Abstract
Pulmonary arterial hypertension (PAH) remains a disease with a poor prognosis, so early detection and treatment are very important. Sensitive and non-invasive markers for PAH are urgently required. This study was performed to identify sensitive markers of the clinical severity and prognosis of PAH. Patients diagnosed with PAH (n = 30) and control participants (n = 15) were enrolled in this observational study. Major EPC and MSC markers (including CD34, CD133, VEGFR2, CD90, PDGFRα, and NGFR) in peripheral blood mononuclear cells (PBMNCs) were assessed by flow cytometry. Associations of these markers with hemodynamic parameters (e.g. mean pulmonary arterial pressure, pulmonary vascular resistance, and cardiac index) were assessed. Patients with PAH were followed up for 12 months to assess the incidence of major adverse events, defined as death or lung transplantation. Levels of circulating EPC and MSC markers in PBMNCs were higher in patients with PAH than in control participants. Among the studied markers, nerve growth factor receptor (NGFR) was significantly positively correlated with hemodynamic parameters. During the 12-month follow-up period, major-event-free survival was significantly higher in patients with PAH who had relatively low frequencies of NGFR positive cells than patients who had higher frequencies. These results suggested that the presence of circulating NGFR positive cells among PBMNCs may be a novel biomarker for the severity and prognosis of PAH.
Introduction
Pulmonary arterial hypertension (PAH) is a rare progressive disease in which pulmonary vascular remodeling leads to intimal hypertrophy of small arteries, resulting in elevated pulmonary vascular resistance (PVR) and pressure; these factors are pivotal in the progression of PAH. 1 Despite advances in PAH-specific therapies, the prognosis remains poor for patients with PAH. At present, the most reliable diagnostic tool for PAH is right heart catheterization (RHC) to evaluate hemodynamic parameters such as mean pulmonary arterial pressure (mPAP), PVR, and cardiac index (CI). However, RHC is an invasive test; moreover, early diagnosis of PAH based solely on hemodynamic parameters obtained from RHC is problematic, because approximately two-thirds of the pulmonary vascular bed has already irreversibly deteriorated by the time of PAH diagnosis. 2 Therefore, identification of biomarkers for the diagnosis of PAH and assessment of its severity at an early stage are important priorities in the field of pulmonary vascular medicine. Vascular remodeling is caused by the proliferation and abnormal signal transduction of vascular endothelium and smooth muscle cells. Recently, several studies have implied that the process of vascular remodeling is driven in part by bone marrow-derived proangiogenic cells,3,4 and that endothelial progenitor cells (EPCs) and mesenchymal stem cells (MSCs) contribute to pulmonary vascular remodeling.5,6 Furthermore, circulating mesenchymal precursor cells accumulate in the walls of remodeled vessels in patients with PAH, thereby contributing to vessel wall thickening. 7 The progression of vascular remodeling leads to the development and aggravation of PAH 8 ; therefore, circulating mesenchymal precursor cells, which play an important role in the initiation of vascular remodeling, may better reflect the pathophysiology of PAH. 9 A low-affinity receptor, nerve growth factor receptor (NGFR), has emerged as a marker for the isolation of highly primitive and proliferative stem cells. 10 NGFR gene expression in peripheral blood mononuclear cells (PBMNCs) is reportedly associated with the progression of vascular remodeling in patients with acute coronary syndrome, 11 suggesting that circulating NGFR positive cells in PBMNC may play important roles in the pathologic mechanisms underlying PAH. We hypothesized that the frequency of circulating EPCs and MSCs including NGFR positive cells in PBMNCs increases in patients with PAH and is associated with hemodynamic measurements that indicate the severity and prognosis of PAH.
Methods
Study design
This observational study was designed to investigate cell-surface antigens in PBMNCs from patients with PAH and was registered in the UMIN Clinical Trials Registry (UMIN 000032832). The study protocol complied with the Declaration of Helsinki and was approved by the local ethics committee. This study included patients from Kanazawa University Hospital in Japan, from July 2016 to March 2020, and informed consent was obtained from each patient prior to enrollment in the study.
Patient population
Patients diagnosed with PAH were selected; a healthy control group was also selected, based on an absence of right heart overload as assessed by echocardiography. In patients with PAH, RHC showed that mPAP was ≥25 mmHg and pulmonary arterial wedge pressure (PAWP) was ≤15 mmHg at rest. Respiratory functional tests revealed forced vital capacity >70% and forced expiratory volume in 1 s (FEV1) >60% of the predicted values, indicating no significant respiratory dysfunction. No abnormalities were observed in pulmonary ventilation blood flow scintigraphy. Blood tests showed no findings of blood, systemic inflammatory, or metabolic disorders. Patients with acute coronary syndrome, pregnancy, malignancy, and infectious diseases were excluded from this study.
Clinical examinations
Patients with PAH underwent RHC at rest to examine hemodynamic parameters, including mean right atrial pressure, mPAP, and PAWP. Cardiac output was determined by the thermodilution method. The CI was calculated by dividing cardiac output by body surface area. PVR was calculated by dividing mPAP by cardiac output. Six-minute walk distance (6MWD) measurements were performed in patients with PAH. Blood samples were obtained and brain natriuretic peptide (BNP) level was measured in all patients.
Flow cytometry
Peripheral blood samples were collected into heparinized tubes; PBMNCs were then isolated using Lymphoprep™ tubes (AXIS-SHIELD PoC AS, Oslo, Norway). 12 Red cell lysis was performed using BD PharmLyse™ (BD Pharmingen, San Diego, CA, USA). IgG was blocked for 10 min at room temperature; subsequently, immunophenotyping of PBMNCs was performed by staining for 15 min on ice in the dark with fluorochrome-labeled monoclonal antibodies, including anti-human CD133-FITC (Miltenyi Biotech, Miltenyi Biotec, Bergisch Gladbach, Germany), VEGFR2-PE (R&D Systems, Minneapolis, MN, USA), CD34-PerCP (BD Pharmingen), CD90-FITC (BD Pharmingen), PDGFRα-PE (BD Pharmingen), and NGFR-APC (Miltenyi Biotech). 13 Isotype-matched control antibodies were used as negative controls to adjust for fluorochrome overlap.
