Abstract
Background:
Prediabetes substantially increases cardiovascular risk. We examined the effect of rosuvastatin on the quantity and quality of low-density lipoprotein cholesterol (LDL-C) in patients with dyslipidemia having impaired fasting glucose (IFG) compared to normoglycemic patients with dyslipidemia.
Methods:
This was a prospective observational study including patients with dyslipidemia and IFG (IFG group, n = 49) matched with normoglycemic patients with dyslipidemia (control group, n = 64). Study participants, following dietary intervention, were prescribed rosuvastatin 10 or 20 mg/d to achieve LDL-C goals. Baseline as well as 24 weeks posttreatment changes in the serum lipid profile were evaluated. Moreover, analysis of the LDL subfraction profile was conducted using a polyacrylamide tube gel electrophoresis method.
Results:
Similar effects were observed in lipid profile in both treatment groups. Patients with IFG experienced a greater decrease in the cholesterol concentration of small dense LDL particles (−65.7%, P < .001 vs baseline) compared to controls (−38.5%, P < .001 vs baseline; P = .018 vs patients with IFG). There was no significant difference in the changes of cholesterol concentration of large and buoyant LDL particles in the IFG group when compared to the control group. A greater increase in the mean LDL particle size (+1.5%, P < .001 vs baseline) was noted in the IFG group compared to the control group at 24 weeks (+0.4%, P = .028 vs baseline; P = .008 vs IFG group).
Conclusion:
Targeting dyslipidemia with rosuvastatin was associated with more favorable changes in the LDL subfraction profile in patients with IFG compared to normoglycemic ones.
Keywords
Introduction
Hypercholesterolemia is an independent risk factor for the development and progression of cardiovascular (CV) disease, which is among the leading causes of mortality globally. 1 Raised low-density lipoprotein cholesterol (LDL-C) levels proportionally increase the risk of CV events. However, this risk cannot be entirely predicted by LDL-C levels. 2 Several lipid contributors associated with the “residual” CV risk include atherogenic dyslipidemia (elevated triglycerides [TGs] together with low high-density lipoprotein cholesterol [HDL-C] levels) and the distribution of low-density lipoprotein (LDL) particles. 3 Moreover, the quality of HDL-C plays an important role in the development of CV disease. 4 Low-density lipoprotein is not homogenous but consists of multiple subfractions with different size, density, and chemical composition. 5 Small dense LDLs (sdLDLs) are particles that appear to possess most of the atherogenic potential of LDL. 6 This is mostly attributed to their capacity to penetrate the vessel wall and undergo oxidative modification. 7
Statins are the cornerstone of hypercholesterolemia treatment. Among statins, rosuvastatin is considered the most potent in beneficially modifying the serum lipid profile 8 and altering LDL subfraction distribution.9,10 We have previously shown that rosuvastatin can reduce the cholesterol content of both large-buoyant LDL (lbLDL) and sdLDL.11,12 Also, rosuvastatin can increase LDL size, especially in patients with raised TG levels at baseline.11,12 On the other hand, statin treatment has been associated with the development of new-onset diabetes mellitus. 13 As a result, the safety and the strength of intervention with statin treatment could be questioned in the subgroup of prediabetic patients who already have a high risk of diabetes development. It is estimated that at least one-third of patients with impaired fasting glucose (IFG) or impaired glucose tolerance (IGT; or more when IFG coexists with IGT) will eventually progress to overt diabetes in the next decade following the primary diagnosis. However, a careful evaluation of the potential risks and benefits with statin administration should be considered. Compared with the CV protection provided by statins, their effect on glucose metabolism is relatively small. In addition, multiple CV risk factors are common among patients with IFG who also frequently fulfill the diagnostic criteria of metabolic syndrome.
To date, it is unclear whether statin intervention in patients with dyslipidemia having IFG better modifies the serum lipid profile as well as the quality synthesis of LDL particles. In this study, we examined the potentially differential effect of rosuvastatin on the quantity and quality of LDL-C in patients with dyslipidemia having IFG compared with normoglycemic patients with dyslipidemia.
