Abstract
Aims
Despite recent medical advances, cardiovascular disease remains the leading cause of death worldwide. As (micro)-calcification is a hallmark of atherosclerosis, this review will elaborately discuss advantages of sodium fluoride positron emission tomography (PET) as a reliable cardiovascular imaging technique for identifying the early onset of vascular calcification (i.e. locking onto the target). We assess state-of-the-art meta-analysis and clinical studies of possible treatment options and evaluate the concept of vitamin K supplementation to preserve vascular health (i.e. loading the bullet).
Methods and results
After a structured PubMed search, we identified 18F-sodium fluoride (18F-NaF) PET as the most suitable technique for detecting micro-calcification. Presenting the pros and cons of available treatments, vitamin K supplementation should be considered as a possible safe and cost-effective option to inhibit vascular (micro)-calcification.
Conclusion
This review demonstrates need for more extensive research in the concept of vitamin K supplementation (i.e. loading the bullet) and recommends monitoring the effects on vascular calcification using 18F-NaF PET (i.e. locking onto the target).
Introduction
Despite recent advances in interventional medicine, cardiovascular disease is still the leading cause of death worldwide, exceeding cancer mortality.1,2 After the recognition of vascular calcification as a hallmark of atherosclerosis, emerging data agree on classifying coronary artery calcification (CAC) score as an independent cardiovascular risk factor. 3 However, the risk ratio of CAC is currently unavailable, because a large patient population falls under low levels of CAC, undetectable by conventional methods (i.e. low dose non-contrast computed tomography (CT) calcium score).
Vascular calcification was considered a passive process; however, recent evidence shows that it is actively regulated with a delicate balance between calcification promoting and inhibiting factors. 4 Most initiators of extracellular matrix mineralization converge to the secretion of extracellular vesicles by synthetic vascular smooth muscle cells and macrophages. 5 These sites provide the perfect nidus for nucleation and elongation of hydroxyapatite crystals, which will eventually destabilize the plaque. 6 Hence, early identification of vascular calcification may allow for a better stratification into high-risk individuals with poor outcomes. 7
In this review, we discuss the most promising methods of vascular imaging (i.e. Part 1) and key emerging concepts in the treatment of vascular calcification (i.e. Part 2).
Part 1: Micro-calcification as an independent risk factor
There is increasing evidence that (micro-)calcification is not merely a passive bystander of atherosclerosis, but actually plays an active role in plaque progression and destabilization. 8 In vitro studies showed that calcium phosphate crystals smaller than 1 µm can activate macrophages and induce vascular smooth muscle cells to undergo programmed cell death. 9 The latter phenomenon further accelerates atherogenesis, by providing a nidus for further calcification and medial degeneration. 10 This vicious cycle increases vulnerability of atherosclerotic plaques. 11 So called ‘spotty calcification’ on CT, which is the imaging equivalent of micro-calcification, has been associated with increased plaque vulnerability in coronary artery disease. 12 On the contrary, in patients with stable coronary artery disease, completely calcified, more solid lesions are more prevalent. 13
Agatston et al. linked the sensitivity, specificity and predictive values of total CAC score with increased age. 14 A high CAC score (≥1000 Agatston units) in asymptomatic patients seems a better predictor for coronary events than severe perfusion abnormalities. 15 Hence, for more than a decade, CAC score has been in clinical practice for detection of subclinical disease and for stratification of asymptomatic individuals. 7 Stratifying based on CAC score reduces the number needed to treat to prevent one ischaemic stroke or transient ischaemic attack from 229 (CAC = 0) to 68 (CAC>100). 16 Moreover, in an observational study of 25,253 asymptomatic patients, a CAC score higher than 1000 was correlated with a 16% lower 10-year survival rate, even after the adjustments for risk factors, such as age, hypercholesterolaemia, diabetes, smoking, hypertension and a family history of premature coronary heart disease. 3
However, the additional risk or the relative risk of CAC is yet unreliable. 17 This is due to the fact that the already established Agatston calcification score is able to assess CAC density, which seems to be inversely associated with cardiovascular risk. 18 Moreover, there is a current paradigm shift from CAC density to CAC volume as a positive predictor for cardiovascular events. Recent studies support the notion that molecular imaging techniques, such as 18F-sodium fluoride (18F-NaF) positron emission tomography (PET), are complementary in detecting vascular calcification and should be introduced to gain further information on active micro-calcification in unstable plaques. 19
Sodium fluoride-18: seeing the unseen
18F-NaF is a PET tracer with an interesting property: the 18F is able to replace the hydroxyl groups of hydroxyapatite, the very building block of (vascular) calcification. This, combined with its small size and its negligible plasma protein binding capacity, results in a high target-to-background ratio (i.e. efficient targeting capability) shortly after intravenous injection.