The fluorescence intensity of cells labeled with fluorochromes was examined using an Accuri C6 flow cytometer (BD Pharmingen). The data were analyzed using FlowJo software v10 (BD Pharmingen).
Outcome assessments
Surface antigens of PBMNCs were compared between patients with PAH and control participants. In patients with PAH, these markers were compared with 6MWD and hemodynamic parameters (mPAP, PVR, and CI) obtained from RHC examinations. In addition, patients with PAH were followed up for 12 months to assess the occurrence of adverse events, such as death or lung transplantation.
Statistical analysis
Statistical analysis and graphs were prepared using Graph Pad Prism 7.0 (Graph Pad Software, La Jolla, CA, USA). All variables are presented as medians with interquartile ranges (IQRs). Statistical significance was defined as P < 0.05 (two-tailed). The Mann–Whitney U test was used to compare quantitative variables between groups. Chi-squared tests were used to compare differences in other variables between the groups. Correlations were analyzed using Spearman’s correlation coefficient. Multiple regression analysis was performed using SPSS for Windows (version 17.0; SPSS Japan Inc., Tokyo, Japan). Kaplan–Meier curves were plotted based on cut-off points for composite endpoints of major adverse events. Comparisons of event-free survival curves were performed using the log-rank test.
Results
Patient characteristics
Thirty patients with PAH were enrolled in the study, along with 15 healthy control participants without findings of right heart overload on echocardiography. The characteristics of enrolled patients are shown in Table 1.
The baseline characteristics in control subjects and patients with PAH.
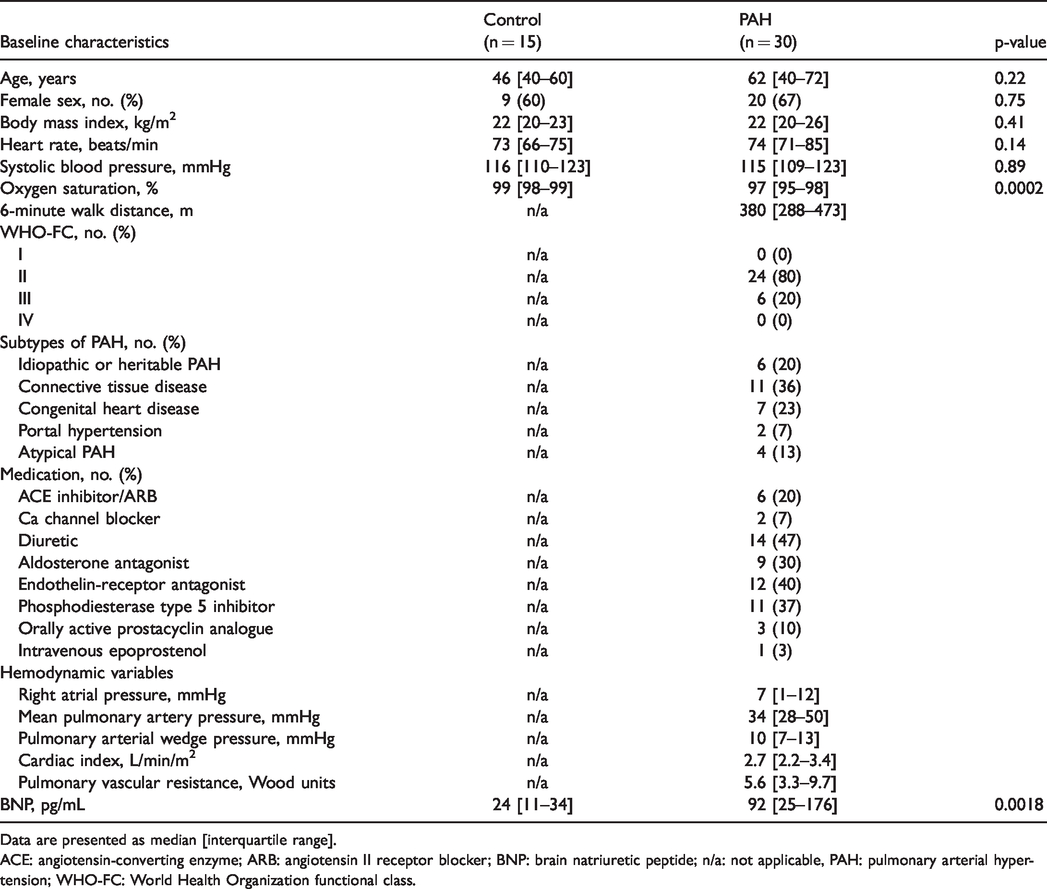
Data are presented as median [interquartile range].
ACE: angiotensin-converting enzyme; ARB: angiotensin II receptor blocker; BNP: brain natriuretic peptide; n/a: not applicable, PAH: pulmonary arterial hypertension; WHO-FC: World Health Organization functional class.
There were significant differences in oxygen saturation and BNP level between control participants and PAH patients, but no differences in age, sex, BMI, or blood pressure. The numbers of patients with PAH caused by idiopathic or heritable PAH, connective tissue disease, congenital heart disease, portal hypertension, and atypical PAH were 6 (20%), 11 (36%), 7 (23%), 2 (7%), and 4 (13%), respectively. At the time of evaluation, the numbers of patients with PAH who had taken endothelin receptor antagonists, phosphodiesterase type 5 inhibitors, orally active prostacyclin analogs, and intravenous epoprostenol were 12 (40%), 11 (37%), 3 (10%), and 1 (3%), respectively. The numbers of patients with PAH classified as World Health Organization Functional Classes I–IV were 0 (0%), 24 (80%), 6 (20%), and 0 (0%), respectively. The median 6MWD in patients with PAH was 380 [IQR 288–473] m.
Frequencies of EPC and MSC marker positive cells in PBMNCs
The gating strategy to identify EPC and MSC marker positive cells in PBMNCs using flow cytometry is shown in Fig. 1.
