Abstract
Spironolactone, a common diuretic used in the treatment of pulmonary arterial hypertension (PAH), improves cardiopulmonary hemodynamics by attenuating the adverse effects of hyperaldosteronism on endothelin type-B receptor function within pulmonary endothelial cells. Spironolactone has demonstrated vascular remodeling properties and reduced all-cause mortality in patients with severe heart failure. Despite widespread use, however, its effect on morbidity and mortality in PAH has not been fully explored. A large cohort of PAH patients from a harmonized dataset from four pivotal trials were analyzed to characterize the patient population and outcomes associated with spironolactone treatment. Of 1229 evaluable patients, 74% were female, mean age of 47 ± 15 years, baseline six-minute walk distance (6MWD) 345 ± 74 m and 75% were World health Organization (WHO) functional class (FC) III/IV. Of the patients receiving spironolactone, 43% were on subcutaneous treprostinil, compared to 29.9% of those patients who were not receiving spironolactone. Long-term spironolactone-treated PAH patients were older (p = 0.01), had lower baseline 6MWD (p = 0.01) and cardiac index (p < 0.01), higher baseline Borg dyspnea score (p = 0.01), WHO functional class, and right arterial pressure (p < 0.01). Additionally, spironolactone-treated patients tended to have higher hazard of clinical worsening (p < 0.01). A propensity analysis was performed and when controlled for disease severity, revealed that spironolactone-treated patients did not demonstrate improved outcomes in terms of survival and clinical worsening. Across studies, the current analysis has demonstrated that the actions of spironolactone within the context of the PAH disease process may differ from its action in CHF patients. Despite reported outcomes of spironolactone use in patients with left heart disease, these findings may not translate to right heart failure and pulmonary vascular pathology, as similar outcomes have not necessarily been recognized in the PAH patient population. Future studies are needed to explore these findings further.
Introduction
Pulmonary arterial hypertension (PAH) is a relatively rare, chronic, and progressive disorder characterized by vascular remodeling of the small pulmonary arteries, resulting in elevated pulmonary vascular resistance and right ventricular failure. 1 , 2 Symptoms of PAH include progressive dyspnea on exertion, fatigue, and exertional chest pain. 3 Despite the development and subsequent approval of PAH therapies, it remains an incurable and severe disease with a poor prognosis. 4
In addition to PAH-specific therapy, current management of patients with moderate to severe PAH and World Health Organization/New York Heart Association (WHO/NYHA) class III–IV symptoms includes diuretic therapy, in an attempt to keep patients in a relatively euvolemic state and delay the development of right heart failure.5–7 Spironolactone, a mineralocorticoid receptor (MR) and androgen receptor (AR) antagonist, is used in approximately one third of PAH patients’ treatment regimen as a diuretic and has also been shown to have vascular remodeling properties, improve endothelial function, and reduce inflammation. 8 , 9 Although spironolactone has been shown to improve heart failure and reduce mortality in patients with severe systolic heart failure, its long-term effect on morbidity and mortality in those with PAH has not been fully explored. 10
Endothelial dysfunction has a key role in the pathogenesis and progression of idiopathic PAH, and inflammation has been associated with driving these dysfunctional processes. 11 , 12 Therefore, targeting endothelial function and pulmonary vascular inflammation by MR and AR antagonism, with spironolactone, may deliver additional benefits to PAH patients.
Here, we describe a large cohort of PAH patients that were enrolled in four pivotal clinical trials, evaluating spironolactone use and outcomes, including clinical worsening and patient survival.
Methods
The objective of the current study was to characterize the patient cohort, including morbidity and mortality outcomes, associated with long-term spironolactone treatment. The primary outcome measures were patient survival and clinical worsening in idiopathic PAH patients. Clinical worsening events were defined as death, lung or heart–lung transplantation, atrial septostomy, hospitalization due to worsening PAH, initiation of new PAH-approved therapy, or worsening WHO functional class. Characterization of duration of spironolactone treatment as short or long-term, referred to the study period noted within the double-blind, placebo-controlled, randomized trials. “Short-term” was defined as 12 to 16 weeks, and “long-term” referred to the period included in the open label extension study. Time to clinical worsening was the time period beginning with the date of administration of the first dose until the date of the clinical worsening event, as defined by each trial protocol.
Study design
We conducted an analysis of a large cohort of patients (n = 1229) with PAH, integrated from four multicenter pivotal trials (PHIRST – Pulmonary Arterial Hypertension and Response to Tadalafil (subcutaneous treprostinil, tadalafil) and FREEDOM trials (oral treprostinil))13–17 (Table 1).
Studies included in analysis.