Methods
Patients
Consecutive patients with dyslipidemia (LDL-C levels >160 mg/dL [4.14 mmol/L]) who attended the Outpatient Lipid and Metabolic Control Clinic of the University Hospital of Ioannina, Greece, from December 2011 to December 2012, were recruited. Patients were excluded if they had any of the following: (1) history of diabetes (fasting plasma glucose >125 mg/dL [7 mmol/L] in 2 consecutive measurements or use of antidiabetic drugs), (2) established CV disease, (3) TG >400 mg/dL (4.52 mmol/L), (4) renal disease (estimated glomerular filtration rate according to the Modification of Diet in Renal Disease [MDRD] formula <60 mL/min/1.73 m2), (5) hypothyroidism (thyroid-stimulating hormone > 5 IU/mL), (6) liver dysfunction (alanine aspartate aminotransferase and/or aspartate aminotransferase activity greater than 3× the upper limit of normal), (7) active lipid-lowering or antihypertensive treatment in the last 3 months prior to recruitment; and (8) childbearing potential (for women). According to the American Diabetes Association (ADA) criteria, prediabetes is defined as IFG with fasting plasma glucose levels between 100 and 125 mg/dL (5.6-6.9 mmol/L) or impaired glucose tolerance (IGT) with 2-hour glucose levels between 140 and 199 mg/dL (7.8-11.0 mmol/L) following an oral glucose tolerance test with 75 g glucose, or both of them. 14 Eligible patients with IFG were compared to a group of patients with dyslipidemia who had normal fasting plasma glucose. All participants gave written informed consent, and the study protocol was approved by our institutional ethics committee.
Study Design
This was a prospective observational study. No extramural funding was used to support this work. The authors are solely responsible for the design and conduct of this study, all study analyses, and drafting and editing of the article. All patients (n = 127) received a 12-week dietary intervention in accordance with the National Cholesterol Education Program Adult Treatment Panel III (NCEP ATP III) guidelines. 15 Patients who continued to meet the inclusion criteria after the dietary intervention period (n = 113; 49 with IFG and 64 controls) were prescribed rosuvastatin 10 or 20 mg/d to achieve the NCEP ATP III LDL-C goals. Adherence with study medication was assessed at week 24 by counting the tablets taken; patients were considered adherent if they took >80% of the prescribed number of tablets.
The primary outcome was the differential change in the LDL subfraction profile in the subgroup of patients with dyslipidemia having IFG compared to those with normal glucose levels, following rosuvastatin treatment for 24 weeks.
Biochemical Parameters
All laboratory determinations were carried out after a 12-hour overnight fasting. Clinical investigators were blinded to laboratory results. Levels of fasting plasma glucose, total cholesterol (TC), HDL-C, and TGs were determined enzymatically in the laboratory of the University Hospital of Ioannina using an Olympus AU 600 analyzer (Olympus Diagnostica GmbH, Hamburg, Germany). Low-density lipoprotein cholesterol was calculated using the Friedewald formula (LDL-C = TC − TG/5 − HDL; provided that TGs were <400 mg/dL [4.52 mmol/L]). Serum apolipoprotein B was measured by immunonephelometry on a Behring Nephelometer BN ProSpec (Dade-Behring, Lieberbach, Germany). Serum concentrations of high-sensitivity C-reactive protein (hsCRP) were measured by the N High Sensitivity CRP Method (Dade Behring Marburg, Germany) based on particle-enhanced immunonephelometry. For all measurements in our laboratory, the coefficients of interassay and intra-assay variation were <5%, and blinded quality control specimens were included in each assay.
