Abstract
Background
Modifiable lifestyle risk factors (modRF) of coronary artery disease (CAD) are associated with increased inflammation represented by elevated C-reactive protein (CRP) levels. Lifestyle changes may influence the inflammatory burden in patients with CAD, relevantly modifying the target population for emerging anti-inflammatory compounds.
Aims
The aims of this study were to analyse the association of modRF and CRP levels in CAD patients, and to define a potential target population for anti-inflammatory treatment with and without the optimisation of modRF.
Methods
We included all patients with angiographically documented CAD from the observational cohort study INTERCATH. Patients with recent myocardial infarction, malignancy, infectious disease, and pre-existing immunosuppressive medication including a history of solid organ transplantation were excluded. Overweight (body mass index (BMI) ≥ 25 kg/m2), smoking, lack of physical activity (PA; <1.5 h/week), and poor diet (≤12 points of an established Mediterranean diet score (MDS), range 0–28 points) were considered as modRF. CRP was measured by a high-sensitivity assay (hsCRP) at baseline. We performed multivariable linear regressions with log-transformed hsCRP as the dependent variable. Based on these associations, we calculated potential hsCRP levels for each patient, assuming optimisation of the individual modRF.
Results
Of 1014 patients, 737 (73%) were male, the mean age was 69 years, and 483 (48%) had an hsCRP ≥ 2 mg/l. ModRF were significantly overrepresented in patients with hsCRP ≥ 2 mg/l compared to patients with an hsCRP < 2 mg/l (BMI ≥ 25 kg/m2: 76% vs 61%; PA < 1.5 h/week: 69% vs 57%; MDS ≤ 12: 46% vs 37%; smoking: 61% vs 54%; p < 0.05 for all). hsCRP increased with the incremental number of modRF present (median hsCRP values for N = 0, 1, 2, 3, and 4 modRF: 1.1, 1.0, 1.6, 2.4, 2.8 mg/l, p < 0.001). Multivariable linear regression adjusting for age, sex, intake of lipid-lowering medication, and diabetes mellitus revealed independent associations between log-transformed hsCRP and all modRF (BMI ≥ 25 kg/m2: exp(ß) = 1.55, p < 0.001; PA < 1.5 h/week: exp(ß) = 1.33, p < 0.001; MDS ≤ 12: exp(ß) = 1.18, p = 0.018; smoking: exp(ß) = 1.18, p = 0.019). Individual recalculation of hsCRP levels assuming optimisation of modRF identified 183 out of 483 (38%) patients with hsCRP ≥ 2 mg/l who could achieve an hsCRP < 2 mg/l via lifestyle changes.
Conclusion
modRF are strongly and independently associated with CRP levels in patients with CAD. A relevant portion of CAD patients with high inflammatory burden could achieve an hsCRP < 2 mg/l by lifestyle changes alone. This should be considered both in view of the cost and side-effects of pharmacological anti-inflammatory treatment and for the design of future clinical trials in this field.
Keywords
Introduction
Key steps in the pathogenesis of atherosclerosis involve inflammatory processes.1–3 The recent CANTOS trial provided proof of principle of specific anti-inflammatory treatment on cardiovascular outcomes in patients with coronary artery disease (CAD) and a high inflammatory burden represented by a C-reactive protein (CRP) ≥2 mg/L. 4 CRP has been shown to be independently associated with an increased incidence of future cardiovascular events in numerous prospective cohort studies, both in populations at risk5,6 as well as CAD populations.7,8 However, CRP is influenced by many factors, and both observational studies as well as interventional studies have shown a significant impact of modifiable lifestyle risk factors (modRF) for CAD on CRP levels. This concerns smoking,9–13 lack of physical activity (PA),14–18 poor diet (e.g. failure to adhere to a Mediterranean diet),19,20 and overweight.21,22
The number of patients considered eligible for anti-inflammatory treatment after the optimisation of modRF remains undetermined. This is of high relevance in light of safety issues and the cost of anti-inflammatory treatment, as well as from a health economics perspective and for the design of future trials in the field.
