Abstract
Guidelines for the treatment of pulmonary arterial hypertension (PAH) recommend sequential add-on therapy for patients who deteriorate or fail to improve clinically. However, it is not known whether these patients also benefit from transitioning from inhaled prostacyclins to parenteral prostacyclins. We sought to characterize PAH patients receiving inhaled treprostinil who were transitioned to parenteral treprostinil. We conducted a multicenter retrospective study at 7 PAH centers and collected reasons, methods, safety, and outcome of patients transitioned from inhaled treprostinil to parenteral treprostinil. Twenty-six patients with pulmonary hypertension in group 1, 4, or 5 transitioned from inhaled treprostinil to parenteral treprostinil (10 intravenous, 16 subcutaneous). Twenty-four patients were also on one or two oral therapies. Reasons for transition were clinical deterioration, lack of clinical improvement, and pregnancy (19, 6, and 1 patients, respectively). Transitions occurred in hospital, clinic, or home (17, 7, and 2 patients, respectively). Parenteral infusion was started after the last inhaled treatment at maintenance dose (13 patients), after the inhaled therapy was downtitrated to 18 μg (6 patients), or with an overlap of inhaled downtitration with parenteral uptitration (7 patients). The transition was safe; side effects included symptoms of prostacyclin overdose. Patients were followed for 3–18 months. At 3 months, 8 patients improved, 17 maintained their functional class, and 1 continued to deteriorate. In conclusion, selected PAH patients can be safely transitioned from inhaled treprostinil to parenteral treprostinil using a variety of methodologies in different settings with the expectation that patients will improve or at least remain clinically stable.
INTRODUCTION
Pulmonary arterial hypertension (PAH) is a progressive disease that leads to right ventricular failure and death. Three classes of drugs are currently available for the treatment of PAH: prostanoids, phosphodiesterase-5 inhibitors, and endothelin receptor antagonists. Current PAH treatment uses a goal-oriented approach, which targets improvements in various markers of disease that are important predictors of outcome, such as New York Heart Association (NYHA) functional class, 1 6-minute walk distance (6MWD), 2 and certain hemodynamic parameters, such as cardiac output and right atrial pressure. 3 Current recommendations include step-up therapy if patients fail to meet targets or deteriorate. 4 In addition, adding or switching to a drug from one class to another and changing the route of administration of drugs that have multiple routes of delivery are considerations when the current treatment is poorly tolerated or ineffective. However, there is no clear consensus on how and when to make these changes or specifically what therapies to add or substitute.
Within the class of prostanoids, three related compounds with different pharmacologic structures are currently available in the United States: epoprostenol, which is administered via continuous intravenous infusion; iloprost, administered via the inhaled route; and treprostinil, administered via inhaled and continuous intravenous or subcutaneous infusion routes. Two open-label multicenter clinical trials supported the safety of transitioning from epoprostenol to intravenous or subcutaneous treprostinil with maintenance of clinical stability in selected patients.5,6 Conversely, similar to a prior report showing that transition from parenteral prostacyclins to oral therapies is not always successful in an otherwise stable patient, 7 transition from parenteral prostacyclins to inhaled treprostinil in stable patients was associated with deterioration in 4 out of the 18 patients studied retrospectively. 8 These reports suggest that certain patients respond differentially not only to different therapies but also to the same drug when administered by a different route. It is also not known whether parenteral forms of treprostinil can still be effective when there has been a lack or loss of response to the inhaled form. In this multicenter retrospective study, we sought to characterize PAH patients receiving inhaled treprostinil who, in the opinion of the investigator, required transition to parenteral treprostinil, with the aim of assessing the safety and clinical efficacy of this transition.