A polyacrylamide tube gel electrophoresis method was used for the separation of LDL subfractions. Low-density lipoprotein subfractions were estimated by the electrophoretic mobility (Rf) between the very low-density lipoprotein (VLDL) fraction (Rf 0.0) and the HDL fraction (Rf 1.0). Low-density lipoprotein subfractions are distributed from Rf 0.32 to Rf 0.64 as 7 bands, whose Rfs are 0.32, 0.38, 0.45, 0.51, 0.56, 0.60, and 0.64 (LDL1 to LDL7, respectively). Both LDL1 and LDL2 are defined as lbLDL, whereas LDL3 up to LDL7 are defined as sdLDL particles. The cholesterol concentration of each LDL subfraction is determined by multiplying the relative area under the curve of each subfraction with the TC concentration of the sample. Intermediate density lipoprotein (IDL) is distributed from Rf 0.09 to Rf 0.27. Standardized quality control material provided by the manufacturer was used as monitor of the methods accuracy and precision. The reproducibility and repeatability of the method were determined by calculating the intra- and interassay coefficients of variation. The coefficient of variation for the intra-assay precision of LDL subfractions is LDL1 = 3.58%, LDL2 = 3.64%, LDL3 = 1.65%, LDL4 = 2.45%, LDL5 = 1.72%, LDL6 = 4.62%, and LDL7 = 17.89%. The coefficient of variation for the interassay precision of LDL subfractions is for LDL1 = 3.67%, LDL2 = 6.73%, LDL3 = 5.59%, LDL4 = 3.45%, LDL5 = 2.58%, LDL6 = 12.06%, and LDL7 = 33.9%. Mean particle size was provided by the polyacrylamide tube gel electrophoresis method.
Statistical Analysis
Values are expressed as mean ± standard deviation (SD) and median (range) for variables with and without normal distribution, respectively. Continuous variables were tested for lack of normality by the Kolmogorov-Smirnov test, and logarithmic transformations were accordingly performed for variables without a normal distribution. The paired-sample t test was used for assessing the effect of treatment in each group. Analysis of covariance, adjusted for baseline values, was used for comparisons between treatment groups. Two-tailed significance was defined as P < .05. We estimated, before study launch, that in order to have 80% power at an α-level <.05, we needed 110 patients to show a 30% difference in cholesterol concentration of sdLDL particles between the groups. Analyses were performed using the SPSS 18.0 (SPSS Inc, Chicago, Illinois).
Results
A total of 127 patients (57 males, mean age 60 years) were enrolled. Of them, 113 (53 males, mean age 60 years) continued to meet the inclusion criteria after the dietary intervention period and were allocated to rosuvastatin treatment (up to 20 mg/d). All patients completed the study without any withdrawals. Adherence was 100% in the total population. The patients between the 2 groups were matched regarding sex, age, smoking status, and weight (Table 1). At baseline, none of study patients received any medication and none had hypertension. As expected, baseline characteristics were significantly different between the 2 groups concerning glucose levels, glycated hemoglobin (HbA1c), prevalence of metabolic syndrome, TC, HDL-C, LDL-C, cholesterol concentration of large LDL, and LDL particle size (Tables 2 and 3). Triglyceride levels and cholesterol concentration of sdLDL were numerically higher in the IFG group compared with the control group, without, however, reaching statistical significance (Tables 2 and 3). No significant change in weight or the presence of metabolic syndrome in any group was observed at the end of the study.
Baseline Demographic Characteristics of Study Participants.a
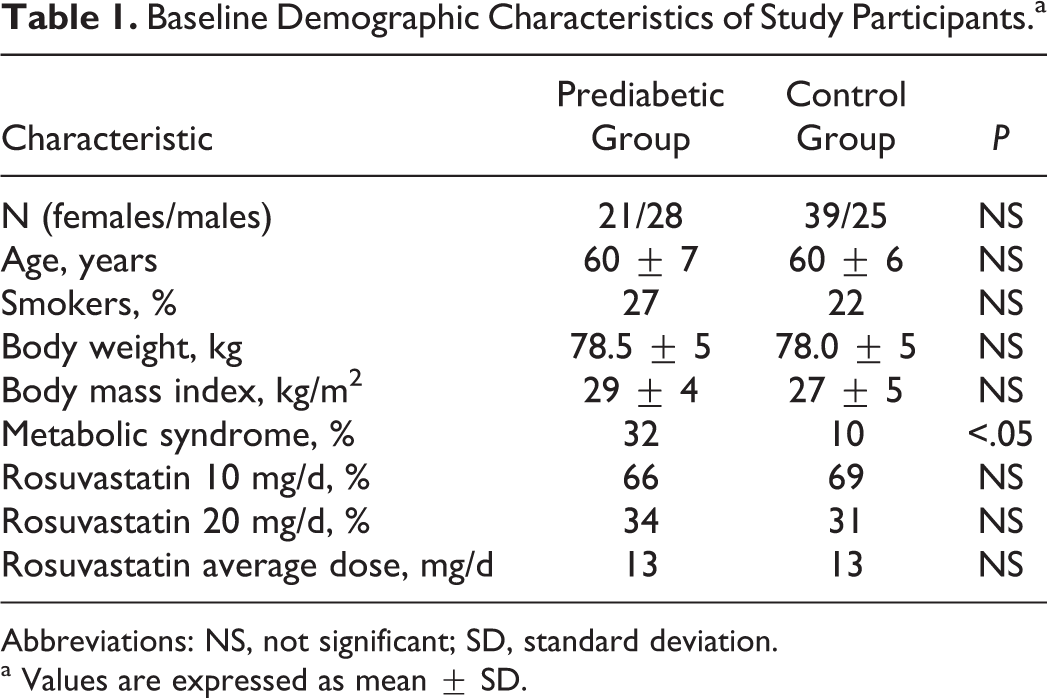
Abbreviations: NS, not significant; SD, standard deviation.