Therefore, our aim was (I) to determine the association between CRP levels and modifiable risk factors in patients with CAD and (II) to evaluate whether the optimisation of modifiable risk factors alone may reduce inflammatory burden as measured by CRP below the threshold of 2 mg/L.
Methods
The INTERCATH study is an observational cohort study ongoing since January 2015 including patients undergoing coronary angiography at the University Heart and Vascular Center Hamburg. The study protocol of the INTERCATH study was approved by the Ethics Committee of Hamburg, Germany (PV4303). Each patient gave written informed consent. Design and rationale have been described previously.23,24 Comorbidities, information on lifestyle risk factors, and medication were assessed by a questionnaire at enrolment as well as written medical reports.
Inclusion and exclusion criteria
Patients <18 years or incapable to give written informed consent or patients without sufficient knowledge of the German language were not considered for inclusion. Furthermore, all patients with cardiogenic shock, life-threating arrhythmias, or other circumstances of hemodynamic instability were not screened for inclusion in the INTERCATH cohort. For the present analysis, all patients with angiographically documented stable CAD (no myocardial infarction at enrolment or within the past 30 days), available measurement of a high-sensitivity assay (hsCRP), and complete information on modRF were included. Amongst patients with hsCRP ≥ 2 mg/L, we excluded patients with at least one of the following: an active infection, an active or recent (within the past five years) malignancy, a history of solid organ transplantation, a pre-existing immunosuppressive medication, a recent postoperative state, or a dynamically changing CRP-value, defined as a pertinent change of more than 50% in serial measurements before or after inclusion if available.
Assessment of modifiable lifestyle cardiovascular risk factors
Smoking, lack of PA, overweight, and poor adherence to a Mediterranean diet were considered as modRF. Smoking was defined as currently smoking or regular smoking within the last five years. Lack of PA was defined as <1.5 h/week of the guideline-recommended amount of exercise. 25 Overweight was defined by a body mass index (BMI) ≥25 kg/m2. To quantify adherence to Mediterranean diet, an established Mediterranean diet score (MDS) was derived, as described previously.24,26 In short, a food frequency questionnaire nearly identical to that used in studies like STABILITY 26 or INTERHEART 27 assesses consumption of six food groups plus alcohol. Based on consumption frequencies, a score ranging from 0 to 28 points is calculated, with 0 points reflecting no adherence to a Mediterranean diet, and 28 points reflecting highest adherence to a Mediterranean diet. Failure to adhere to a Mediterranean diet was defined as a MDS ≤12, a threshold which was recently shown to be predictive for the severity of CAD and prognosis for patients with stable CAD.24,26
Cardiovascular risk factor assessment
Patients taking antihypertensive medication or with arterial hypertension documented as a diagnosis in a written medical report were classified to have arterial hypertension. Patients were defined to have diabetes mellitus in the case of intake of oral blood-glucose-lowering therapy, regular substitution of insulin, HbA1c levels >6.5%, or documented diabetes in a written medical report.
Laboratory methods
All blood samples were drawn from a peripheral vein at fasting state before coronary angiography. Blood counts, renal function parameters, CRP, HbA1c, and lipid parameters at baseline were determined directly after sampling by standardised routine laboratory methods. CRP was measured by an hsCRP (CardioPhase® hsCRP assay).
Statistical analyses
Continuous variables are shown as mean ± standard deviation or as median (25th percentile, 75th percentile) and were compared with the Mann–Whitney test, whereas binary variables are shown as absolute numbers and percentages and were compared with the χ 2 test. The Kruskal–Wallis test was used to compare hsCRP levels of the groups defined by the number of modifiable risk factors present. To analyse the association between modifiable risk factors and hsCRP, we performed uni- and multivariable linear regression analyses with log-transformed hsCRP as the dependent variable. A transformation was necessary since hsCRP was not normally distributed. The log-transformation was chosen after a Box–Cox analysis. For multivariable analyses, we constructed, assisted by a Directed Acyclic Graphs (DAG) analysis, a model adjusting for age and sex (Model 1) and a model adjusting for age, sex, diabetes, and intake of cholesterol-lowering drugs (Model 2). A complete data set was present for model 1, whilst for model 2 less than 1% of patients lacked full information for the adjusting variables, thus ruling out missing values as a significant source of bias to our results. Based on the associations in the multivariable linear regression, we recalculated the potential hsCRP levels assuming optimisation of the modifiable risk factors. For each patient, we multiplied the measured hsCRP by the exposed sum of the betas of the present risk factors. Non-existent risk factors were assumed to stay constant. Results were considered as statistically significant at a significance level of a two-sided alpha <0.05. All statistical tests were computed using R version 3.5.2 (2018-12-20).