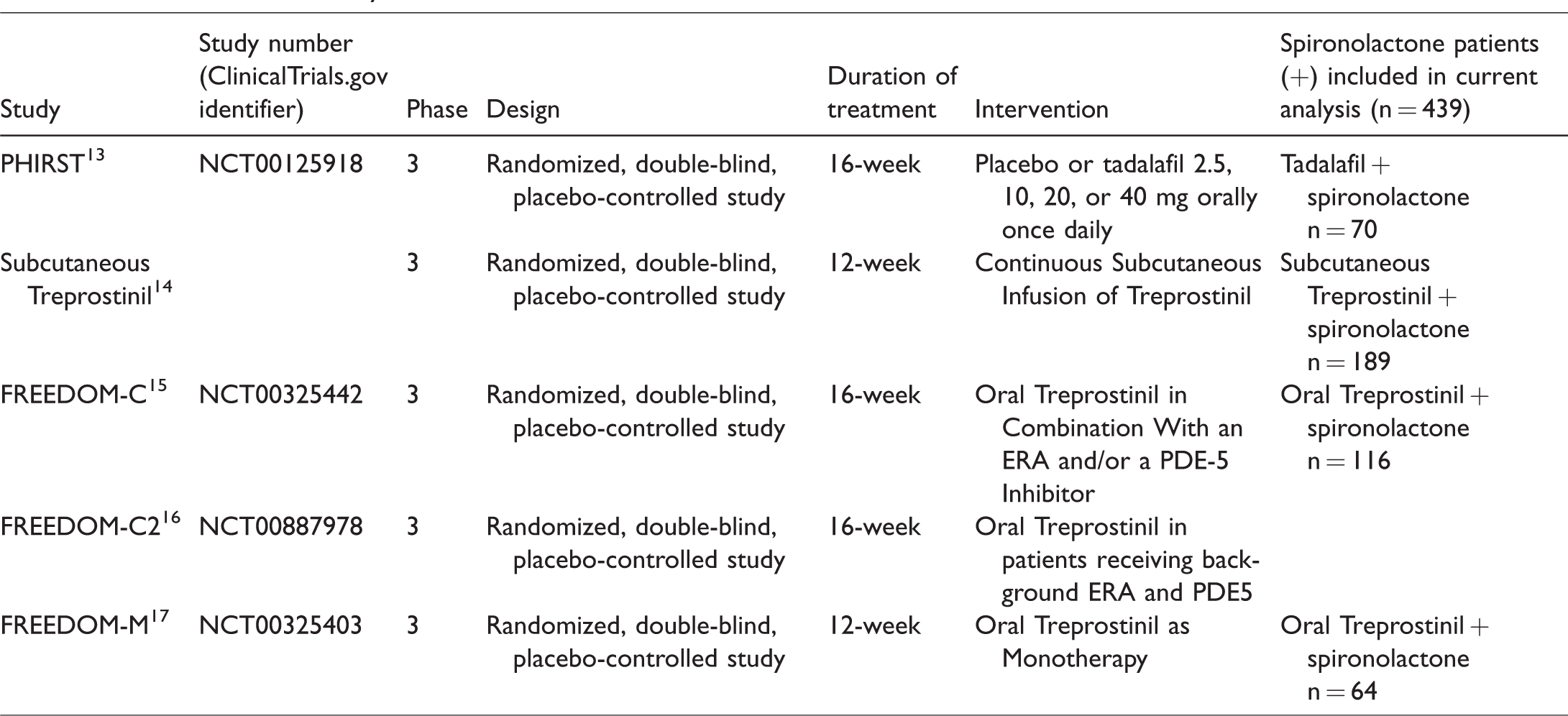
All studies were conducted with institutional review board or independent ethics committee approval and all patients provided written informed consent. All data was collected based on each study’s individual protocol and was anonymized when extracted for the current analysis.
Study sample
The analysis set was comprised of any patients that were included in the locked dataset for the PHIRST and FREEDOM trials (n = 1858). In order to limit the influence of confounders in the current analysis, any patients with an etiology other than idiopathic PAH were excluded, which resulted in 1229 patients being included, 439 that had received spironolactone, and 790 that had not.
Of the 1229 patients identified for inclusion, baseline demographics and disease characteristics, including hemodynamic data and spironolactone dosing were collected. Patients that received spironolactone and were also on background therapy (35.8%) were included in the analysis, and of these, 14.1% received ERA as background therapy, 8.0% received PDEi-5, and 13.7% were receiving both ERA and PDEi-5 background therapy. Of those patients that did not receive spironolactone, 39.9% were on background therapy, 16.1% having received an ERA as background therapy, 11.5% received PDEi-5, and 12.3% were receiving both ERA and PDEi-5 background therapy. The differences in background therapy between the two groups were not statistically significant. Outcome measures included assessment of clinical worsening, hemodynamic variables, and SF-36 (Physical and Mental component) scores.
Data management and statistical analysis
All baseline patient and disease characteristics were analyzed. Patient demographics and hemodynamics were expressed as means ± standard deviation, and categorical data were expressed as values and percentages. A two-sample Student’s t-test was used to compare means of continuous variables for normally distributed data between the two groups, while Wilcoxon’s rank-sum test was used for non-parametric distributions. T-tests were also used to determine if any trends exist in hemodynamic parameters at baseline and end of the study. Linear regression models, both simple and multivariate, were used to evaluate the association between short-term or long-term use of spironolactone and SF-36 (Physical and Mental component) scores. A log-rank test was used to compare survival times and times to clinical worsening between the two groups. Proportional hazard survival models, both simple and multivariate, were used to evaluate the association between short-term or long-term use of spironolactone and clinical worsening, as defined above.
For the multivariate model, potential confounders were identified and included age, gender, race, 6MWD, WHO/NYHA functional class, concomitant PAH medications, background therapy, potassium, sodium, and eGFR at baseline. Listings of comorbidities and dosing of non-PAH concomitant medications, including spironolactone, were not available within the dataset. For the combined population, active drug indicator (SC-TRE, tadalafil, oral treprostinil), background therapy (none, monotherapy endothelin receptor antagonist (ERA), phosphodiesterase-5 inhibitor (PDE5i), and combined therapy with ERA + PDE5i) were considered. For the purposes of analysis, bosentan in the PHIRST study population was categorized as an ERA.
A propensity score matching procedure was used to determine whether the probability of treatment assignment, or another potential unbalanced effect, is conditional on observed baseline characteristics.
The propensity score matching procedure contains several steps: (1) Run logistic regression between dependent variables and the covariates to be adjusted; (2) Calculate the estimated propensity score, which turned out to be the log odds; (3) Match each participant to one or more participants by propensity score using optimal fixed ratio matching with one matching unit and 0.9 caliper width; (4) Verify that the covariates are balanced across two groups on matched samples; (5) Carry out the analysis based on new matched samples.