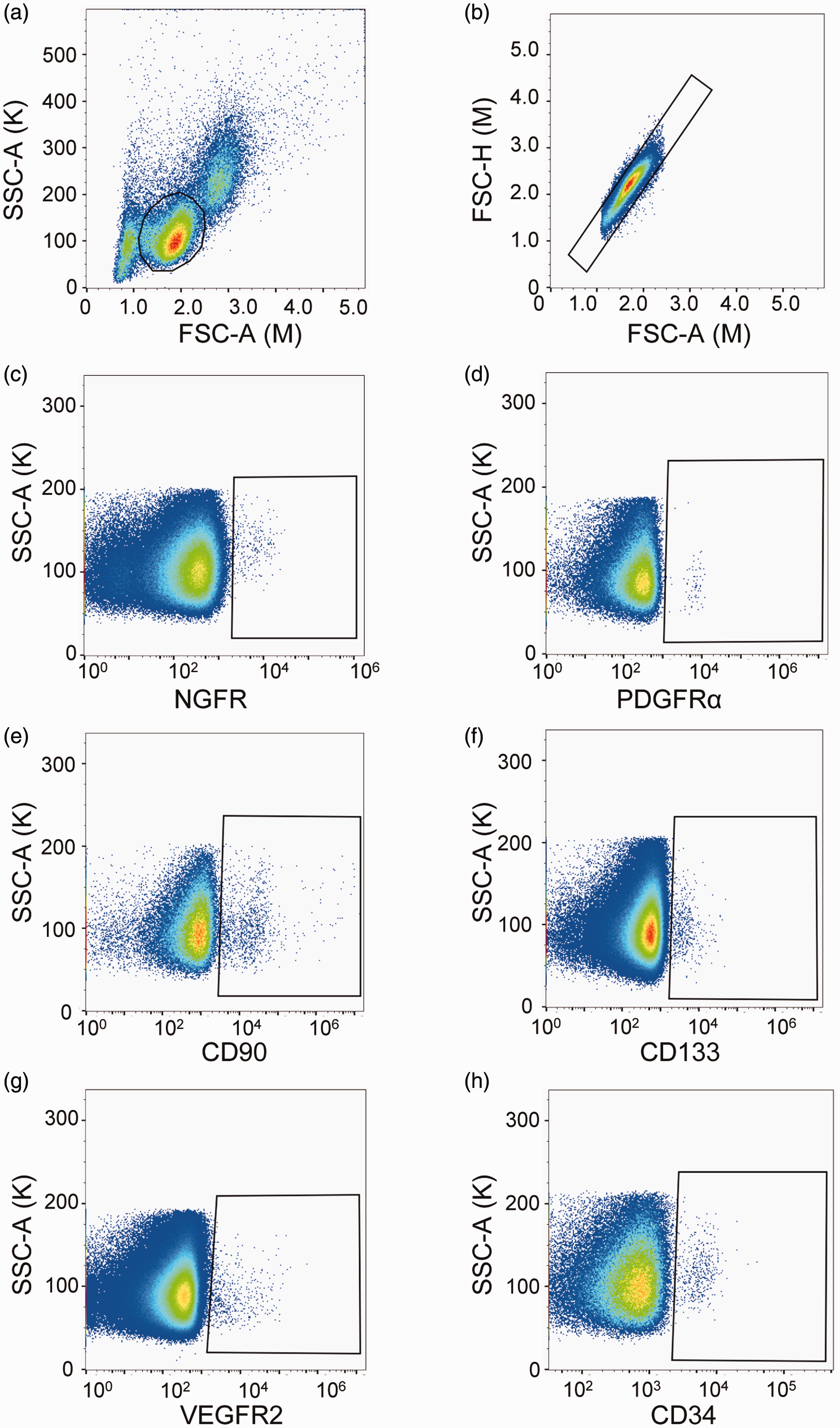
Gating strategy for flow cytometry assay. Flow cytometry was used to assess the frequencies of NGFR, PDGFRα, CD90, CD133, VEGFR2, and CD34 positive cells in peripheral blood mononuclear cells from patients with PAH and control participants. (a) Mononuclear cells. (b) Single cells. (c) NGFR positive cells. (d) PDGFRα positive cells. (e) CD90 positive cells. (f) CD133 positive cells. (g) VEGFR2 positive cells. (h) CD34 positive cells.
Comparisons of EPC and MSC marker positive cells in the PAH and control groups are shown in Fig. 2.
