Abstract
There are limited data regarding the feasibility of transitioning from intravenous prostacyclins to selexipag in pulmonary arterial hypertension patients. We present a case series of successful transitions from intravenous prostacyclins to selexipag in the majority of carefully selected five stable pulmonary arterial hypertension patients using a standardized protocol in the outpatient setting.
Keywords
Introduction
Our center uses the United States Registry to Evaluate Early and Long-term pulmonary arterial hypertension (PAH) disease management (REVEAL) risk score calculator 2.0 as part of multi-faceted risk assessment to help guide PAH treatment because it has better risk discrimination ability compared with other PAH risk assessment scores.
1
High-risk PAH patients (REVEAL score 2.0 ≥ 10) are treated upfront with combination therapy including intravenous (IV) prostacyclin therapy, while low risk (REVEAL score 2.0 ≤
However, limited data are available for the transition to selexipag in patients in whom parental prostacyclin was initially indicated.4–6 We have previously reported our successful initial experience of transition from IV prostacyclin to oral selexipag using implantable wireless pulmonary artery pressure (PAP) monitoring. 7 The current study is a single-center case series detailing the feasibility of transition in carefully selected patients using an outpatient transition protocol at a tertiary care PAH referral center.
Case description
Case 1
A 24-year-old woman with idiopathic pulmonary arterial hypertension (iPAH) was diagnosed in April 2017 at the age 21 in the setting of syncope. Her REVEAL score was 8 and World Health Organization functional class (WHO FC) III symptoms. She was started on IV epoprostenol, titrated to 36 ng/kg/min, ambrisentan 10 mg daily, and tadalafil 20 mg daily. Repeat risk assessment in September 2017 suggested low risk (REVEAL score 3) and normal right ventricle (RV) systolic function (Table 1).
Summary of the patient’s characteristics.
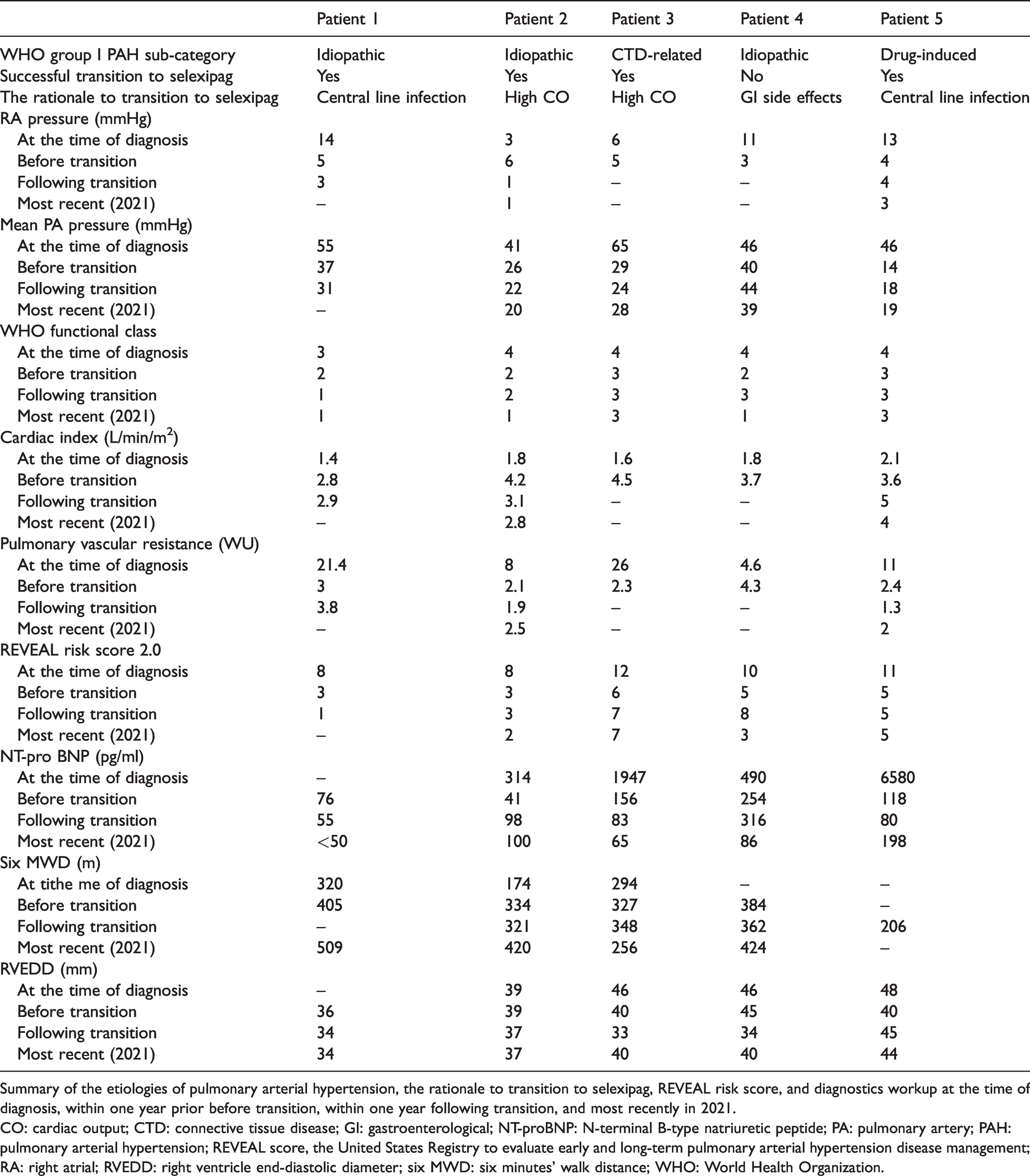
Summary of the etiologies of pulmonary arterial hypertension, the rationale to transition to selexipag, REVEAL risk score, and diagnostics workup at the time of diagnosis, within one year prior before transition, within one year following transition, and most recently in 2021.
CO: cardiac output; CTD: connective tissue disease; GI: gastroenterological; NT-proBNP: N-terminal B-type natriuretic peptide; PA: pulmonary artery; PAH: pulmonary arterial hypertension; REVEAL score, the United States Registry to evaluate early and long-term pulmonary arterial hypertension disease management; RA: right atrial; RVEDD: right ventricle end-diastolic diameter; six MWD: six minutes’ walk distance; WHO: World Health Organization.
In 2018, she developed recurrent methicillin-susceptible Staphylococcus aureus bacteremia due to central line infections. Given the recurrent and severe central line infections, she was transitioned to selexipag in April 2018. The epoprostenol dose was down-titrated, and selexipag was up-titrated using our outpatient protocol shown in Table 2. She tolerated selexipag 1600 mg twice daily without significant side effects. A year later, the REVEAL score was 1 and the right ventricle ejection fraction (RVEF) of 56%.
Conversion protocol from intravenous prostacyclin to selexipag.
