Abstract
Background
Despite numerous studies and meta-analyses the prognostic effect of cardiac rehabilitation is still under debate. This update of the Cardiac Rehabilitation Outcome Study (CROS II) provides a contemporary and practice focused approach including only cardiac rehabilitation interventions based on published standards and core components to evaluate cardiac rehabilitation delivery and effectiveness in improving patient prognosis.
Design
A systematic review and meta-analysis.
Methods
Randomised controlled trials and retrospective and prospective controlled cohort studies evaluating patients after acute coronary syndrome, coronary artery bypass grafting or mixed populations with coronary artery disease published until September 2018 were included.
Results
Based on CROS inclusion criteria out of 7096 abstracts six additional studies including 8671 patients were identified (two randomised controlled trials, two retrospective controlled cohort studies, two prospective controlled cohort studies). In total, 31 studies including 228,337 patients were available for this meta-analysis (three randomised controlled trials, nine prospective controlled cohort studies, 19 retrospective controlled cohort studies; 50,653 patients after acute coronary syndrome 14,583, after coronary artery bypass grafting 163,101, mixed coronary artery disease populations; follow-up periods ranging from 9 months to 14 years). Heterogeneity in design, cardiac rehabilitation delivery, biometrical assessment and potential confounders was considerable. Controlled cohort studies showed a significantly reduced total mortality (primary endpoint) after cardiac rehabilitation participation in patients after acute coronary syndrome (prospective controlled cohort studies: hazard ratio (HR) 0.37, 95% confidence interval (CI) 0.20–0.69; retrospective controlled cohort studies HR 0.64, 95% CI 0.53–0.76; prospective controlled cohort studies odds ratio 0.20, 95% CI 0.08–0.48), but the single randomised controlled trial fulfilling the CROS inclusion criteria showed neutral results. Cardiac rehabilitation participation was also associated with reduced total mortality in patients after coronary artery bypass grafting (retrospective controlled cohort studies HR 0.62, 95% CI 0.54–0.70, one single randomised controlled trial without fatal events), and in mixed coronary artery disease populations (retrospective controlled cohort studies HR 0.52, 95% CI 0.36–0.77; two out of 10 controlled cohort studies with neutral results).
Conclusion
CROS II confirms the effectiveness of cardiac rehabilitation participation after acute coronary syndrome and after coronary artery bypass grafting in actual clinical practice by reducing total mortality under the conditions of current evidence-based coronary artery disease treatment. The data of CROS II, however, underscore the urgent need to define internationally accepted minimal standards for cardiac rehabilitation delivery as well as for scientific evaluation.
Keywords
Introduction
Within the past 25 years, cardiovascular morbidity and mortality after acute coronary syndromes (ACSs) have shown a remarkable decrease which is associated with the implementation of acute coronary revascularisations as well as the application of effective acute and long-term pharmacotherapy. 1 Supporting these results from the United States 1 the French FAST-MI registry revealed a mortality reduction 6 months after ST-segment elevation myocardial infarction (STEMI) and non-ST-segment elevation myocardial infarction (NSTEMI) from 17.2% to 5.3% and 6.3%, respectively. 2 Moreover, on the basis of the SWEDEHEART registry a marked improvement of 2 years’ survival was found, but strictly associated with the use of acute coronary interventions and evidence-based long-term secondary prevention. 3 Accordingly, current evidence-based treatment modalities of ACS and coronary artery disease (CAD) do have a large impact on the acute and long-term success of care delivered to these patients. Against this background, the effects of special treatment modalities such as cardiac rehabilitation (CR) need to be re-evaluated in the light of their added short and long-term clinical and prognostic benefit. The Cardiac Rehabilitation Outcome Study (CROS) aimed to evaluate the prognostic effect of CR after ACS and coronary artery bypass grafting (CABG) in the modern era of cardiovascular treatment modalities. On the basis of predominantly controlled observational studies including a large number of patients, CROS confirmed a beneficial effect of CR (i.e. reduced all-cause mortality after ACS and after CABG). 4 However, with CROS it became apparent that minimal requirements for CR delivery (based on published standards and core components)5–8 had to be fulfilled to reach effectiveness. These minimal requirements have been addressed by other recent meta-analyses9–13 with a focus on the volume and intensity of exercise sessions and treatment of cardiovascular risk factors during CR. Not meeting these minimal requirements may explain in part the negative results of some recent studies and meta-analyses.14–16
Against this background, the aim of this CROS update was to re-evaluate the results of CROS I critically in the light of newly published CR studies meeting the strict CROS inclusion criteria. Moreover, the aim of this update was to elucidate further the CR effect on secondary and non-fatal clinical endpoints representing a heterogeneous field in clinical CR research. By evaluating controlled observational studies the CROS data finally reflect everyday clinical care thereby allowing an estimation of how guideline standards are actually translated into clinical practice.
Methods
This review was conducted and reported according to the PRISMA statement (preferred reporting items for systematic reviews and meta-analyses), and the MOOSE statement (meta-analysis of observational studies in epidemiology).17,18 The core methods used were essentially unchanged compared to the 2016 publication. The study protocol was prospectively published in PROSPERO (CRD42014007084). 19
Study eligibility criteria
Randomised controlled trials (RCTs) as well as prospective controlled cohort studies (pCCS) and retrospective controlled cohort studies (rCCS) of multi-component CR versus usual care, with a follow-up period of at least 6 months, were investigated. We included men and women of all ages after hospitalisation for ACS or CABG, respectively. In addition, we included studies enrolling mixed populations of patients after ACS and/or after CABG as a basic requirement, as well as patients with chronic stable CAD with or without elective percutaneous coronary intervention (PCI). Patient enrolment had to be carried out by 1995 or later. The primary endpoint was total mortality. Secondary endpoints mainly included non-fatal cardiovascular events, hospital readmissions and mixed endpoints. The detailed study selection criteria were presented previously (see Supplementary Material (SM), Table SM 1). 4
Search methods and identification of studies
For the previous review 4 highly sensitive search strategies were developed to identify two types of studies: RCT and CCS regardless of the studies’ current status (published, unpublished, finished or ongoing). A detailed description of the elaboration of the search strategy is available in the previous review. 4
For this update, we restricted our search to the following four databases: PubMed, Embase, Cochrane Central Register of Controlled Trials and the World Health Organization’s International Clinical Trials Registry Platform (ICTRP). Databases which did not contribute studies for inclusion in the previous review were no longer deployed. The search informing this update comprised the period 23 December 2015 to 4 September 2018. No language restrictions were applied. Details of all search strategies are documented in Supplementary Material (see Table SM 2). In addition to searching electronic databases, the references of recent systematic reviews were screened.
Study selection
The titles and abstracts of all references were independently evaluated by at least two members of the reference selection board (AS, CHD, BR). Abstracts of potential interest were re-evaluated and selected for full text evaluation (FTE) and structured study evaluation (SSE), respectively, consented within the whole board. FTE for assessing main inclusion criteria and SSE with quality assessment was performed and consented within an extended reference selection board (AS, CHD, BR, PD) including a biometrician (KJ). The primary reasons for study exclusion are given in Table SM 4.
