Abstract
Objective
Cardiac structure and function are important predictors for cardiovascular disease in adults. Not much is known about tracking of cardiac measures, other than left ventricular mass, from early life onwards. We examined whether and to what extent cardiac measures track from infancy into school-age.
Methods
We performed a population-based prospective cohort study among 1072 children. Aortic root diameter, left atrial diameter, left ventricular mass, relative wall thickness and fractional shortening were measured repeatedly by echocardiography. We explored tracking between infancy (1.5, six and 24 months) and school-age (six and 10 years).
Results
Of all cardiac measures, aortic root diameter, left atrial diameter and left ventricular mass were significantly correlated between infancy and school-age (
Conclusion
Our results suggest moderate tracking of structural cardiac measures from early infancy until school-age, which become stronger at older ages, but not of relative wall thickness or fractional shortening. Moderate tracking of cardiac structures suggests that cardiac structures are at least partly determined in early life.
Introduction
Cardiovascular disease is a major public health problem and seems to originate at least partly in early life.1,2 Common risk factors for cardiovascular disease, including blood pressure and lipid levels, track from childhood to adulthood.3,4 Tracking represents the maintaining of a given rank order relative to peers over time. 2 Previous studies have shown that left ventricular mass (LVM) tracks from childhood to adulthood.1,2 Longitudinal studies on tracking of LVM in children from the age of seven years until the age of 22 years show correlation coefficients in the range of 0.4 to 0.7. 1 Previously, we have reported that tracking of LVM is also present during the first two years of life. 5 Increased LVM is an independent predictor of cardiovascular disease and mortality in adults.6,7 Next to LVM, an increase in aortic root diameter (AOD) is associated with increased risk for heart failure, whereas an increase in left atrial diameter (LAD) is associated with cardiovascular events, such as stroke, and cardiovascular mortality in adults.8,9 The predictive value of increased LVM for cardiovascular events is higher when combined with information about relative wall thickness (RWT). 7 To the best of our knowledge, no previous studies have analysed tracking of these different cardiac structural and functional measures from infancy to childhood.
We hypothesize that structural and functional cardiac measures already track from infancy onwards. Therefore, we examined the extent of tracking from infancy into school-age in a population-based prospective cohort study among 1072 children followed from foetal life to the age of 10 years. We measured cardiac structure and function repeatedly with echocardiography at the ages of 1.5, six and 24 months, and six and 10 years. Measures included LVM, AOD, LAD, RWT and fractional shortening.
Methods
Design and study population
This study was embedded in the Generation R Study, a population-based, prospective cohort study from foetal life onwards in Rotterdam, The Netherlands. 10 All children were born between 2002 and 2006. Details of this study have been described previously. 10 Detailed cardiovascular measures were performed in a subgroup of 1106 Dutch children. 10 Of the total of 1079 live born singleton children, we excluded seven children from the analysis due to cardiac abnormalities (flowchart given in Supplemental Material Figure S1 online). Echocardiograms were successfully performed in 85%–95% of the participating children at the different ages, with 24 months being the least successful. Missing echocardiograms were mainly due to crying or unavailability of equipment or echocardiographer. Written informed consent was obtained from parents of participants. The study has been approved by the local Medical Ethics Committee.
