Abstract
Oral treprostinil has recently been shown to delay disease progression in patients with pulmonary arterial hypertension in a long-term outcomes study. The potential advantages of an oral formulation have resulted in patients transitioning from inhaled to oral treprostinil. The current study reports a retrospective analysis of patients who transitioned from treatment with inhaled to oral treprostinil. A multicenter retrospective chart review was conducted for 29 patients with pulmonary hypertension that transitioned from inhaled to oral treprostinil. Data were collected from inhaled treprostinil initiation and patients were followed until discontinuation of oral treprostinil or the end of the observation period. Persistence was calculated using Kaplan–Meier estimates. Prior to transition to oral treprostinil, patients had received inhaled treprostinil for a median of 643 (IQR: 322–991) days and 52% of patients were New York Heart Association/World Health Organization Functional Class III. For patients that cross-titrated between formulations, the median time to complete the cross titration was 24 (IQR: 1–57) days. At 16- and 24-weeks post-transition, oral treprostinil persistence was 86 and 76%, respectively. Persistence was 59% at 52 weeks post-transition. Clinical stability for the majority of patients at first follow-up post-transition was suggested based on available New York Heart Association/World Health Organization Functional Classification. Transitions from inhaled to oral treprostinil appeared safe and tolerable in the short-term. Additional prospective studies are needed to fully evaluate the safety and efficacy of transitions from inhaled to oral treprostinil.
Introduction
Pulmonary arterial hypertension (PAH) is a rare progressive disease that leads to right heart failure and often death. 1 While treatment options have expanded over the past 20 years to target three pharmacologic pathways, prostacyclins and prostacyclin analogues remain robust treatment options in the management of PAH.2–5 Current guidelines recommend prostacyclin analogues as initial treatment in patients with more severe and rapidly progressive disease (World Health Organization (WHO)-Functional Class (FC) III or IV) and in patients with persistent intermediate risk at subsequent evaluations as add-on therapy to existing treatment.6,7
Treprostinil is a prostacyclin analogue available in three different formulations with four routes of administration (subcutaneous, intravenous, inhaled, and oral). Each route of administration is associated with unique pharmacokinetics, dosing considerations, and adverse effects. 8 Clinicians may select an appropriate route of administration based on the patient’s clinical status, health literacy, quality of life considerations, and comorbidities. Considerations for transitioning patients from one treatment to another include patient preference, patient characteristics, disease severity, treatment complications (such as blood stream infections from intravenous administration), intolerable side effects (based on the route of administration), efforts to improve compliance by simplifying dosing, and pressure from external forces, such as third-party payers. 9
Inhaled treprostinil (Tyvaso; United Therapeutics Corp, Silver Spring, MD) was approved for the treatment of PAH in 2009 based on results of the TRIUMPH I study.10,11 In the 12-week, randomized, placebo-controlled TRIUMPH I study, the addition of inhaled treprostinil in patients already receiving PAH monotherapy resulted in a median 20 m (p < 0.001) improvement in six-minute walk distance (6MWD) compared with placebo.10,11 Subsequently, oral treprostinil (Orenitram; United Therapeutics Corp, Silver Spring, MD) was approved for treatment of PAH in 2013, based on results from the FREEDOM-M study.12,13 More recently, in the event-driven, FREEDOM-EV trial, oral treprostinil was shown to reduce the risk of the composite endpoint of clinical worsening by 25% compared to placebo (p = 0.039). 14 These results were largely driven by a delay in disease progression, with oral treprostinil reducing the risk of disease progression by 61% compared to placebo (HR = 0.39 (95% CI: 0.23–0.66); p = 0.0002). 14
The potential advantages of oral treprostinil over inhaled therapy include the ability to titrate with no maximum labeled dose and no device requirement. These theoretical benefits have resulted in some patients transitioning from inhaled treprostinil to oral treprostinil; however, limited data are available describing the transition. The largest retrospective study published to date included seven patients. 15 Remaining retrospective studies included one to two patients transitioning from inhaled to oral treprostinil, with limited findings on transition times, persistence, clinical follow-up, and adverse events (AEs).16–18
The current observational retrospective chart review was designed to characterize real-world transitions from inhaled to oral treprostinil by investigating the following: (1) details of the transition (reason, duration, dosing, and titration schedule), (2) oral treprostinil dosing and persistence on therapy, (3) observed efficacy assessments, and (4) concomitant medications and AEs. The aim of the study was to investigate and analyze real-world practices for transition between these two formulations of treprostinil, assess the safety and efficacy of oral treprostinil post-transition, and evaluate oral treprostinil dosing over time. The overarching goal is to provide a framework to safely and effectively transition from one treatment to another while maintaining patient stability.
Methods
Study design and patient population
This retrospective, observational, multicenter, chart review study was designed to characterize real-world transitions from treatment with inhaled to oral treprostinil. The study protocol was reviewed and approved by a central Institutional Review Board and delegated authority from each individual institution’s Institutional Review Board.
Medical records were assessed to determine patient eligibility. Eligible patients were using inhaled treprostinil for at least four weeks prior to the transition to oral treprostinil and were 18 years of age or older at the time of the transition. Patients intentionally treated with both inhaled and oral treprostinil (excluding those patients who received both drugs during the transition period) were ineligible for the study. Patients who had more than seven days between the complete discontinuation of therapy with inhaled treprostinil and the initiation of therapy with oral treprostinil were excluded from the study.
Following confirmation of eligibility, a retrospective chart review was performed for patients who transitioned from inhaled to oral treprostinil between June 2014 and August 2017. Data collection began with the date of the first known dose of inhaled treprostinil therapy. The Inhaled Treprostinil Maintenance Dosing Period began once a stable dose was achieved and continued until the last known stable dose of inhaled treprostinil prior to either the start of down titration or switch to oral treprostinil. Baseline data were abstracted while a patient was on inhaled treprostinil and reflected the most recent available clinical data prior to the initiation of oral treprostinil. The transition period began with the initial dose of oral treprostinil and concluded at the final dose of inhaled treprostinil. Transitions were categorized as direct transitions or cross titrations. Cross titrations received both treatments, decreasing the inhaled treprostinil dosage while increasing oral treprostinil over time. Direct transitions switched directly from inhaled to oral therapy with no treatment overlap and were not considered to have a transition period. The time of duration of oral treprostinil commenced post-transition and concluded at the final dose of oral treprostinil or the last time point of data collection (End of Study). See Fig. 1 for a summary of the transition timeline. Transition schedules and methodology were at the discretion of the prescribing physician due to the real-world nature of this chart review.
