preparation, approval, and decision to submit the manuscript for publication were made by the coauthors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