20
Thin nano-sized hydroxyapatite crystals provide a higher surface area for 18F-NaF to bind to, in contrast to the macroscopic counterpart, in which the tracer cannot enter the inner core.
20
Irkle et al. showed that fluoride is better adsorbed by micro-calcified plaques (i.e. nodules <50 μm) when compared with macro-calcifications (i.e. nodules >50 μm).
19
In the same study, 18F-NaF PET demonstrated a higher sensitivity then CT, by detecting larger areas of active micro-calcification sites.
19
This makes 18F-NaF suitable for detecting vulnerable sites by visualizing active micro-calcification in contrast to stable macro-calcified plaques, which are better detected by CT (Figure 1). Moreover, 18F-NaF uptake in the coronary arteries is in close agreement with CT markers for plaque vulnerability (e.g. plaque attenuation <30 Hounsfield units).
21
The superiority of locking with 18F-NaF and loading with vitamin K.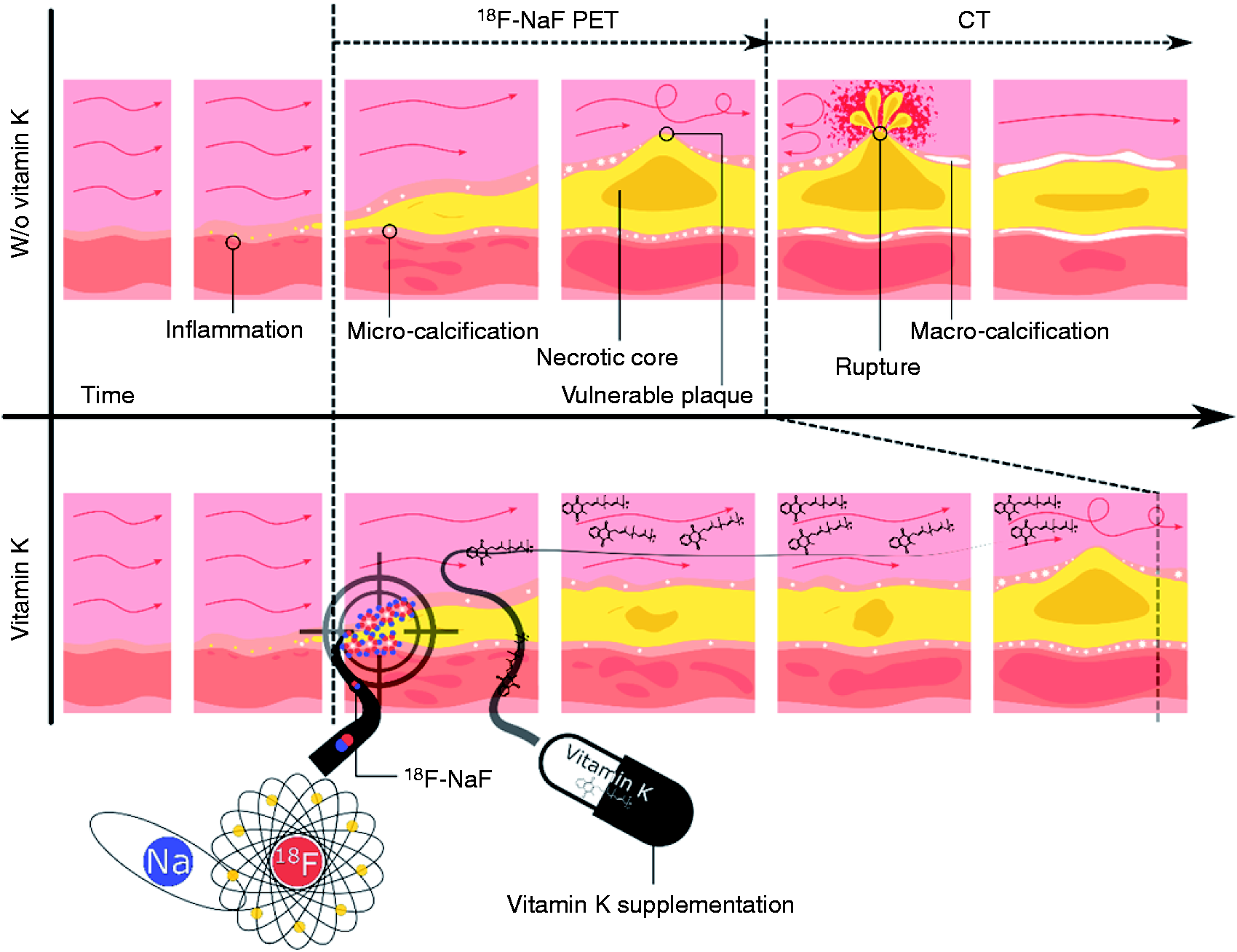
Indeed, the feasibility of 18F-NaF PET for in vivo quantification of micro-calcification in the aorta as well as in the coronary and carotid arteries as a feature of culprit plaques has already been proven (Figure 2).22,23 In patients with myocardial infarction, 18F-NaF was able to discriminate between culprit and non-culprit plaques.