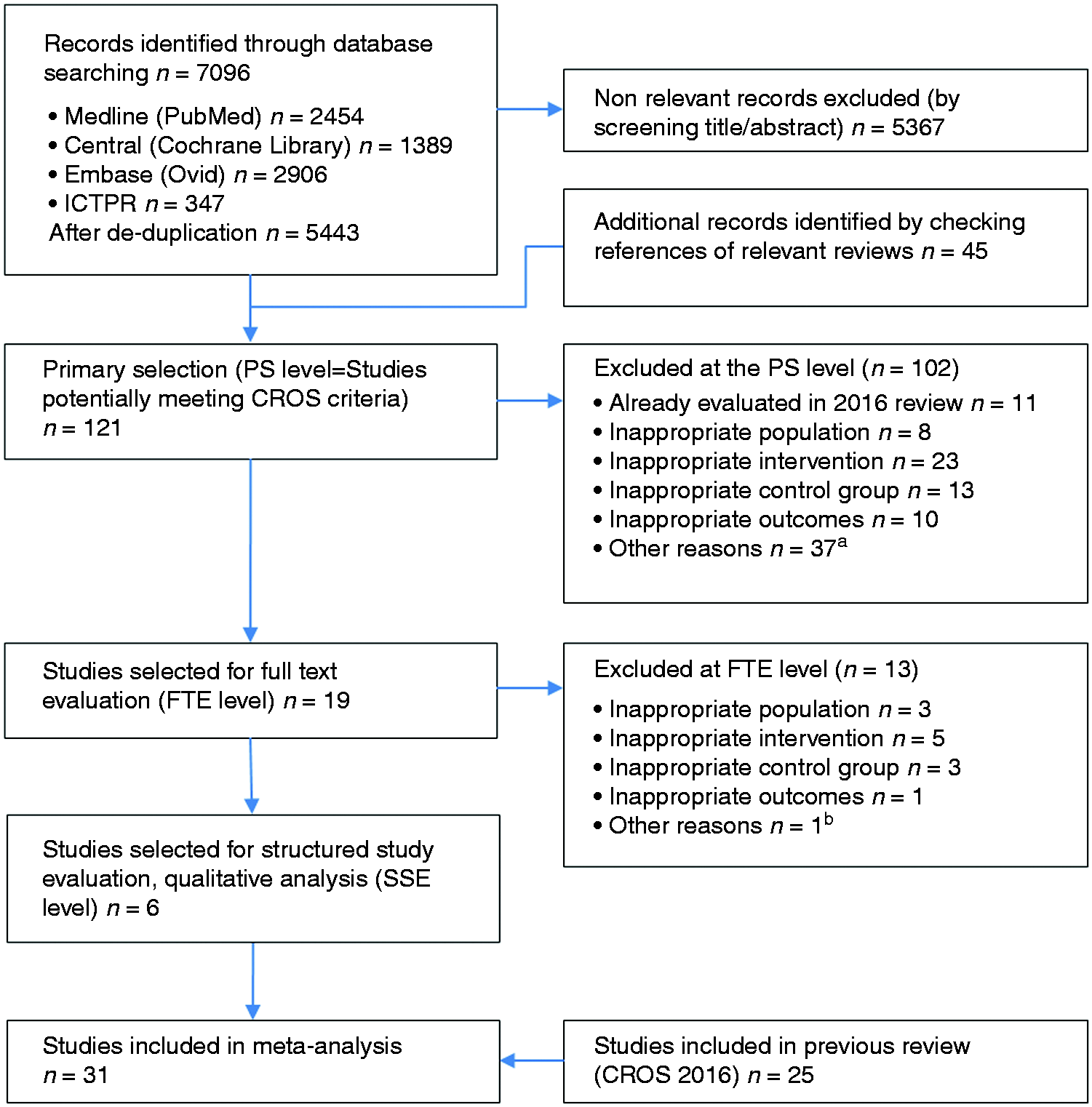
For the meta-analysis, the studies resulting from the SSE process of the current update were merged with the selected studies from the 2016 publication. The study selection process is outlined in Figure 1.
Study selection flow chart. aOther reasons PS level: reviews, letters, study protocol, only abstract available; bOther reasons FTE level: referral only, no information about CR enrollment and adherence available. ICTRP: International Clinical Trials Registry Platform; PS: primary selection of extracted studies; FTE: full-text evaluation; CR: cardiac rehabilitation; SSE: structured study evaluation and quality analysis according to the checklist of methodological issues on non-randomised studies.
20
Study evaluation process
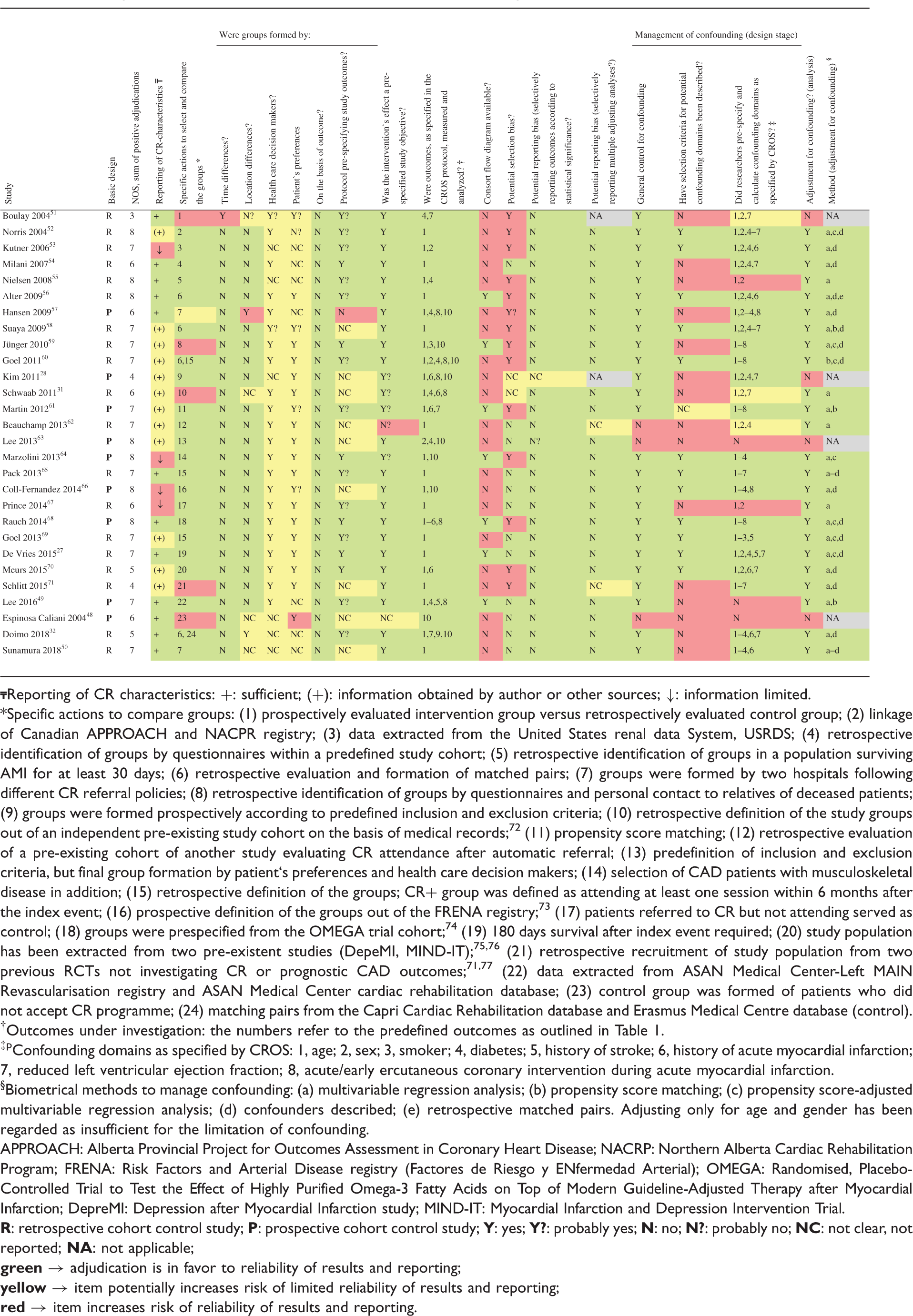
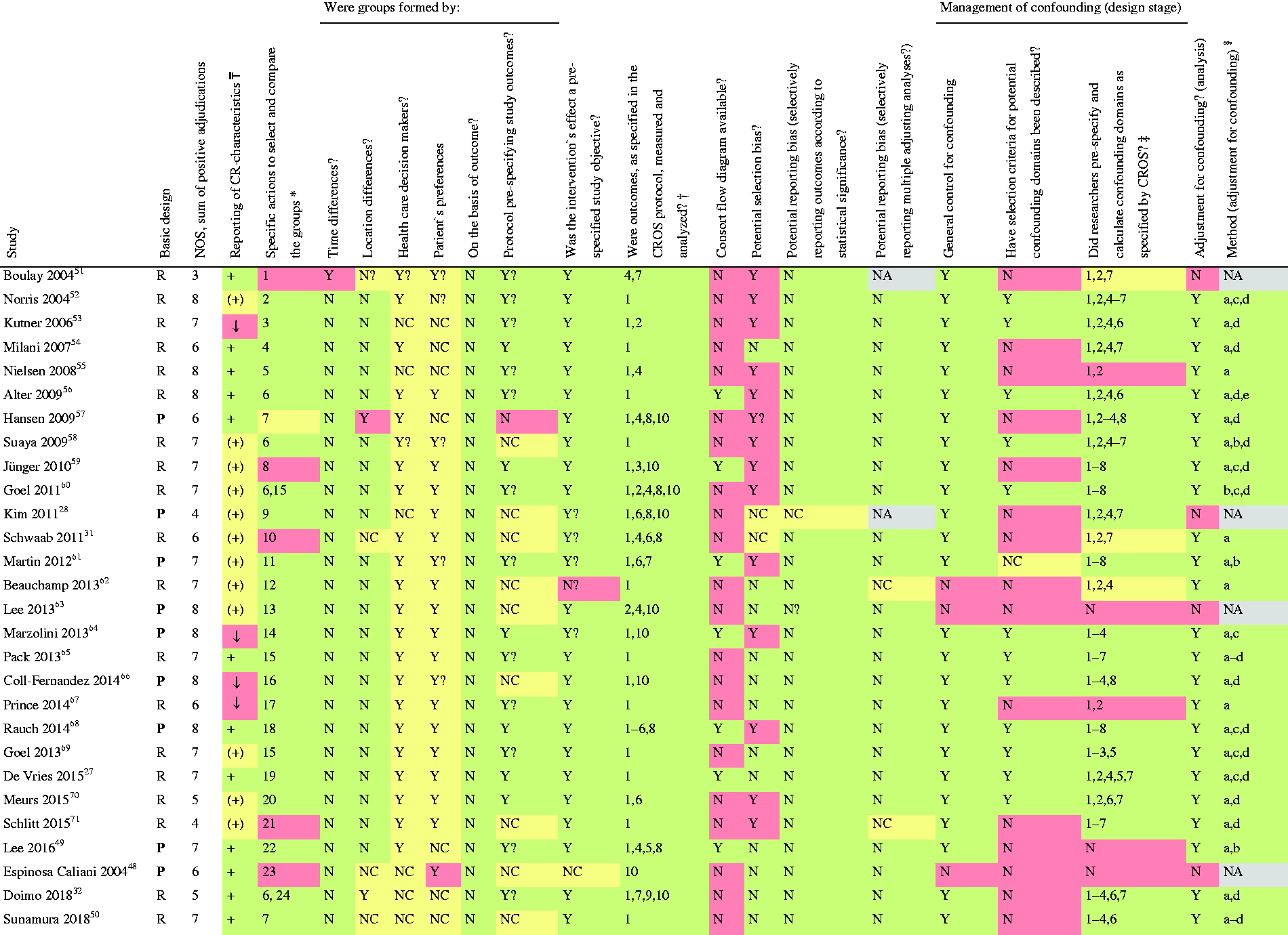
The study evaluation included design, data sources, information on population, interventions, controls, calculation and presentation of outcomes and handling of bias. For RCTs the Cochrane risk of bias table (http://tech.cochrane.org/revman/download), and for the CCSs the checklists of methodological issues on non-randomised studies,20,21 and the Newcastle Ottawa Scale (NOS) were used. 22 To facilitate the study evaluation with respect to the management of confounding, age, gender, smoking, diabetes, history of stroke, history of acute myocardial infarction (AMI), reduced left ventricular ejection fraction and acute or early PCI during AMI have been prespecified as potential confounders.
Data extraction
Data extraction was performed by two biometricians independently (KJ, MH), using a standardised extraction form. Disagreements were solved by consensus. We extracted the following information from each eligible article: name of first author, year of publication, study location (country), study design, data source, number of participants, population (ACS, CABG or mixed), inclusion period, inclusion criteria, follow-up time, mean age of participants, proportion of men, intervention characteristics, control characteristics, reported outcomes, information on outcomes, data on outcomes, covariates included in the adjusted models.
Statistical analysis