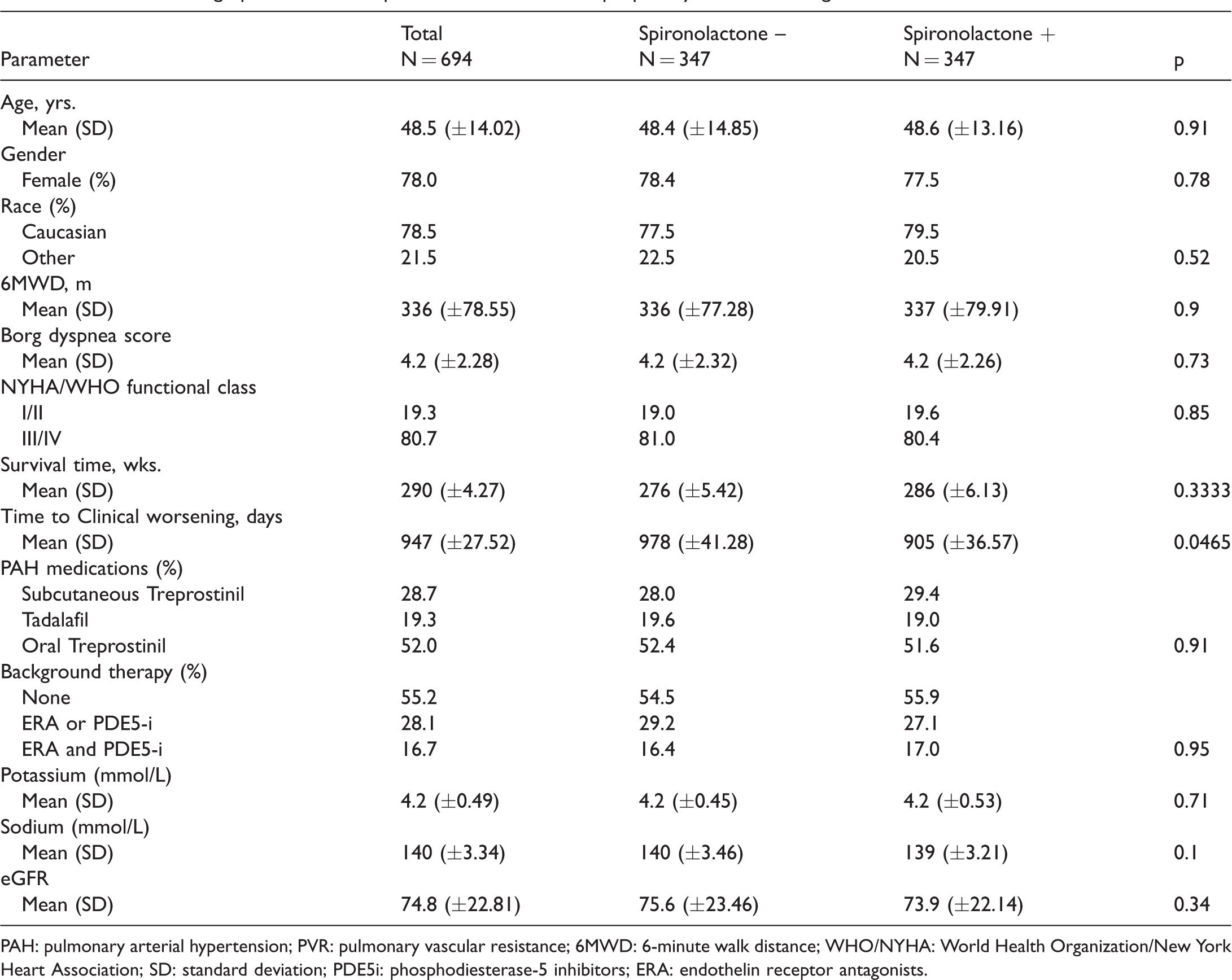
Propensity score matching was performed using the “proc psmatch” procedure in SAS. After propensity score matching, the total number of patients included was reduced to 694 (347 receiving spironolactone, 347 that had not received spironolactone), while the number of patients with non-missing baseline RAP was reduced to 231. Propensity score matching variables included baseline 6MWD, background therapy, NYHA functional class, and Borg dyspnea score. Multivariate and Univariate proportional hazard survival models were then used to evaluate the association between the use of spironolactone and clinical worsening.
Statistical significance was set at p < 0.05 and analyses were performed using the Statistical Analysis System (SAS) Enterprise Guide, version 8.3. SAS Institute Inc., Cary, NC, USA.
Results
Baseline characteristics
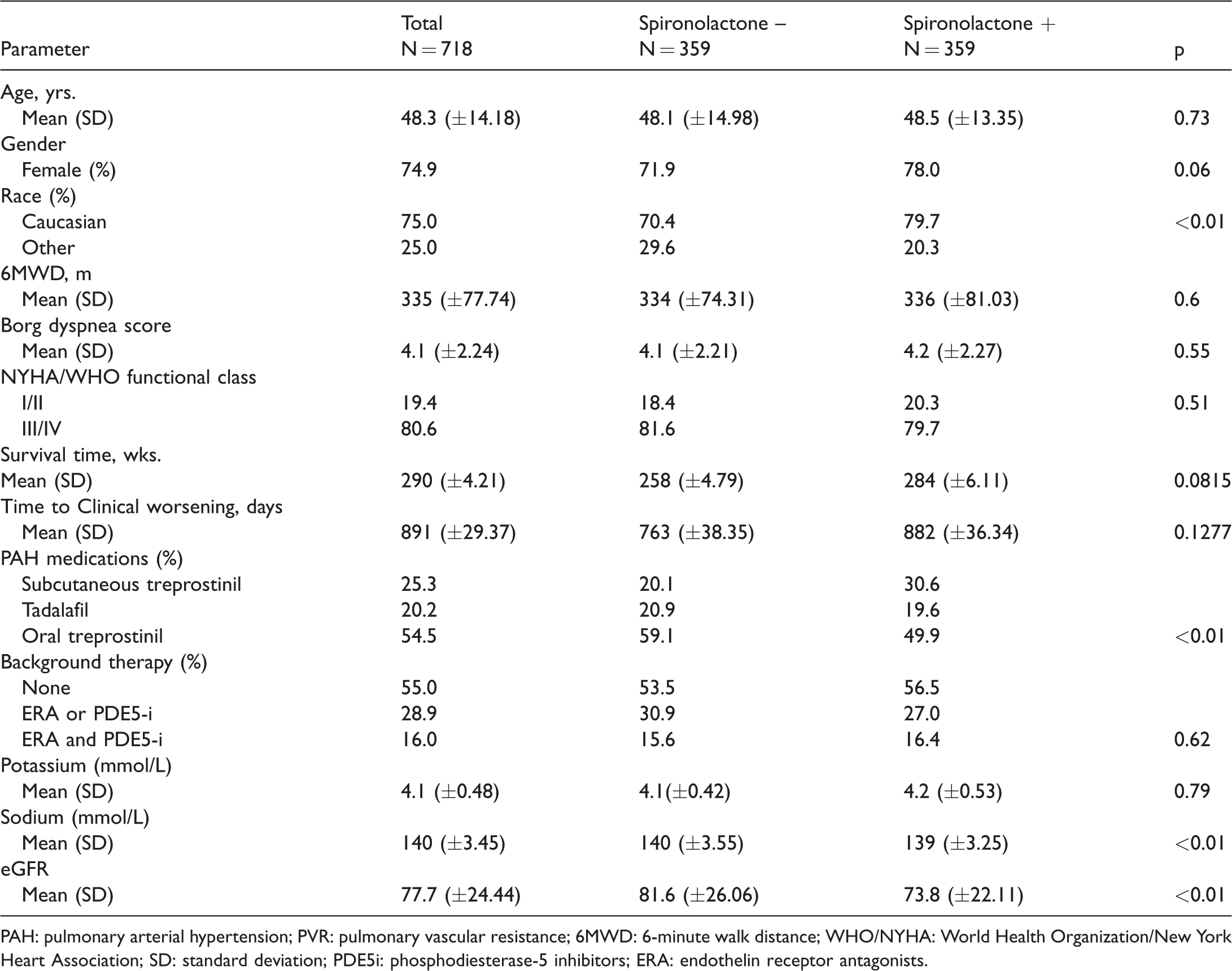
A total of 1229 PAH patients were included in the analysis, of which 439 were receiving spironolactone and 790 were not. Baseline demographic and clinical parameters are shown in Table 2. The mean patient age was 47 ± 15 years, with a median age of 47 years and 74% of patients were female. The mean 6MWD was 345 ± 74 meters and 75% were WHO/NYHA functional class III/IV at baseline. Comparison of the baseline and demographic characteristics demonstrated that patients that had been receiving long-term spironolactone therapy were older (p = 0.01), with lower baseline 6MWD (p = 0.01), higher baseline BORG dyspnea score (p = 0.01), and higher baseline WHO/NYHA functional class. Of the patients receiving spironolactone, 43% were on subcutaneous treprostinil, compared to 29.9% of patients who were not receiving spironolactone.
