Abstract
Chronic thromboembolic pulmonary hypertension is considered as a rare but severe complication after acute pulmonary embolism and is potentially curable by pulmonary endarterectomy. We aimed to evaluate, over an 11-year period, time trends of in-hospital outcomes of pulmonary endarterectomy in chronic thromboembolic pulmonary hypertension patients and to investigate predictors of the in-hospital course. We analyzed data on the characteristics, comorbidities, treatments, and in-hospital outcomes for all chronic thromboembolic pulmonary hypertension patients treated with pulmonary endarterectomy in the German nationwide inpatient sample between 2006 and 2016. Overall, 1398 inpatients were included. Annual number of pulmonary endarterectomy increased from 67 in 2006 to 194 in 2016 (P < 0.001), in parallel with a significant decrease of in-hospital mortality (10.9% in 2008 to 1.5% in 2016; P < 0.001). Patients’ characteristics shifted slightly toward older age and higher prevalence of chronic renal insufficiency and obesity over time, whereas duration of hospital stay decreased over time. Independent predictors of in-hospital mortality were age (OR 1.03 (95%CI: 1.01–1.05); P = 0.001), right heart failure (2.55 (1.37–4.76); P = 0.003), in-hospital complications such as ischemic stroke (6.87 (1.06–44.70); P = 0.044) and bleeding events like hemopneumothorax (24.93 (6.18–100.57); P < 0.001). Annual pulmonary endarterectomy volumes per center below 10 annual procedures were associated with higher rates of adverse in-hospital outcomes. Annual numbers of chronic thromboembolic pulmonary hypertension patients treated with pulmonary endarterectomy increased markedly in Germany between 2006 and 2016, in parallel with a decrease of in-hospital mortality. Our findings suggest that perioperative management of pulmonary endarterectomy, institutional experience, and patient selection is crucial and has improved over time.
Keywords
Introduction
Chronic thromboembolic pulmonary hypertension (CTEPH) is considered as a rare but severe complication after acute pulmonary embolism with a high mortality rate and poor prognosis if untreated. 1 , 2 CTEPH is potentially curable if treated by pulmonary endarterectomy (PEA). A multicenter, international prospective study of long-term outcome in CTEPH demonstrated that patients who underwent PEA had a better three-year survival (89%) compared with patients without PEA (70%). 2 However, PEA is a complex procedure including the removal of fibrous obstructive tissue from the pulmonary arteries during circulatory arrest under deep hypothermia and is recommended as the gold standard by current guidelines to ameliorate right ventricular compromise caused by high pulmonary vascular resistance (PVR). 3 The in-hospital mortality rate after PEA is ranging between 2.2 and 16.0%. 2 ,4– 6 Recently, the European CTEPH registry demonstrated that in PEA centers with < 10 PEAs per year, mortality both during in-hospital stay and at long-term was two-fold higher than in centers with a larger operation volume. 2 Notably, a greater proportion of CTEPH patients per one million inhabitants referred for surgery in the UK and Europe compared to the United States. 7 , 8 Although the principal technique of endarterectomy has not changed, 9 PAH-specific medical therapy is frequently used as an additional treatment approach before PEA and the complex and close collaboration of a multidisciplinary team including pulmonologists, cardiothoracic surgeons, radiologists, and anesthesiologists became an increasingly important factor. 10 , 11
Progress in surgical experience and perioperative management as well as improvement in medical therapy over the past decade has considerably improved the outcome of patients with CTEPH, but a large study investigating time trends of patient characteristics and predictors of in-hospital outcomes for CTEPH patients treated with PEA are still missing. Thus, we aimed to evaluate CTEPH patients treated with PEA to investigate time trends and predictors of the in-hospital course.
Methods
Data source
The computed study analyses were performed on our behalf by the Research Data Center of the Federal Statistical Office and the Statistical Offices of the federal states in Wiesbaden, Germany (source: DRG Statistics 2006–2016, own calculations). The Research Data Center provided us aggregated statistical results on the basis of SPSS codes (SPSS® software, version 20.0, SPSS Inc., Chicago, IL), which were sent by us to the Research Data Center. 12
Diagnoses, procedural codes, and definitions
In Germany, diagnoses are coded according to the International Classification of Diseases and Related Health Problems, 10th Revision with German Modification (ICD-10-GM), and diagnostic, surgical, or interventional procedures according to the German Procedure Classification (OPS, surgery and procedures codes (Operationen- und Prozedurenschlüssel)). All Diagnosis Related Groups diagnoses and OPS codes of hospitalized patients are gathered by the Federal Statistical Office of Germany. Thereby, we were able to identify all hospitalized CTEPH patients (based on the ICD-code I27.20) with a procedure of PEA (based on the OPS code 5-381.42).