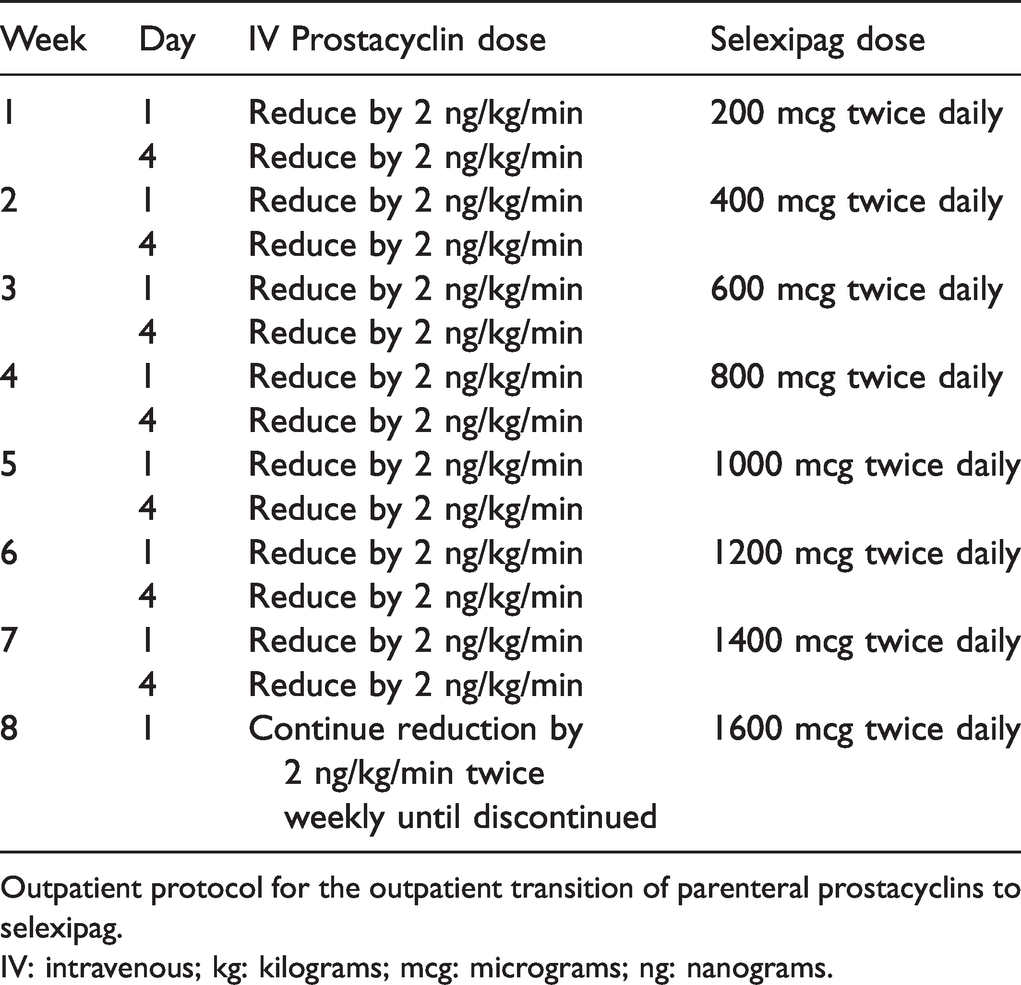
Outpatient protocol for the outpatient transition of parenteral prostacyclins to selexipag.
IV: intravenous; kg: kilograms; mcg: micrograms; ng: nanograms.
Case 2
A 48-year-old man was diagnosed with iPAH in December 2014 at the age of 42. REVEAL score was 8 and RVEF 36%. He received dual therapy of IV treprostinil 43 ng/kg/min and sildenafil initially, which was later switched to riociguat 2.5 mg three times daily with subsequent improvement in his functional capacity. Risk assessment was repeated in November 2016 which suggested the REVEAL risk score was 3 and RVEF was 60%. Given the high cardiac output (CO), the treprostinil dose was weaned by 2 ng/kg/min every week to 20 ng/kg/min with no significant change in hemodynamics or functional capacity. Hence, selexipag 200 mcg twice daily was initiated in February 2017 and was titrated to a total dose of 1600 mcg twice daily while weaning off treprostinil using our outpatient protocol. Repeat risk assessment in February 2018 showed a low REVEAL score of 3 and a normal RVEF of 65%.
Case 3
A 59-year-old woman with PAH related to scleroderma was diagnosed in October 2014 in the setting of right-sided heart failure along with concomitant interstitial lung disease with diffusing capacity for carbon monoxide (DLCO) 25% predicted. She was at high mortality risk (REVEAL score of 12) and RVEF was 29%. She was treated with triple therapy of IV treprostinil 47 ng/kg/min, ambrisentan 10 mg daily, and tadalafil 40 mg daily. Risk assessment was repeated in April 2015; REVEAL score was 6 and RVEF was 56%. CardioMEMSTM was implanted for remote wireless PAP monitoring as part of an ongoing national heart, lung, and blood institute (NHLBI)-funded pilot study of this technology in PAH.