METHODS
A multicenter retrospective chart review was performed at 7 PAH centers with extensive experience in using all PAH-specific therapies, including inhaled and parenteral prostanoids for the management of PAH. The study was approved by the institutional review board (IRB) at Tufts Medical Center, and local IRB approvals were obtained by each center prior to collection of data. Clinical data were collected between April 2007 and March 2012 from consecutive adult patients with PAH with World Health Organization groups 1, 4, and 5 4 who were transitioned from inhaled treprostinil to parenteral treprostinil (intravenous or subcutaneous routes). The reason and method for transition were determined at each center by the local PAH team, without a prespecified protocol. Data included baseline demographics, location of transition, reasons and methods for transition, assessments of PAH severity within 1 month before and 3 to 6 months after transition, adverse events encountered during and after transition, and long-term outcomes. In our statistical analyses, demographic and clinical characteristics are presented in a descriptive fashion as means ± standard deviations and ranges.
RESULTS
Baseline characteristics
Twenty-six patients underwent transition from inhaled treprostinil to parenteral treprostinil. Demographic data are presented in Table 1. The majority of patients had idiopathic, heritable, or drug- and toxin-associated PAH; were treated with inhaled treprostinil in combination with one or two oral PAH therapies (Table 2); and had poor functional capacity as evidenced by a low 6MWD and an advanced NYHA functional class prior to transition (Table 1). At the time of transition, patients had been on inhaled treprostinil for at least 2 months, 23 of them were receiving the recommended dose of 54 μg (9 puffs) 4 times a day or higher, and most were on a combination with one or two oral therapies (Table 2). The most common reason for transition to parenteral treprostinil was clinical deterioration, followed by lack of clinical improvement on the current regimen (Table 1). No patients were transitioned to infusion therapy because of intolerance or noncompliance with inhaled treprostinil. One patient became pregnant while on a combination of inhaled treprostinil and sildenafil without experiencing clinical deterioration or an inadequate response. She was transitioned to intravenous treprostinil until her successful delivery and then was switched back to the inhaled route.
Demographic and clinical characteristics of patients transitioned

