Abstract
Few meta-analyses evaluated the efficacy and safety of pulmonary vasodilators in patients with Eisenmenger syndrome. Recently, some studies have reported conflicting results regarding improvements in exercise capacity. This study evaluated the efficacy and safety of pulmonary vasodilators in patients with Eisenmenger syndrome. Relevant studies were identified by searching major databases. Pooled outcomes were used to assess the efficacy and safety of pulmonary vasodilators. In total, five studies with 508 patients were included. Meta-analysis indicated that the pulmonary vasodilators reduced the mortality (odd risk (OR) = 0.35; 95% CI, 0.13 to 0.95;
Introduction
Eisenmenger syndrome (ES) is the most advanced form of pulmonary arterial hypertension (PAH) associated with congenital systemic‐to‐pulmonary shunts leading to a severe increase in pulmonary vascular resistance and a reversed (pulmonary‐to‐systemic) or bidirectional shunt flow. 1 Clinically, ES is a multisystem disorder associated with a large number of complications, which greatly affect the functional capacity, quality of life, and survival of patients. The only definitive treatment for ES is heart–lung transplantation. 2 However, the general lack of organs and the survival rates of 80% at 30 days and 70% after one year underline the need for alternative therapeutic option. 3
In the past, treatment for patients with ES has given priority to palliative and supportive management options. At present, pulmonary vasodilators have been introduced to treat patients with ES. Pulmonary vasodilators are mainly classified into three pharmaceutical groups: endothelin receptor antagonists (ERAs), phosphodiesterase-5 inhibitors (PDE-5i), and prostanoids. Riociguat, a soluble guanylate cyclase stimulator targeting the same pathway as PDE-5i, is also available.
A number of pulmonary vasodilators are used in clinical practice to treat patients with ES, including ERAs, PDE-5i, and prostanoids. Four randomized clinical trials (RCTs) 4–7 and prospective, open-label, and retrospective studies previously investigated the efficacy of pulmonary vasodilators and reported an improvement in 6-min walk distance (6MWD). However, the latest MAESTRO study 8 (Macitentan in Eisenmenger Syndrome to Restore Exercise Capacity), another randomized, double-blind, placebo-controlled, multicenter study that investigated a pulmonary vasodilator in ES patients, showed a different insight that Macitentan has no superiority over placebo on the primary end point of change from baseline to week 16 in exercise capacity in patients with ES. In addition, other studies obtained conflicting results on the efficacy of pulmonary vasodilators in improving the systemic oxygen saturation (SO2)4–6,8 and mortality6,9–15 of patients with ES. In addition, the relatively small number of patients enrolled in each study may be a limiting factor for the evaluation of the efficacy and safety of the procedure. Therefore, we sought to evaluate the efficacy and safety of pulmonary arterial hypertension-targeted drug (pulmonary vasodilators) therapy in patients with ES by conducting a meta-analysis of RCTs.
Methods
Search strategy
We performed a review of the literature and a meta-analysis of studies that reported the effects and safety of pulmonary vasodilators in ES patients. Relevant studies were identified by searching the PubMed, Cochrane library, Chinese National Knowledge Infrastructure (CNKI), WanFang, and VIP databases using the following search terms: (“Eisenmenger syndrome” or “Eisenmenger complex”) and (“drug therapy” or “drug” or “pharmac agent” or “medication” or “administration” or “endothelial receptor antagonist” or “bosentan” or “ambrisentan” or “macitentan” or “phosphodiesterase type 5 inhibitor” or “sildenafil” or “tadalafil” or “vardenafil” or “Viagra” or “prostacyclin” or “Epoprostenol” or “Treprostinil” or “Remodulin” or “iloprost” or “riociguat” or “nitric oxide” or “NO”). The reference lists of the studies were retrieved from the databases, and conference reviews of ES and medical management of patients with ES were evaluated. This meta-analysis was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement. 16 All studies included in this meta-analysis were published from February 1955 to February 2019.