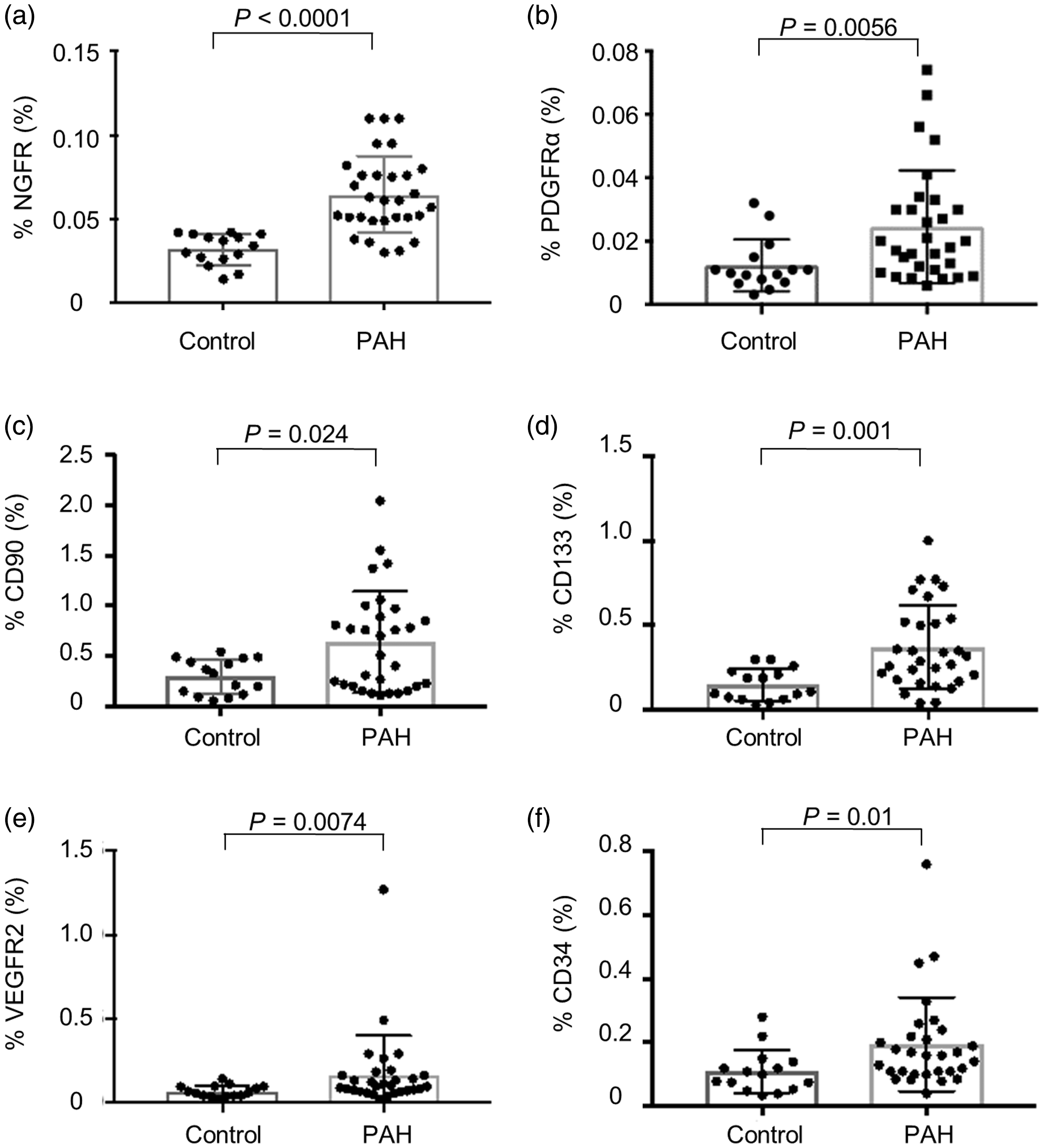
Frequencies of mesenchymal stem cell and endothelial progenitor cell marker positive cells in control participants and patients with pulmonary arterial hypertension. The frequencies of MSC and EPC marker positive cells were higher in patients with PAH than in control participants. (a) % NGFR. (b) % PDGFRα. (c) % CD90. (d) % CD133. (e) % VEGFR2. (f) % CD34. Center lines show medians, bars indicate interquartile range (IQR).
The frequencies of EPC and MSC marker positive cells were significantly higher in patients with PAH than in the control participants. The median (interquartile range) frequencies of each marker in patients with PAH and in the control participants were as follows: VEGFR2, 0.098% (0.071–0.16) vs. 0.062% (0.040–0.094), P = 0.011; CD34, 0.16% (0.11–0.22) vs. 0.1% (0.65–0.13), P = 0.01; CD133, 0.31% (0.19–0.52) vs. 0.11% (0.073–0.22), P = 0.001; CD90, 0.61% (0.21–0.88) vs. 0.33% (0.14–0.46), P = 0.024; PDGFRα, 0.019% (0.011–0.03) vs. 0.0099% (0.0075–0.013), P = 0.0056; NGFR, 0.061% (0.051–0.076) vs. 0.034% (0.027–0.04), P < 0.0001.
Frequencies of EPC and MSC marker positive cells in PBMNCs correlated with hemodynamic severity of PAH
The relationships between the frequencies of EPC and MSC marker positive cells or BNP level and hemodynamic parameters (mPAP, PVR, and CI) or 6MWD are shown in Table 2.
Univariate correlations between the frequency of MSC and EPC markers or BNP level and hemodynamic parameters or 6MWD.
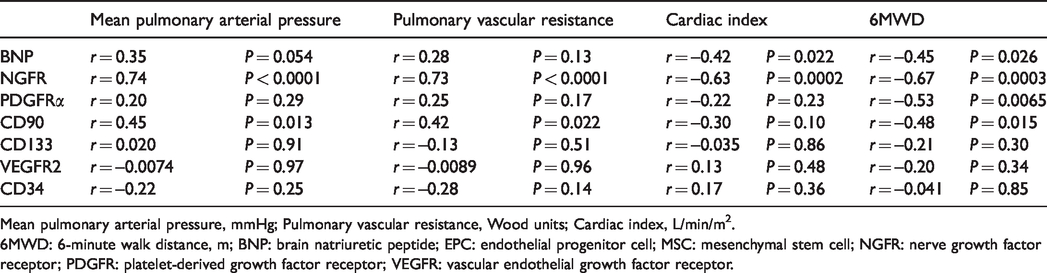
Mean pulmonary arterial pressure, mmHg; Pulmonary vascular resistance, Wood units; Cardiac index, L/min/m2.
6MWD: 6-minute walk distance, m; BNP: brain natriuretic peptide; EPC: endothelial progenitor cell; MSC: mesenchymal stem cell; NGFR: nerve growth factor receptor; PDGFR: platelet-derived growth factor receptor; VEGFR: vascular endothelial growth factor receptor.
Among the studied markers, the frequency of NGFR positive cells in PBMNCs was strongly correlated with several hemodynamic parameters: mPAP (R = 0.74, P < 0.0001), PVR (R = 0.73, P < 0.0001), and CI (R = −0.63, P = 0.0002) (Fig. 3).