Left cardiac structures until the age of 10 years
Two-dimensional M-mode echocardiograms were performed when the children were aged 1.5, six and 24 months and at the ages of six and 10 years in our dedicated research centre. We used methods recommended by the American Society of Echocardiography. 11 Intraobserver and interobserver intraclass correlation coefficients (ICCs) were calculated previously in 28 children with a median age of 7.5 years (interquartile range 3.0–11.0) and varied between intraobserver ICC 0.91 to 0.99 and interobserver ICC 0.78 to 0.96. 12 We measured AOD, LAD, left ventricular end diastolic diameter (LVEDD), left ventricular posterior wall thickness (LVPWT), and interventricular septum thickness (IVS) and calculated fractional shortening and LVM.11,13 To assess left ventricular concentricity, we calculated RWT as (2*LVPWT)/LVEDD. 14
To account for differing body sizes, we additionally standardized all cardiac outcomes on body surface area (BSA) using Generalized Additive Models for Location, Size and Shape using R, version 3.2.0 (R Core Team, Vienna, Austria). 15 These models enable flexible modelling, taking into account the distribution of the response variable. 16 Worm plots and Akaike Information Criterion were used in sensitivity analyses to obtain the best model fit. Weight and length were measured at the cardiac ultrasound. BSA was computed using the Haycock formula (BSA (m2) = 0.024265 × weight (kg)0.5378 × height (cm)0.3964. 15
Statistical analysis
First, we used one-way analysis of variance and Chi-square tests to compare childhood characteristics between boys and girls. Second, to examine whether children maintain their position in the distribution of the different cardiac structure measures, we estimated the Pearson’s correlation coefficients. AOD, LAD and LVM were standardized on BSA to account for differing body sizes. Since RWT and fractional shortening are ratios between cardiac measures and not dependent on BSA, we constructed standard deviation scores (SDSs) using this formula: (observed value-mean)/standard deviation. Third, we categorized the cardiac outcomes in quartiles and calculated the percentages of children that remained in the lowest or highest quartile between the measures at 1.5 months and 10 years. Finally, we performed conditional regression analyses to identify the independent associations of the cardiac measures at the different age windows with cardiac measures at age 10 years. We used standardized residuals obtained from regression of the cardiac structure measure at a specific age window on the previous measures.
17
These standardized residuals are independent from each other and can be used in a regression model together. The
Results
Structural and functional cardiac measures in boys and girls.
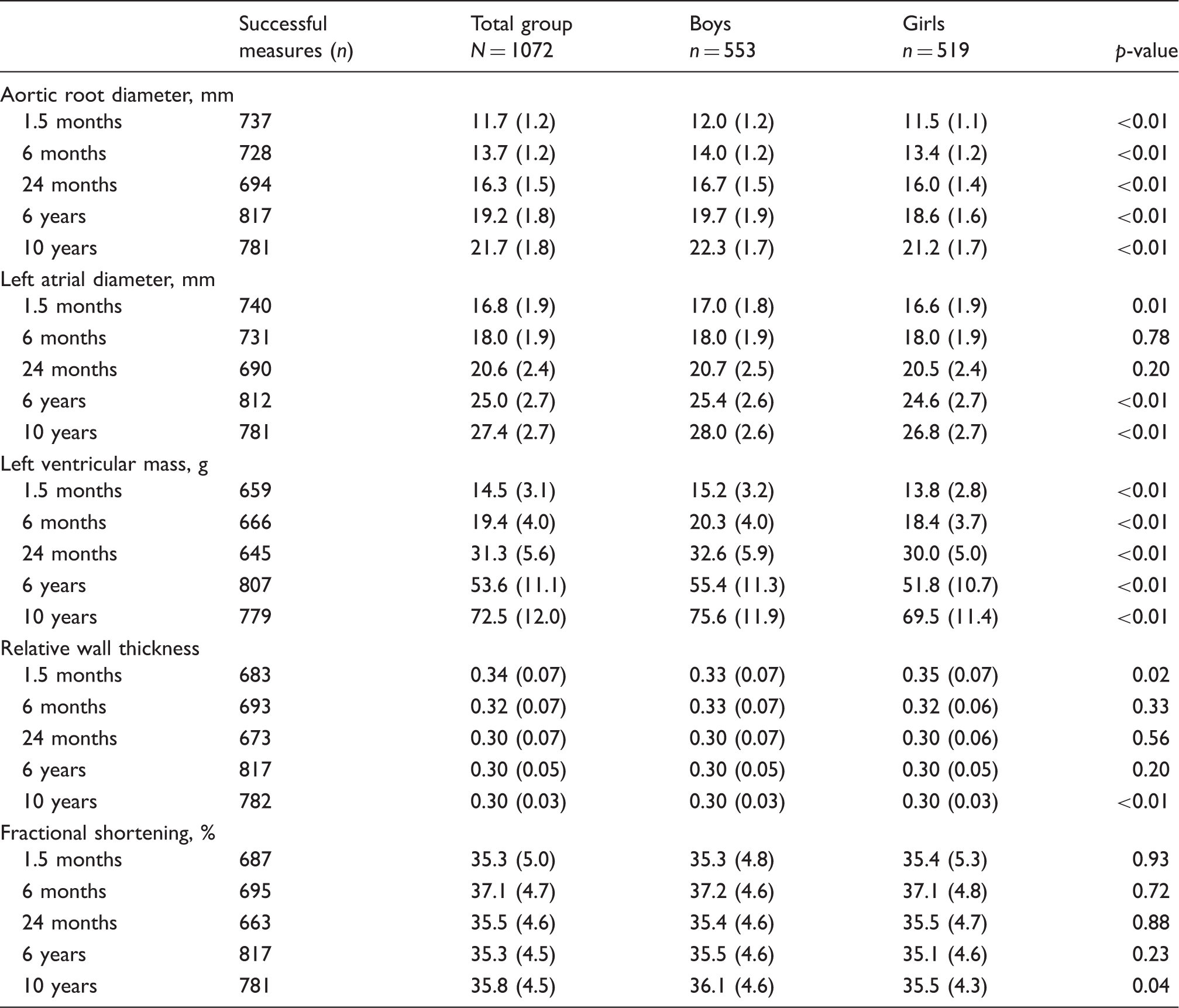
Values are means (SD).