Selection criteria
Studies were included by screening “titles/abstracts and full texts” if they meet all of the following criteria: (i) the patients enrolled in the studies had ES; (ii) the patients were receiving pulmonary vasodilators, and the efficacy or safety of the medication was evaluated; (iii) control groups, including non-treatment or placebo groups, were enrolled in the studies; (iv) the outcomes included mortality, hemodynamics, New York Heart Association (NYHA) functional assessments, 6MWD, SO2, and exercise capacity, and adverse effects were measured; and (v) the study design was an RCT. No restrictions concerning the publication language were found. We excluded self-controlled design studies, case reports, and conference abstracts.
Data extraction
The following information was independently extracted by two authors (Y.L.Hou and T.T.Shu): first author, publication year, study design, drug, number of patients, age, follow-up time, outcomes, and conclusions.
Assessment of study quality
The risk of bias within each study was assessed using the Cochrane risk of bias tool, which contained the following seven aspects: random sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias), and other bias. 17 Each item is evaluated by “low risk of bias”, “unclear risk of bias”, or “high risk of bias”. Quality assessment was independently conducted by two authors (Y.L. Hou and T.T. Shu), and a third author intervened if a consensus could not be reached.
Statistical analysis
The pooled treatment effects, point estimate of the mortality, mPAP, PVRi, 6MWD, and SO2 were calculated using the Review Manager software (Version 5.3). A
We used Cochran’s χ2-based Q test and I-squared test to assess inter-study heterogeneity.
18
If no significant heterogeneity (defined as
Results
Literature search