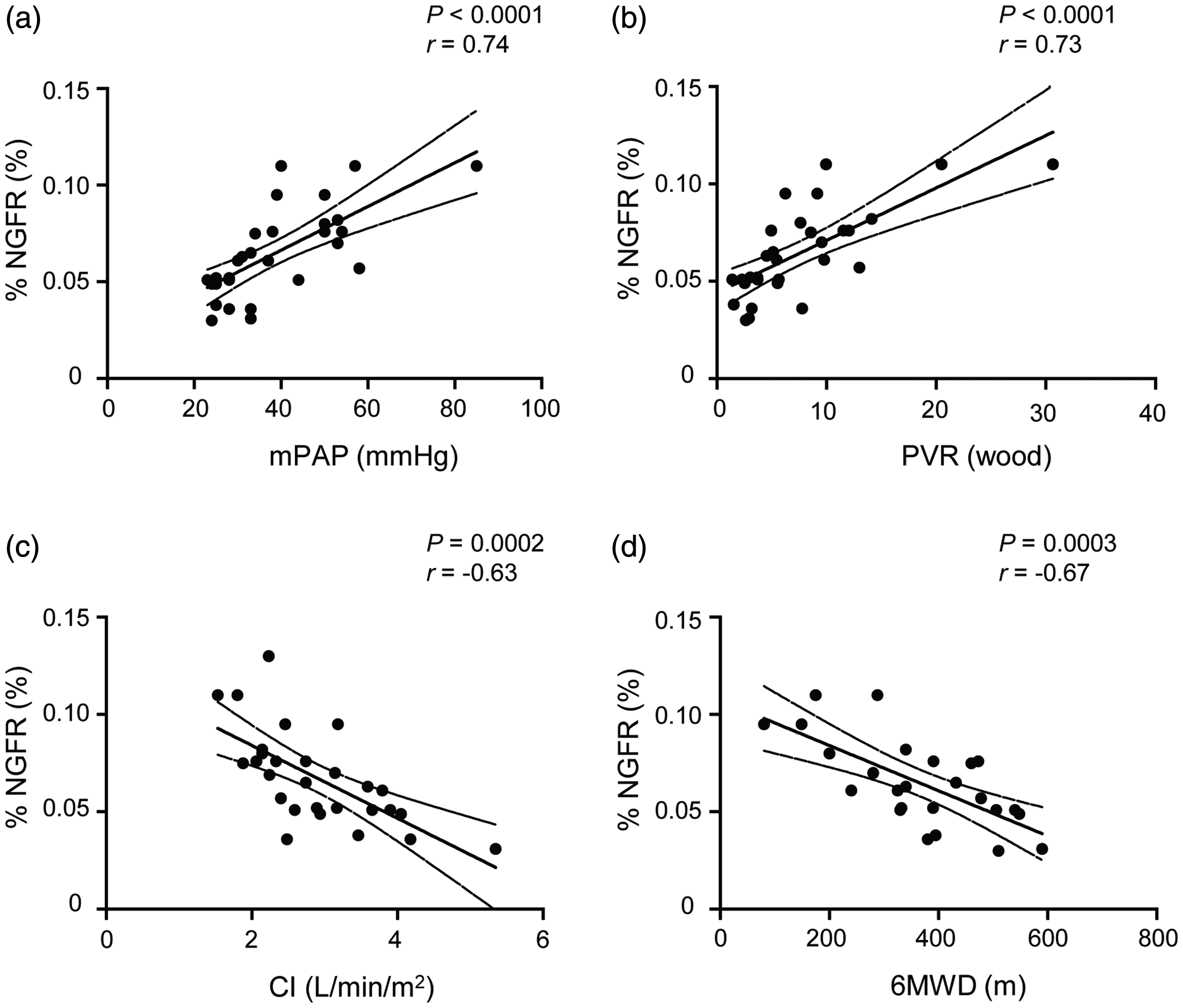
Correlations between frequency of nerve growth factor receptor positive cells in patients with pulmonary arterial hypertension and hemodynamic parameters or 6-minute walk distance. The frequency of NGFR positive cells in patients with PAH was significantly associated with hemodynamic parameters (mPAP (a), PVR (b), and CI (c)) and 6MWD (d).
The frequencies of NGFR positive cells in patients with different subtypes of PAH are also shown in the Supplemental Figure. The frequencies were significantly higher in patients with idiopathic or heritable PAH, connective tissue disease, and atypical PAH than in controls. However, we found no difference in the frequency of NGFR between patients with different PAH subtypes.
The frequency of CD90 positive cells in PBMNCs was also positively correlated with mPAP (R = 0.45, P = 0.013) and PVR (R = 0.42, P = 0.022), but not with CI (R = −0.3, P = 0.1). BNP level was correlated with CI (R = −0.42, P = 0.022), but not with mPAP (R = 0.35, P = 0.054) or PVR (R = 0.28, P = 0.13) (Table 2). 6MWD was also negatively correlated with BNP level (R = −0.45, P = 0.026) and MSC marker positive cells, such as those positive for NGFR (R = −0.67, P = 0.0003), PDGFRα (R = −0.67, P = 0.0065), and CD90 (R = −0.53, P = 0.015). However, the frequencies of EPC marker positive cells were not correlated with hemodynamic parameters or 6MWD (Table 2).
Regression analysis of hemodynamic parameters
Regression analysis with multiple variables was performed on the frequencies of NGFR and CD90 positive cells, which were correlated with hemodynamic parameters based on univariate analysis. Table 3 shows the results of regression analysis of these parameters.
Results of regression analysis of NGFR and CD90 in mPAP.
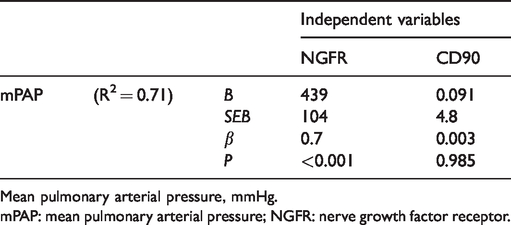
Mean pulmonary arterial pressure, mmHg.
mPAP: mean pulmonary arterial pressure; NGFR: nerve growth factor receptor.
The results indicated that the correlation between NGFR and mPAP was significantly stronger than that between CD90 and mPAP (P < 0.001).
Correlations of the frequencies of NGFR positive cells in PBMNCs with major adverse events
Patients with PAH were followed up for 12 months to determine the incidence of major adverse events, defined as death or lung transplant. The baseline characteristics of patients with PAH with and without major adverse events are shown in Table 4.