Results
Baseline characteristics
Baseline characteristics of the study cohort.
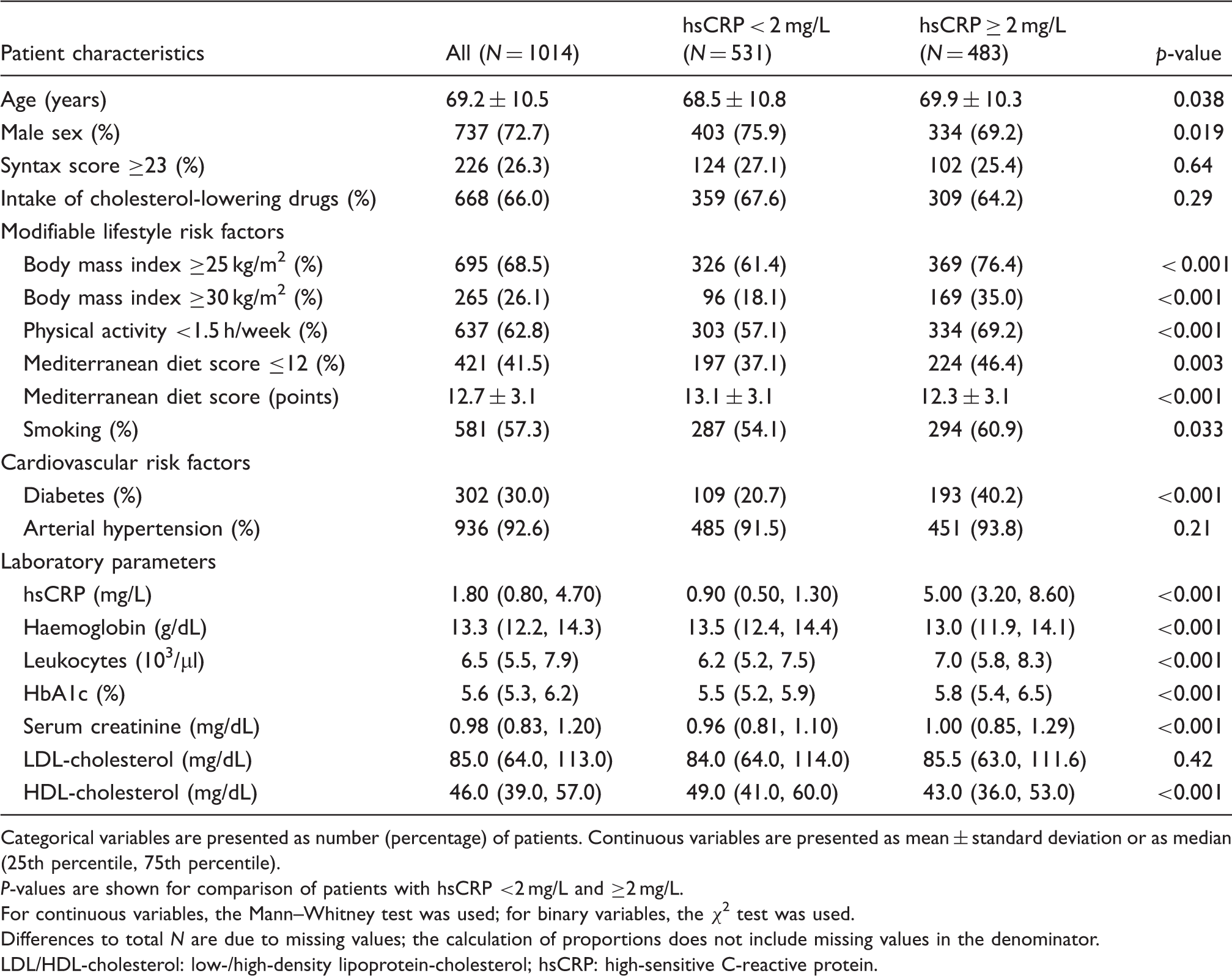
Categorical variables are presented as number (percentage) of patients. Continuous variables are presented as mean ± standard deviation or as median (25th percentile, 75th percentile).