Baseline demographic and clinical parameters.
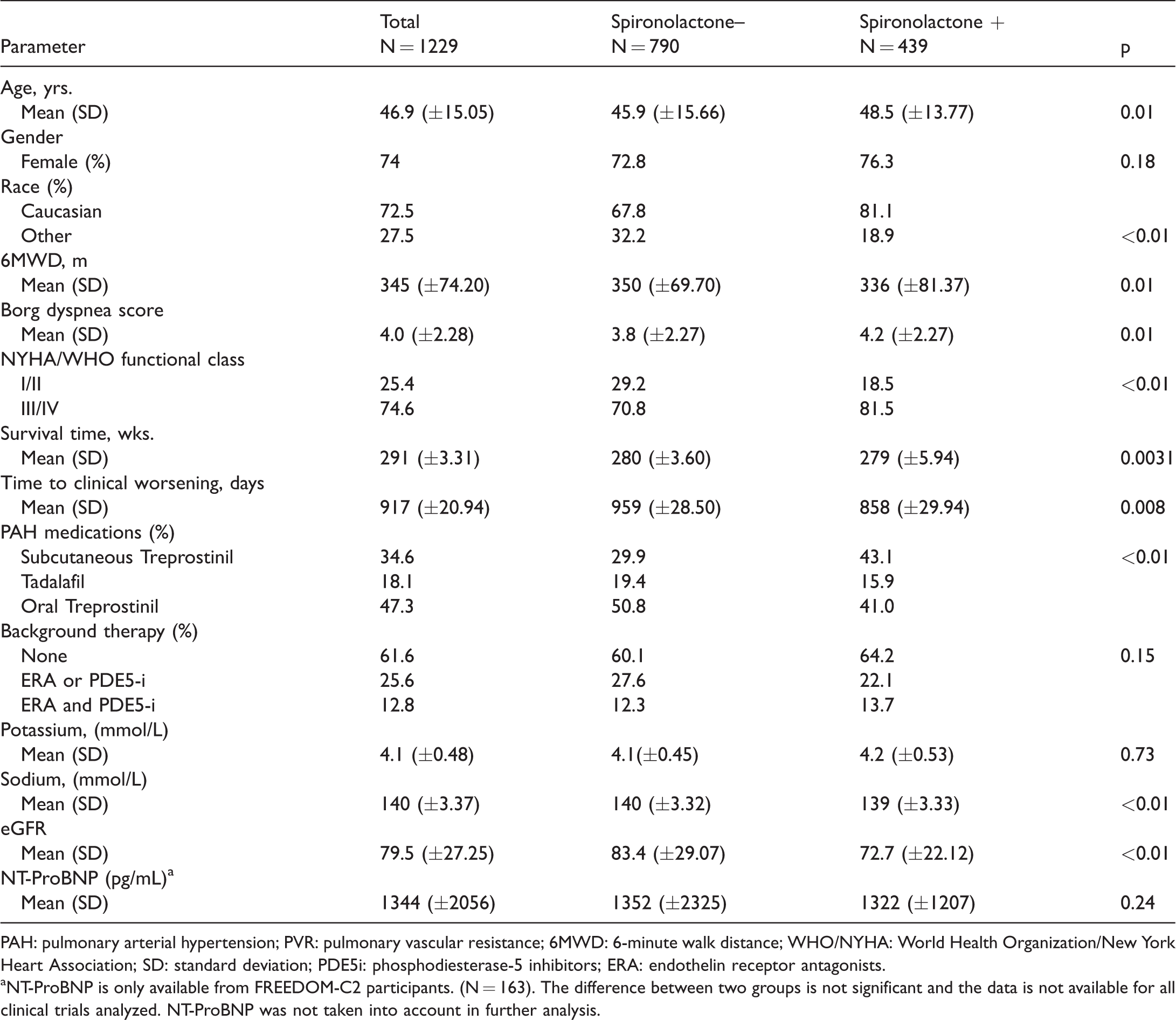
PAH: pulmonary arterial hypertension; PVR: pulmonary vascular resistance; 6MWD: 6-minute walk distance; WHO/NYHA: World Health Organization/New York Heart Association; SD: standard deviation; PDE5i: phosphodiesterase-5 inhibitors; ERA: endothelin receptor antagonists.
aNT-ProBNP is only available from FREEDOM-C2 participants. (N = 163). The difference between two groups is not significant and the data is not available for all clinical trials analyzed. NT-ProBNP was not taken into account in further analysis.
Hemodynamic data was available from patients that participated in the subcutaneous treprostinil infusion study and PHIRST. The hemodynamic parameters at baseline and at the end of the study period are shown in Table 3, as well as the calculated change in each parameter. At baseline, for the entire cohort, the mean pulmonary artery pressure (mPAP) was 58.4 mmHg, cardiac index (CI) was 2.3 ± 0.75 L/min/m2 and pulmonary vascular resistance (PVR) was 13.5 ± 6 Wood Units. Patients on long-term spironolactone therapy were shown to have significantly higher baseline right atrial pressure (RAP) (11.6 ± 5.21 vs. 9.1 ± 5.12 mmHg, p < 0.01) and significantly lower baseline CI (2.4 ± 0.75 vs. 2.1 ± 0.73 L/min/m2, p < 0.01). The patients receiving spironolactone demonstrated worsening in RAP and a trend towards improved CI at the end of the study period; however, this result was not statistically significant (Table 3).