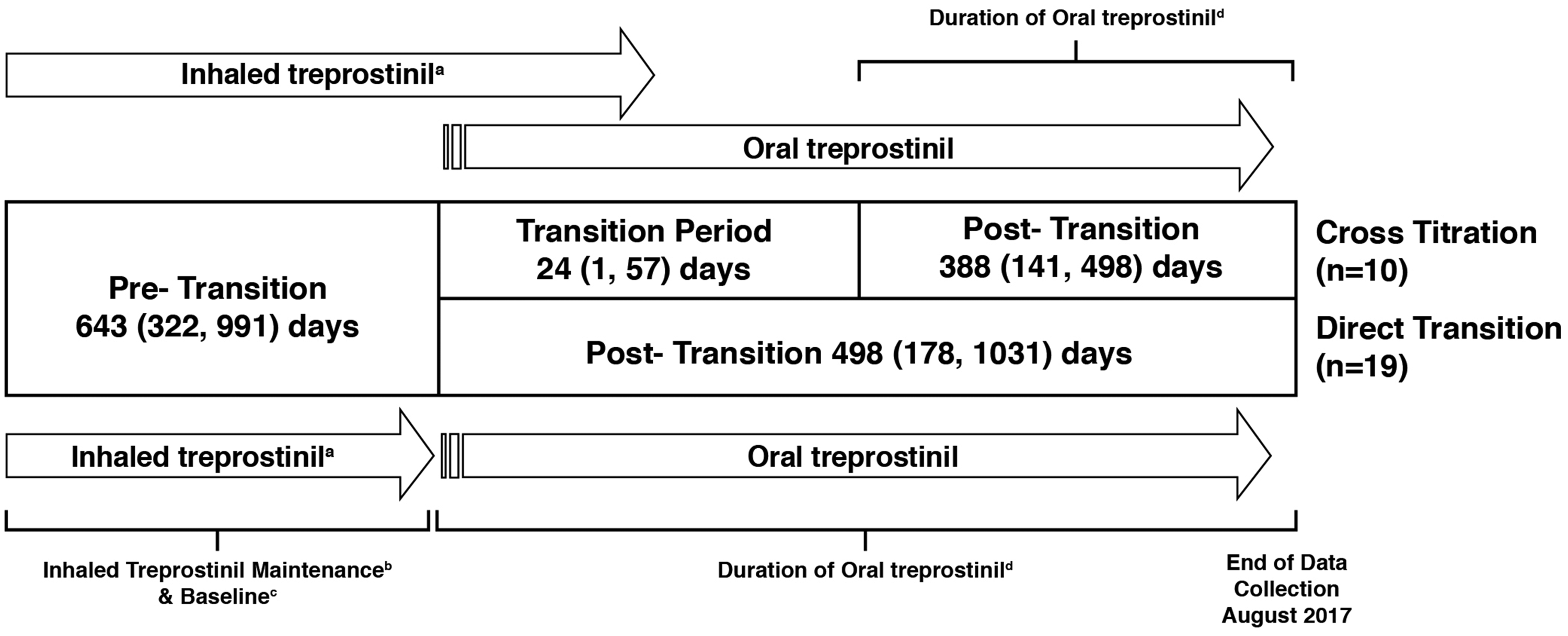
Study timeline. Times represented are median (IQR). aInhaled treprostinil initiation was the first known start date and dose of inhaled treprostinil therapy. bInhaled treprostinil maintenance included the first to last date of stable, chronic inhaled treprostinil dose prior to any down-titration or transition to oral treprostinil. cBaseline was the date of the last known stable, chronic inhaled treprostinil dose prior to any down titration or transition to oral treprostinil. dEnd of study was the date of oral treprostinil discontinuation or the last date of data collection.
Data abstracted
Patient demographic information, clinical characteristics, and inhaled treprostinil therapy history were collected as available at baseline. Transition assessments, including details regarding the transition (reason, time period, and strategies used) and dosing information for inhaled and oral treprostinil, were collected. Further dosing information for oral treprostinil following the transition until the end of the study was also included.
Information regarding FDA-approved PAH therapy and supportive therapy for pulmonary hypertension were collected from baseline through the end of study as available. AEs of interest and serious adverse events (SAEs) were also recorded. AEs of interest were defined as those possibly attributable to inhaled treprostinil and/or oral treprostinil therapy, as assessed by the investigator, beginning four weeks before baseline through study end, which also required treatment or intervention, resulted in the inability to titrate therapy, dose reduction, or therapy discontinuation. The incidence of AEs of interest and SAEs were retrospectively collected for all patients, coded using the Medical Dictionary for Regulatory Activities (MedDRA) current at the end of the study, and summarized by MedDRA primary system organ class and preferred term.
Available efficacy assessments were collected from baseline through the end of the study and included New York Heart Association (NYHA)/WHO-FC, exercise capacity as assessed by the six-minute walk test, hemodynamic measurements, echocardiographic assessments, and laboratory assessments, as available in the medical record.
Statistical analyses
All efficacy and safety data were summarized with descriptive statistics. No adjustments were made for missing data. Persistence on oral treprostinil therapy was evaluated by Kaplan–Meier analysis. Data from two patients were censored prior to one-year post-transition, as the data collection time period had ceased and the patients were still receiving treatment with oral treprostinil. A Cox proportional hazards model was constructed to identify predictors of persistence. Persistent use was calculated using Kaplan–Meier estimates starting at the first time point post-transition.