Correlation tables of different age windows of structural and functional cardiac measures.
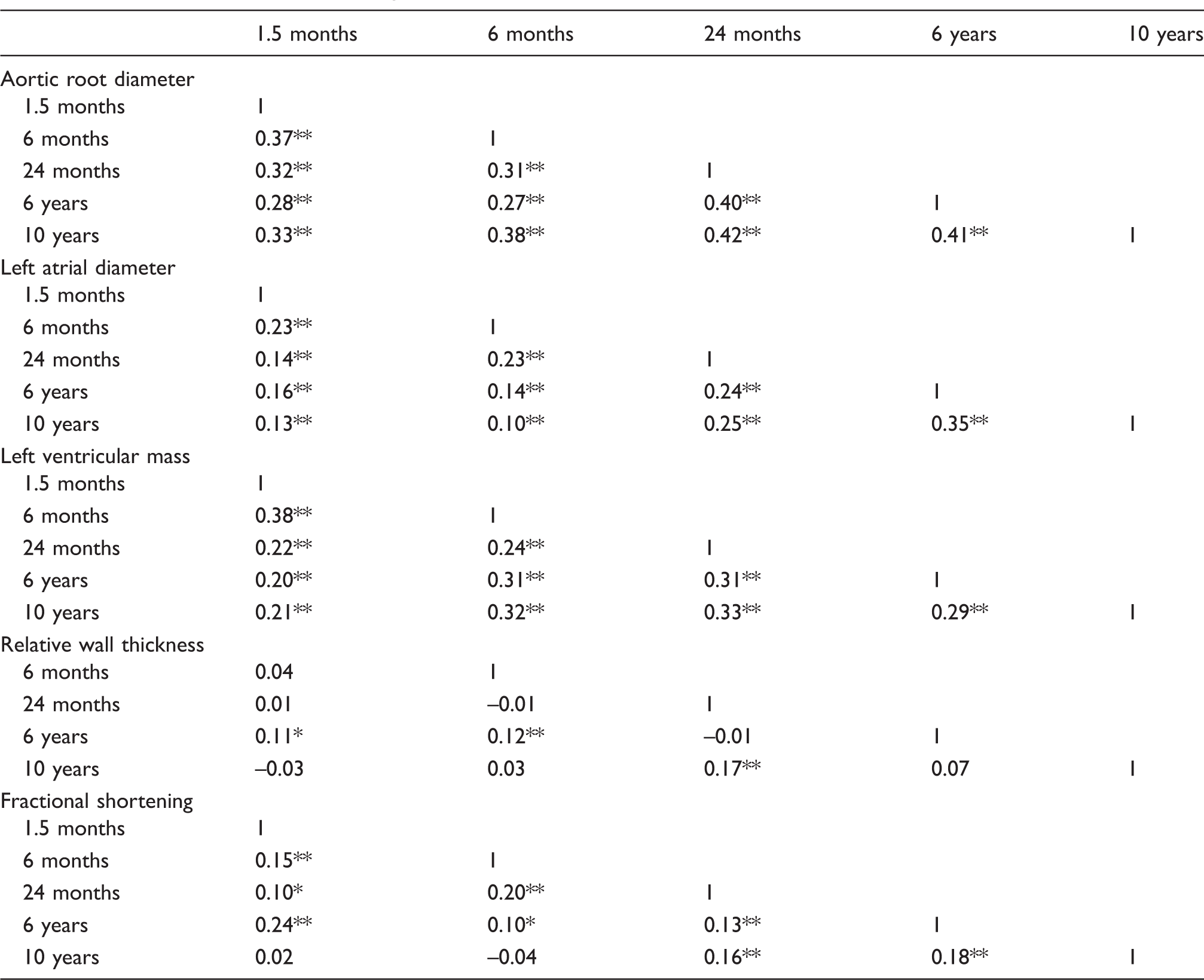
Numbers are Pearson’s correlation coefficients.