The analyses were separated with regard to population (AMI, CABG or mixed) and study design (RCT, pCCS and rCCS). For time-to-event outcomes, the hazard ratio (HR) with its 95% confidence interval (CI) was chosen as the effect measure per study. If possible, log HRs and their standard errors were extracted directly, preferably from an adjusted model and matched-group analysis. If they were not reported but adequate univariate analyses were available, an indirect estimation method was used.23,24 In some study publications, instead of HR adjusted odds ratios (ORs) at the end of follow-up or only absolute numbers of events to calculate ORs were reported. HRs and ORs were reported and pooled separately in the present review. 25 For dichotomous outcomes, the OR with its 95% CI was used as the effect measure per study. If no event occurred in one or in both arms, a continuity correction of 0.5 per cell was applied. For consistency, we re-calculated the treatment effect to be in the same direction, as necessary, with an HR or OR above 1 indicating a higher risk of CR with respect to each outcome. HRs were combined using the generic inverse variance method. ORs were pooled using the Mantel–Haenszel method or the generic inverse variance method. The latter one was used when at least one study reported an adjusted OR. Random effects models were used to calculate overall effect estimates and CIs because we assumed heterogeneity between the ‘true’ effects of the different CR programmes used in the studies. All results were investigated for statistical heterogeneity by I2 statistics with 0–30% representing no or only small, 30–60% moderate, 50–90% substantial and 75–100% considerable heterogeneity. 26 A statistical investigation of potential publication bias based on a test of funnel plot asymmetry could not be done because of too few studies per single meta-analysis. 26 Nevertheless, sensitivity analyses for the outcome total mortality have been performed with respect to extracted results of alternative analysis techniques (e.g. independent groups instead of matched groups). There are some deviations from the review protocol published in PROSPERO. 19 ORs instead of relative risks were used as effect measure for dichotomous outcomes because in some studies adjusted ORs and no absolute numbers are reported. Furthermore, it was not possible to undertake the planned subgroup analyses due to the small number of studies in each subgroup. R version 3.5.1 (R Foundation for Statistical Computing, 2015) and the R ‘meta’ package version 4.9-2 (developed by Guido Schwarzer) was used for all statistical analyses.