The baseline characteristics of patients with PAH with and without adverse events.
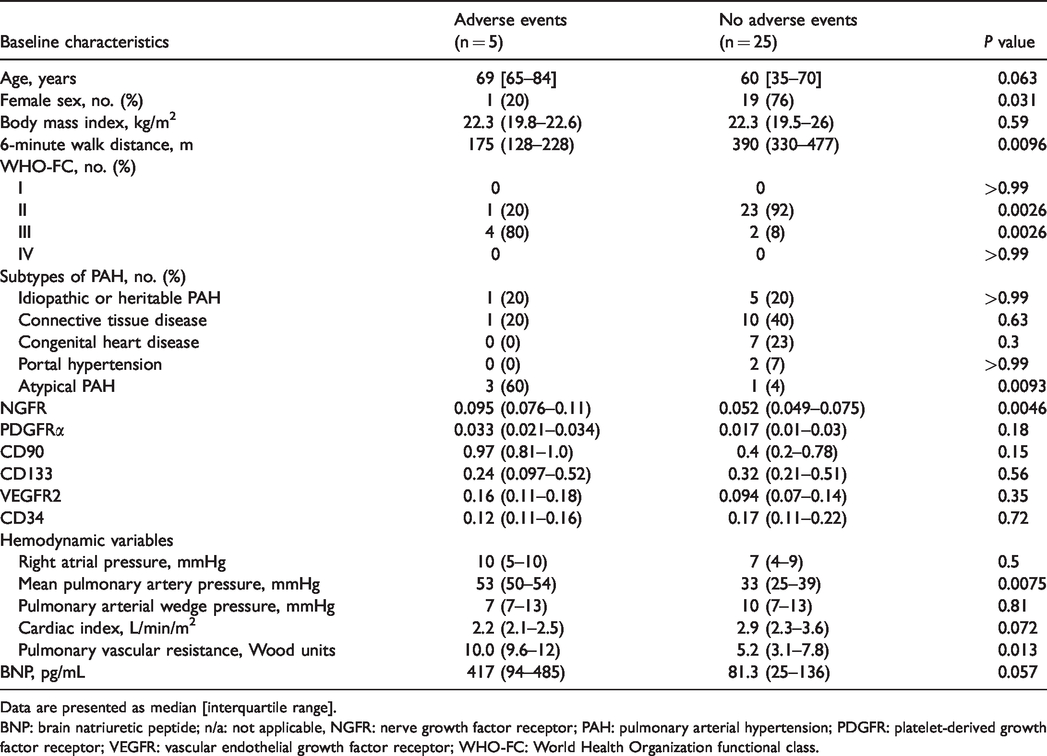
Data are presented as median [interquartile range].
BNP: brain natriuretic peptide; n/a: not applicable, NGFR: nerve growth factor receptor; PAH: pulmonary arterial hypertension; PDGFR: platelet-derived growth factor receptor; VEGFR: vascular endothelial growth factor receptor; WHO-FC: World Health Organization functional class.
Major adverse events occurred in five patients (16.7%): death in five (100%) and lung transplant in none (0%). The cause of death in all cases was heart failure. Receiver operating characteristic (ROC) curves predicting major adverse events based on the frequencies of NGFR positive cells are shown in Fig. 4a. The AUC was 0.88 (95% CI 0.76–1.0). The cut-off point was 0.067 (specificity = 100%, sensitivity = 72%). The frequencies of NGFR-positive cells were <0.067 in 18 PAH patients and >0.067 in the other 12. Kaplan–Meier curves for patients with PAH with and without major adverse events were constructed based on a cut-off frequency of 0.067 for NGFR positive cells (Fig. 4b).
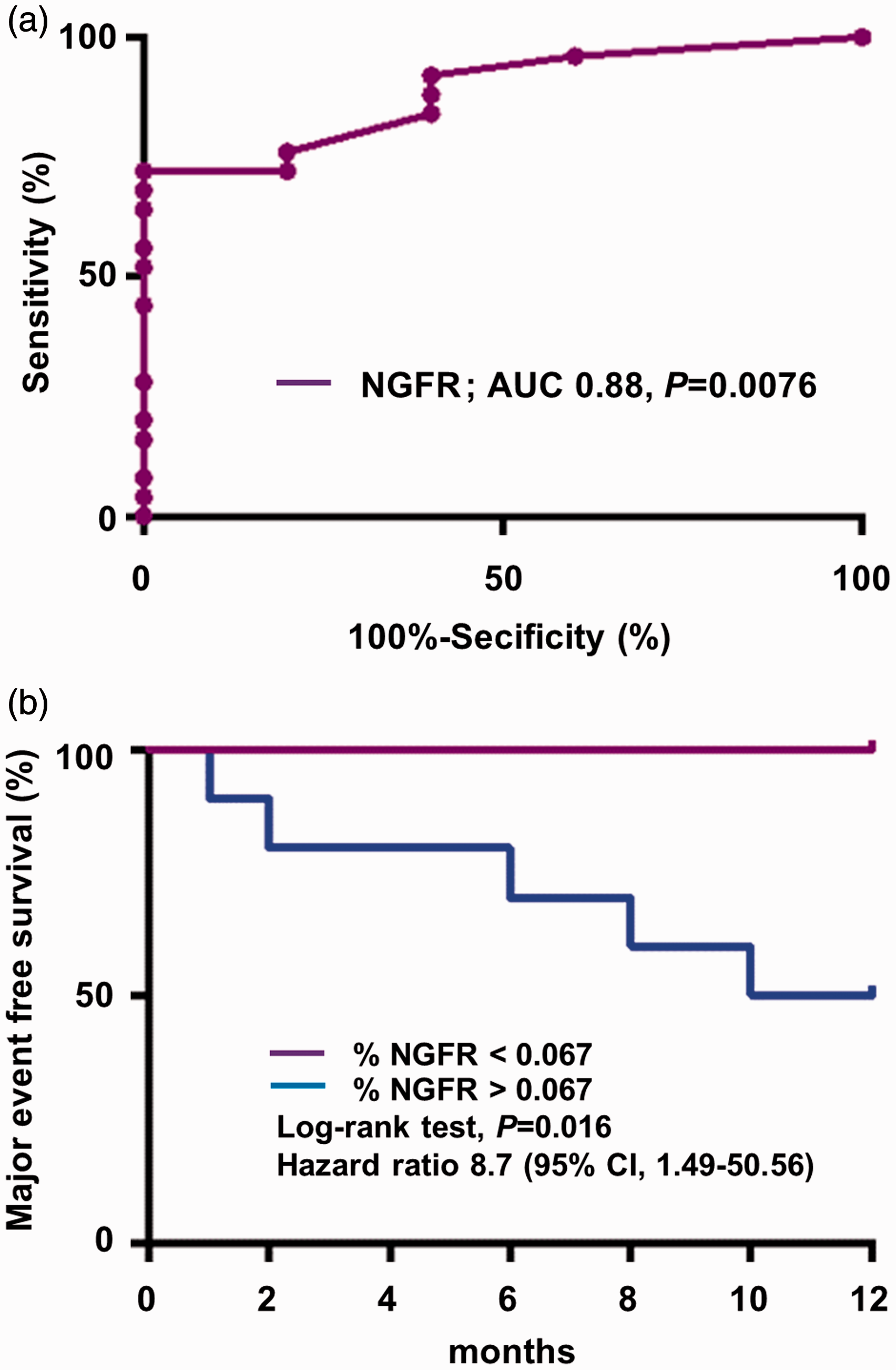
(a) Receiver operating characteristic curves predicting major adverse events based on the frequencies of NGFR positive cells. ROC analysis revealed that NGFR was a predictive biomarker for major adverse events, with an area under the curve of 0.88 (P < 0.01). (b) Kaplan–Meier curves of survival without major adverse events based on the frequency of nerve growth factor receptor positive cells. Kaplan–Meier curves according to the frequency of NGFR positive cells (cut-off level, 0.067) are shown. Major-event-free survival was significantly better in pulmonary arterial hypertension patients with NGFR positive cell frequencies < 0.067 than in patients with NGFR-positive cell frequencies > 0.067 (P = 0.016).
During the 12-month follow-up period, major-event-free survival was significantly better in patients with PAH who had frequencies of NGFR positive cells <0.067 than in patients who had frequencies of NGFR positive cells >0.067 (cut-off point, 0.067%; hazard ratio, 8.7; 95% confidence interval, 1.49–50.56; P = 0.016).
Discussion
In this study, we demonstrated that the frequencies of EPC and MSC marker positive cells among PBMNCs were higher in patients with PAH than in control participants. Among the EPC and MSC markers, NGFR was significantly more abundant in patients with PAH than in control participants, and this cell subset had the strongest correlation with hemodynamic parameters and clinical severity. The BNP level was reported to be correlated with PVR, CI, and mPAP in patients with primary pulmonary hypertension (PH). 14 In the present study, the frequency of NGFR positive cells was more strongly associated with mPAP than the BNP level. Moreover, in patients with PAH, the incidence of major adverse events was correlated with the frequency of NGFR positive cells. These results indicate that NGFR positive cells may be a useful marker for severity and prognosis in patients with PAH.