Like the previous case, IV treprostinil was gradually weaned to 20 ng/kg/min, given the high CO as an outpatient, and selexipag was up-titrated to 1600 mcg twice daily. She tolerated full-dose selexipag without side effects. PAP was monitored throughout the transition process and remained ∼37/17 (24) mmHg. REVEAL score one year later was 7 and RVEF was 56% (Table 1).
Case 4
A 51-year-old woman had iPAH diagnosed in 1999. At that time, the REVEAL risk score was 10 and moderately reduced RV systolic function. She has been treated with bosentan 125 mg twice daily, IV treprostinil 26 ng/kg/min, and tadalafil 40 mg daily and improved her functional capacity. In August 2014, she underwent CardioMEMSTM placement as part of the NHLBI-funded pilot study as detailed above. Repeat risk assessment at that time showed a REVEAL score of 5 and RVEF of 50% (Table 1).
Given the severe and recurrent treprostinil-related gastrointestinal side effects, she was transitioned to selexipag at the beginning of August 2016 while monitoring PAP using the remote monitoring device. The dose of IV treprostinil was gradually weaned down to 16 ng/kg/min. However, her side effects continued, so she was transitioned to selexipag using our outpatient protocol (Table 2). However, at a dose of 16 ng/kg/min, she was hospitalized for right-sided heart failure with WHO FC III and moderate RV systolic dysfunction. Wireless PAP monitoring showed PAP 67/31 (44) mmHg, and her REVEAL score was 8 during the hospital stay; as she failed to transition from IV treprostinil to selexipag, selexipag was down-titrated. IV treprostinil was up-titrated to 29 ng/kg/min while in the hospital. On follow-up six months later, REVEAL score was 5, RV systolic function was normal, and PAP was 76/40 (54) mmHg on CardioMEMSTM.
Case 5
A 75-year-old woman with drug-induced PAH was diagnosed in October 2012 with concomitant chronic hypoxemia in the absence of obstructive or restrictive lung disease. REVEAL score was 11, WHO FC IV, and RVEF 29% at the time of diagnosis. She was treated with triple therapy of IV treprostinil 36 ng/kg/min, ambrisentan 10 mg daily, and tadalafil 40 mg daily. Repeat risk assessment in May 2014 showed a low REVEAL risk score of 5 and RVEF of 60% (Table 1).
Transitioning to selexipag began in September 2016 following recurrent cellulitis at the site of her central line. Using our outpatient protocol, selexipag was up-titrated initially to 1200 mcg twice daily then subsequently decreased to 600 mcg twice daily due to tremors and palpitations. Risk assessment following the transition in December 2017 showed a REVEAL score of 5 and RVEF of 67%.
Discussion
We report a series of five cases of transition from IV prostacyclins to oral selexipag using a protocol specified for the outpatient setting that includes slow down-titration of IV prostacyclin and up-titration of selexipag with mean follow-up time of 4.9 ± 0.9 years. All patients were WHO FC II or III with relatively low REVEAL risk profile at the time of transition. Four patients were successfully transitioned to selexipag in the setting of central line complications or high CO, and one patient with intractable adverse gastrointestinal effects clinically deteriorated during the transition, and IV treprostinil was restarted. We only had one patient who was on IV epoprostenol at the time of transition. Hence, we are unable to draw conclusions in comparing differences between transitioning from IV treprostinil or IV epoprostenol.
Patients who remain at high risk of one-year mortality (REVEAL score 2.0 > 10) should not be transitioned to selexipag, and worsening REVEAL score 2.0 after transition implies the need to reintroduce IV prostacyclin therapy. Our patients had moderate or low REVEAL scores 2.0 and relatively low IV prostacyclin doses, with most patients receiving less than 40 ng/kg/min before transition. Previous studies have shown that in some patients who fail transition from IV prostacyclins to oral therapy, they cannot be rescued by re-initiation of parenteral prostacyclins. 8 While case 4 failed transition to IV prostacyclin, she continues to do well without recent hospitalization following IV prostacyclin re-initiation, with a low-risk REVEAL score on the yearly assessment and lower mean pulmonary artery pressure of 39 mmHg on CardioMEMSTM.