Initially, 430 studies were identified using the above search strategies, and 89 studies were duplicates and removed. After reading titles and/or abstracts, 287 studies that met the exclusion criteria were removed, and 54 studies that met the primary inclusion criteria were considered. Full-text reviews were conducted for the 54 remaining studies, and 49 studies did not fulfill the inclusion criteria. The studies were excluded in this analysis for the following reasons: (i) 11 studies did not focus on patients with ES; (ii) one was not about pulmonary vasodilators; iii) one used pulmonary arterial hypertension-targeted drug (pulmonary vasodilator) therapy in the control and experimental groups; (iv) one reported the outcome without evaluating the efficacy and safety of the pulmonary vasodilators; and (v) 33 were non-RCTs. Therefore, five RCTs4–8 were ultimately included in the present meta-analysis (Fig. 1).
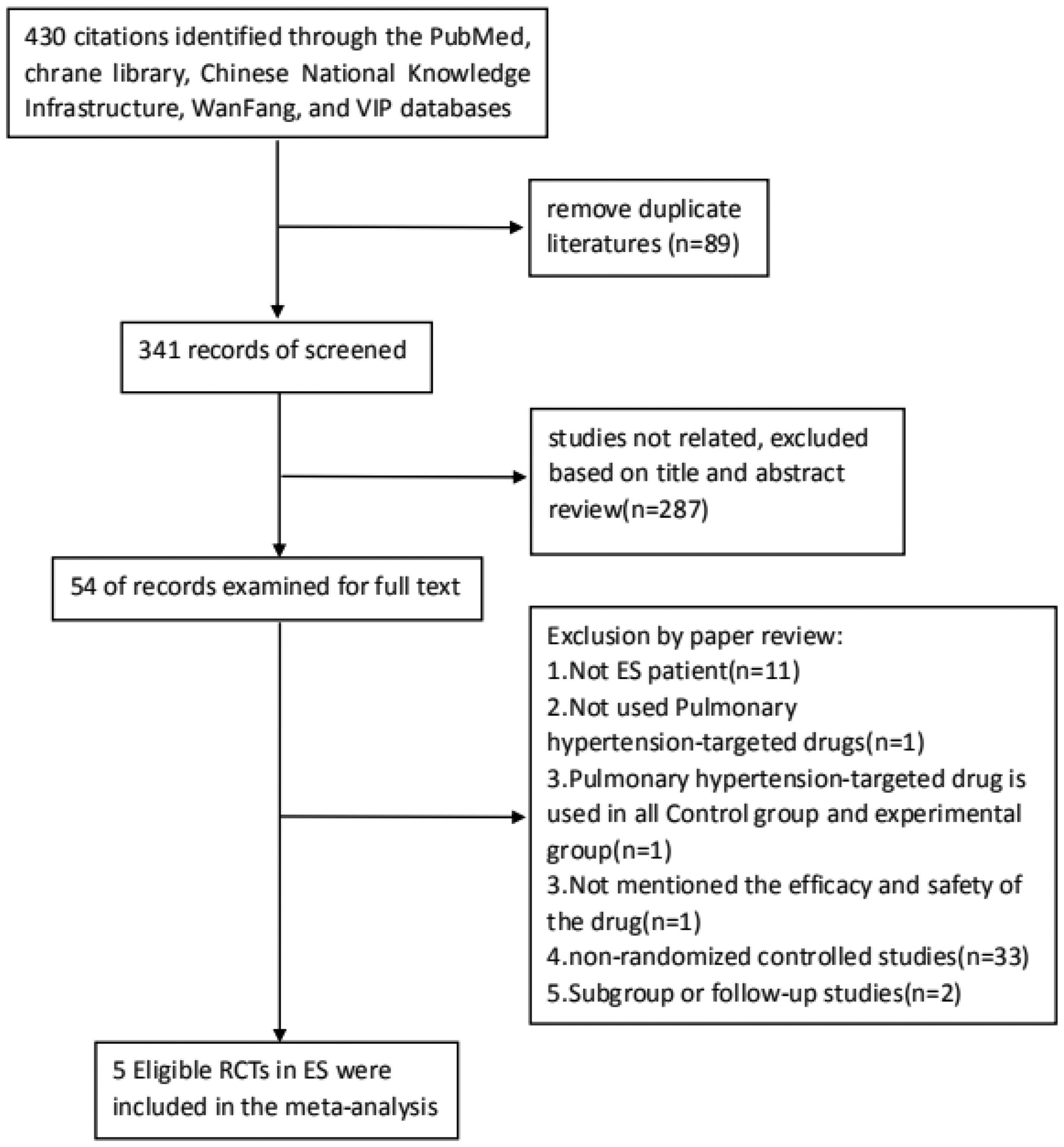
Flow diagram of the study selection. The flow diagram shows the literature search for the relevant studies of the effect on and safety of pulmonary vasodilators for ES patients. RCTs: randomized controlled trials; ES: Eisenmenger syndrome.
Eligible studies
The baseline characteristics of the five included studies are indicated in Table 1. The latest study was published in 2019. A total of 508 patients were assigned to the experimental and control groups in the five RCTs. The age of the patients ranged from 17 years to 53 years. The follow-up time was 6 weeks to 154 weeks. During the studies, five evaluated mortality,4–8 three reported mPAP,5,7,8 two reported PVRi,7,8 four assessed the 6MWD,4,5,7,8 and four reported the SO24,5,7,8
Characteristics of the trials included in the meta-analysis.
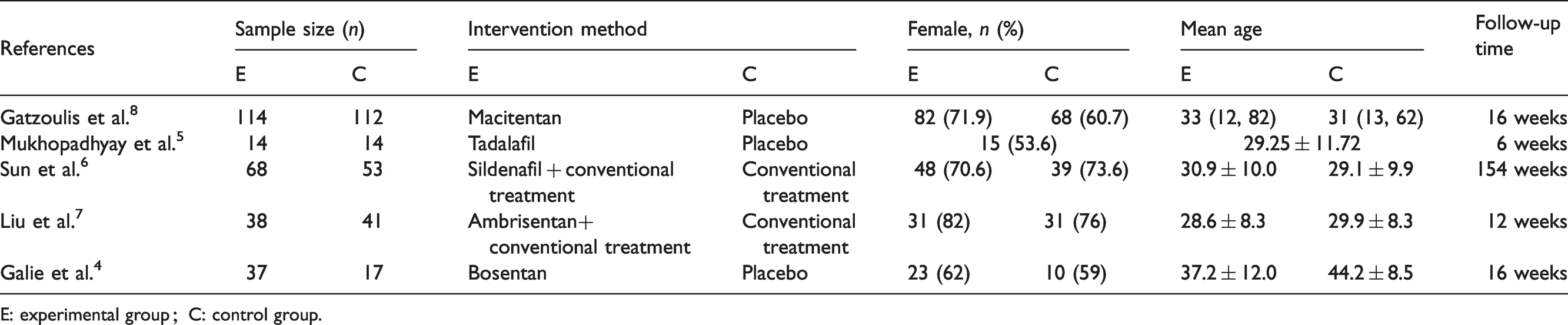
E: experimental group;C: control group.