The pathophysiology of PAH, including environmental and genetic factors that promote vascular remodeling, is not completely understood.15–18 Risk assessment using multiple parameters has been recommended in clinical settings. 19 However, several parameters proposed for risk stratification in patients with PAH do not clearly reflect vascular remodeling. A non-invasive biomarker reflecting the pathophysiology of PAH may play an important role in treatment. Recently, MSCs with pluripotency have been reported to be involved in vascular remodeling, and showed associations with pathological changes and therapeutic effects. 7 In response to tissue injury, circulating MSCs are recruited to the injury site and contribute to the tissue remodeling process. In animal models, circulating MSCs are essential for maintenance of vascular homeostasis in cardiovascular disorders 4 ; administration of MSCs is also reportedly beneficial for PH.20,21 These results suggest that the frequency of circulating MSCs may be an excellent biomarker reflecting vascular remodeling in patients with PAH. Therefore, we focused on the main markers of MSCs and showed that levels of circulating MSCs were significantly elevated in patients with PAH compared to control participants. Among these MSC markers, we found that NGFR positive cells were significantly positively correlated with parameters indicating the severity of PAH, including mPAP. In this study, we identified NGFR as one of the major markers of MSC, but it is remains controversial whether NGFR positive cells in PBMNCs are MSCs.
NGFR is involved in several pathological and physiological processes,22,23 such as cell development, survival, and differentiation. It was initially reported to be expressed on cells of the central and peripheral nervous systems. 24 NGFR was also reported to be expressed in various tissues in the immune system, and in peripheral blood, and plays a role in inflammatory reactions and repair processes in damaged tissue. 25 Recently, NGFR has been employed as a surface marker for mesenchymal progenitor cells with proliferative and multipotential differentiation abilities. 10 To our knowledge, circulating NGFR positive MSCs have not yet been reported to be associated with PAH pathogenesis or progression. However, Iso et al. reported that circulating NGFR positive MSCs increased in abundance and promoted angiogenesis after acute myocardial infarction. 26 Some studies have reported that MSCs are induced in peripheral blood in response to hypoxia; subsequently, they promote vascular remodeling and lead to PAH pathogenesis. 7 The results of the present study support the suggestion that NGFR, as an MSC marker, may be related to pathological states in PAH.
Chronic hypoxia has emerged as a well-established independent cause of vascular remodeling in PAH. 27 HIF-1α functions as a master regulator of oxygen homeostasis and hypoxic adaptation in the lung. Early studies demonstrated that the activation of HIF-1α plays a key role in vascular remodeling by promoting enhanced arterial wall thickness under hypoxia in experimental animals. 3 Previous findings showed that an interaction between NGFR and Sah2 regulates the expression of HIF-1α under hypoxic conditions. 28 On the basis of these observations, we speculate that the NGFR-HIF-1α axis might constitute a promising marker and therapeutic target for PAH.