P-values are shown for comparison of patients with hsCRP <2 mg/L and ≥2 mg/L.
For continuous variables, the Mann–Whitney test was used; for binary variables, the χ2 test was used.
Differences to total N are due to missing values; the calculation of proportions does not include missing values in the denominator.
LDL/HDL-cholesterol: low-/high-density lipoprotein-cholesterol; hsCRP: high-sensitive C-reactive protein.
Distribution of modifiable risk factors and their effect on hsCRP levels
Figure 1 shows hsCRP levels according to the number of modifiable risk factors present. Only 41 patients had no modifiable risk factors (4.0%), whilst about three-quarters of the patients presented with at least two modifiable risk factors (N = 769; 75.8%). We found similar hsCRP levels between patients with n = 0 modifiable risk factors and those with n = 1 modifiable risk factors, but a significant increase in hsCRP levels with each incremental modifiable lifestyle risk factor >1 (median hsCRP for 1–4 modifiable risk factors: 1.0, 1.6, 2.4, 2.8 mg/L, respectively; p < 0.05).
hsCRP in dependence of the number of modifiable risk factors.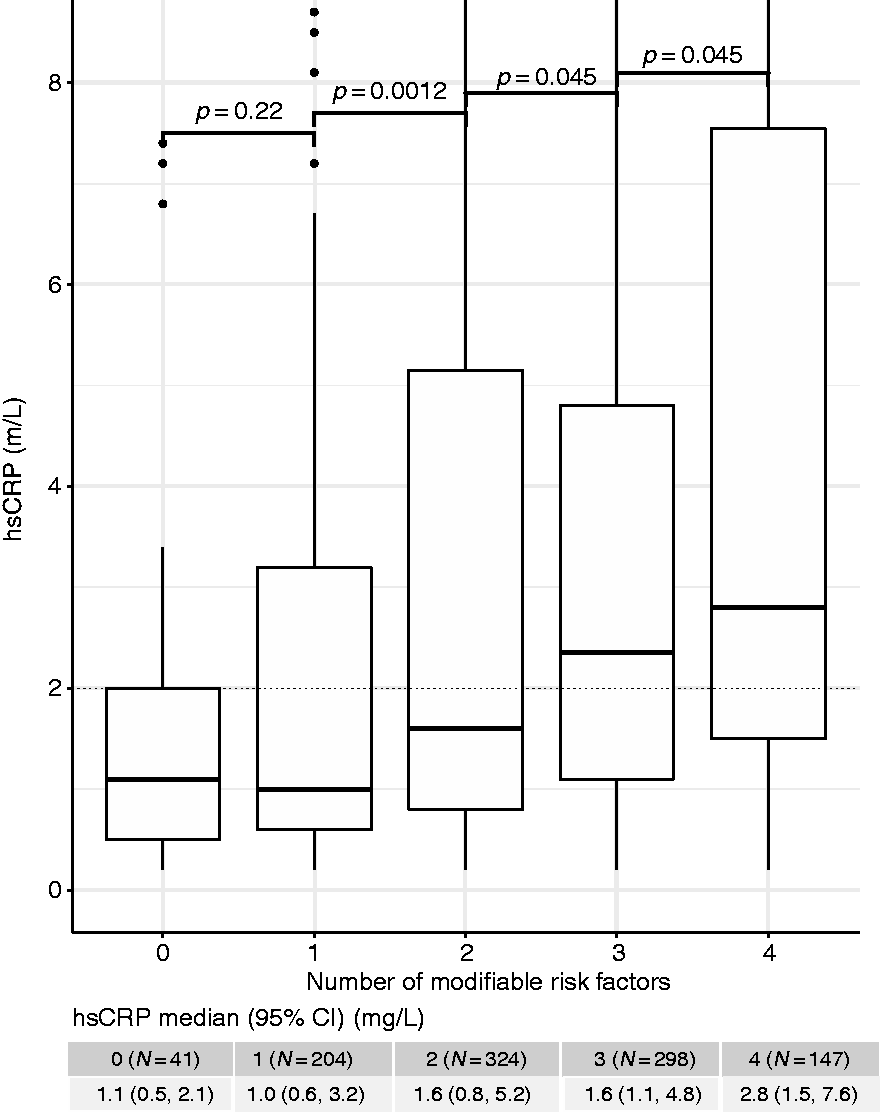
Association of hsCRP with modRF