a Values are expressed as mean ± SD.
Metabolic Parameters at Baseline and After 6 Months of Treatment.a

Abbreviations: IFG, impaired fasting glucose; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; Glc, glucose; hsCRP, high-sensitivity C-reactive protein; SD, standard deviation.
a Values are expressed as mean SD (except for triglycerides that are expressed as median [interquartile range])
b P < .05 for the comparison of baseline values between the 2 groups, accordingly.
c P < .001 versus baseline.
LDL Subfraction Cholesterol Concentration and LDL Size at Baseline and After 6 Months of Treatment.a

Abbreviations: IFG, impaired fasting glucose; LDL, low-density lipoprotein; IDL, intermediate-density lipoprotein; SD, standard deviation.
a Values are expressed as mean SD (except for small dense LDL that are expressed as median [interquartile range]). The change in all parameters was adjusted for baseline values.
b P < .05 for the comparison of baseline values of large LDL cholesterol, IDL cholesterol, and LDL particle size between the 2 groups, accordingly.
c P < .001 versus baseline.
d P = .018 versus control group.
e P = .008 versus control group.
f P = .028 versus baseline.
The administration of rosuvastatin significantly decreased TC, TG, and LDL-C levels to a similar extent in both the groups (Table 2). Glucose levels, HbA1c as well as hsCRP remained unchanged in both the groups. Moreover, a similar decrease in apolipoprotein B levels as well as non-HDL-C was observed in both the groups. In contrast, there was no significant alteration in HDL-C levels in any of the groups (Table 2). No patient among study population developed new-onset diabetes mellitus after 6 months of statin treatment.
A similar reduction in the cholesterol concentration of lbLDL particles at 24 weeks, after adjustment for baseline levels, was noted in both IFG (−34.6%; P < .001 vs baseline) and normoglycemic patients (−40.5%; P < .001 vs baseline; not significant difference [NS] between groups) with dyslipidemia (Table 3). The IFG group exhibited a greater decrease in the cholesterol content of sdLDL particles at 24 weeks, after adjustment for baseline levels, compared with the control group (−65.7% vs −38.5%, P < .001 vs baseline for both changes; P = .018 for the comparison between groups; Table 3). In this context, a greater increase in LDL size, after adjustment for baseline levels, was observed in the IFG compared with the control group at 24 weeks (IFG group: +1.5%, P < .001 vs baseline, control group: +0.4%, P = .028 vs baseline; P = .008 for the comparison between groups; Table 3).
At baseline, the percentage of LDL-C distribution among sdLDL was numerically higher in the IFG group without, however, reaching statistical significance, while among lbLDL was similar between the 2 groups (Table 4). The IFG group showed a significant decrease in LDL-C percentage among sdLDL subfractions (−39.5%; P < .001 vs baseline; P = .014 vs control group), while a nonsignificant increase (18.3%) was seen in the control group. Both IFG and control groups showed a significant increase in LDL-C percentage among lbLDL by 21.8% (P < .001 vs baseline; P = .002 vs control group) and 7.7% (P = .017 vs baseline), respectively. These differences indicate a redistribution from sdLDL to lbLDL subfractions observed in the IFG group.