Results
Baseline characteristics during inhaled treprostinil therapy
Twenty-nine eligible patients on inhaled treprostinil from four centers in the United States were included in the data analyses (Table 1). All patients (n = 29) were determined to have WHO Group 1 PAH at baseline. The patient cohort had a mean age of 59.6 years and was comprised of predominantly women (76%). Thirty-one percent (n = 9) of patients had idiopathic or heritable PAH and 69% (n = 20) had associated PAH (connective tissue disease, human immunodeficiency virus, congenital heart disease, etc.). The mean time since diagnosis at the time of oral treprostinil initiation was 4.5 (0.7–18.2) years.
Baseline characteristics.
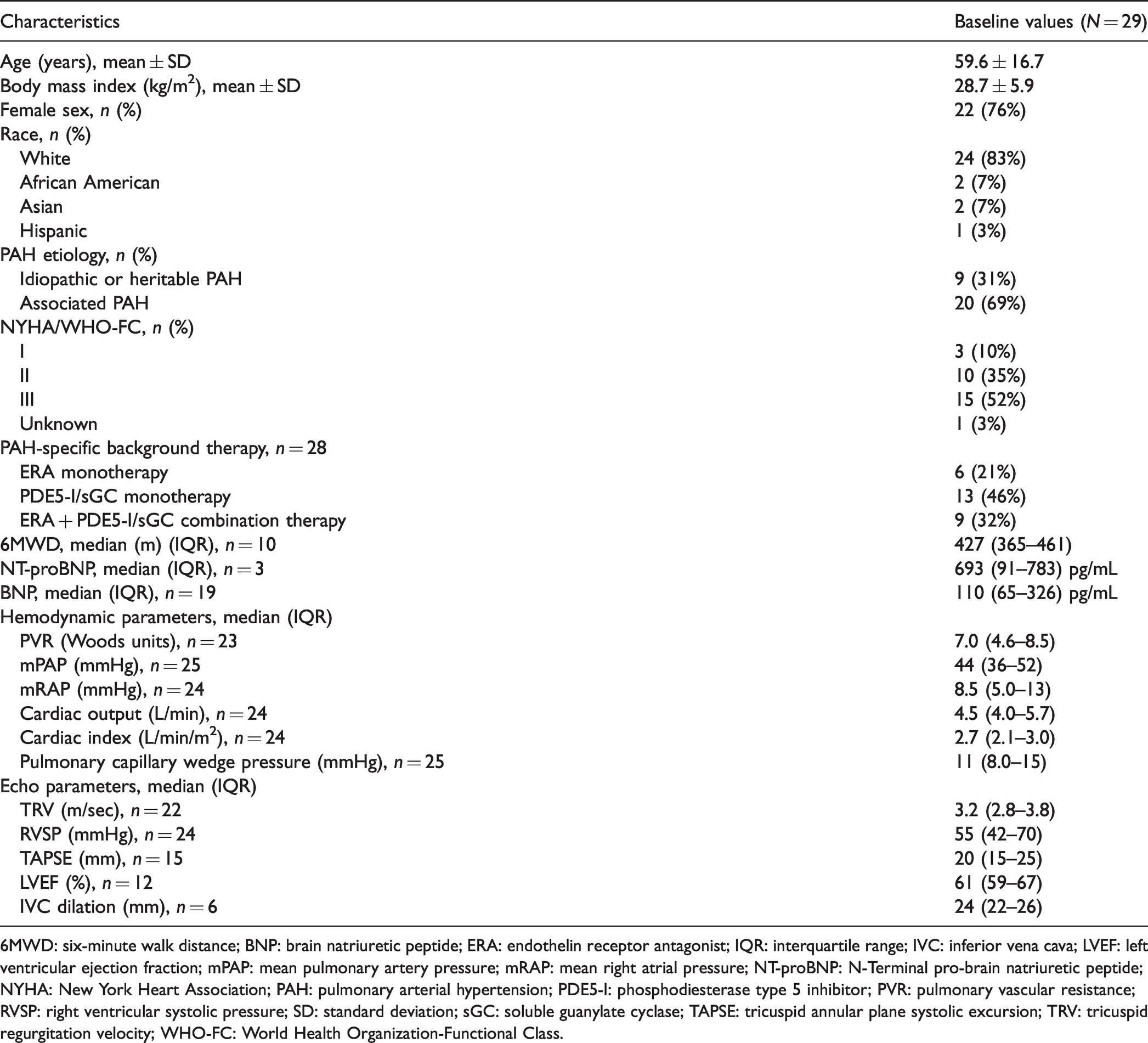
6MWD: six-minute walk distance; BNP: brain natriuretic peptide; ERA: endothelin receptor antagonist; IQR: interquartile range; IVC: inferior vena cava; LVEF: left ventricular ejection fraction; mPAP: mean pulmonary artery pressure; mRAP: mean right atrial pressure; NT-proBNP: N-Terminal pro-brain natriuretic peptide; NYHA: New York Heart Association; PAH: pulmonary arterial hypertension; PDE5-I: phosphodiesterase type 5 inhibitor; PVR: pulmonary vascular resistance; RVSP: right ventricular systolic pressure; SD: standard deviation; sGC: soluble guanylate cyclase; TAPSE: tricuspid annular plane systolic excursion; TRV: tricuspid regurgitation velocity; WHO-FC: World Health Organization-Functional Class.
Baseline characteristics were collected prior to transitioning to oral treprostinil but while on stable doses of inhaled treprostinil (Table 1). Baseline NYHA/WHO-FC of patients at the time of the transition was I (10%), II (35%), and III (52%). For 1 patient, the functional class was unknown. A majority of patients were on PAH-specific background therapy, including endothelin receptor antagonist (ERA) monotherapy (21%), phosphodiesterase type 5 inhibitor (PDE5-I)/soluble guanylate cyclase (sGC) stimulator monotherapy (46%), and ERA and PDE5-I/sGC stimulator combination therapy (32%). Prior to transitioning, available baseline clinical characteristics included a median 6MWD of 427 m, median pulmonary vascular resistance of 7.0 Wood units, median mean pulmonary arterial pressure of 44 mmHg, median mean right atrial pressure of 8.5 mmHg, median cardiac output of 4.5 L/min, median cardiac index of 2.7 L/min/m2, and median pulmonary capillary wedge pressure of 11 mmHg (Table 1).