Univariable linear regression results are demonstrated in Supplementary Table 1. We found significant associations between all modifiable risk factors and elevated hsCRP levels (BMI ≥ 25 kg/m2: exp(ß) = 1.63 (1.40, 1.89); PA < 1.5 h/week: exp(ß) = 1.58 (1.37, 1.82); MDS ≤ 12: exp(ß) = 1.31 (1.14, 1.51), all p < 0.001; smoking: exp(ß) = 1.16 (1.01, 1.34), p = 0.040). A strong association with increased hsCRP levels was also observed for the presence of diabetes.
In the multivariable linear regression analysis adjusted for age and sex (Model 1), lack of PA, failure to adhere to a Mediterranean diet, and overweight, but not smoking, were significantly associated with hsCRP levels (Supplementary Table 2). In the model fully adjusted for age, sex, diabetes, and intake of cholesterol-lowering drugs (Model 2), all modifiable cardiovascular lifestyle risk factors were significantly associated with elevated hsCRP levels, as demonstrated in Figure 2 and Supplementary Table 3: BMI ≥ 25 kg/m2: exp(ß) = 1.55 (1.34, 1.80); PA < 1.5 h/week: exp(ß) = 1.33 (1.15, 1.53), both p < 0.001; MDS ≤ 12: exp(ß) = 1.18 (1.03, 1.36), p = 0.018; smoking: exp(ß) = 1.18 (1.03, 1.36), p = 0.019.
Forest plot demonstrating exp(β) of each modifiable risk factor from a multivariable linear regression analysis, with hsCRP as the dependent variable after adjustment for age, sex, diabetes, and intake of cholesterol-lowering drugs.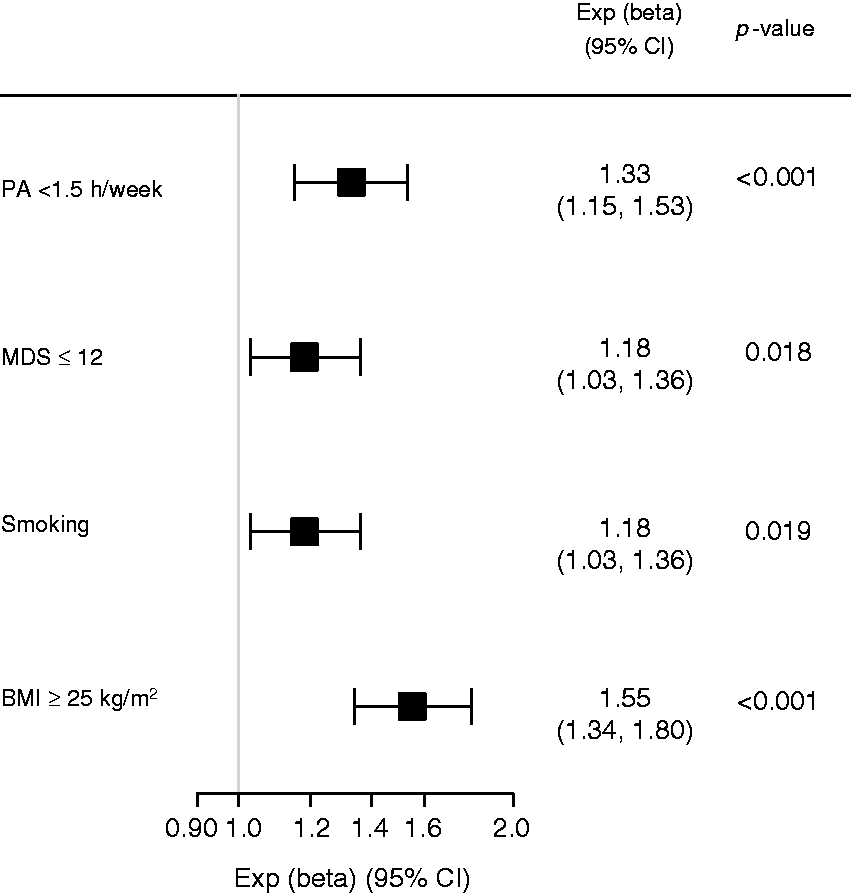