Methodological quality assessment
Two studies6,7 did not randomly allocate, concealed treatment allocation, or did not provide the blinding methods. Two studies5,8 did not adequately describe the methods used for concealed treatment allocation. No risk of attrition bias, reporting bias, or other bias concerning the company sponsors was found among the studies (Fig. 2).
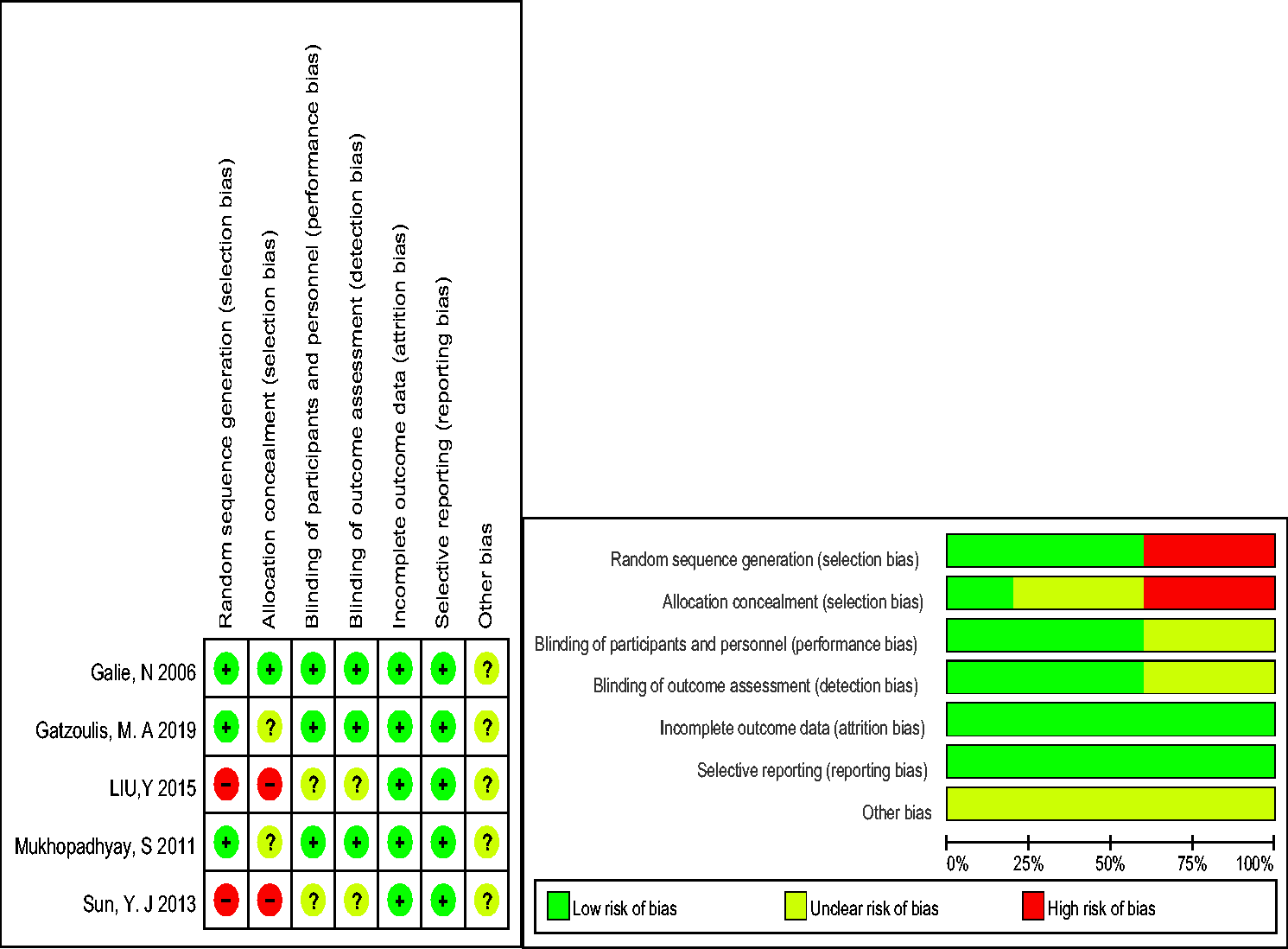
Quality assessment of each included study. (a) Risk of bias summary, (b) risk of bias graph.
Mortality