Figure 1 shows the distribution of children in quartiles of cardiac measures at the age of 10 years for the children who were in the lowest quartile of the measure at the age of 1.5 months, and for the children who were in the highest quartile at 1.5 months. Of the children who were in the lowest quartile of AOD at 1.5 months 36% remained in the lowest quartile at the age of 10 years, while 8% changed to the highest quartile. Of the children who were in the highest quartile at 1.5 months, 45% remained in the highest quartile, while 10% changed to the lowest quartile. AOD showed the strongest trend. The trends of children remaining in the lowest (30%) or highest (29%) LAD quartile and LVM quartiles (29% and 37%) from 1.5 months to 10 years were less clear. The distribution of RWT and fractional shortening did not show the same clear trend. Distribution for all infant quartiles is shown in Supplemental Material Table S2. Trends found for 24 months were stronger than those observed for 1.5 months (Supplemental material Figure S2).
Distribution of cardiac measures in school-age for children who were in the lowest or highest quartile at 1.5 months.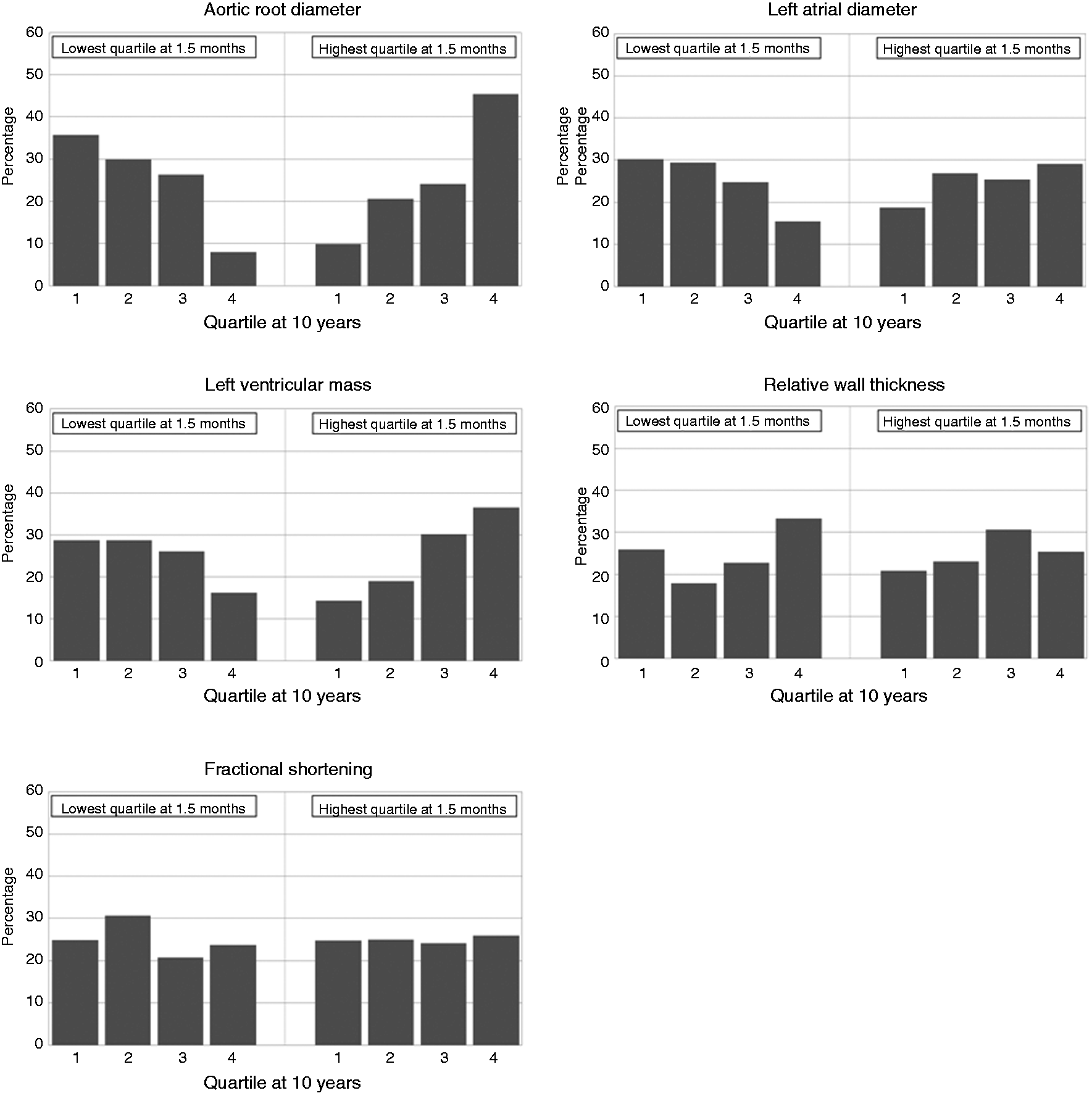
The results of the conditional regression analyses focused on identification of specific age windows for the cardiac outcomes at age 10 years did not show one clear age window for all outcomes (Supplemental Material Figure S3). AOD at 1.5 months had the strongest, independent association with AOD at 10 years. The other periods each had an additional, but less strong effect. The explained variability (
Discussion
In this population-based prospective cohort study, we observed moderate tracking of AOD, LAD and LVM between the ages of 1.5 months and 10 years. Around 30% of the children who were in the lowest or highest quartiles at the age of 1.5 months remained in the same quartile at the age of 10 years. Tracking was not consistently seen in RWT and fractional shortening.
Interpretation of main findings
Tracking can be defined as the stability of a child’s rank in a distribution over time.
18
Tracking of structural and functional cardiovascular measures suggests that cardiac structure originates at least partly in early life. Adverse cardiac structure in childhood could possibly place individuals at greater risk for cardiovascular disease in later life. Tracking can also be important for identifying individuals at risk for cardiovascular disease early in life.
18
Longitudinal studies have shown tracking of common risk factors for cardiovascular disease including blood pressure, lipid levels and LVM from childhood to adulthood.2–5 Tracking of cholesterol (
Previously, we reported moderate tracking of cardiac structures and function between the ages of 1.5 and 24 months in the same study group as in the current study. 5 Another study describes tracking of LVM in adolescents. This study followed 231 normotensive adolescents’ years and reported a tracking coefficient for LVM of 0.41 between the ages of 11 and 17 years. 2 In line with this study, we observed moderate tracking of LVM. In our study we observed slightly lower tracking coefficients of LVM than in the study on adolescents. This phenomenon has also been described in tracking of blood pressure.3,18 Baseline age was an important predictor of tracking of blood pressure, with stronger tracking in (late) adolescence than in childhood.3,18