LDL Subfraction Cholesterol Concentration Distribution at Baseline and After 6 Months of Treatment.a

Abbreviations: LDL, low-density lipoprotein; IFG, impaired fasting glucose; SD, standard deviation.
a Values are expressed as mean SD (except for small dense LDL cholesterol percentage that is expressed as median [interquartile range]). The change in all parameters was adjusted for baseline values.
b P < .001 versus baseline.
c P = .014 versus control group.
d P = .002 versus control group.
e P = .017 versus baseline.
Discussion
In the present study, we examined the differential effects of the most potent statin, rosuvastatin, on the serum lipid and LDL subfraction profile in patients with dyslipidemia having normal fasting glucose or IFG. Both groups exhibited similar alterations in TC, TG, LDL-C, apolipoprotein B, glucose, HbA1c, hsCRP, and nonHDL-C levels. Interestingly, a greater decrease in the cholesterol content of sdLDL particles was noted in the IFG group compared with the normoglycemic group. As a result, a greater increase in LDL size was noted in the clinical setting of IFG.
Impaired fasting glucose has been associated with insulin resistance and a highly atherogenic lipid profile. Among the various metabolic factors influencing both insulin sensitivity and lipid metabolism, β2 adrenergic receptors play a significant role.16–18 Indeed, β2 adrenergic receptors are present in important tissues such as adipose tissue, 19 liver, 20 and pancreas that contribute to the regulation of both glucose homeostasis and lipid metabolism. 21 Santulli et al have shown the significance of β2 adrenergic receptor in age-related impaired insulin secretion and glucose homeostasis. 22 Adult β2 adrenergic receptor-null C57Bl/6 N mice had glucose intolerance, and an impaired glucose-induced insulin release was observed in pancreatic islets isolated from these animals. Moreover, a reduced expression of peroxisome proliferator activated receptor γ, pancreatic duodenal homeobox 1, and glucose transporter 2 was observed in those animals. Adenovirus-mediated gene transfer of human β2 adrenergic receptor reversed these defects.
Impaired fasting glucose-associated dyslipidemia is characterized by raised TG and low HDL-C levels as well as the predominance of sdLDL particles. Moreover, we have previously shown that patients with IFG exhibit increased proportion of small HDL3 particles combined with decreased activity of the antiatherogenic HDL-associated lipoprotein-associated phospholipase A2. 23 In this context, patients with IFG are considered as high risk for CV events. 24 A meta-regression analysis reported a progressive relationship between glucose levels and CV risk even in nondiabetic patients. 25 Moreover, the Australian Diabetes, Obesity, and Lifestyle Study (AusDiab) showed that IFG was an independent predictor for CV mortality after adjustment for traditional CV risk factors. 26
Statins are the most widely used evidence-based drugs for the management of dyslipidemia and the reduction in risk of CV events. It was suggested that LDL-C lowering is the main mechanism explaining a statin-associated reduction in CV risk. In addition, statin treatment can decrease the cholesterol concentration of both sdLDL and lbLDL in concert as well as increase LDL size, especially in patients with elevated TG levels. 11
Although numerous studies assessed the effect of statin treatment on LDL subfraction profile in patients with dyslipidemia, few studies have focused on IFG. A small study showed that cerivastatin, a statin withdrawn from the market several years ago, decreased cholesterol concentration of sdLDL particles in patients with IFG. 27 Data from the Scandinavian Simvastatin Survival Study showed that simvastatin reduced CV-related hospitalizations, major coronary events, revascularizations, and total coronary mortality when compared to placebo in patients with IFG.28,29 Likewise, the Long-Term Intervention with Pravastatin in Ischemic Disease (LIPID) study showed that pravastatin was associated with reduced risk of any major vascular event in patients with IFG. 30 Studies evaluating the effect of statins on the LDL subfraction profile in diabetic patients have reported mixed results, ranging from neutral to beneficial effects on sdLDL particles and LDL size. 31 Of note, these studies have used various methods to evaluate the LDL subfraction profile such as gradient gel electrophoresis, ultracentrifugation, density gradient ultracentrifugation, nuclear magnetic resonance (NMR), or the polyacrylamide tube gel electrophoresis method. In our study, the polyacrylamide tube gel electrophoresis method was used, which correlates well with other methods.32,33 However, none of these major trials examined the exact effect of statin treatment on LDL subfractions in IFG compared to matched normoglycemic patients. Our results could help explain this well-established statin benefit in the clinical setting of IFG.