Transition to oral treprostinil
Details regarding transition characteristics are provided in Table 2. The median time on inhaled treprostinil prior to the transition was 643 (IQR: 322–991) days and the mean dose was 11.8 (8–18) breaths per session four times daily (QID) for the 29 eligible patients (Table 2). The primary reason for the transition was intolerance to inhaled therapy or AE (62%), followed by worsening PAH (14%), other reasons (10%), patient preference (10%), and noncompliance (3%). Most patients (79%) completed the transition at their home while the remaining patients completed the transition in the hospital setting. A majority (62%) of patients transitioned within six months of oral treprostinil becoming commercially available and 76% transitioned within one year of availability.
Transition characteristics.

PH: pulmonary hypertension; IQR: interquartile range; SD: standard deviation.
aOther reasons included unclear, elevated pulmonary arterial pressure, switch to solo drug therapy, or higher prostacyclin dose needed.
One-third (34%) of patients cross-titrated (i.e. received both inhaled and oral treprostinil during the transition), with 66% directly transitioning from inhaled to oral treprostinil. The median time for patients that completed cross titration was 24 (IQR: 1–57) days. The mean total daily dose (TDD) of oral treprostinil at the start of the transition was 2.5 mg; patients who directly transitioned, not surprisingly, started with a higher mean TDD of oral treprostinil (3.3 mg) than patients who cross-titrated (1.1 mg). For patients who cross-titrated, the mean oral treprostinil dose at the end of the transition period was 3.6 mg/day. The mean TDD of oral treprostinil at 24 weeks post-transition was 5.3 mg (1.5–9) for patients that directly transitioned and 8.0 mg (3.4–18) for patients that cross-titrated. For all patients, the mean TDD was 5.6 mg (1.5–12.5), 6.2 mg (1.5–18), and 8.7 mg (2.1–18) at 16, 24, and 52 weeks, respectively, following the end of the transition period. TDD from time of oral treprostinil initiation is shown in Fig. 2.
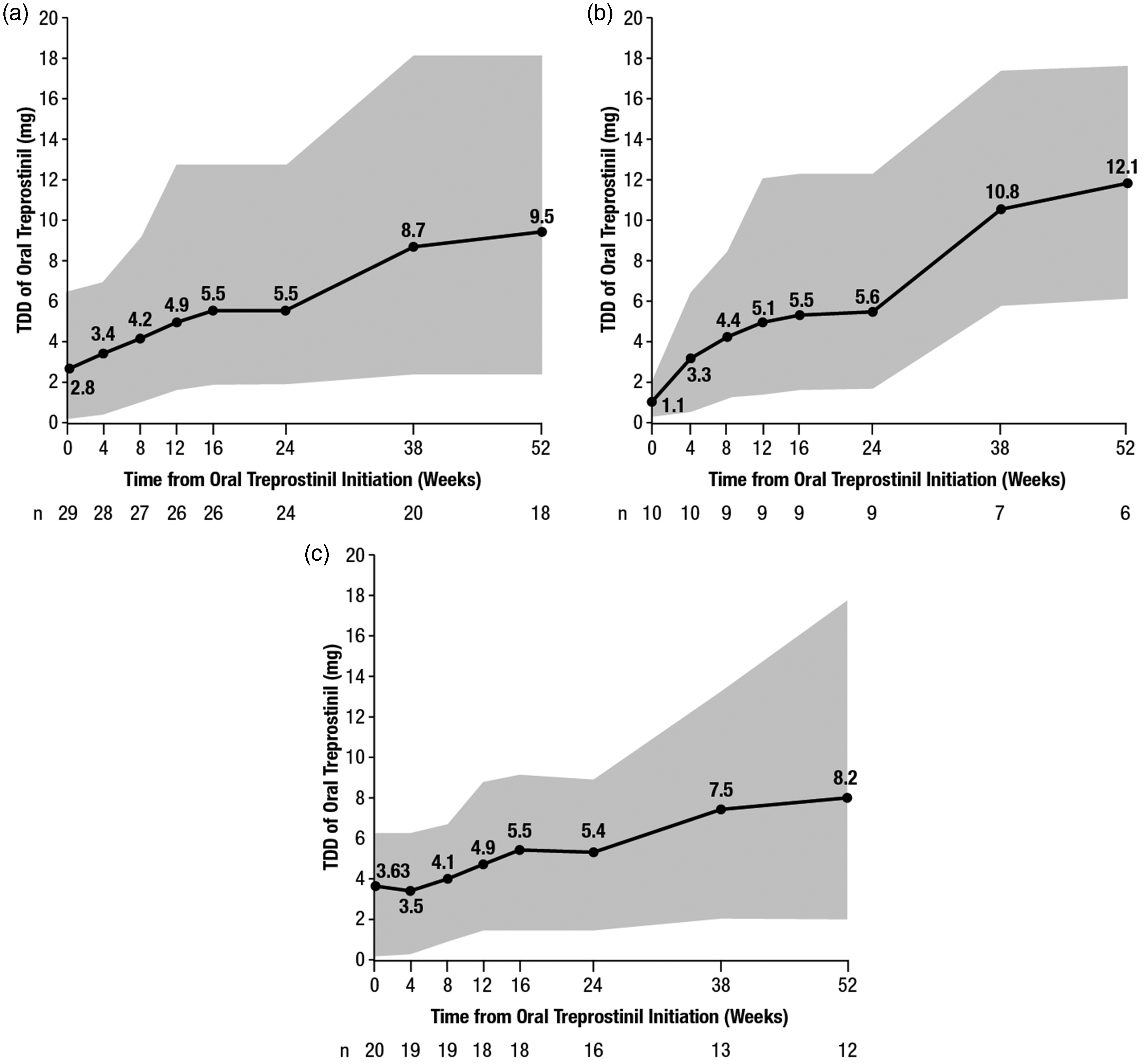
Total Daily Dose (TDD) of oral treprositinil from time of oral treprostinil initiation. (A) Total population, (B) cross-titrated, and (C) Direct transition. Mean TDD is indicated in black. Gray area indicates upper and lower dose ranges.