Mortality was reported in five studies with 508 patients. Compared with those in the control group, the patients using pulmonary vasodilators demonstrated significant decrease in mortality (odd risk (OR) = 0.35; 95% CI, 0.13 to 0.95;
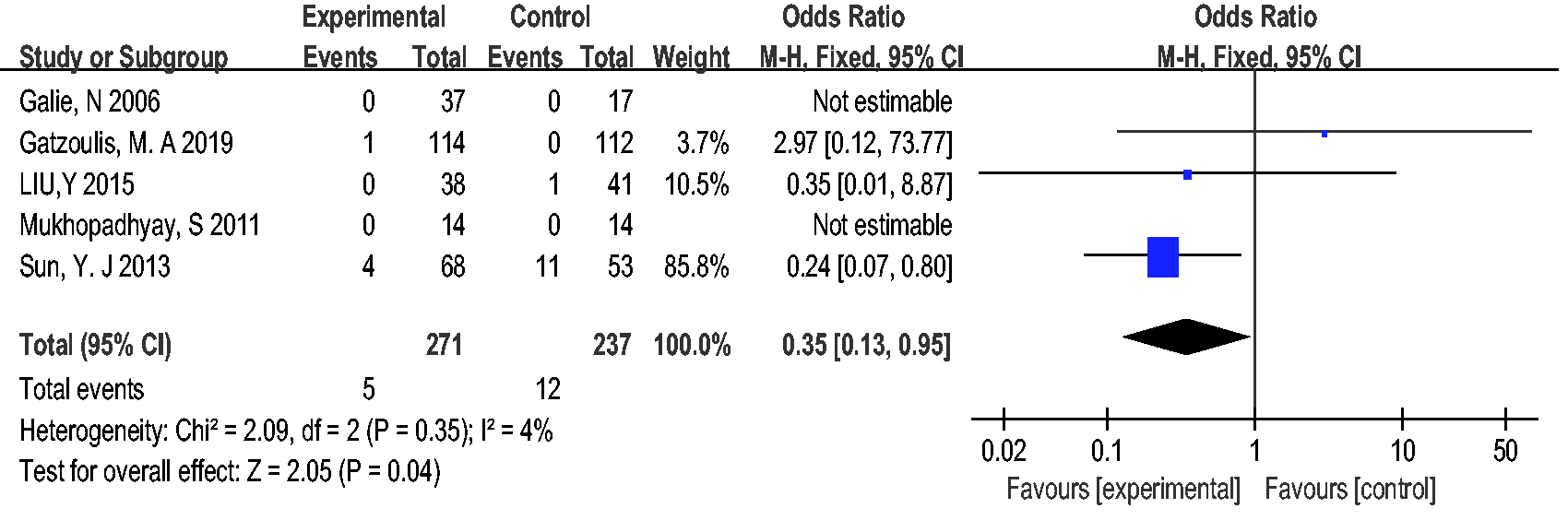
Forest plot of mortality. The squares indicate the odds ratio (OR) and the horizontal lines indicate the 95% confidence interval (CI) for each trial included; the statistical weight of a trial in the meta-analysis is proportional to the size of each square; diamond indicates pooled risk ratio and 95% confidence interval with the center indicating the point estimate and the left and the right ends the 95% CI.