Statin therapy has been associated with disadvantageous effects on glucose homeostasis and insulin resistance. 34 In this context, we have previously shown a dose-dependent increase in insulin resistance following rosuvastatin treatment in patients with IFG. 35 However, statin use should not be discouraged since CV risk reduction associated with statins is relatively high. In this respect, an analysis of the Justification for the Use of Statins in Primary Prevention: An Intervention Trial Evaluating Rosuvastatin trial (JUPITER) trial showed that the benefits of statin therapy in major CV events outweighed any possible risk of diabetes. 36 Furthermore, we have demonstrated a reversal of the disadvantageous effects of rosuvastatin on insulin resistance when combined with telmisartan in patients with mixed dyslipidemia, IFG, and hypertension. 37
In the present study, even after adjustment for baseline values, a greater decrease in sdLDL cholesterol and increase in LDL size was observed in the IFG group with dyslipidemia compared with normoglycemic individuals. Therefore, in the setting of IFG, statin treatment may induce a redistribution of LDL-C from the atherogenic sdLDL to lbLDL particles in patients with IFG. As a result, an aggressive statin treatment may yield added CV benefits in prediabetic patients with dyslipidemia. However, in light of the association of statins with new-onset diabetes development, the query arises of whether prediabetic patients should receive aggressive statin treatment. Currently, diabetic patents have lower LDL-C goals compared with prediabetic subjects, according to NCEP ATP III, ADA, or European society of cardiology/European atherosclerosis society guidelines. Recently, the American College of Cardiology and the American Heart Association released new guidelines for the prevention of cardiovascular disease and management of elevated blood cholesterol. 38 These guidelines leave behind the need to define any LDL-C goals. Instead, specific risk groups of patients have been identified that need to be prescribed either moderate- or high-intensity statin therapy, regardless of their baseline LDL-C levels and without aiming for a particular predefined LDL-C target. Diabetic patients, being at high CV risk, are among the identified risk groups of patients in whom the benefits outweigh the risks. Further studies are needed to determine whether there is a need for a more aggressive statin treatment in the often overlooked subgroup of patients with IFG, which are at the doorstep of diabetes. Taking into consideration our findings, the association of statins with new-onset diabetes and the CV benefits from statin treatment, we encourage that lipid management should still remain in accordance with the current guidelines.
Study Limitations
This was an open-label study, but the end points were blindly assessed. The power estimation was based on published data, which we recognize are limited. A polyacrylamide tube gel electrophoresis method was used which provides cholesterol concentration of LDL subfractions and no particle numbers. Although prediabetic patients were selected only with the measurement of fasting plasma glucose and not with an oral glucose tolerance test, it is by far the simpler way to characterize an individual as prediabetic in the everyday clinical practice. We acknowledge that our study represents a pilot study on the effects of rosuvastatin in LDL subfraction profile of prediabetic patients. Larger randomized control trials are needed to evaluate the effects of statins in prediabetic patients and clarify the uncertainty of aggressive LDL-C reduction with statin treatment in this important population subgroup.
Conclusion
Rosuvastatin was associated with a greater decrease in the cholesterol content of sdLDL as well as an increase in LDL size in patients with IFG compared to normoglycemic patients with dyslipidemia.
Footnotes
Acknowledgments
The authors would like to thank the Atherothrombosis Research Centre of the University of Ioannina for providing access to the laboratory equipment and facilities.
Author Contribution
Christos V. Rizos contributed to conception and design of the study, substantial data acquisition, analysis and interpretation of the data, drafting of the manuscript, and critical revision of the manuscript for intellectual content; Michael S. Kostapanos contributed to conception and design of the study, data acquisition, analysis and interpretation of the data, drafting of the manuscript, and critical revision of the manuscript for intellectual content; Evangelos C. Rizos contributed to conception and design of the study, data acquisition, analysis and interpretation of the data, drafting of the manuscript, and critical revision of the manuscript for intellectual content; Alexandros D. Tselepis contributed to conception and design of the study, data acquisition, analysis and interpretation of the data, drafting of the manuscript, and critical revision of the manuscript for intellectual content; and Moses S. Elisaf contributed to conception and design of the study, data acquisition, analysis and interpretation of the data, drafting of the manuscript, and critical revision of the manuscript for intellectual content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