The approach to the follow-up outcome assessment was not consistent across sites and has limited the value of these measures to inform efficacy. The exception was NYHA/WHO-FC which was more widely collected. Post-transition NYHA/WHO-FC assessment was available for 76% (n = 22) of patients. The majority of patients with available data (59%, n = 13) had no change in their NYHA/WHO-FC at first follow-up post-transition (Fig. 3). Improvements were seen in 32% of patients (n = 7), whereas worsening was seen in 9% of patients (n = 2) at first follow-up post-transition.
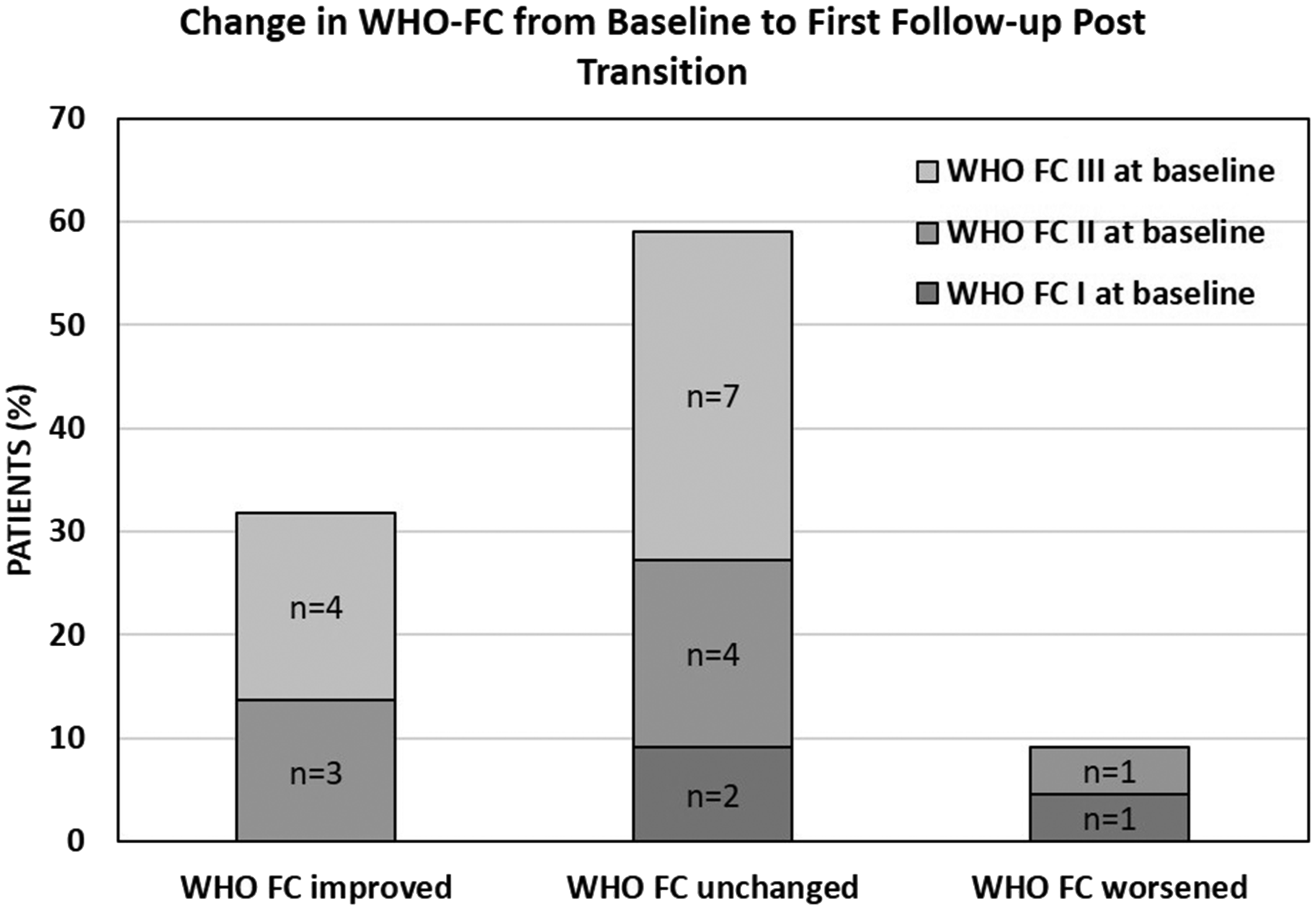
Change in New York Heart Association (NYHA)/World Health Organization (WHO)-Functional Classification (FC) from baseline to first follow-up post-transition. The first NYHA/WHO-FC follow-up post-transition ranged from two to seven months post-transition completion.
Safety and tolerability
AEs potentially related to treatment with inhaled or oral treprostinil were experienced by 83% of patients overall (up to 485 days post-transition); 55% of patients experienced AEs prior to and up to the end of the transition period and 69% of patients experienced AEs on or after Day 1 of the post-transition period (Table 3). Cough was the most frequently experienced AE overall and prior to and up to the end of the transition period (45 and 45% of patients, respectively); however, cough resolved on or following post-transition Day 1. AEs experienced after transition to oral treprostinil were consistent with known AEs and the most common AE on or after post-transition Day 1 was headache (35%).
Adverse events (> 5% of total population).