Exercise capacity
The 6MWD was reported in four studies with 386 patients. The 6MWD was notably improved by (mean difference (MD) = 28.38 m; 95% CI, 2.99 to 53.77;
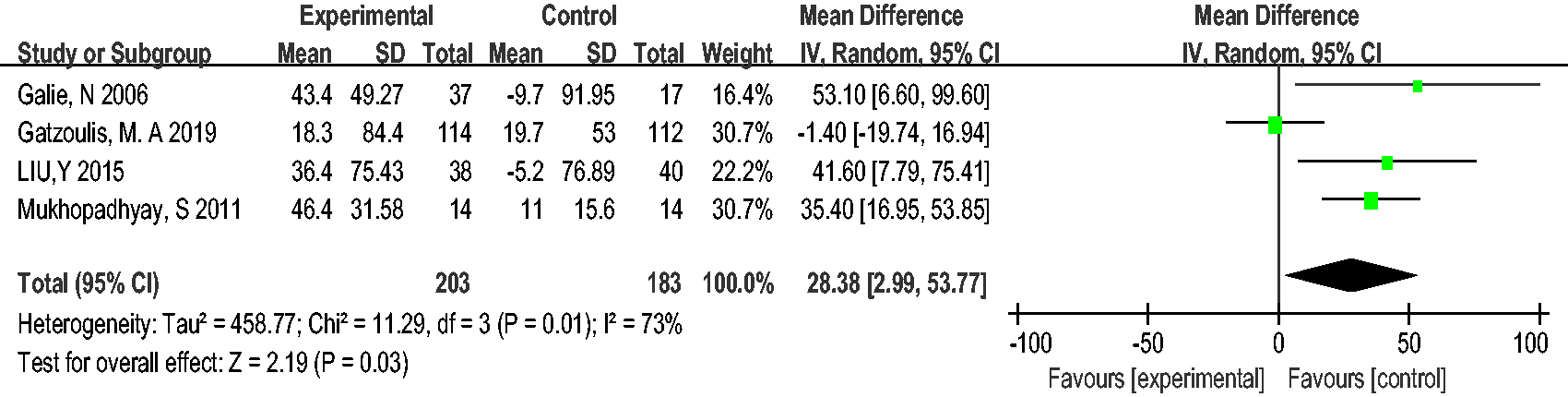
Forest plot of 6MWD. The squares indicate the mean difference (MD), and the horizontal lines indicate the 95% confidence interval (CI) for each included trial. The statistical weight of a trial in the meta-analysis is proportional to the size of each square. The diamonds indicate the pooled risk ratio and 95% confidence interval, with the center indicating the point estimate, and the left and right ends indicating the 95% CI.
Systemic oxygen saturation
The systemic oxygen saturation (SO2) was reported in four studies with 384 patients. Compared with the control group, SO2 at rest was elevated (MD = 1.00%; 95% CI, 0.12 to 1.88;
Hemodynamic parameters
Concerning the hemodynamic parameters during right cardiac catheterization, the mPAP significantly reduced in the experimental group (MD = −4.35mmHg; 95% CI, −7.19 to −1.50;
Side effects
Side effects were reported in four of the included studies,4,5,7,8 which occurred in 135 patients (66% in the pulmonary vasodilators group). ES patients with ERAs were reported mild adverse effects, primarily peripheral edema 17 (9%), headache 25 (13%), and dizziness 12 (6%), while the PDE-5i could mainly contribute to headache 2 (14%), nasal stuffiness 2 (14%). And serious adverse events occurred in 12 patients (only 6% in the pulmonary vasodilators group). The serious adverse events were defined as those that led to premature discontinuation of study or a need for intervention therapy, including liver function abnormalities 2 (1%), biliary tract disease include cholelithiasis and biliary colic 2 (1%), cardiac disorders include arrhythmias, angina pectoris and heart failure 5 (2%), infection 2 (1%), and transient ischemic attack 2 (1%). Actually, most of the serious adverse events were rare and curable. Overall, the pulmonary vasodilators were well tolerated.