To our knowledge, tracking of AOD, LAD, RWT and fractional shortening in children has not been studied before. In our study, we observed tracking of AOD and LAD, but we did not find consistent tracking of RWT and fractional shortening. Since AOD and LAD correlate with LVM, we expected these measures to track. Tracking of AOD was stronger than tracking of LAD and LVM. Echocardiography of LAD and LVM shows more intraobserver and interobserver variation than the measures on AOD. 12 Larger measurement error in repeated measures causes underestimation of the true tracking coefficients, which could explain the observed differences. 18 RWT is used in clinic as an extension to LVM to determine geometry of the heart. It represents the ratio between LVPWT and LVEDD, and both are dependent on growth. We would have expected that the ratio between these two measures would be constant in a healthy child and would show tracking. However, this was not the case. The same was observed for fractional shortening. This measure is the percentage change in cavity diameter. It is possible that there is very limited variation of these measures between persons in this relatively healthy population, and that there is a high variability of the repeated measurements in a participant, due to factors such as measurement error, heart rate variability and blood pressure variability. This within person variability could be large enough to obscure any possible real tracking. Also, the explained variability of the first four measures on RWT and fractional shortening at the age of 10 years was very low. This would indicate that not the measures at earlier ages, but other factors at the time of the measurement can explain the variability. Factors associated with RWT and fractional shortening could be BMI, exercise, heart rate and blood pressure. 19
Various factors may affect tracking of cardiac structures. In adults, cardiac remodelling is varied between the sexes. 20 However, even though a study in 231 adolescents found that boys have a larger LVM than girls, the degree of tracking was not influenced by sex. 2 The results are comparable to our results. We also found that boys have larger cardiac structures, but no differences in the degree of tracking. In childhood and adolescence, most variation in cardiac size can be explained by lean mass and not by cardiovascular risk factors.21,22 In our study, boys had a higher BSA and higher lean mass index than girls, which can explain the larger cardiac structures. 23
To determine the most important age window for cardiac tracking, we used conditional analyses. With these analyses, we could determine the effect of a measure at a given age on the measure at 10 years, independent of the effect of the measures at the other ages. We did not find one age window to be consistently more strongly correlated than the other age windows. Our results suggest that the measures at 24 months seem to be a stronger predictor in infancy for the measures at the age of 10 than do the measures at 1.5 months. This finding may reflect stability of cardiac structures after the first two years of life, or may reflect just a shorter time interval between the ages. However, we did not observe stronger correlations between six and 10 years.