Modelling the effect of modifiable risk factor optimisation on hsCRP levels
Assuming optimisation of each patient’s modifiable risk factors, we calculated the individual hsCRP-values based on the strength of the associations in the fully adjusted multivariable linear regression. The original hsCRP distribution with a median of 1.80 mg/L (0.80, 4.70) was shifted to lower levels with a recalculated hsCRP distribution displaying a median of 0.90 mg/L (0.50, 2.40). Of the initial 483 patients with hsCRP levels ≥2 mg/L, 183 (37.9%) reached an hsCRP <2 mg/L after assumed optimisation. Figure 3 demonstrates the redistribution of CRP values of patients with high inflammatory status.
hsCRP distribution assuming optimisation of modifiable lifestyle risk factors.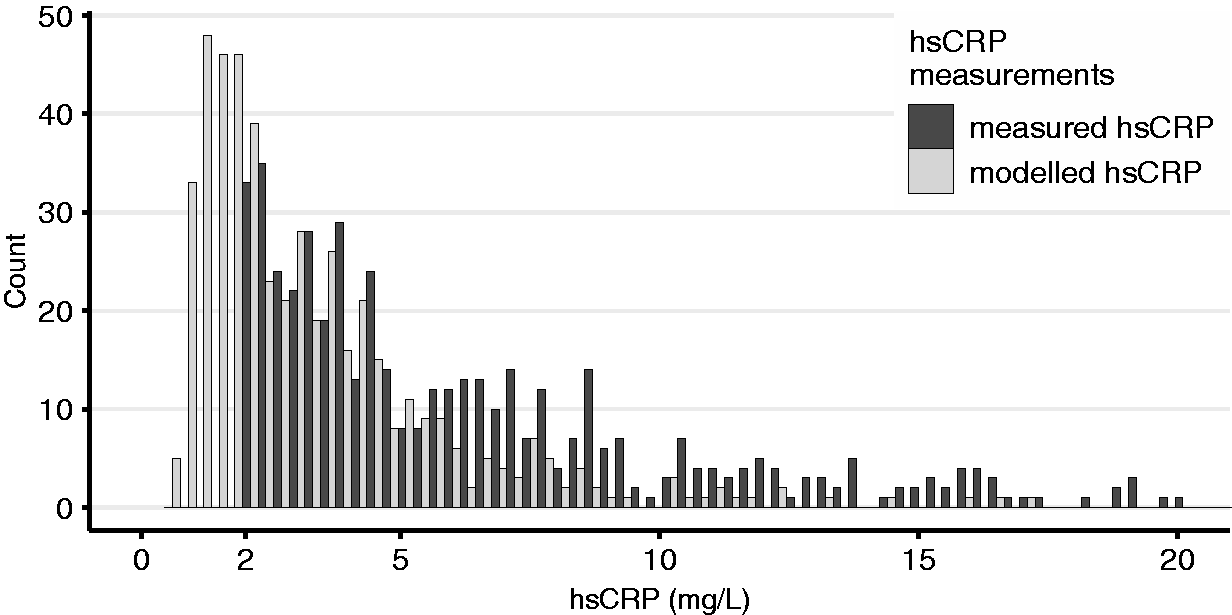
Discussion
The main findings of the present study are: (I) CAD patients display a lifestyle risk profile that leaves significant room for improvement; (II) there are strong and independent associations between modRF and CRP levels in patients with CAD; and (III) optimisation of lifestyle risk factors could relevantly reduce the size of the target population for anti-inflammatory treatment.