Sensitivity analysis
We performed sensitivity analysis to identify the potential sources of heterogeneity in the efficacy of pulmonary vasodilators in patients with ES. For the 6MWD, when one study was omitted in turn, the pooled improvement changed from (MD = 28.38 m; 95% CI, 2.99 to 53.77;
Discussion
The present meta-analysis, which included five RCTs, examined the efficacy and safety of pulmonary vasodilators in 508 patients with ES. The present study demonstrated that pulmonary vasodilators (primarily include ERAs, PDE-5i) significantly decreased mortality, mPAP, and PVRi and improved 6MWD and SO2.
Previous studies showed that pulmonary vasodilators improve 6MWD in patients with ES.4–7 However, a recent MAESTRO study 8 has reported conflicting results that Macitentan does not show superiority over placebo in exercise capacity in patients with ES. The results of the present meta-analysis showed a significant improvement in 6MWD. Thus, we proposed the following reasons for the discrepancy in exercise capacity. First, the patients in MAESTRO included simple and complex CHD (simple septal defects and patent ductus arteriosus were classified as simple CHD, and other conditions were classified as complex CHD), whereas only simple CHD was included in the Bosentan Randomized Trial of Endothelin Antagonist Therapy-5 (BREATHE-5) study. The clinical phenotype, hemodynamics, and outcomes of ES differ according to defect location.20–22 In addition, the response to pulmonary vasodilators may differ according to defect location. The outcomes varied with defect location. 23 Second, the patients in MAESTRO include World Health Organization functional class II to IV symptoms, and 59.4% of patients were functional class II. However, the patients in the BREATHE-5 study were restricted to WHO FC III. Patients with worse functional class are likely to improve with pulmonary vasodilators. 24 In addition, the effectiveness of pulmonary vasodilators in ES may be greatest in the WHO-FC III and IV classes. 20 Third, MAESTRO also enrolled the patients with background therapy (27% were receiving background phosphodiesterase type 5 therapy). With regard to the inclusion of patients receiving PDE-5i, PAH studies have demonstrated that background therapy may attenuate the treatment effects on exercise capacity. 25 Meanwhile, a randomized, placebo-controlled, double-blind trial involving patients with ES found that adding sildenafil to bosentan cannot improve hemodynamic and functional variables. 26 Fourth, pulmonary vasodilators may have systemic vasodilator potential in patients with ES. The addition of background pulmonary vasodilator therapy may have a similar effect on the combination of multiple drugs, which may aggravate the vasodilation of systemic blood vessels, increase right-to-left shunting, and exaggerate cyanosis, thereby hampering an additional increase in function due to therapy. 8 Moreover, a significant improvement was observed in patients without background treatment. Fifth, Down syndrome was enrolled in MAESTRO but was not included in the BREATHE-5 study. Previous studies showed that the efficacy of pulmonary vasodilators is particularly limited in ES patients with Down syndrome.27–31
Previous studies showed that the physicians were usually reluctant to apply pulmonary vasodilators to patients with ES for the following reasons. 2 First, the increased pulmonary blood flow caused by pulmonary vasodilators may reactivate the pathogenesis for pulmonary vascular disease. 2 Second, pulmonary vasodilators may also reduce systemic vascular resistance while reducing pulmonary vascular resistance, which may cause desaturation. 2 Previous studies showed that SO2 did not deteriorate with pulmonary vasodilators,32–34 but no consensus on the improvement of SO2 was reached. Increased SO2 was observed in ES patients with pulmonary vasodilators in the study, which is in accordance to the findings reported in another related systematic review and meta-analysis.2,35 This result may be attributed to the fact that pulmonary vasodilators exert a similar impact on systemic and pulmonary circulations, resulting in a limited influence on the net right-to-left shunt. 4 And PAH is an independent risk factor for poor prognosis in patients with CHD, which can increase the risk of all-cause death by more than four times. 21 The present study shows that pulmonary vasodilators decrease the mortality and improve the hemodynamics and exercise capacity of patients with ES. And a 20-year follow-up study showed that the long-term prognosis of patients with ES treated with pulmonary vasodilators was significantly improved than that of the patients without the treatment. 21 Therefore, pulmonary vasodilators should be recommended for patients with ES in the clinical practice.