The observed moderate tracking is important from an aetiological view point. It suggests that variation in cardiac structure partly originates in early life and might put individuals at risk for later cardiovascular disease. However, based on this research, we cannot determine whether tracking alone provides enough evidence to identify the individuals at risk in early life. More research and longer follow-up is needed to explain the variation in cardiac structure and to study whether this variation indeed leads to increased cardiovascular risk later in life, before predictive models can be created.
Study limitations
The main strength of this study is its population-based prospective study design starting from early foetal life. Also, we were able to perform echocardiography repeatedly in a large cohort of children over a time period of 10 years. Another strength is that we standardized the cardiac structural measures on BSA; this way we created SDSs that were independent from body size at the time of measuring. This ensured that we measured cardiac tracking, as opposed to tracking of linear growth in childhood, since cardiac structures in childhood are mainly dependent on body size. 21 This study was performed in a Dutch population, making it less generalizable to other ethnicities. A limitation of our study is that for each time point 15–25% of the children did not visit the research. Of the children who did visit, we could not obtain cardiac measures in 5–15% of the children. Missing values were because of the child being uncooperative at time of measure, or because of defective equipment or absent echocardiographer. However, we do not think these missing values lead to bias, because it is very unlikely that the correlation coefficients we found would be different in the children in whom we were not able to obtain cardiac measures. As mentioned previously, measurement error in repeated measures causes underestimation of the true tracking coefficients. 18 Since measurement error is more likely in the younger children, who have smaller hearts and are less cooperative, this could have underestimated the tracking coefficients we found within infancy and from infancy to childhood. Also, measurement inaccuracies of the ventricular diameter and wall thickness could increase measurement error in the calculated measures, such as LVM, RWT and fractional shortening. Studies on tracking of cardiac structure with more precise methods, such as cardiac magnetic resonance imaging or speckle-tracking echocardiography for cardiac function, could be an interesting addition to this research field. 24
Conclusion
Our study indicates that children who have a larger cardiac size measured by LVM, AOD and LAD compared with their peers in infancy are also more likely to have a larger cardiac size in school-age. The strongest period for tracking across infancy to school-age seems to be between the ages of 24 months and 10 years. Our results suggest moderate tracking of structural cardiac measures from early infancy until school-age, which become stronger at older ages, but not of fractional shortening or RWT. Moderate tracking of cardiac structures suggests that cardiac structures are at least partly determined in early life. Whether early cardiac structure and functional development predicts later life cardiac disease should be further studied.
Footnotes
Author contribution
LT, RG, LO, AH and VJ contributed to conception or design of the work. All authors contributed to acquisition, analysis or interpretation of data for the work. LT and VJ drafted the manuscript, and all authors critically revised the manuscript and gave final approval.
Acknowledgements
We gratefully acknowledge the contribution of participating mothers, general practitioners, hospitals, midwives and pharmacies in Rotterdam.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Generation R Study is made possible by financial support from the Erasmus Medical Centre, Rotterdam, the Erasmus University Rotterdam and the Netherlands Organization for Health Research and Development. Vincent Jaddoe received an additional grant from the Netherlands Organisation for Scientific Research (NWO-VIDI 016.136.361) and a Consolidator Grant from the European Research Council (ERC-2014-CoG-648916).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