Study cohort, hsCRP distribution, and presence of modifiable risk factors
Our study cohort reflects a typical, contemporary CAD population, with predominantly male and older patients and with both conventional and lifestyle risk factors in high prevalence. The cohort’s median hsCRP of 1.8 mg/L is of similar magnitude to that reported in other CAD cohorts.7,8,28 The subgroup of patients with hsCRP ≥ 2 mg/L compares well with the study population from the CANTOS trial, 4 notable differences being older age and slightly higher hsCRP values (median 5.0 vs 4.2 mg/L). Overall, our study cohort can be considered a representative population of CAD patients.
As expected, we observed an increase in hsCRP with the number of present modifiable risk factors. Importantly, a very high percentage of patients (about 75%) displayed at least two modifiable risk factors. Failure to meet secondary prevention targets, including goals on lifestyle changes, is common in CAD patients, as demonstrated by recent European data. 29 Unlocking the potential health benefit inherent in meeting these targets remains a challenge.
Association of lifestyle risk factors with CRP
In our study cohort, we found significant and independent associations between the presence of lifestyle risk factors and inflammatory burden. Many previous studies examined the isolated association of a certain lifestyle risk factor on CRP levels. However, most of the risk factors correlate with each other and an adjustment for all lifestyle risk factors is mandatory to determine the true association for each single risk factor. A particular strength of this study is the simultaneous consideration of all lifestyle risk factors on CRP levels. We shall briefly discuss the results for each risk factor.
Smoking
Our data support the previously described association between smoking status and raised levels of most inflammatory biomarkers, including CRP. 10 However, the relationship between smoking and CRP is complex: associations are reported for smoking intensity 12 as well as cumulative smoking exposure. 11 Time elapsed since the last cigarette also influences CRP levels, both on the short scale of hours 10 as well as on the scale of years in the case of smoking cessation: former smokers have persistently elevated CRP levels, which gradually decrease over time, with some studies reporting a return to never-smoker levels after five years.9,13 Therefore, our smoking variable (a combination of current and recently ceased smoking) offers a more generalised depiction of the association between smoking and CRP.
On the cellular level, the numerous constituents of cigarette smoke act in a complex fashion to drive vascular inflammation. Key processes are endothelial activation, including increased expression of adhesion molecules, the nuclear factor-kappa B-mediated production of pro-inflammatory cytokines, endothelial dysfunction with reduced nitric oxide production, leading to increased lipid oxidation via reactive oxygen species, macrophage activation, and, thus, increased foam cell formation. 30
PA
We found a significant association between regular PA and lower CRP, in line with data from previous observational studies.14,15 Interventional studies reveal a more complex picture, as there is uncertainty whether changes in CRP after PA interventions are down to changes in body weight or body fat alone.31,32 However, two recent meta-analyses of interventional studies found a significant reduction in CRP through exercise,17,18 the first both in healthy adults and those with heart disease, the second observing the largest decreases in CRP when accompanied by changes in body weight or body fat. In our results, the association between PA and hsCRP is attenuated in the multivariable regression analysis compared to the univariable results, whilst the association of BMI with hsCRP remains largely unaltered. This suggests that the association of PA with inflammation might be at least partly driven by body weight.
The exact mechanism by which PA reduces CRP independent of changes in body weight or body fat composition is incompletely understood. Proposed mechanisms include an altered, more anti-inflammatory cytokine production pattern of blood mononuclear cells, 33 improved endothelial function, 34 and an antioxidant effect with reduced oxidation of low-density lipoprotein (LDL). 35 Recent data on the association of PA with cardiovascular biomarkers independent of body fat might yield mechanistic insight in future studies. 36
Diet
We could confirm the findings from observational and interventional studies that a more stringent adherence to a Mediterranean diet is associated with or leads to a lower CRP,19,20 Similar to our results for PA, the association with hsCRP by multivariable regression was attenuated compared to univariable regression results. This is likely due to the covariates diabetes and overweight, since both represent diseased states strongly driven by diet.