22
Over the years, many studies used 18F-NaF to acquire information about morphological and functional properties of calcified plaques. All of them provided increasing evidence that this technique represents a feasible option for imaging active micro-calcification.
24
Increased 18F-sodium fluoride (18F-NaF) uptake in culprit coronary artery lesion. In a patient with coronary artery disease, invasive coronary angiography (a) showed non-obstructive disease in the right coronary artery. Corresponding 18F-NaF positron emission tomography/computed tomography (b) showed a region of increased 18F-NaF activity (positive lesion, red line) in the mid-right coronary artery (tissue-to-background ratio, 3.13) and a region without increased uptake in the proximal vessel (negative lesion, yellow line). Radiofrequency intravascular ultrasound shows that the 18F-NaF negative plaque (c) is principally composed of fibrous and fibro-fatty tissue (green) with confluent calcium (white with acoustic shadow) but little evidence of necrosis. On the contrary, the 18F-NaF positive plaque (d) shows high-risk features such as a large necrotic core (red) and micro-calcification (white).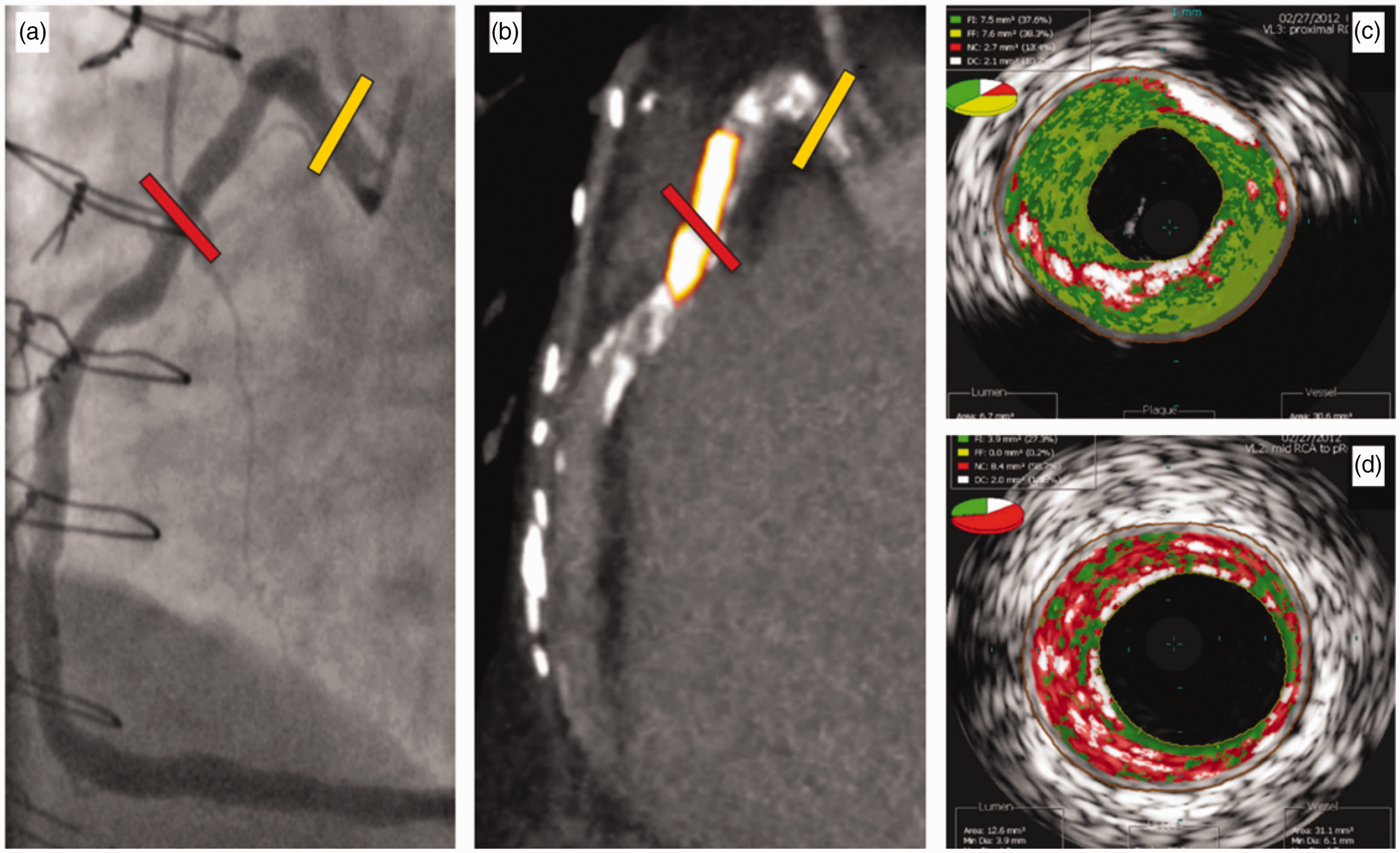
When compared with 18F-fluorodeoxyglucose, 18F-NaF showed improved detection of culprit plaques in thoracic aorta and coronary arteries.22,25 Therefore, active micro-calcification, rather than vascular inflammation, should be associated more strongly with cardiovascular risk. This led Dweck et al. to conclude that 18F-NaF PET was the only currently available clinical imaging approach that can non-invasively detect vascular micro-calcifications. 26
By implementing hybrid PET/magnetic resonance imaging (MRI), 18F-NaF has the opportunity to fully exhibit its potential, by delivering data about the state of micro-calcification alongside high-resolution magnetic resonance images that can give a detailed description of plaque burden (e.g. juxtaluminal lesions, intra-plaque haemorrhage, lipid-rich necrotic core and fibrous cap status) (Figure 1). CT fails to detect many subtle Pseudoxanthoma elasticum-related abnormalities, such as calcifications in the endo- or myocardium. 27 Although, 18F-NaF PET/MRI is currently most used in studies that concern bone pathologies, cardiovascular research has adapted this technique. 28 Ongoing trials are using hybrid 18F-NaF PET/MRI, for example, BASIK-2 (NCT02917525), which assesses vitamin K influence on calcific aortic valve stenosis.