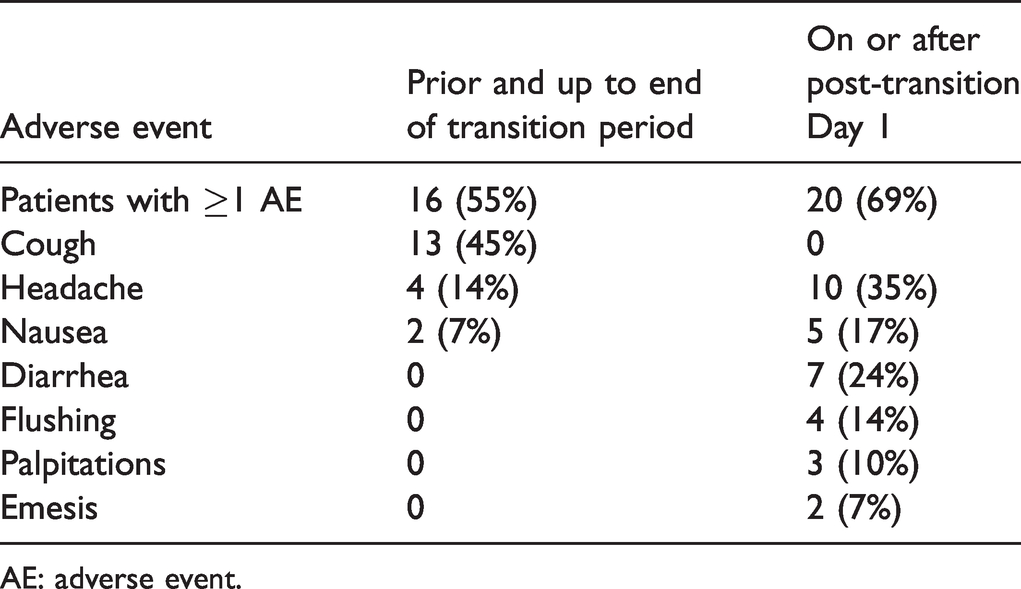
AE: adverse event.
Persistence after transition
Patients were followed on treatment with oral treprostinil for a median of 443 (IQR: 178–1001) days post-transition until discontinuation of oral treprostinil or end of study observation, whichever occurred first. Persistence was defined as patients who remained on oral treprostinil continuously following the end of the transition period; Kaplan–Meier estimates were 86, 79, and 65% for Weeks 16, 24, and 52 post-transition, respectively (Fig. 4). Data from two patients were censored (141 days and 359 days), as the data collection period had completed prior to the Week 52 time point while they were still receiving treatment with oral treprostinil. Twelve patients (41%) continued oral treprostinil use past the study end date; therefore, the duration on oral treprostinil was artificially shortened and difficult to compare with the length of time that these patients were on inhaled treprostinil. Cox proportional hazards did not suggest that age, gender, body mass index, NYHA/WHO-FC, background therapy, time on therapy with inhaled treprostinil, or type of switch were associated with the durability of oral treprostinil use to a year or longer.
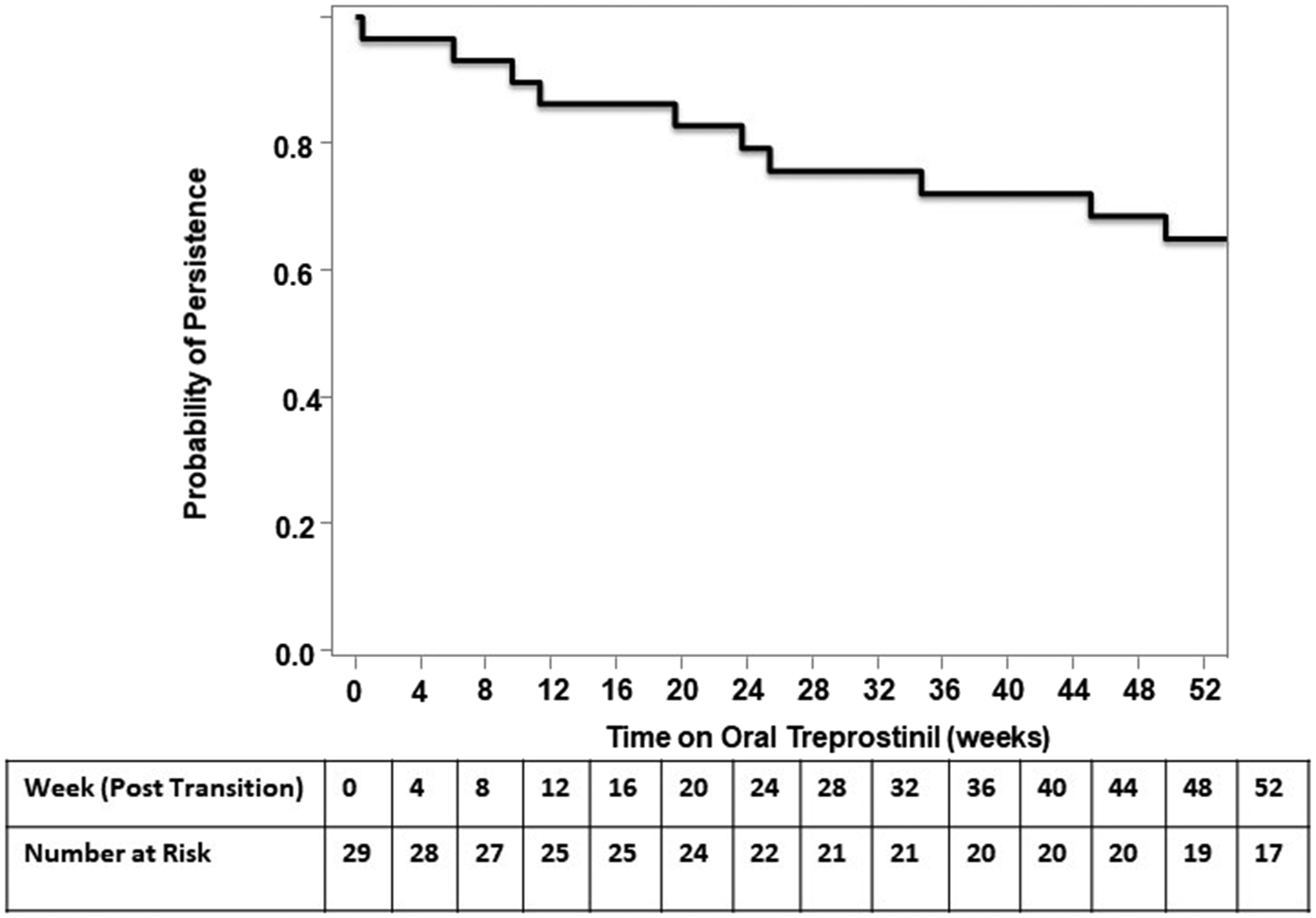
Kaplan–Meier estimate of persistence on oral treprostinil therapy post-transition. Data from two patients remaining on oral treprostinil at the end of the data collection time period were censored.