Hemodynamic parameters at baseline and at the end of the study period.
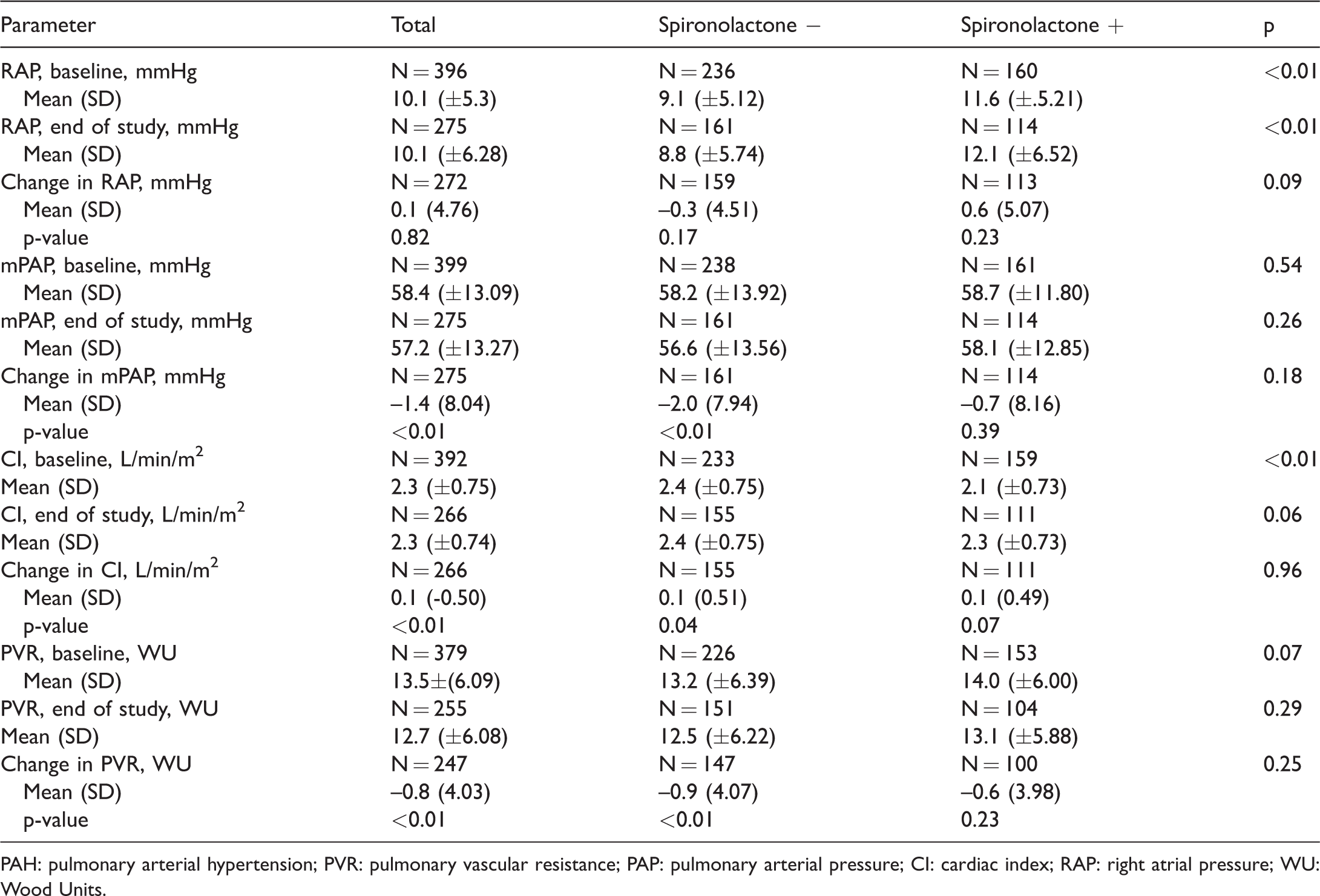
PAH: pulmonary arterial hypertension; PVR: pulmonary vascular resistance; PAP: pulmonary arterial pressure; CI: cardiac index; RAP: right atrial pressure; WU: Wood Units.
Quality of life
Univariate linear regression analysis of SF-36 (Physical and Mental component) scores indicated that there is no significant association between long-term spironolactone use and change in mental the component summary score (MCS) (p = 0.77) and physical component summary scores (PCS) (p = 0.54). Additionally, multivariate analysis demonstrated no difference in SF-36 (PCS and MCS) scores in patients on spironolactone, compared to those who were not (p = 0.70 and p = 0.95, respectively).
Clinical worsening and survival
Proportional hazard survival analysis indicated that patients receiving spironolactone tended to have poorer survival and an increased likelihood of clinical worsening than those patients who did not receive spironolactone (p < 0.01) (Fig. 1a–b).