Results
Study characteristics
Newly identified studies selected for quantitative analysis; baseline study characteristics and overall results.
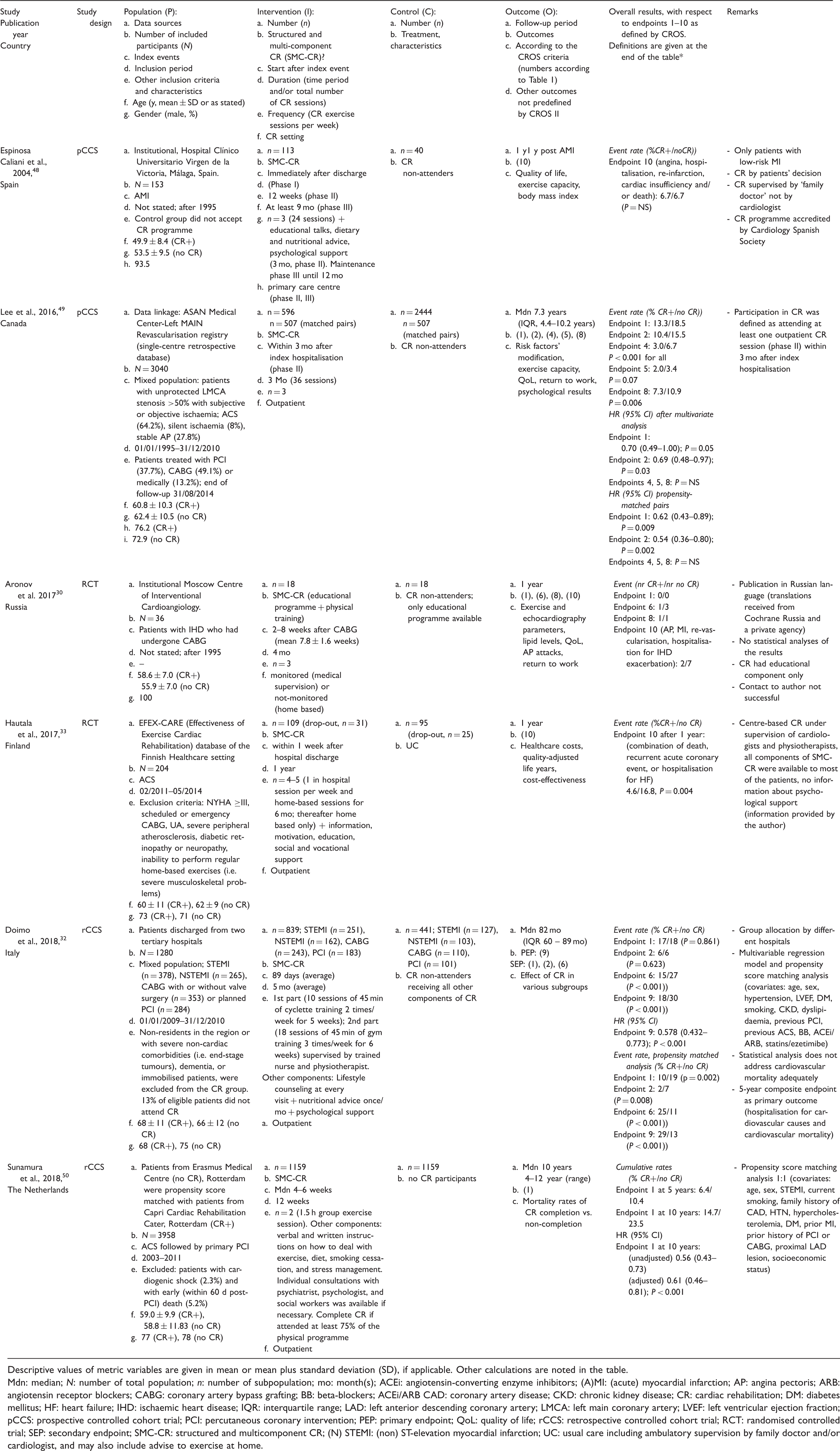
Descriptive values of metric variables are given in mean or mean plus standard deviation (SD), if applicable. Other calculations are noted in the table.
Mdn: median; N: number of total population; n: number of subpopulation; mo: month(s); ACEi: angiotensin-converting enzyme inhibitors; (A)MI: (acute) myocardial infarction; AP: angina pectoris; ARB: angiotensin receptor blockers; CABG: coronary artery bypass grafting; BB: beta-blockers; ACEi/ARB CAD: coronary artery disease; CKD: chronic kidney disease; CR: cardiac rehabilitation; DM: diabetes mellitus; HF: heart failure; IHD: ischaemic heart disease; IQR: interquartile range; LAD: left anterior descending coronary artery; LMCA: left main coronary artery; LVEF: left ventricular ejection fraction; pCCS: prospective controlled cohort trial; PCI: percutaneous coronary intervention; PEP: primary endpoint; QoL: quality of life; rCCS: retrospective controlled cohort trial; RCT: randomised controlled trial; SEP: secondary endpoint; SMC-CR: structured and multicomponent CR; (N) STEMI: (non) ST-elevation myocardial infarction; UC: usual care including ambulatory supervision by family doctor and/or cardiologist, and may also include advise to exercise at home.
Three new studies enrolled 4315 patients after ACS (total of 15 studies; n = 50,653 patients), one additional study included 36 patients after CABG (total of 10 studies; n = 14,583 patients), while two newly identified studies recruited 4320 patients in ‘mixed populations’ (total of 11 studies; n = 163,101 patients).
The CR setting was ‘outpatient’ in all new studies (total of 27) and the CR duration varied from 12 weeks to 12 months, thereby not changing the range of 3–4 weeks up to 12 months identified in the previous CROS study. Moreover, the previously reported ‘CR intensity’ ranging from two up to more than five exercise sessions per week plus motivation, information, education and psychosocial interventions with variable intensities and combinations remained unchanged.
Notably, the included studies reveal a considerable heterogeneity not only with respect to the predefined study designs (RCT, pCCS, rCCS), and populations (after ACS, after CABG, mixed CAD populations), but also with respect to study endpoints and biometrical evaluation (Tables 2, 3 and 4 and Figure 2). For this reason, the majority of the secondary endpoints predefined by CROS could not be integrated into a meta-analysis (Table 2, Figure 2).