Note: 6MWD: 6-minute walk distance; CTEPH: chronic thromboembolic pulmonary hypertension; F: females; HPAH: heritable pulmonary arterial hypertension; IPAH: idiopathic pulmonary arterial hypertension; IV: intravenous; M: males; NYHA FC: New York Heart Association functional class; PAH: pulmonary arterial hypertension; PAH-CHD: congenital heart disease–associated pulmonary arterial hypertension; PAH-CTD: connective tissue disease–associated pulmonary arterial hypertension; PoPH: portopulmonary hypertension; SD: standard deviation; SQ: subcutaneous; WHO: World Health Organization.
The two patients in group 5 had sarcoidosis and polyneuropathy, organomegaly, endocrinopathy, monoclonal protein, and skin changes (POEMS) syndrome, respectively.
One patient who was transitioned for inadequate response also complained of cough.
Background therapies of patients transitioned
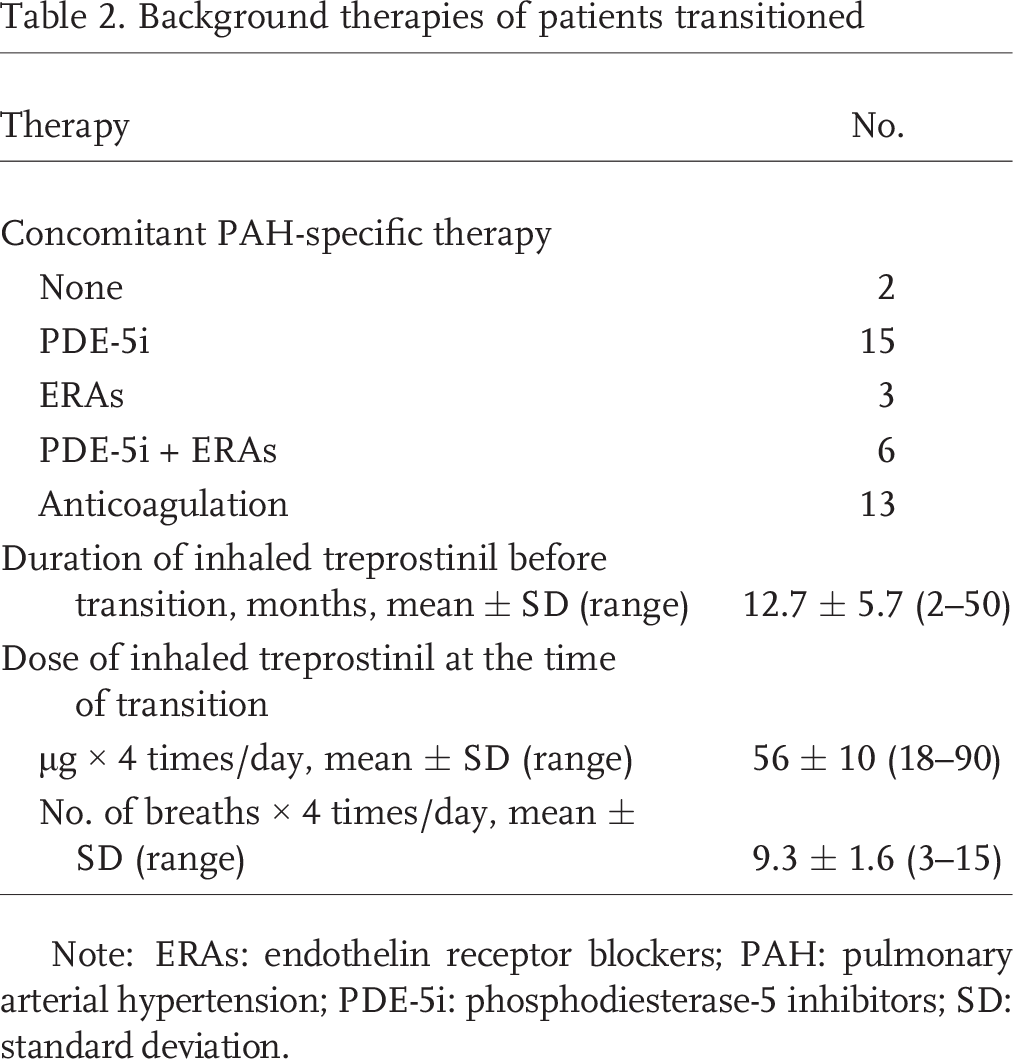
Note: ERAs: endothelin receptor blockers; PAH: pulmonary arterial hypertension; PDE-5i: phosphodiesterase-5 inhibitors; SD: standard deviation.
Transition methods
More than half of transitions occurred in the hospital (Table 1). The majority of patients started the transition while they were on 54 μg 4 times a day of inhaled treprostinil. The method of transition was highly variable from center to center. In 13 patients, the parenteral infusion started at 2–4 ng/kg/min 6 hours after the last inhalation at the maintenance dose, and it was followed by uptitration of the parenteral therapy. In an additional 6 patients, inhaled treprostinil was weaned off over several days, and parenteral infusion was started at 2–4 ng/kg/min after the last inhalation of 18 μg of treprostinil (3 breaths). In the remaining 7 patients, the inhaled dose was downtitrated while the parenteral infusion was started at 2–4 ng/kg/min and uptitrated. Inhalation was then stopped when the parenteral infusion rate reached 10–20 ng/kg/min, an overlap process that took 1–13 days. All oral PAH-specific therapies were continued throughout the transition to parenteral treprostinil.
Follow-up after transition
At 3 months after transition, 8 patients had improved their NYHA functional class (Fig. 1A; 7 patients improved from functional class 3 to 2 and 1 from 4 to 3), and 17 maintained their functional class. One patient with portopulmonary hypertension remained in functional class 4 and died from end-stage liver failure soon after the transition. Among the 12 patients who had 6MWDs 1 month before and 3–6 months after transition, 4 had at least a 10% improvement, another 4 maintained their walks within 10% pretransition, and the last 4 had a decline of at least 10% (Fig. 1B). Brain natriuretic peptide (BNP) was measured in 13 patients prior to transition (average 496 ± 388, range 20–1,201 pg/mL). Among the 9 patients who had BNP measurements 1 month before and 3–6 months after transition, 8 patients showed improvements (Fig. 1C).