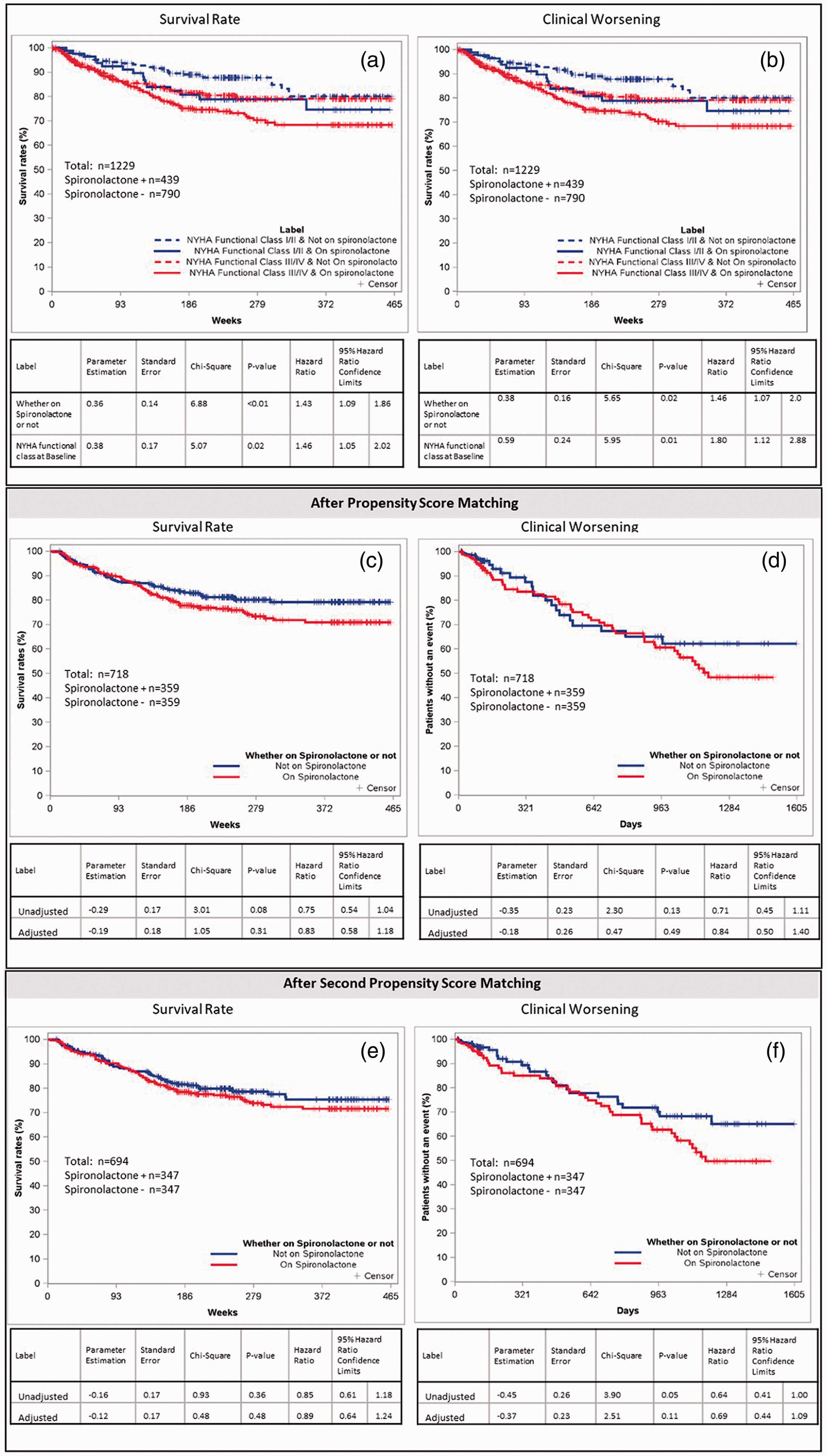
(a) Kaplan-Meier plot of patient survival (weeks) by spironolactone administration and NYHA functional class at baseline. (b) Kaplan-Meier plot of clinical worsening (days) by spironolactone administration and NYHA functional class at baseline. (c) Kaplan-Meier plot of patient survival (weeks) by spironolactone administration after propensity score matching. (d) Kaplan-Meier plot of clinical worsening (days) by spironolactone administration after propensity score matching. (e) Kaplan-Meier plot of patient survival (weeks) by spironolactone administration after second propensity score matching. (f) Kaplan-Meier plot of clinical worsening (days) by spironolactone administration after second propensity score matching.
The unadjusted hazard ratio for survival analysis was 0.674 (p = 0.0033) which was significant; however, the adjusted hazard ratio, 0.818, did not demonstrate statistical significance (p = 0.2045). The unadjusted hazard ratio for clinical worsening was 0.659 (p = 0.0084) was significant, while the adjusted hazard ratio was non-significant at 0.739 (p = 0.1688) (Table 4).
Unadjusted and adjusted hazard ratios for survival and clinical worsening.
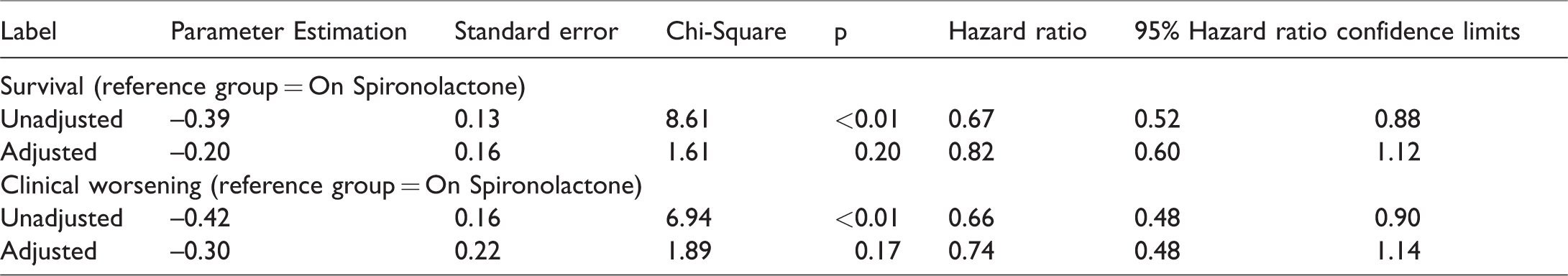
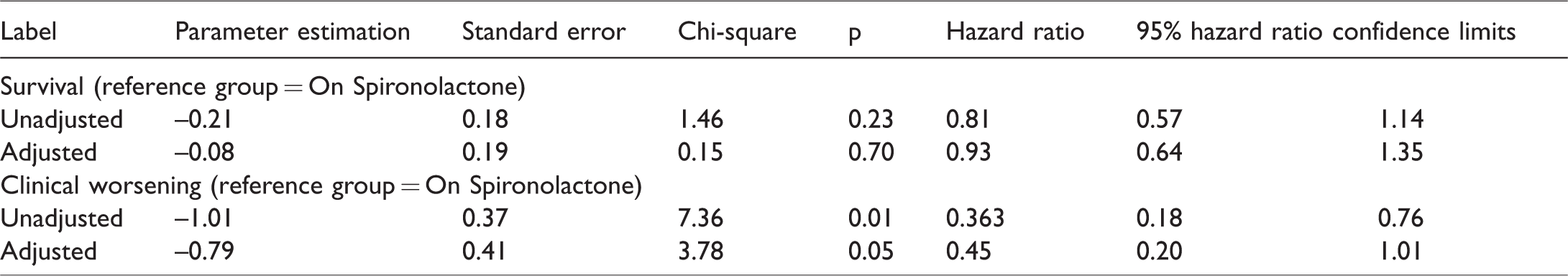
It may have been expected that the patients included in the study of subcutaneous infusion of treprostinil may have been sicker; therefore, a sensitivity analysis that excluded this group was performed. Patients that were treated with only oral therapy, with or without spironolactone, demonstrated statistically significant differences in the time to clinical worsening, based on the unadjusted hazard ratio (Table 5) (p = 0.01); however, the adjusted hazard ratio for clinical worsening was not significant, although it neared significance (p = 0.05) and in terms of survival, the differences were no longer significant.