Previous studies reported no specific safety concerns with the use of pulmonary vasodilators in ES patients.4,5,7,8 The safety in the meta-analysis is consistent with previous studies.
For survival, contemporary data in this regard are diverse. Numerous studies showed that the mortality rate of patients using pulmonary vasodilators is much lower than that of patients without advanced therapy, which is in line with the findings of our meta-analysis.6,9,11–15 Conversely, the association between survival and pulmonary vasodilators on multivariable analysis has not been found in a recently published multicenter study of >1000 patients with ES 10 . The discrepancy may be the result of the relatively short follow-up time.
Due to the heterogeneity observed among the included studies, pooled estimates were calculated using different effects models. For the assessments of mPAP and PVRi, no heterogeneity was found (I2 = 0.0%), indicating the reliability of the results. For mortality (I2 = 4%) and SO2 (I2 = 26%), mild heterogeneity was found. We used a fixed model to assess the outcomes. However, the 6MWD showed moderate heterogeneity with I2 = 73%, which was largely attributed to one study after the sensitivity analysis; 8 thus, the randomized model was chosen, assuming that underlying true effects differed between studies.
Some limitations should be noted in the study. First, the results were limited by the small number of studies and the heterogeneity observed. Thus, the analysis results should be interpreted with caution. Therefore, RCTs with larger sample size and long-term follow-up are needed to evaluate the efficacy and safety of pulmonary vasodilators in patients with ES. Second, this meta-analysis did not evaluate the efficacy and safety of the combined therapy and the long-term effects of pulmonary vasodilators on patients with ES. Third, not all outcome indicators described in this study were reported in five articles, so there is a lack of consistency regarding the inclusion of studies between meta-analysis of them.
Conclusion
Pulmonary vasodilators (primarily the PDE-5 inhibitor and endothelin-1 receptor antagonist) significantly decrease the mortality and improve the hemodynamics and exercise capacity of patients with ES. Pulmonary vasodilators are well tolerated. However, this conclusion should be considered carefully and confirmed with large RCTs.
Supplemental Material
sj-pdf-1-pul-10.1177_20458940211015823 - Supplemental material for Efficacy and safety of pulmonary vasodilators in the patients with Eisenmenger syndrome: a meta-analysis of randomized controlled trials
Supplemental material, sj-pdf-1-pul-10.1177_20458940211015823 for Efficacy and safety of pulmonary vasodilators in the patients with Eisenmenger syndrome: a meta-analysis of randomized controlled trials by Yulin Hou, Li Wen, Tingting Shu, Rong Dai and Wei Huang in Pulmonary Circulation
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Authors’ contributions
Yulin Hou, Li Wen, Tingting Shu were responsible for the study screening, data extraction, data analysis, and writing the manuscript. Rong Dai and Wei Huang were responsible for checking and reviewing the final manuscript.
Acknowledgments
N/A.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chongqing municipal health and Health Committee (ZQNYXGDRCGZS2019001, No.2019ZY3340, No. 2016HBRC001).
Guarantor
Wei Huang, Rong Dai.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