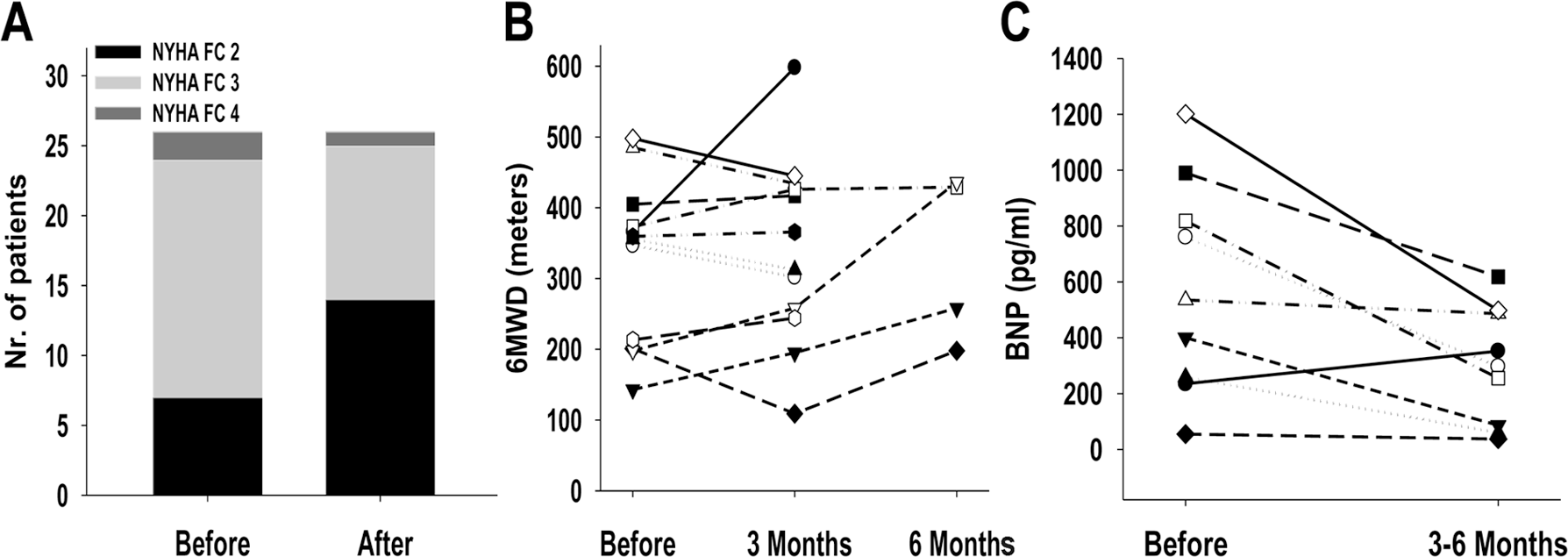
Clinical characteristics before and after transition. A, All 26 patients had New York Heart Association functional class (NYHA FC) assessments before and 3 months after transition. B, Among the 12 patients who underwent a 6-minute walk distance (6MWD) test, 8 showed stability or improvement. C, Brain natriuretic peptide (BNP) levels were measured in 9 patients before and 3–6 months after the transition.
Hemodynamic assessments
Fifteen patients underwent hemodynamic assessment by right heart catheterization 0–60 days before transition, with 12 of the 15 being performed within 30 days of transition (Table 3). The 15 patients in whom hemodynamics were assessed were in NYHA functional classes 2 (6 patients), 3 (7 patients), and 4 (2 patients). The majority of patients had moderate to severe hemodynamic impairment, with 11 of the 15 having a pulmonary vascular resistance greater than or equal to 8 Wood units. Eleven patients had right heart catheterizations 5 ± 3 months after transition (range 0.5–10 months). Of those 11 patients, 7 were evaluated before (0.5 ± 0.4 months) and after (5.6 ± 3 months; range 2–10 months) the transition (Fig. 2). Six of the 7 patients showed improved hemodynamics at follow-up. Patient 3, who had PAH associated with rheumatoid arthritis, improved from NYHA functional class 4 to NYHA functional class 3 for 6 months after the transition, after which she deteriorated, showed severe hemodynamic impairment at follow-up assessment, and underwent bilateral lung transplantation.