Unadjusted and adjusted hazard ratios for survival and clinical worsening excluding subcutaneous treprostinil.
A subgroup analysis, stratified patients by WHO functional class, showed poorer survival and increased likelihood of clinical worsening for those receiving spironolactone within WHO functional class III/IV (p = 0.03 and 0.04, respectively, Fig. 1a–b).
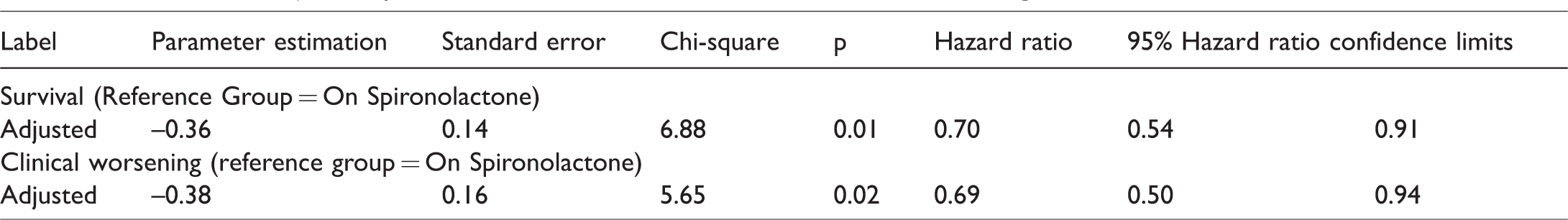
Additionally, when looking at hazard ratio adjusted by WHO functional class (class I/II vs. III/IV), those patients receiving spironolactone had poorer survival, and an increased likelihood of clinical worsening (p < 0.01 and 0.02, respectively) (Table 6).
Hazard ratios adjusted by WHO functional class for survival and clinical worsening.
After propensity score matching
A propensity analysis was conducted to ascertain the effect of spironolactone treatment on outcomes by starting with a balanced group. Baseline demographic and clinical parameters after propensity score matching are shown in Table 7. The analysis showed no statistically significant difference between the groups; however, there was a trend toward worse outcomes in the spironolactone treatment group. The hazard ratios for the survival rate analyses were not statistically significant. The unadjusted hazard ratio for survival analysis was 0.748 (p = 0.0826) and the adjusted hazard ratio was 0.831 (p = 0.3050) (Fig. 1c). Similarly, hazard ratios for clinical worsening were also not statistically significant, with an unadjusted hazard ratio of 0.707 (p = 0.1297) and the adjusted hazard ratio was 0.835 (p = 0.4913) (Fig. 1d).
Baseline demographic and clinical parameters after propensity score matching.
PAH: pulmonary arterial hypertension; PVR: pulmonary vascular resistance; 6MWD: 6-minute walk distance; WHO/NYHA: World Health Organization/New York Heart Association; SD: standard deviation; PDE5i: phosphodiesterase-5 inhibitors; ERA: endothelin receptor antagonists.
After second propensity score matching
A second propensity score analysis was implemented in order to adjust for sodium level and eGFR as well as the degree of disease severity. Baseline demographic and clinical parameters after the second propensity score matching are shown in Table 8. The analysis implied that with further control of sodium levels and eGFR, the curve for the survival time and the time to clinical worsening between two groups, those receiving spironolactone and those who were not, were no longer significantly different. Also, the spironolactone use indicator was no longer a significant variable within the Cox proportional hazards regression.
Baseline demographic and clinical parameters after second propensity score matching.
PAH: pulmonary arterial hypertension; PVR: pulmonary vascular resistance; 6MWD: 6-minute walk distance; WHO/NYHA: World Health Organization/New York Heart Association; SD: standard deviation; PDE5i: phosphodiesterase-5 inhibitors; ERA: endothelin receptor antagonists.
The hazard ratios for the survival rate analyses following the second propensity score matching were not statistically significant. The unadjusted hazard ratio for survival analysis was 0.851 (p = 0.3354) and the adjusted hazard ratio was 0.888 (p = 0.4863) (Fig. 1e). Finally, hazard ratios for clinical worsening were also not statistically significant, with an unadjusted hazard ratio of 0.641 (p = 0.0484) and the adjusted hazard ratio was 0.692 (p = 0.1130) (Fig. 1f).
Discussion
Spironolactone, commonly prescribed for use as a diuretic in PAH treatment, competitively binds to the MR, a ligand-activated transcription factor present in distal convoluted renal tubes, is activated by aldosterone and induces diuresis. Takeda et al. determined that following a myocardial infarction in rats, expression of MR mRNA and also of 11β-HSD2 mRNA were significantly upregulated and that spironolactone, as an aldosterone antagonist, inhibited myocyte apoptosis, and improved post-infarct ventricular remodeling by modulating the MR-mediated aldosterone signaling. 18
In humans, spironolactone also has multiple effects on vascular remodeling. Administration of spironolactone over a period of one-month improved forearm blood flow in response to acetylcholine in chronic heart failure (CHF) patients, indicating an improvement in endothelial dysfunction. MacDonald et al. examined whether spironolactone, in combination with ACE inhibitors, would have favorable effects in 43 less severe (NYHA class I-II) CHF patients, over a three-month period. They concluded that spironolactone improved vascular function (endothelial function, vascular ACE activity) and other markers of prognosis in asymptomatic or mild CHF, already receiving optimal treatment with β blockade. 10 Furthermore, the circulating concentration of N-terminal propeptide of procollagen type III (PIIINP), a marker of vascular collagen turnover, was reduced by spironolactone in stable CHF patients. 19 , 20
Our group had previously demonstrated a significant correlation between PIIINP levels and PAH disease severity, as well as a negative correlation between cardiac index and 6MWD. 21 We have also shown that PIIINP is strongly related to health-related quality of life scores in PAH patients, further suggestive of its usefulness as a determinant or confirmation of disease severity. 22 Recently, we published results of a cross-over placebo-controlled clinical trial of PAH patients that received spironolactone and placebo in a randomized order for a minimum of eight weeks. 23 The primary objective of the study was to detect changes in collagen biomarker levels in this population, specifically looking at PIINP levels. Although treatment with spironolactone in this population was found to be safe and well tolerated, no significant changes in collagen biomarkers, PIIINP levels, or improvements in PAH disease parameters were observed. 23 The lack of changes observed could have been attributed to the short duration of treatment exposure or perhaps spironolactone acts differently in PAH than observed in patients with CHF. 23
The Randomized Aldactone Evaluation Study (RALES) demonstrated that the addition of low-dose spironolactone to an angiotensin-converting enzyme (ACE) inhibitor and loop diuretic for 12 weeks improved heart function in severe systolic heart failure patients and treatment for 24 months reduced morbidity and mortality, despite normal circulating aldosterone levels. 24 , 25 It is proposed that the effects of aldosterone vary in relation to the underlying oxidative states and that in cases of pathological vascular conditions, accompanied by high oxidative stress, aldosterone receptor blockage may be beneficial. Following the publication of results from the RALES trial, Juurlink et al. published their examination of trends in the rate of spironolactone prescriptions and the rate of hospitalization for hyperkalemia. 24 , 26 They noted that in addition to the increased rate of prescriptions for spironolactone following RALES, there was also an increase in hyperkalemia-associated morbidity and mortality. 26 If it had been available within the current dataset, analysis, adjusting for potassium and renal function, would have been interesting to determine if these parameters are potential mediators of worse outcomes in spironolactone users.