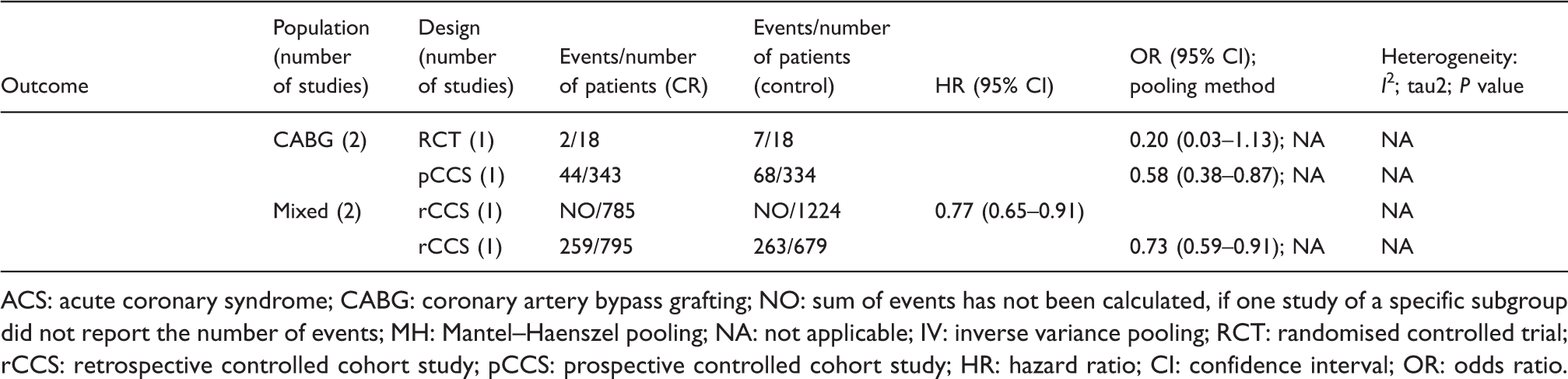
Analysis of total mortality. Forest plots presenting the evaluation of the endpoint ‘total mortality’. HR: hazard ratio; OR: odds ratio; MH: Mantel–Haenszel pooling method; CR: cardiac rehabilitation; no CR: no cardiac rehabilitation (control); CI: confidence interval; Events: number of events in the evaluated group; Total: number of patients in the evaluated group; Start (w): start of cardiac rehabilitation after hospital discharge in weeks; Follow-up: follow-up in years. Summary of results. ACS: acute coronary syndrome; CABG: coronary artery bypass grafting; NO: sum of events has not been calculated, if one study of a specific subgroup did not report the number of events; MH: Mantel–Haenszel pooling; NA: not applicable; IV: inverse variance pooling; RCT: randomised controlled trial; rCCS: retrospective controlled cohort study; pCCS: prospective controlled cohort study; HR: hazard ratio; CI: confidence interval; OR: odds ratio. Specific actions to compare groups: (1) prospectively evaluated intervention group versus retrospectively evaluated control group; (2) linkage of Canadian APPROACH and NACPR registry; (3) data extracted from the United States renal data System, USRDS; (4) retrospective identification of groups by questionnaires within a predefined study cohort; (5) retrospective identification of groups in a population surviving AMI for at least 30 days; (6) retrospective evaluation and formation of matched pairs; (7) groups were formed by two hospitals following different CR referral policies; (8) retrospective identification of groups by questionnaires and personal contact to relatives of deceased patients; (9) groups were formed prospectively according to predefined inclusion and exclusion criteria; (10) retrospective definition of the study groups out of an independent pre-existing study cohort on the basis of medical records;
72
(11) propensity score matching; (12) retrospective evaluation of a pre-existing cohort of another study evaluating CR attendance after automatic referral; (13) predefinition of inclusion and exclusion criteria, but final group formation by patient`s preferences and health care decision makers; (14) selection of CAD patients with musculoskeletal disease in addition; (15) retrospective definition of the groups; CR+ group was defined as attending at least one session within 6 months after the index event; (16) prospective definition of the groups out of the FRENA registry;
73
(17) patients referred to CR but not attending served as control; (18) groups were prespecified from the OMEGA trial cohort;
74
(19) 180 days survival after index event required; (20) study population has been extracted from two pre-existent studies (DepeMI, MIND-IT);75,76 (21) retrospective recruitment of study population from two previous RCTs not investigating CR or prognostic CAD outcomes;71,77 (22) data extracted from ASAN Medical Center-Left MAIN Revascularisation registry and ASAN Medical Center cardiac rehabilitation database; (23) control group was formed of patients who did not accept CR programme; (24) matching pairs from the Capri Cardiac Rehabilitation database and Erasmus Medical Centre database (control). Outcomes under investigation: the numbers refer to the predefined outcomes as outlined in Table 1. Confounding domains as specified by CROS: 1, age; 2, sex; 3, smoker; 4, diabetes; 5, history of stroke; 6, history of acute myocardial infarction; 7, reduced left ventricular ejection fraction; 8, acute/early ercutaneous coronary intervention during acute myocardial infarction. §Biometrical methods to manage confounding: (a) multivariable regression analysis; (b) propensity score matching; (c) propensity score-adjusted multivariable regression analysis; (d) confounders described; (e) retrospective matched pairs. Adjusting only for age and gender has been regarded as insufficient for the limitation of confounding. APPROACH: Alberta Provincial Project for Outcomes Assessment in Coronary Heart Disease; NACRP: Northern Alberta Cardiac Rehabilitation Program; FRENA: Risk Factors and Arterial Disease registry (Factores de Riesgo y ENfermedad Arterial); OMEGA: Randomised, Placebo-Controlled Trial to Test the Effect of Highly Purified Omega-3 Fatty Acids on Top of Modern Guideline-Adjusted Therapy after Myocardial Infarction; DepreMI: Depression after Myocardial Infarction study; MIND-IT: Myocardial Infarction and Depression Intervention Trial. Quality evaluation of randomised controlled trials included into meta-analysis (according to the Cochrane risk of bias table).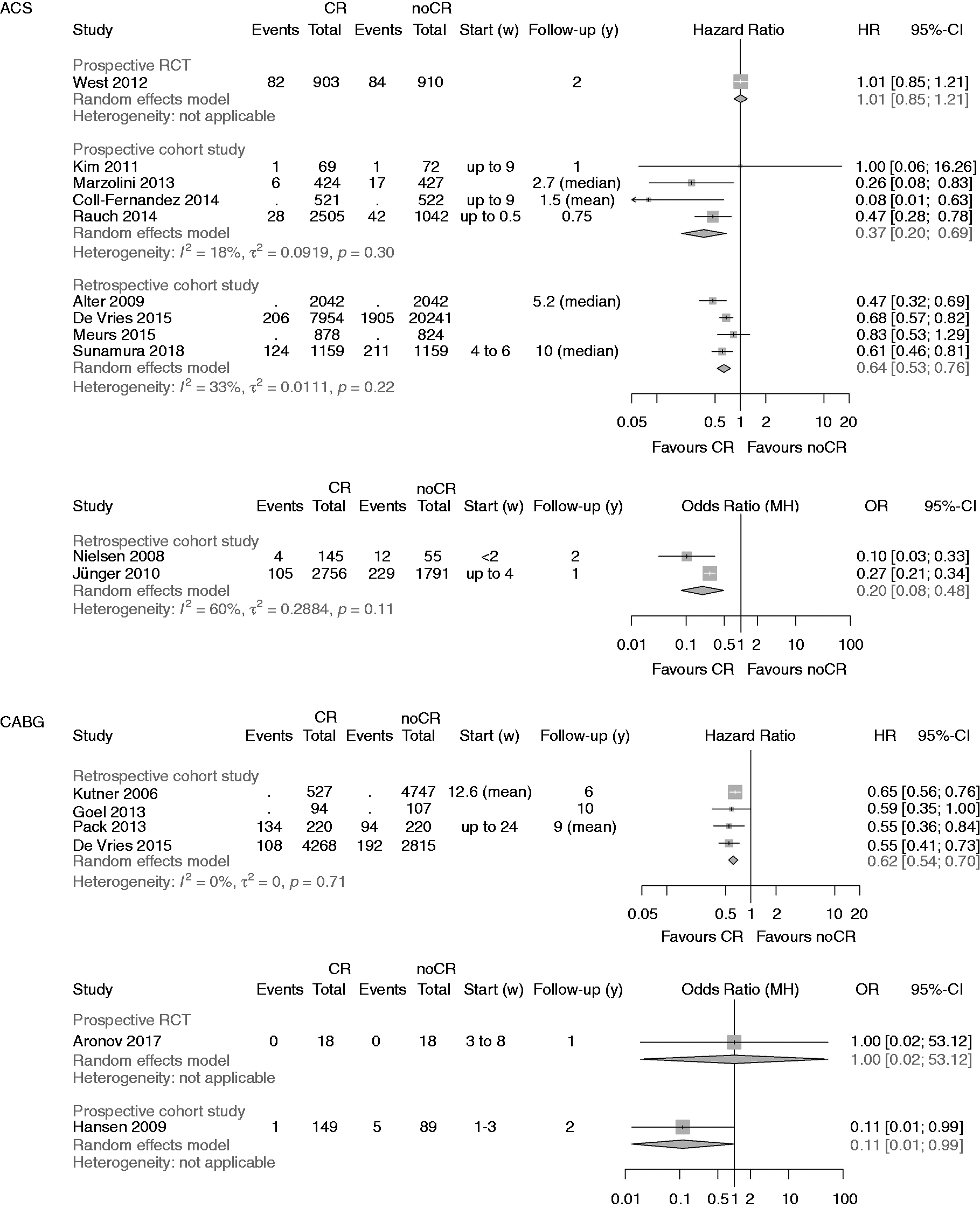
 Reporting of CR characteristics: +: sufficient; (+): information obtained by author or other sources; ↓: information limited.