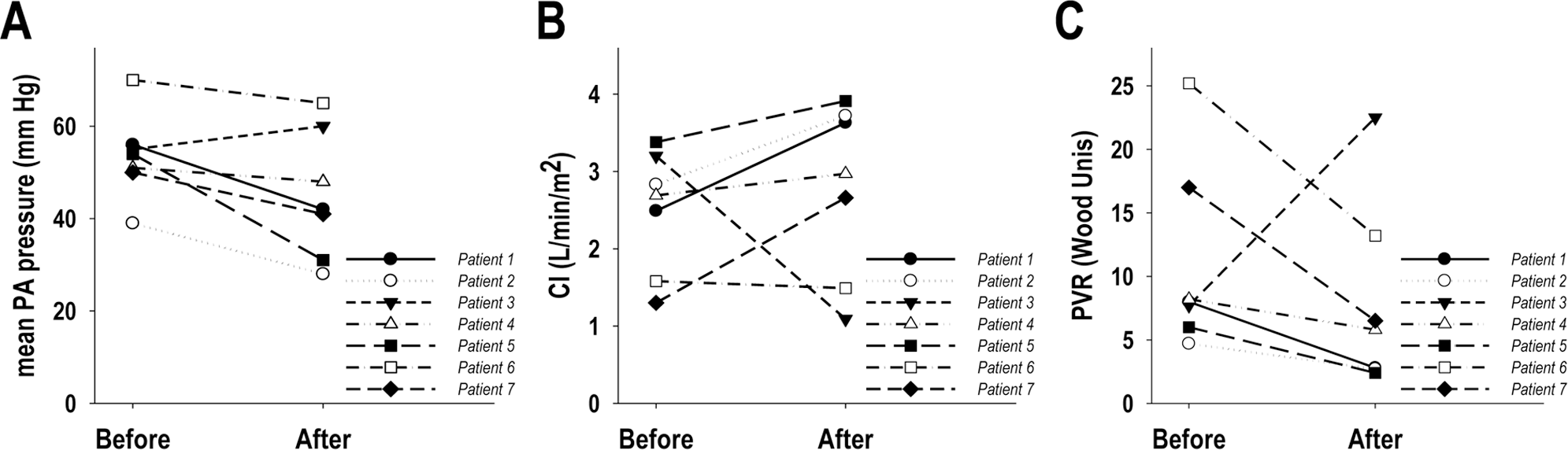
Hemodynamic characteristics before and after transition in 7 patients. Shown are changes in mean pulmonary artery (PA) pressures (A), cardiac index (CI; B), and pulmonary vascular resistance (PVR; C). Follow-up assessment occurred 2–10 months after the transition.
Hemodynamic characteristics of the 15 patients tested before transition

Note: CI: cardiac index; CO: cardiac output; mPAP: mean pulmonary artery pressure; mRAP: mean right atrial pressure; PAWP: pulmonary artery wedge pressure; PVR: pulmonary vascular resistance; SD, standard deviation.
Long-term outcomes
Patients were followed for 3–18 months. The dose of parenteral treprostinil at the last assessment was 26 ± 20 ng/kg/min (range 2–78 ng/kg/min). Three patients died 3, 3.8, and 6.8 months after transition, of metastatic carcinoid with pulmonary embolism, end-stage liver disease, and pulmonary embolism, respectively. One patient underwent lung transplantation after 10 months of intravenous therapy, and another patient with portopulmonary hypertension underwent liver transplantation.
Safety of the transition
The transition process was not associated with any serious adverse events, and there were no hemodynamic adverse outcomes. Three of the 7 patients who had an overlap transition experienced prostacyclin overdose symptoms, which were controlled by decreasing more rapidly or permanently discontinuing the inhaled form. Six of the 19 patients in whom the transition did not involve an overlap had overdose symptoms and the infusion was temporarily downtitrated to control the side effects. These symptoms included nausea, vomiting, headaches, diarrhea, or jaw pain. An additional side effect was pain at the site of the subcutaneous infusion (5 patients).