This protocol was based on our clinical experience of both IV and oral prostacyclins tolerability and safety. Given that parenteral prostacyclins and selexipag are generally adjusted on a weekly to twice weekly basis, we decided upon once weekly selexipag dose adjustments with twice weekly prostacyclin dose adjustments. Of note, dual therapy of IV prostacyclins and selexipag was not associated with a higher incidence of adverse events.
Routine right heart catheterization (RHC) was not part of our protocol post transition in patients with CardioMEMSTM because we have demonstrated previously that monitoring PAH therapy using CardioMEMSTM is safe and feasible. 9 In addition, cases 3 and 4 were part of NHLBI-funded pilot study in which we did have access to a proprietary CardioMEMSTM-derived stroke volume and CO algorithm, which allowed calculation of total pulmonary resistance (TPR), a close surrogate of pulmonary vascular resistance (PVR) in patients with normal wedge pressure. Thus, we were able to monitor CO and TPR in close to real-time during the transition. Unfortunately, the CO, TPR data were not available retrospectively for inclusion in the manuscript. In addition, serial PAP measurements as measured by CardioMEMSTM could provide some insight into changing PVR and CO. Finally, patient resistance to having an invasive RHC amongst those with a previously implanted CardioMEMSTM device also dictated some of the protocol.
To date, there have been four case reports that have suggested a protocol for the transition from parenteral prostacyclin to selexipag with a high success rate similar to our protocol.4–7 Our group published an initial case series in February 2019 reporting the feasibility of transitioning from IV prostacyclin to oral selexipag using indwelling hemodynamic monitoring in patients treated with low-dose IV prostacyclin. 7 Attempt to transition was successful in two patients, while the third patient (case 4 in our case series) was transitioned back to IV prostacyclin. Here, we expand upon our entire center’s experience.
Similar protocols for transition have been published by Holthaus et al. of five patients with 100% success rate and Parikh et al. of 14 patients with 93% success rate.5,6 These patients were transitioned in a hospital setting and on higher doses of IV prostacyclins at the time of transition. While these two studies did not calculate the REVEAL score at the time of transition, authors have closely monitored its components during and after the transition. Yanaka et al. have reported a successful transition to oral selexipag in eight patients; all patients were at low risk when assessed using the French invasive and non-invasive approaches and the Swedish Pulmonary Arterial Hypertension Register (SPAHR)/Comparative, Prospective Registry of Newly Initiated Therapies for Pulmonary Hypertension (COMPERA) method. 4
This emphasizes that successful transition to selexipag is possible and feasible when patients selected for transition are at low risk of cardiac adverse events at the time of transition and closely followed during the transition period and immediately after transition. This is a single-center experience with a small number of patients at a tertiary care PAH referral center.
Conclusion
We describe a protocol for the outpatient transition of carefully selected patients with PAH from parenteral prostacyclins to selexipag when indicated by either complication of the central line used for continuous prostacyclin therapy, high CO on a small dose of IV prostacyclin, or intolerable side effects due to IV prostacyclin. This case report adds to current literature regarding the feasibility of transition to selexipag in highly selected cases with low-risk REVEAL scores. However, further research is required to validate the results of this case series.
Footnotes
Authors’ contributions
The authors confirm contribution to the paper as follows: study conception and design: Nael Aldweib, Nathan J. Verlinden, Hayah Kassis-George, Amresh Raina; data collection: Nael Aldweib, Nathan J. Verlinden; analysis and interpretation of results: Nael Aldweib, Nathan J. Verlinden, Hayah Kassis-George, Amresh Raina; draft manuscript preparation: Nael Aldweib, Nathan J. Verlinden, Hayah Kassis-George, Amresh Raina. All authors reviewed the results and approved the final version of the manuscript.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