Reporting of CR characteristics: +: sufficient; (+): information obtained by author or other sources; ↓: information limited.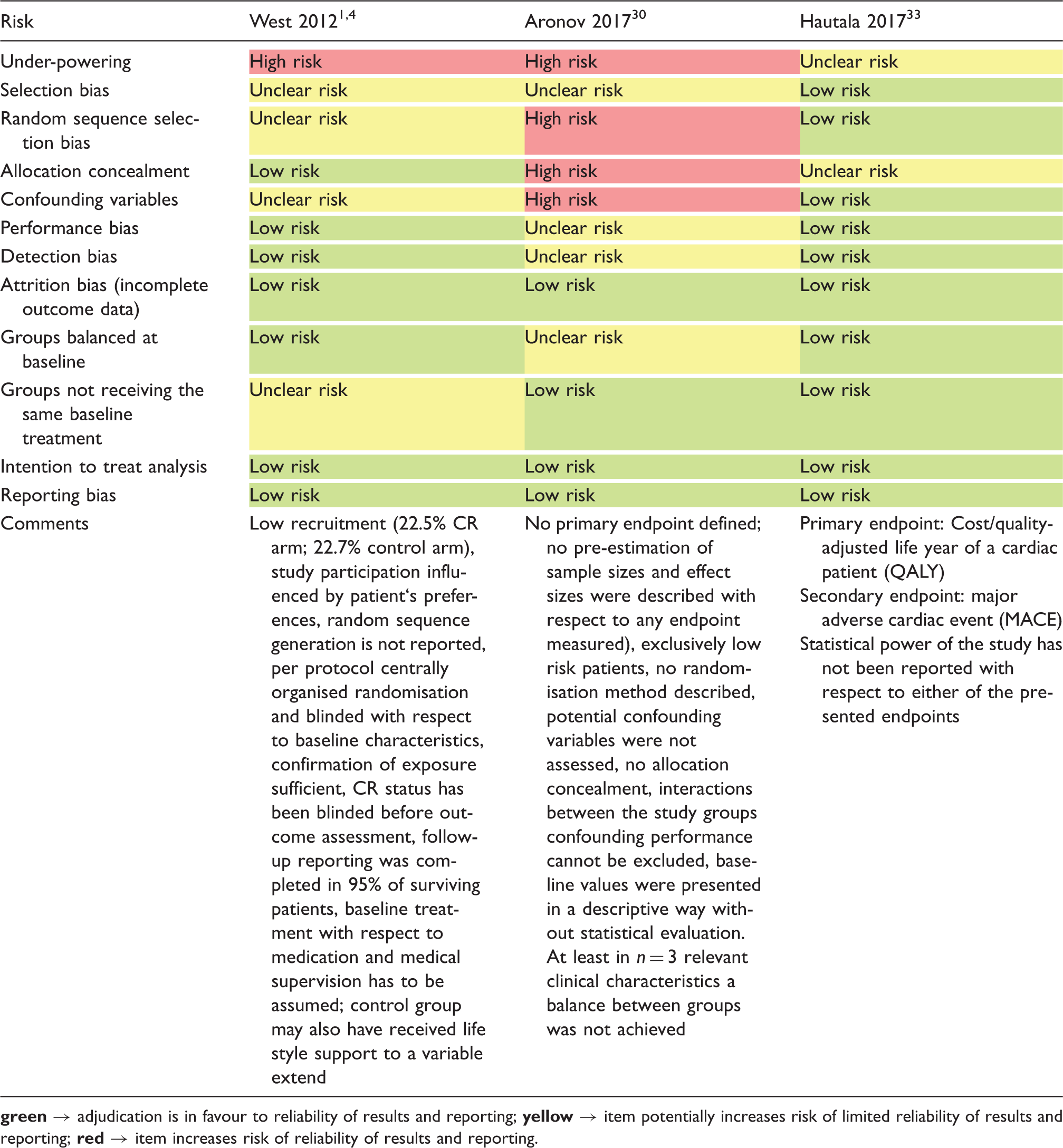
Primary endpoint ‘total mortality’
A summary of the clinical outcomes is shown in Table 2. The primary endpoint ‘total mortality’ was evaluated in 27 studies, one of them evaluating both mortality after ACS and after CABG (Figure 2). 27 Participation in CR was associated with a significant reduction of total mortality in all but six studies.14,28–32
After ACS a significant reduction in total mortality was confirmed by the newly added pCCSs (four studies; HR 0.37, 95% CI 0.20–0.69; I2 = 28%) and even strengthened by the newly added rCCSs (four studies; HR 0.64, 95% CI 0.53–0.76; I2 = 33%).
After CABG, the newly identified single RCT was small, only enrolling 36 low-risk patients. During a follow-up period of one year, no deaths occurred, and the risk of ‘underpowering’ has to be regarded as high in this study (see Table 4, Figure 2). No additional rCCSs or pCCSs were identified; consequently, the previous positive results on mortality reduction remained unchanged in this population.
In ‘mixed populations’ the addition of one more pCCS confirmed the significant mortality reduction in CR participants (two studies; HR 0.66, 95% CI 0.55–0.79) with zero heterogeneity. No additional rCCSs calculating HR within the mixed populations could be included by the current search (HR 0.52, 95% CI 0.36–0.77, I2 = 84%). The single rCCS newly added within the group calculating ORs did not change the neutral result reported before in this group (three studies; OR 0.68, 95% CI 0.34–1.37) but heterogeneity was high (I2 = 94%). Sensitivity analyses did not change the overall results.