DISCUSSION
We report here the experience of 7 experienced PAH centers that successfully transitioned selected PAH patients from inhaled treprostinil to parenteral treprostinil. We chose to include transition to both intravenous treprostinil and subcutaneous treprostinil, since the two forms of parenteral delivery provide similar pharmacokinetic profiles. 9
Selected patients had moderate to severe functional and hemodynamic impairment at the time of transition, despite therapy with a combination of inhaled treprostinil and one or two oral therapies in the majority of cases. The assessment of lack of clinical improvement or worsening of the disease varied from center to center and from patient to patient, with only selected patients undergoing complete noninvasive and invasive testing. All had reported NYHA functional class in combination with at least one other test: 6MWD, serologic (BNP), or hemodynamic assessment.
The location of transition varied among centers. In all patients transitioned to intravenous treprostinil, the transition occurred in the hospital or outpatient setting, while transition to the subcutaneous form occurred in the inpatient setting, outpatient clinic, or home. Similarly, the transition method consisted of an overlap of the two drugs, stopping the inhaled form and starting the parenteral form at low dose, or downtitrating the inhaled form to the lowest dose and then starting the parenteral form at a low dose followed by uptitration. Regardless of the method of transition, all patients tolerated the process quite well, experiencing only occasional minor symptoms of prostacyclin excess and having no serious adverse events. Most importantly, most patients stabilized or improved clinically.
The introduction of inhaled prostanoids (iloprost and treprostinil) has widened the choices of treatment for PAH patients. How the pharmacokinetics and the effectiveness of inhaled forms compare to parenteral forms of prostanoids at the currently approved doses, however, has not been adequately studied. Comparing blood levels during infusion and after inhalation may be misleading because lung tissue levels after inhaled delivery are probably higher than circulating blood levels. Therefore, clinicians need to rely on clinical assessment as proof of response to therapy. In our cohort of 26 patients who failed to respond or experienced a decline on inhaled treprostinil, one-third of patients improved their functional class and only 1, who was in functional class 4 at the time of transition, failed to improve. This might indicate that lack or loss of response to the inhaled form suggests that the route of delivery or the dose currently used is somehow limiting efficacy. Conversely, in selected patients intolerant of parenteral treprostinil infusion, transition to inhaled treprostinil can also be achieved. Of note, however, clinicians should exercise caution when making such a transition, as one small series reported a 22% rate of clinical worsening despite continuation of concomitant oral nonprostanoid therapies among patients transitioned from the parenteral route to the inhaled route. 8
Our report supports the notion that inhaled treprostinil and parenteral treprostinil are not always equivalent. As the maximum studied dose of inhaled treprostinil (54 μg 4 times a day) is thought to be equivalent to approximately a 10-ng/kg/min infusion, 10 patients who require low doses of prostanoids may remain stable or improve on the inhaled form, whereas those who require higher doses of prostanoid may benefit from transition to parenteral forms.
Our study is limited by the retrospective design, lack of a standardized method of transition, small number of patients, and missing data, which precluded us from performing meaningful statistical analyses. On the other hand, data on the transition from inhaled treprostinil to parenteral treprostinil are sparse, and to our knowledge, this is the only report thus far in the literature. Our data suggest that in selected patients who do not achieve and/or maintain satisfactory improvements on inhaled treprostinil in combination with oral therapies, transition to parenteral treprostinil may be beneficial. In addition, our findings support the notion that a number of different approaches to transitioning are safe and well tolerated and can take place in the hospital, outpatient clinic, or home, the latter for the subcutaneous route.
In conclusion, our report provides important information to clinicians who treat patients with advanced PAH and are contemplating transitions from inhaled prostanoids to parenteral prostanoids. These can be done safely by experienced PAH teams, using a variety ofmethodologies in a number of different settings with the expectation that patients will improve or at least remain clinically stable for months afterward. Future studies evaluating criteria for patient selection and timing of transitions as well as comparing different methods are needed.
Footnotes
ACKNOWLEDGMENTS
Preliminary results of this manuscript were presented at the 2011 American College of Chest Physicians meeting.