However, additional scientific efforts are still required until 18F-NaF can enter routine clinical practice as a tracer for vascular micro-calcification, but, as mentioned above, available studies reveal a promising future (Figure 2). 22
Part 2: Available treatment options against micro-calcification
Micro-calcifications have long been proposed as a marker for vulnerable plaques. In silico models predict that inclusions, located in a thin fibrous cap in an area of high circumferential stress, can double the intensity of initial stress. 29 The most likely candidate for these inclusions is micro-calcification. Bobryshev et al. showed by quantification in ultrathin sections that micro-calcifications are more present in vulnerable compared with stable plaques. 30 Based on data generated by studies using 18F-NaF PET, active micro-calcifications are now an established hallmark of atherosclerosis and plaque vulnerability. 17
Promising therapies for the future of treatment of vascular calcification.

Bisphosphonates and phosphate binders are an attractive choice for their promise to attack the building blocks of micro-calcification, namely calcium and phosphate. Bisphosphonates bind free calcium, whereas phosphate binders prevent the absorption of dietary phosphate, making it unavailable for incorporation into hydroxyapatite. A systematic review suggests that bisphosphonates are able to favourably influence calcium homeostasis within the vessel wall. 32 However, long-term administration of bisphosphonates may cause severe adverse reactions, like osteonecrosis of the jaw, probably by reducing macrophage and osteoclast viability. 43 In the case of phosphate binders, a meta-analysis that examined 104 studies involving 13,744 adults found no cardiovascular protection for dialysis patients and uncertain effects for the rest of a chronic kidney disease population. 33
Over recent years, the focus of potential cardiovascular protective medications was set on statins. Besides lowering cholesterol, numerous medical trials showed positive, pleiotropic, effects of statins on plaque stabilization and anti-inflammatory properties. However, several studies revealed that they seem to negatively influence the progression of calcification. 34 Moreover, recent studies suggest that statins promote formation of hydroxyapatite crystals within the plaque by inhibiting activity of vitamin K2. 35
Considering that bisphosphonates, phosphate binders and statins have already been extensively studied, sowing inconsistent results (i.e. phosphate binders) or detrimental side effects (i.e. bisphosphonates), vitamin K supplementation might be considered as a safe, cost-effective alternative for inhibiting vascular calcification. Therefore, research evaluating potential beneficial effects of vitamin K on vascular calcification has gained more and more interest over recent years. 44
Vitamin K: the magic bullet to fight vascular calcification?
Vitamin K is paramount for the biological activation of an array of proteins which are able to bind free calcium via their γ-carboxyglutamate-rich domains. Amongst these vitamin K dependent proteins (VKDPs) are liver-derived proteins with a significant role in haemostasis (i.e. factors II, VII, IX and X and proteins C, S and Z). In the last decade, several extra-hepatic VKDPs have been discovered, with dispersed functions, including bone metabolism (e.g. osteocalcin) and inhibition of ectopic calcification (e.g. matrix γ-carboxyglutamate protein (MGP)).
Vitamin K is an unequivocal cofactor in the activation of VKDPs and it comes in two flavours: phylloquinone (i.e. vitamin K1) and menaquinones (MKs, i.e. vitamin K2). For purposes of nomenclature, MKs are further sub-classified according to the number of repeating prenyl units (i.e. MK-n, where n is the number of repeating units).
In the case of vitamin K deficiency, a high-dose supplementation of vitamin K carboxylates (i.e. activates) VKDPs with anti-calcification properties. Plasma levels of dephosphorylated uncarboxylated (i.e. inactive) MGP rapidly change after increasing MK-7 intake. 45 It is worth mentioning that vitamin K supplementation does not induce a state of hypercoagulability. 46 Thus, a higher intake of vitamin K is not associated with any negative reaction on the coagulation cascade.