Secondary endpoints
The results of CROS II with respect to the secondary endpoints are shown in Table 2, differentiating between the various study designs, populations and biometrical approaches. These results are summarised as follows:
Regarding the secondary endpoints ‘CV mortality’ (three additional studies, seven studies in total) and ‘MACCE’(major adverse cardiovascular and cerebrovascular events) (three studies, unchanged) all selected studies considerably differed with respect to populations and designs, and a ‘matching’ of these studies for meta-analysis was not possible (Table 2). Focusing on the endpoint ‘CV mortality’ and based on the two large controlled observational studies (pCCS, rCCS) there might be a trend in favour of CR participation after ACS and after CABG. With regard to the endpoint MACCE, however, the selected studies do not allow a final conclusion on the effect of CR participation (Table 2).
The outcomes ‘non-fatal MI’ (total seven studies) and ‘non-fatal stroke’ (total three studies) also did not show a clear trend, but all studies varied in design and population thus hindering a further evaluation by meta-analysis.
The same is true for studies investigating the variably predefined endpoints for ‘hospital readmission’ (endpoints 6–9, see Methods). Most of these studies had heterogeneous designs, and matching of the studies for meta-analysis was not possible (Table 2).
In a descriptive way the results on ‘hospital readmission’ may be summarised as follows: all studies included in CROS either showed a reduction of hospital readmissions in favour of CR participation, or there was a neutral result. In 12 studies, combined endpoints with various components were evaluated. One more RCT has been identified showing a statistically reduced combined endpoint (death, recurrent acute coronary events, or hospitalisation for HF) after CR participation compared to usual care (HR 0.26, 95% CI 0.09–0.73). 33
Quality evaluation of the studies
The sum of positive adjudications estimated by NOS is presented in Table 3 (for details see online version, Supplementary Material, Supplementary Table 5). Four additional studies were graded within a range of 5–7. In total, five out of 28 studies (18%) were graded with 5 points or less. Limitations were found with respect to representativeness (six studies), comparability of the cohorts (three studies), adequacy of follow-up (five studies) and the assessment of outcomes (two studies).
On the basis of the checklist of methodological issues on non-randomised studies the following limitations were identified (Tables 3 and 4):
Three studies were based on a secondary analysis of original studies with different original objectives. In three studies, either time or location differences between the study groups were apparent. In most studies, the group formation was potentially influenced by healthcare decision-makers and patient preferences. The majority of the studies had unclear study protocols and a consort flow diagram was presented in only seven out of 28 studies. Management of confounding was not reported in three studies, whereas the description of potential confounding domains remained unclear or has not been reported in 16 studies. Predefinition and calculation of all confounding domains as prespecified by CROS (see Materials and methods) were performed to various degrees. In only four studies all eight predefined confounders were considered for adjustment. Moreover, six studies only considered three or even fewer confounders as predefined by CROS. In general, adjustment for confounding was performed in 24 CCSs with four studies not applying adequate biometrical methods. Both RCTs evaluating the primary endpoint ‘total mortality’ do have a considerable risk of being underpowered (Table 4).14,30,33
Discussion
This update of CROS II confirms the beneficial prognostic effect of CR in CAD patients by significantly reducing the primary endpoint ‘total mortality’ especially after ACS or CABG. However, the effects of CR participation on secondary endpoints such as ‘CV mortality’, ‘non-fatal myocardial infarction’, ‘non-fatal stroke’, ‘combined endpoints’ and various forms of ‘hospital readmission’ remain less clear. This at least in part is due to a considerable heterogeneity of the selected studies with respect to design, populations, predefined endpoints and biometry. Inconsistent results may be due to the kind of selected endpoints including ‘weak’ endpoints with increased risks of confounding. This is particularly true for the variable forms of ‘hospital readmission’, which may be influenced by local routines in medical services, individual comorbidities not necessarily associated with CVDs, and the individual’s disease perception. Moreover, a longer survival of patients after AMI/CABG may reveal other diseases that primarily determine the number of hospital admissions during prolonged follow-up.
With regard to the secondary endpoint ‘non-fatal AMI’ an overall ‘neutral’ effect has also been reported by Cochrane (Anderson et al.). 9 As AMI and death are closely interrelated clinical events one might speculate that CR participation effectively prevents death initiated by AMI, but also reduces the incidence of AMI (fatal plus non-fatal) per se, resulting in an apparent ‘neutral effect’ with respect to non-fatal AMI occurrence. Unfortunately, the data sources presently available for CROS do not allow for further evaluation of this hypothesis.
One of the major strengths of this study is its robust approach to CR intervention aligned with published national CR standards and core components.5–7 Our strict definition of a comprehensive multicomponent CR underscores the importance of the amount of physical exercise provided, the adherence to exercise intervention and the adherence to non-exercise components on the patients’ prognosis. The results of recently published meta-analyses (some of them including studies of the modern era of novel medication and interventions) seem to support this approach and somehow elucidate our results. Thus, van Halewijn et al. have shown that a significant reduction in all-cause mortality was feasible in CAD patients only under the condition of a comprehensive CR programme managing six or more cardiovascular risk factors, 10 while the recently published EU-CaRE study showed positive effects of comprehensive CR in 58% of older patients with three or more uncontrolled risk factors before CR. 34 These findings, coupled with CROS II results, strengthen clinical recommendations that comprehensive CR is preferable to standalone exercise-based CR in reducing total and cardiac mortality, in post-myocardial infarction patients. 13 The effectiveness of a comprehensive CR programme is increased by the patients’ adherence and by the shared effort consequently to assess and treat the majority of all individual cardiovascular risk factors.
With regard to the importance of the CR dose, Santiago de Araujo Pio et al. established that total mortality reduction was only possible in CVD patients experiencing medium and high doses of CR. 12 Similar CR dose and volume-related effects on mortality have been published.9,35 Finally, in a systematic review of multicomponent CR, applying almost all CROS inclusion criteria, the study by Sumner et al. carried out a meta-analysis of observational studies published after the year 2000, concluding that all-cause and cardiac mortality were reduced in AMI patients following a CR programme. 36
Still, one has to keep in mind that this beneficial effect of CR participation as shown in CROS may not apply to special subgroups such as elderly and frail patients who need a particularly personalised approach. 37 According to Deaton, 38 however, the average age of the CROS study population reflects actual clinical reality. Likewise, CR participation of patients with severe systolic heart failure may not result in mortality reduction as shown in previous meta-analyses.39–41
Apart from these limitations, CROS II presents a timely account of the effectiveness of CR when delivered to agreed published standards including scientifically confirmed CR core components.5–7 Utilising a strict approach to CR intervention study inclusion we can report a significant benefit (Table 2 and Figure 2) in favour of CR with respect to all-cause mortality. However, at the same time this approach might be viewed as a significant weakness as it makes our findings almost incompatible with previous reviews, which have been much more inclusive of CR interventions often defined by innovations in CR being evaluated as part of clinical trials rather than informed by interventions based on published CR programme standards and core components. Only three RCTs were selected for CROS II compared to 63 in the most recent Cochrane review which reported a significant reduction in cardiovascular mortality but not in all-cause mortality. 9 We are not suggesting that previous trial-based reviews are erroneous. On the contrary, we agree that robust trials-based reviews remain top of the evidence base hierarchy. What we are proposing is that the CROS II approach differs to the extent that it should be viewed as an additional form of evidence that utilises registry-based research reflecting a broader population in the modern cardiology era from 1995 onwards.