Circulating EPCs have also been reported to be associated with PAH, so we evaluated major EPC markers in this study.29,30 In the present study, although the EPC marker positive cells were elevated in patients with PAH compared with control participants, they were not correlated with the severity of PAH. Asosingh et al. reported that circulating CD34+CD133+ bone marrow-derived proangiogenic precursors were more abundant in patients with PAH than in healthy control participants, and were correlated with pulmonary artery pressure. 29 Another study reported that circulating EPCs, defined as CD34+CD133+VEGFR2+ cells, were induced to a greater extent in patients with PAH than in healthy controls. 30 These reports support the present findings and suggest that circulating EPCs may play an important role in PAH.
Endothelial cell apoptosis is an important factor in the initiation of PH following a reduction of monolayer integrity and disruption of barrier function. 31 NGFR is rarely expressed in healthy endothelial cells, and is strongly induced in capillary endothelial cells under conditions of ischemia and diabetes. 32 NGFR signaling in pathological endothelial cells promotes apoptosis through inhibition of the VEGF-A/Akt/eNOS/NO pathway. The increase in the number of NGFR positive cells in PAH patients may reflect endothelial cell apoptosis.
Dysregulated endothelial cell proliferation and complex vascular lesions also play critical roles in the progression of PH. 33 Occlusive vascular lesions contain proliferating endothelial cells, smooth muscle cells, and inflammatory cells, including T and B lymphocytes and macrophages. Recent studies indicated that resident stem/progenitor cells also accumulate and contribute to blood vessel remodeling, leading to occlusive vascular lesion formation. 34
The expression of NGFR has been shown to be associated with a proliferative state, and increased migration/invasion in several primary and metastatic human cancers. NGFR controls cell survival via the recruitment of receptor-interacting serine/threonine-protein kinase 2-mediated activation of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB). 35 NGFR also triggers the mitogen-activated protein kinase pathway, leading to signaling processes mediated by extracellular-regulated kinases or phosphatidylinositol 3-kinases promoting cell proliferation and survival. 36 Here, we showed that circulating NGFR positive cell expression was positively correlated with the severity of PAH. These cells may be sources of proliferating endothelial cells, and show crosstalk with local cells involved in occlusive vascular lesions. NGFR has been reported to regulate neuronal survival and apoptosis. 37 The contrasting effects are due to the dependence of NGFR signaling on co-receptors and intracellular ligand availability. Further basic studies are needed to clarify the roles of circulating NGFR positive cells in the pathogenesis of PH.
This study had several limitations. First, because PAH is a rare disease, only 30 patients with PAH participated in the study. The small number of patients in this study is not uncommon; other studies have each included approximately 20 patients with PAH.29,30 Although our sample size was sufficient to identify crude trends among patients with PAH, the number of cases was not sufficient to allow assessment of the differences in PAH subcategories. We found that the frequencies of NGFR positive cells, mPAP, and major adverse events tended to be higher in the atypical PAH group, perhaps because these patients were older and had more complications such as heart and respiratory diseases. Further studies including greater numbers of patients are also required to identify factors that affect NGFR positive cell expression, such as subtypes and treatments of PAH. Furthermore, our subjects had mild-to-moderate PAH; studies including patients with severe PAH are required to facilitate more accurate prediction of survival and adverse events.
Second, only major markers of EPCs (CD34, VEGFR2, and CD133) and MSCs (NGFR, PDGFRα, and CD90) were investigated in this study. Recently, CD146, an MSC marker, has been shown to be involved with vascular remodeling and the progression of PAH in animal models. 38 In addition, the changes in single marker positive EPCs and MSCs did not directly reflect changes in EPC and MSC expression in PBMNCs. Further study is required regarding other representative markers of EPCs and MSCs, and the functions of marker positive cells in PAH.
Furthermore, this study compared only the BNP level with EPC and MSC markers. However, various other biomarkers (e.g. IL-6, osteopontin, and N-terminal propeptide of procollagen III) are also reportedly correlated with PAH. 2 Therefore, the abilities of EPC and MSC markers to predict the severity and prognosis of PAH should also be compared with the biomarkers mentioned above. Finally, although NGFR is reportedly expressed on the surface of MSCs, the present study did not demonstrate that circulating NGFR positive cells have properties of MSCs. Further studies are needed to investigate how circulating NGFR positive cells are involved in vascular remodeling in patients with PAH.
Conclusion
In conclusion, levels of circulating EPC and MSC markers in PBMNCs were elevated in patients with PAH. Among these markers, NGFR was the most strongly correlated with disease severity, and may be a prognostic factor for PAH. These results imply that circulating NGFR positive cells in PBMNCs may be a useful novel biomarker for severity and prognosis in patients with PAH.
Supplemental Material
sj-pdf-1-pul-10.1177_2045894021990525 - Supplemental material for Circulating nerve growth factor receptor positive cells are associated with severity and prognosis of pulmonary arterial hypertension
Supplemental material, sj-pdf-1-pul-10.1177_2045894021990525 for Circulating nerve growth factor receptor positive cells are associated with severity and prognosis of pulmonary arterial hypertension by Chiaki Goten, Soichiro Usui, Shin-ichiro Takashima, Oto Inoue, Hirofumi Okada, Masaya Shimojima, Kenji Sakata, Masaaki Kawashiri, Shuichi Kaneko and Masayuki Takamura in Pulmonary Circulation
Footnotes
Conflict of interest
The author(s) declare that there are no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Ministry of Education, Culture, Sports, Science and Technology Japan Society for the Promotion of Science; Grant-in-Aid for Scientific Research(C)/19K08486; Grant-in-Aid for Young Scientists (B)/19K17670.
Ethical approval
This study was approved by the Ethics Committee of Kanazawa Medical University (Kanazawa, Japan).
Authors’ contributions
CG performed the experiments, analyzed the data, and wrote the manuscript. SU designed the research studies and wrote the manuscript. SIT and OI provided technical support. HO and MS analyzed and interpreted the data. KS, MK, SK, and MT critically revised the manuscript with respect to the intellectual content. All authors read and approved the final manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