We previously investigated the plasma aldosterone levels of 125 PAH patients and determined that although the aldosterone level was not associated with increased mortality in PAH, it was a marker of disease severity. 27 Interestingly, the patients in that study had the contradictory finding that those on spironolactone had a higher RAP than those who were not. This finding may indicate that those receiving spironolactone may have worse disease. In a retrospective analysis of the ARIES study, spironolactone was shown to be beneficial with improved hemodynamics. 28 However, it is important to note that the 10 patients in the ambrisentan + spironolactone group had higher WHO/NYHA functional class and worse 6MWD than the group receiving only ambrisentan. Ambrisentan is an endothelin A receptor blocker, and PAH patients have demonstrated increased aldosterone levels; hence, the addition of an aldosterone receptor antagonist may be expected to mitigate otherwise adverse clinical outcomes.
Studies have shown that spironolactone also has anti-fibrotic effects. de Man et al. assessed systemic and pulmonary renin-angiotensin-aldosterone system (RAAS) activity from blood samples of 58 idiopathic PAH patients. They hypothesized that these patients have low cardiac output and in order to compensate, neurohormonal systems like RAAS and the sympathetic nervous system are upregulated, having negative long-term effects on the progression of idiopathic PAH. It was found that both systemic and pulmonary RAAS-activities, elevated renin, angiotensin I and angiotensin II activity, were found in PAH patients and associated with increased pulmonary vascular remodeling and disease progression. Notably, 26 (45%) of the 58 patients included were receiving spironolactone therapy; however, it is not clear how this contributed to the increased RAAS activity. In a study of aldosterone levels in acutely decompensated heart failure, high-dose spironolactone added to standard therapy induced increases in renin and aldosterone levels. 29
In this present study of PAH patients enrolled in clinical trials, spironolactone was prescribed in 36%, which aligns with other studies in which spironolactone had been used in approximately 20–30%. 5 We noted a minimally higher proportion of patients receiving spironolactone that were being treated with subcutaneous treprostinil (43%) as compared to oral treprostinil (41%), although we are uncertain about the interpretation of this finding. By excluding patients that were receiving subcutaneous infusion of treprostinil, our results still showed shorter time to clinical worsening event; however, patient survival rates were not influenced.
Recently, Lahm et al. published their retrospective study of the Veterans Affairs CART Database, investigating relationships between angiotensin converting enzyme inhibitors, angiotensin receptor blockers, and aldosterone antagonists and mortality in pulmonary hypertension. 30 They reported that ACEI/ARB use was associated with lower mortality in their population, noting that aldosterone antagonist use was a marker of disease severity in pulmonary hypertension. This aligns with previously published data and with our current findings. Interestingly, spironolactone was more commonly used in patients with poorer cardiac output, and as expected, these patients experienced more clinical worsening. We speculate that this may be because these patients have severe disease and were therefore prescribed spironolactone as a diuretic. Following completion of our propensity analysis, even when we controlled for the degree of disease severity, patients receiving spironolactone did not demonstrate survival benefit or delayed clinical worsening. This suggests that the protective effects of spironolactone observed in patients with left heart failure may not translate to patients with right heart failure and pulmonary vascular pathology.
It may be expected that targeting endothelial function and pulmonary vascular inflammation by MR and AR antagonism with spironolactone may deliver additional benefits to PAH patients. Despite the results reported with spironolactone use in patients with left heart disease, similar outcomes have not necessarily been recognized in the PAH patient population. As supported by the current analysis, the actions of spironolactone within the context of the PAH disease process may differ from its action in CHF. Our findings should be examined within the context of several potential limitations. Propensity score matching relies on matching patients based on known variables and those available to use within each of the study datasets. Despite robust propensity-matching and multiple sensitivity analyses, other differences may exist between matched populations that cannot be observed, which may cause some degree of unmeasured, residual confounding. Also, not all patients were able to be matched within this analysis, reducing the number of patients in matched groups. Additionally, a potential weakness of the study may be due to its retrospective nature, which did not permit for any clarification or alternative characterization or classification (e.g. comorbidities) from the original datasets. Another limitation of this dataset was that within the conduct of the original studies, dose ranges for spironolactone were not collected, as it was regarded as a concomitant medication rather than a “medication of interest,” as well, BNP was only available for participants in the FREEDOM-C2 study.
The studies included in the analysis were conducted over different periods of time; therefore, the analysis may be confounded by potential differences in standard of care management over the time periods in which the studies were conducted. Finally, extending our findings from the analysis of patients participating in controlled, randomized trials, to mimic real-world clinical decision-making during the course of PAH treatment will require further study.
Footnotes
Consent to participate
All participants provided written informed consent to be included in pivotal studies that were analyzed in the current study.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Ethical approval
Pivotal trials included in current analyses were conducted with institutional review board or independent ethics committee approval.
Guarantor
Not applicable.
Authors’ contribution
Conception and design: ZS; analysis and interpretation: ZS and EC; drafting of the article: ZS.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