The mechanism explaining the anti-inflammatory effect of a Mediterranean diet is not fully understood. A favourable impact on oxidised LDL levels, 37 a known inflammatory stimulus in the vascular wall, as well as anti-inflammatory and antioxidant effects of polyphenols present in virgin olive oil 38 are being discussed.
Overweight
The association observed between a BMI ≥ 25 kg/m2 and hsCRP was the strongest amongst the studied modifiable risk factors. This mirrors findings in diverse populations, where a raised BMI and particular central adiposity are associated with an inflammatory state. 22 Furthermore, studies concerning the effect of weight loss on CRP demonstrate a decrease in CRP, 39 irrespective of whether this loss is achieved by conservative40,41 or interventional/surgical means.42,43
Adipose tissue is known to secrete pro-inflammatory cytokines such as IL-6 or TNF-α, thereby causing subclinical inflammation. Weight loss dampens these inflammatory pathways. 44
Estimating the anti-inflammatory potential of lifestyle optimisation
We estimate that more than a third of patients with high inflammatory burden could lower their hsCRP by lifestyle changes alone below 2 mg/L, the threshold commonly considered as raised or worthy of anti-inflammatory treatment. 4 This represents a relevant portion of CAD patients, offering a significant secondary prevention opportunity that is both cost-effective and largely free of side-effects. Considering that approximately three-quarters of the subgroup with an hsCRP≥2 mg/L display a BMI≥25 kg/m2, and that the association of BMI and hsCRP was the strongest of the studied risk factors, obesity seems the key variable for optimisation of inflammatory status.
Strengths and limitations
Strengths of the study are the use of a very contemporary and well-characterised CAD cohort, and the fact that multiple lifestyle risk factors were investigated simultaneously.
Some caveats should be considered: this study presents observational, cross-sectional data, meaning no causality between the observed associations can be deduced. Furthermore, we cannot rule out that these associations might have been confounded by unmeasured variables. The results from modelling the effect of risk factor optimisation on hsCRP levels should, therefore, be treated with particular caution. For this calculation, it was assumed that the associations between lifestyle risk factors and hsCRP could be translated into causal relationships with similar effect sizes. In light of the evidence from both longitudinal and numerous interventional studies demonstrating the impact of lifestyle changes on CRP, as well as plausible mechanistic considerations, this assumption seems reasonable.
A further point concerns the unspecific nature of CRP elevations. Although we took greatest care, there remains a risk that a small minority of patients were erroneously included in the analysis with, for example, undiagnosed infections or occult malignancy, thereby potentially confounding our results.
Conclusion
We demonstrate strong and independent associations between the presence and number of modRF and hsCRP levels in CAD patients. Therefore, comprehensive lifestyle risk factor control might represent an effective strategy to lower inflammatory burden, thus significantly impacting the target population for emerging anti-inflammatory compounds. This should be considered both in view of the cost and side-effects of pharmacological anti-inflammatory treatment and for the design of future clinical trials in this field.
Supplemental Material
CPR885458 Supplemental material - Supplemental material for Modifiable lifestyle risk factors and C-reactive protein in patients with coronary artery disease: Implications for an anti-inflammatory treatment target population
Supplemental material, CPR885458 Supplemental material for Modifiable lifestyle risk factors and C-reactive protein in patients with coronary artery disease: Implications for an anti-inflammatory treatment target population by Christopher Blaum, Fabian J Brunner, Friederike Kröger, Julian Braetz, Thiess Lorenz, Alina Goßling, Francisco Ojeda, Lukas Koester, Mahir Karakas, Tanja Zeller, Dirk Westermann, Renate Schnabel, Stefan Blankenberg, Moritz Seiffert and Christoph Waldeyer in European Journal of Preventive Cardiology
Footnotes
Author contribution
CB, FB, and CW contributed to the design and conception of the work. MK, TZ, DW, RS, SB, MS, and CW contributed to the conception and design of the study. CB, FB, FK, JB, TL, AG, FO, and LK contributed to the acquisition, analysis, or interpretation of data for the work. CB and FB drafted the manuscript. CW critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Acknowledgement
This work has not been presented previously. A poster presentation has been accepted for the ESC Congress 2019, Paris, France.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