Reasons for discontinuation of oral treprostinil are summarized in Table 4. During the data collection period, 17 patients (59%) discontinued treatment with oral treprostinil, of which 10 patients discontinued within one-year post-transition. The most common reasons for discontinuation (patients may have had more than one reason for discontinuation) were AEs associated with oral treprostinil or initiation of alternative prostacyclin class therapy (six patients each (21%) during the data collection period, including four patients each (14%) within one-year post-transition). Four of the patients who initiated alternative therapies transitioned to parenteral prostacyclin. Notably, one patient (3%) discontinued treatment to initiate end-of-life care. Four patients discontinued oral treprostinil therapy due to evidence of restrictive cardiomyopathy. Analyses excluding these four patients did not show meaningful differences in baseline characteristics (Table s1), transition characteristics (Table s2), follow-up parameters collected (Table s3), and persistence (Figure s1).
Overall reasons for oral treprostinil discontinuation during the observation period.

AE: adverse event; IV: intravenous; SC: subcutaneous.
Discussion
This retrospective study examined the characterization of patients prior, during, and following transition from inhaled to oral treprostinil. Most patients transitioned directly from inhaled to oral treprostinil without cross titration or taper of either product. The vast majority (28/29) patients transitioned to three times a day oral therapy. Oral treprostinil doses achieved at Week 24 post-transition were modest (mean TDD 6.2 mg); however, patients who employed a cross titration technique achieved higher doses compared with those who had direct titration. The achieved TDD is in line with initial oral treprostinil studies, which may have influenced targets for clinician dosing.13,19,20 In comparison, titration of oral treprostinil in our data set was relatively modest compared to recent clinical trials; the median TDD dose achieved at Week 24 in this cohort of patients was lower than the median TDD achieved in FREEDOM-EV at Week 24 of approximately 10.68 mg TDD. 14
Oral therapy was moderately well tolerated, demonstrated by 86, 79, and 65% persisting at 16, 24, and 52 weeks, respectively, following transition. This result should be taken in context with the fact that patients in this analysis were receiving inhaled treprostinil for a median of almost two years prior to transitioning to oral treprostinil and 52% of patients were NYHA/WHO-FC III prior to initiating oral treprostinil. Other factors that may have increased the frequency of post-transition discontinuations included one patient who initiated end-of-life care and four who were ultimately diagnosed with restrictive cardiomyopathy. Clinical stability at first follow-up may be suggested based on stability in NYHA/WHO-FC data seen in the subset of the cohort with baseline and follow-up measures.
This study suggests that one of the primary reasons that patients transition from inhaled to oral therapy is because of AEs associated with the route of administration. 8 Treatment with inhaled treprostinil requires inhalation QID at equally spaced intervals (approximately four hours) via an ultrasonic nebulizer and may be associated with adverse effects such as cough, throat irritation, and pharyngolaryngeal pain related to the inhalation procedure. 21 In the TRIUMPH-1 study with inhaled treprostinil, the most common AEs were cough, headache, throat irritation/pharyngolaryngeal pain, nausea, flushing, and syncope. 10 We also found that the most common reported AE prior to and up to the end of the transition was cough (45% of patients) and AEs or intolerance to inhaled treprostinil were listed as a transition reason for 62% of patients. It is important to note that despite this, patients were on inhaled therapy for a median of almost two years. Although quite speculative, this may indicate a reluctance to transition to parenteral therapy, which was the only other option at the time. However, on or after post-transition Day 1 to oral treprostinil, cough was no longer reported.
AEs reported in patients after the transition to oral therapy were consistent with the most common AEs associated with oral treprostinil including headache, diarrhea, nausea, vomiting, flushing, pain in jaw, pain in extremity, hypokalemia, and abdominal discomfort. 12 Unlike cough, which improved following the transition, these AEs appeared to be more common following the transition. In this study, the most common reported AEs with oral treprostinil were consistent with those observed in the FREEDOM-M study. 13 The ability to confidently assess changes in the frequency, severity, or pattern of AEs following transition from inhaled to oral treprostinil was limited due to the retrospective nature of this current study. This is because there was no predefined time point or procedures for collection of events, which limited the available data for analysis.