The detrimental effect of vitamin K antagonists (e.g. warfarin) on cardiovascular and renal systems has already been revealed. 47 A considerable number of studies over the last two decades showed an association between vitamin K supplementation and positive outcomes in calcium metabolism. In most post-menopausal women and chronic kidney disease patients there is a paradoxical decline in bone calcium content, paralleled by an increase in vascular calcification. Vitamin D is frequently used in combination with calcium supplementation to protect against bone disease; however, this treatment might accelerate vascular calcification. 48 By activating MGP, vitamin K supplementation may be the way out of this paradox. Indeed, supplementation with minerals (i.e. calcium, magnesium and zinc), vitamin D and K showed beneficial effects on arterial elasticity in post-menopausal women after a follow-up of three years. 49
Vitamin K seems to have beneficial effects on vascular calcification and thus indirectly fights cardiovascular disease, yet supporting vascular health (Figure 1). Poor vitamin K status correlated with intensive CAC in patients with high blood pressure, even when under antihypertension medication. 36 Meanwhile, when compared with control, vitamin K1 supplementation slows down the progression of calcification in the coronary arteries after three years and in the aortic valve after one year.37,50 Moreover, as it is known that bone morphogenetic proteins promote the inflammation in atherogenic conditions, vitamin K supplementation may also improve vascular health by directly activating MGP, the natural inhibitor of the pro-apoptotic bone morphogenetic protein 2. 51
Prospective population-based trials linked the increased consumption of MKs with a reduced relative risk of coronary heart disease mortality (risk ratio = 0.43 with a 95% confidence interval of 0.24–0.77). 38 Moreover, a recent meta-analysis confirmed these findings and concluded that higher dietary vitamin K consumption is associated with a lower risk of coronary heart disease. 52 A more in-depth study also observed an inverse association between MK intake (but not for vitamin K1 intake) and the risk of coronary heart disease; this association was mainly due to subtypes MK-7, MK-8 and MK-9. 39 Indeed, MK-7 supplementation was also associated with improved MGP levels in chronic kidney disease patients and with decreased arterial stiffness in healthy postmenopausal women.40,41 Moreover, MK-7 supplementation showed a beneficial effect on arterial stiffness in renal transplant recipients with stable graft function. 42 However, available data from randomized clinical trials is insufficient to argue in favour of an increase in the recommended daily dose of vitamin K or its introduction in clinical practice. With progress in ongoing clinical trials new supporting information will come to light on how beneficial vitamin K supplementation (i.e. vitamin K1 and MK-7) is in cardiovascular disease (i.e. VitaK-CAC NCT01002157, iPACK-HD NCT01528800, VitaVasK NCT01742273 and BASIK2 NCT02917525).
Conclusion: locking with 18F-NaF and loading with vitamin K
Currently, the significant impact of 18F-NaF PET in non-invasively identifying the early onset of vascular calcification is gaining more and more attention. In addition, emerging data are positive about the up-and-coming concept of vitamin K supplementation to combat micro-calcification. Here, we raise awareness of the need for more extensive clinical trials using vitamin K to promote vascular health. These studies should consider the use of 18F-NaF PET for monitoring active micro-calcifications.
Vitamin K emerges as a suitable, cost-effective bullet that can and should be loaded after targeting micro-calcification with 18F-NaF PET.
One-sentence summary
Vitamin K emerges as a suitable, cost-effective, bullet that can and should be loaded after targeting micro-calcification with 18F-NaF PET.
Footnotes
Author contribution
AF, AM, JB, LJS and FMM contributed to the conception or design of the work. AF drafted the manuscript. AM, JB, LJS and FMM critically revised the manuscript. All gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Acknowledgements
AF, LJS and FMM receive research funding from the ITN INTRICARE of European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie (grant 722609). LJS receives research funding from the ITN EVOluTION of European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie (grant 675111). AM and FMM receive research funding from the RTG 2375 of the German Research Foundation (grant 331065168). The authors do hereby declare that Figure 1 in the manuscript is entirely original and does not require reprint permission. Figure 2 in the manuscript is used with permission from Dr Nikhil V Joshi, the corresponding author of the article in question.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