For a critical estimation of the CROS II results, the following aspects have to be emphasised:
CR participation after ACS or CABG is associated with reduced total mortality if delivered on top of the current evidence-based treatment modalities (medication and acute coronary interventions). CR participation therefore may contribute to treatment adherence and further add effective individual lifestyle changes necessary to reduce patients’ cardiovascular risk significantly.42–46 This positive effect of CR participation obviously works in current clinical practice of different countries provided a minimum of CR volume and intensity is delivered. This especially refers to the individually adapted and supervised exercise training and a rigorous treatment of all individual cardiovascular risk factors. 9,12,13,47
Unfortunately, these prerequisites of successfully delivered CR – although outlined in detail in many position papers – are not necessarily followed in clinical practice. As noted in CROS II, these prerequisites are not sufficiently described in many clinical studies evaluating CR effectiveness. Therefore, there is an urgent need to translate these well-known and evidence-based minimal standards effectively into all-day clinical practice wherever CR is offered. Moreover, these clinical standards need to be the adamant basis of future CR outcome studies. To this end, minimal standards for CR interventions in clinical practice and clinical trials should be based on robust published guidelines and research. We offer the CROS II definition and criteria as a useful guide for optimal CR intervention content and delivery; including multidisciplinary and multicomponent programmes with structured, supervised exercise training delivered at least twice per week in combination with motivational techniques, risk factor modification education, dietary advice, psychosocial and vocational support delivered at least once per week. The CR setting could be inpatient, outpatient or mixed but the time between hospital discharge and CR initiation should be as low as possible, preferably within 3 months.
From this background it is one of CROS’s aims not only to evaluate the results and clinical outcomes of the studies included, but also to evaluate critically the strengths and deficiencies in detail of each single study included in the meta-analysis (see Table 3). As in the first evaluation in CROS, this update uncovers considerable deficits in current CR studies that need to be addressed and prevented in future. These deficits include predominantly insufficient description of CR content (e.g. applied components), frequency and volume of exercise sessions, CR initiation (i.e. after hospital stay for an acute cardiac event) and duration, absence of CR adherence at follow-up as well as methodological issues such as the inadequate consideration of confounding parameters at the stage of study and statistical analysis design.
Clinical implications
Together with the results of other recent reviews, minimal requirements for a successful CR after ACS or CABG are apparent and need to be ensured in clinical practice:4,9,10,12,13,45
CR is multicomponent including consequent treatment of the individual’s cardiovascular risk factors, individually adapted physical exercise, information, motivation as well as individualised psychosocial support.
4
The individualised approach also reflects gender, age, frailty, heart failure, concomitant diseases, psychosocial background and effectors of the individual’s health and capabilities. CR is supervised and carried out by adequately trained health professionals including cardiologists.
4
During CR the ‘dose’ of exercise training (number of weeks of exercise training × average number of sessions/week × average duration of session in minutes) exceeds 1.000.
9
The number of CR sessions (including physical exercise, information, education and psychosocial support) needs to exceed 36.
12
During CR all individually recognised cardiovascular risk factors need to be addressed and treated.
10
Consequently, future studies on the effect of CR need to report in detail whether these minimal requirements were rigorously followed by the participating CR centres.
Conclusions
CROS II confirms the effectiveness of CR participation after ACS and after CABG in actual clinical practice by reducing total mortality under the conditions of current evidence-based CAD treatment. The CROS approach to more strictly predefined CR intervention and to include controlled registry-based studies represents a valid hybrid approach that has clear utility in clinical decision-making.
Supplemental Material
CPR905719 Supplemental Material - Supplemental material for Effectiveness of comprehensive cardiac rehabilitation in coronary artery disease patients treated according to contemporary evidence based medicine: Update of the Cardiac Rehabilitation Outcome Study (CROS-II)
Supplemental material, CPR905719 Supplemental Material for Effectiveness of comprehensive cardiac rehabilitation in coronary artery disease patients treated according to contemporary evidence based medicine: Update of the Cardiac Rehabilitation Outcome Study (CROS-II) by Annett Salzwedel, Katrin Jensen, Bernhard Rauch, Patrick Doherty, Maria-Inti Metzendorf, Matthes Hackbusch, Heinz Völler, Jean-Paul Schmid and Constantinos H Davos in European Journal of Preventive Cardiology
Footnotes
Acknowledgements
EAPC Cardiac Rehabilitation Section, Nucleus members:
• Ana Abreu, Cardiology Department, Hospital Santa Marta, Lisbon, Portugal
• Marco Ambrosetti, Cardiovascular Rehabilitation Unit, ‘Le Terrazze’ Clinic, Cunardo, Italy
• Thomas Berger, Cardiomed Linz, Austria
• Véronique Cornelissen, Research Group for Rehabilitation in Internal Disorders, KU Leuven, Belgium
• Wolfram Doehner, Charité-Universitätsmedizin Berlin, Germany
• Ines Frederix, Faculty of Medicine and Life Sciences, Hasselt University, Belgium
• Andreas Gevaert, Laboratory of Cellular and Molecular Cardiology, Universität Antwerpen, Belgium
• Dominique Hansen, Biomedisch Onderzoeksinstituut (BIOMED), Universiteit Hasselt, Belgium
• Marie-Christine Iliou, Cardiac Rehabilitation and Secondary Prevention, Corentin Celton Hospital, APHP, Paris, France
• Hareld Kemps, Máxima Medisch Centrum, The Netherlands
• Nicolle Kraenkel, Medizinische Klinik für Kardiologie, Charité-Universitätsmedizin Berlin, Germany
• Jari Laukkanen, University of Jyväskylä and Central Finland Health Care District, Finland
• Roberto Pedretti, Clinic for Pressure Monitoring and Hypertension, Pavia, Italy
• Maria Simonenko, Federal Almazov North-West Medical Research Centre, St Petersburg, Russian Federation
• Heinz Völler, University of Potsdam, Human Sciences Faculty, Potsdam, Germany
• Matthias Wilhelm, University Hospital of Cardiology, Bern, Switzerland
Author contribution
All authors participated in designing the study, generating hypotheses, interpreting data, and critically reviewing the report. The special responsibilities were as follows: Initiation, organisation and leading of the project: BR, CHD, PD, JPS, HV; literature search and search strategies: MIM, BR; study selection: AS, CHD, PD, BR; study evaluation: AS, CHD, BR, KJ; statistical and biometrical analyses: KJ, MH; writing: AS, HV, CHD, PD, KJ, MIM, BR; internal reviewing: JPS, BR, HV, AS, PD, CHD, and the nucleus members of the secondary prevention and rehabilitation section of the European Association of Preventive Cardiology (EAPC).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Pfizer AG Switzerland (unrestricted grant), Deutsche Herzstiftung eV (German Heart Foundation), Deutsche Gesellschaft für Prävention und Rehabilitation von Herz-Kreislauferkrankungen eV (DGPR; German Society of Cardiovascular Prevention and Cardiac Rehabilitation), Schweizer Arbeitsgruppe für kardiovaskuläre Prävention, Rehabilitation und Sportkardiologie (SCPRS; Swiss Working Group for Cardiovascular Prevention, Rehabilitation and Sports Cardiology). The sponsors did not have any influence on study initiation, conducting and reporting.
Previous review version
Rauch B, Davos CH, Doherty P, et al. The prognostic effect of cardiac rehabilitation in the era of acute revascularisation and statin therapy: a systematic review and meta-analysis of randomized and non-randomized studies – the Cardiac Rehabilitation Outcome Study (CROS). Eur J Prev Cardiol 2016; 23: 1914–1939. doi: 10.1177/2047487316671181.
Systematic review registration
PROSPERO International prospective register of systematic reviews (registration number: CRD42014007084): http://www.crd.york.ac.uk/prospero/review_print.asp?RecordID=7084&UserID=5736.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.