The transition of patients with PAH from inhaled treprostinil to oral prostacyclin-class therapy has been examined in a recent prospective study, 22 which investigated the transition from inhaled treprostinil to oral selexipag, a prostacyclin receptor agonist. Of the 34 patients enrolled, 32 (94%) continued to receive selexipag at 16 weeks after initiating the transition which included a median time of 6.2 (1–14.9) weeks to complete down-titration of inhaled treprostinil and 28 (82%) met the predefined criteria for a successful transition. The mean dose of inhaled treprostinil was 59.3 ± 10.0 µg, approximately 9.9 breaths QID, and the median selexipag dose at Week 16 was 1200 (400–1600) µg twice a day. The conclusions of the study were that transition from inhaled treprostinil to oral prostacyclin (IP)-receptor agonist therapy was generally successful and safe, with comparable clinical outcomes of WHO-FC and 6MWD. 22 Remaining investigations of inhaled to oral prostacyclin-class therapy transitions are restricted to limited case studies.16,17 Other investigations of inhaled to oral prostacyclin-class therapy transitions have been mostly limited to small retrospective case studies with limited comparison value.15–18
Results from our retrospective study are consistent with those observed in the prospective TRANSIT-1 study, which transitioned patients with PAH from inhaled treprostinil to an oral IP-receptor agonist. 22 The sample sizes were similar in both studies as was the pattern of AEs, with the most common events after the transition being headache and gastrointestinal events. The mean dose of inhaled treprostinil prior to transition was higher in the present study, 11.8 breaths QID compared to 9.9 breaths QID in the TRANSIT-1 study. 22 Notably, the transition was different in the two studies, as a cross titration over 12 weeks was used in the TRANSIT-1 study, whereas the majority of patients had a direct transition in this real-world observational study. In addition, the majority of patients with available data in both studies had no change in their NYHA/WHO-FC at follow-up (67.6% in TRANSIT-1 and 59.1% in this study). Persistence on selexipag at 16 weeks post-initiation, which included a median time of 6.2 weeks to down-titrate inhaled treprostinil in the TRANSIT-1 study, was similar to persistence rates on oral treprostinil at Week 16 in the present study, which excluded transition time. However, long-term data beyond Week 16 were not reported in the TRANSIT-1 study. 22 In a related real-world, retrospective study of specialty pharmacy data of primarily FC III and IV patients who transitioned from oral to inhaled treprostinil, persistence on inhaled treprostinil was 87% (13 of 15) at 12 weeks and 80% (12 of 15) at 24 weeks. 23 While this transition was in the opposite direction (oral to inhaled) when compared to our study, the persistence at these time points were similar. Alongside the TRANSIT-1 data, this may suggest similar persistence of use when transitioning between non-parenteral forms of prostacyclin-class therapies.
Data from a retrospective study examining the transition from inhaled to oral treprostinil based on pharmacy records from 275 patients who had oral treprostinil dispensed 60 days or less after a prescription for inhaled treprostinil have previously been presented. 24 Although there was a trend toward prescribing higher initial oral treprostinil doses for patients receiving higher inhaled treprostinil doses, there was no correlation between prior duration or dose of inhaled treprostinil and initial dose of oral treprostinil.
The limitations of the current study are that a small number of patients were included from four centers with the majority of patients (69%) coming from a single center. It is noteworthy that over half the patients had advanced disease as suggested by their NYHA/WHO-FC and yet were transitioning from inhaled to oral treprostinil after nearly two years on inhaled therapy. It might be expected that such a patient population would instead transition to parenteral treprostinil at this juncture. This may suggest a selection bias reflecting a group of patients who may have had socioeconomic barriers or were particularly resistant to transitioning to parenteral therapy, which may make them less representative of PAH patients at-large. The inference on clinical outcomes after transition in this retrospective study was limited by the lack of paired data before and after transition for many important outcome measures which likely reflects real-world clinical practice variation. Although the functional class data suggests stability, these important limitations could be overcome with a well-designed prospective study that could compare risk scores or other clinical outcomes.
Although our study gave general context to transition reasons, dosing, AEs, persistence, and NYHA/WHO-FC outcomes, several important questions remain. For instance, although most patients underwent a direct transition, there was substantial variation in the transition approach. Although there is some suggestion that patients who underwent cross titration had a higher dose at the end of the transition, it is unknown whether the transition approach or patients’ clinical status influences long-term clinical outcomes, achieved dose of oral therapy, or persistence of oral treprostinil therapy after transitioning from inhaled treprostinil. These questions have even greater importance in the current treatment environment where multiple inhaled, oral, and parenteral prostacyclin options exist.
In conclusion, this retrospective study provides the first multicenter, published real-world characterization of transitions from inhaled to oral treprostinil, suggesting that transition can be completed directly or via cross titration. Despite the limitations of the study, the data presented suggest that patients can be safely and effectively transitioned from treatment with inhaled treprostinil to oral treprostinil. Additional prospective studies are needed to fully evaluate the safety and efficacy of transition from inhaled to oral treprostinil and better define best practices.
Supplemental Material
sj-pdf-1-pul-10.1177_2045894021998203 - Supplemental material for A multicenter retrospective study of patients with pulmonary hypertension transitioned from inhaled to oral treprostinil
Supplemental material, sj-pdf-1-pul-10.1177_2045894021998203 for A multicenter retrospective study of patients with pulmonary hypertension transitioned from inhaled to oral treprostinil by Diane L. Zwicke, Ricardo Restrepo-Jaramillo, Hassan Alnuaimat, Kathryn Gordon, Meredith Broderick, Lisa D. Edwards, Andrew Allmon and Peter J. Leary in Pulmonary Circulation
Footnotes
Ethical approval
This study was approved by an Institutional Review Board.
Guarantor
Dr Diane L. Zwicke
Contributorship
United Therapeutics Corporation participated in the design of the study, data analyses, interpretation, and preparation of the manuscript. D.L.Z., R.R.-J., P.J.L., and H.A. collected data, participated in the preparation of the manuscript, and offered critical feedback. M.B. and K.G. participated in the design of the study, interpretation, and preparation of the manuscript. L.D.E. and A.A. performed statistical analyses.
Conflict of interest
M.B., K.G., and L.D.E. are employees of United Therapeutics Corporation. P.J.L. has received grant support from the NHLBI, AHA, and Chest foundation. He is also the site PI for industry-sponsored trials and/or registries by Actelion, United Therapeutics, and Bayer. R.R.-J. has received research grants from United Therapeutics Corporation and Johnson and Johnson Actelion Pharmaceuticals Ltd along with personal fees from United Therapeutics Corporation, Johnson and Johnson Actelion Pharmaceuticals Ltd, and Bayer for participation in speaker bureaus and advisory boards. D.L.Z. is the site PI for industry sponsored studies by United Therapeutics Corporation and has received personal fees from United Therapeutics Corporation for consulting and participation in speaker bureaus and advisory boards.
Acknowledgements
Medical writing support was provided by Carol L. Sable, PhD and Natalie Patzlaff, PhD.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by United Therapeutics Corporation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