Coded parameters and study outcomes
Analyzed variables as comorbidities and clinical presentation comprised coronary artery disease (ICD-code I25), cancer (ICD-codes C00-C97), left heart failure (ICD-code I50.1), right heart failure (ICD-code 50.0), atrial fibrillation/flutter (ICD-code I48), chronic obstructive pulmonary disease (COPD; ICD-code J44), thrombophilia (ICD-code D68.5, D68.6), arterial hypertension (ICD-code I10), chronic renal insufficiency (ICD-code N18.3, N18.4, N18.5, N18.83, N18.84, N18.9), diabetes mellitus (ICD-code E10, E11, E12, E13, E14), tachycardia (ICD-code I47, R00.0), syncope (ICD-code R55), and York Heart Association (NYHA) classes III (ICD-code I50.13) and IV (ICD-code I50.14). Outcomes of this study comprised death of any cause during the hospital stay (all-cause in-hospital death), major adverse events such as ischemic stroke (ICD-code I63) or clinically relevant bleeding events (hematopneumothorax (ICD-code J942), hemopericardium (ICD-code I31.2), transfusion of erythrocyte concentrates (OPS code 8-800), intracerebral bleeding (ICD-code I61), or gastrointestinal bleeding (ICD-code I60)). The primary outcome is all-cause in-hospital death (ICD-10-GM code I61). Conditions and outcomes observed in two or fewer patients for each subgroup were censored to prevent re-identification.
Ethical aspects
Since this study did not involve direct access to data of individual patients by the investigators, approval by an ethics committee and informed consent were not required, in accordance with German law.
Statistical methods
While continuous variables are presented as median and interquartile range (IQR), categorical variables were provided and reported as absolute numbers and corresponding percentages. Comparison of survivors versus non-survivors were performed using the Mann–Whitney U test for continuous variables and the Fisher’s exact or chi-square test, as appropriate, for categorical variables. The total numbers, proportion, incidence, relative mortality rate, and length of in-hospital stay for CTEPH patients undergoing PEA were calculated annually and linear regressions were used to assess trends over time. The results were presented as beta (β) and corresponding 95% confidence intervals (CIs). Patients undergoing PEA during the observational period 2006–2016 were stratified for procedural volumes per center. Patients were divided in the ones treated in centers with an average annual PEA volume of ≤10 over the 11-year period (≤110 procedures overall) and those in centers with > 10 procedures annually (>110 overall). We further stratified the study population into the patients treated in centers with an annual average PEA volume of ≤25 versus >25 procedures per year; and into ≤50 versus >50 procedures. Groups were compared for in-hospital adverse events.
Univariate and multivariate logistic regression models were performed to investigate the impact of age, comorbidities, and clinical conditions on the mortality during hospitalization (in-hospital mortality). Results were presented as Odds Ratios and corresponding 95% CIs. Multivariate logistic regression models included in a first analysis age and sex. In a second model cancer (ICD-codes C00-C97), coronary artery disease (ICD-code I25), heart failure (ICD-code I50), COPD (ICD-code J44), essential arterial hypertension (ICD-code I10), diabetes mellitus (ICD-codes E10-E14), and chronic renal insufficiency (comprised diagnosis of chronic renal insufficiency stages 3–5 with glomerular filtration rate <60 ml/min/1.73 m2, ICD-code N18.3, N18.4, N18.5, N18.83, N18.84, N18.9) were added to the model with age and sex.
The software SPSS (SPSS® software, version 20.0, SPSS Inc., Chicago, IL) was used for statistical analysis. P values of < 0.05 (two-sided) were considered to be statistically significant.
Results
Baseline characteristics
We identified 1398 hospitalized CTEPH patients treated with PEA between 2006 and 2016 in Germany. The majority of patients were women (56.8%), had a median age of 62 years and a median hospital stay of 15 days (Table 1). Cardiovascular comorbidities were common in these patients: overall, 21.7% were diagnosed with coronary artery disease and 31.0% with arterial fibrillation/flutter. In addition, 11.9% of the patients had a diagnosis of renal insufficiency and 14.2% were coded with COPD. In total, 18.1% of the patients reported severe dyspnoea of NYHA class III or IV (Table 1).
Baseline characteristics, medical history, and presentation of the 1398 CTEPH patients with pulmonary endarterectomy (PEA) (cumulative data of the years 2006–2016).
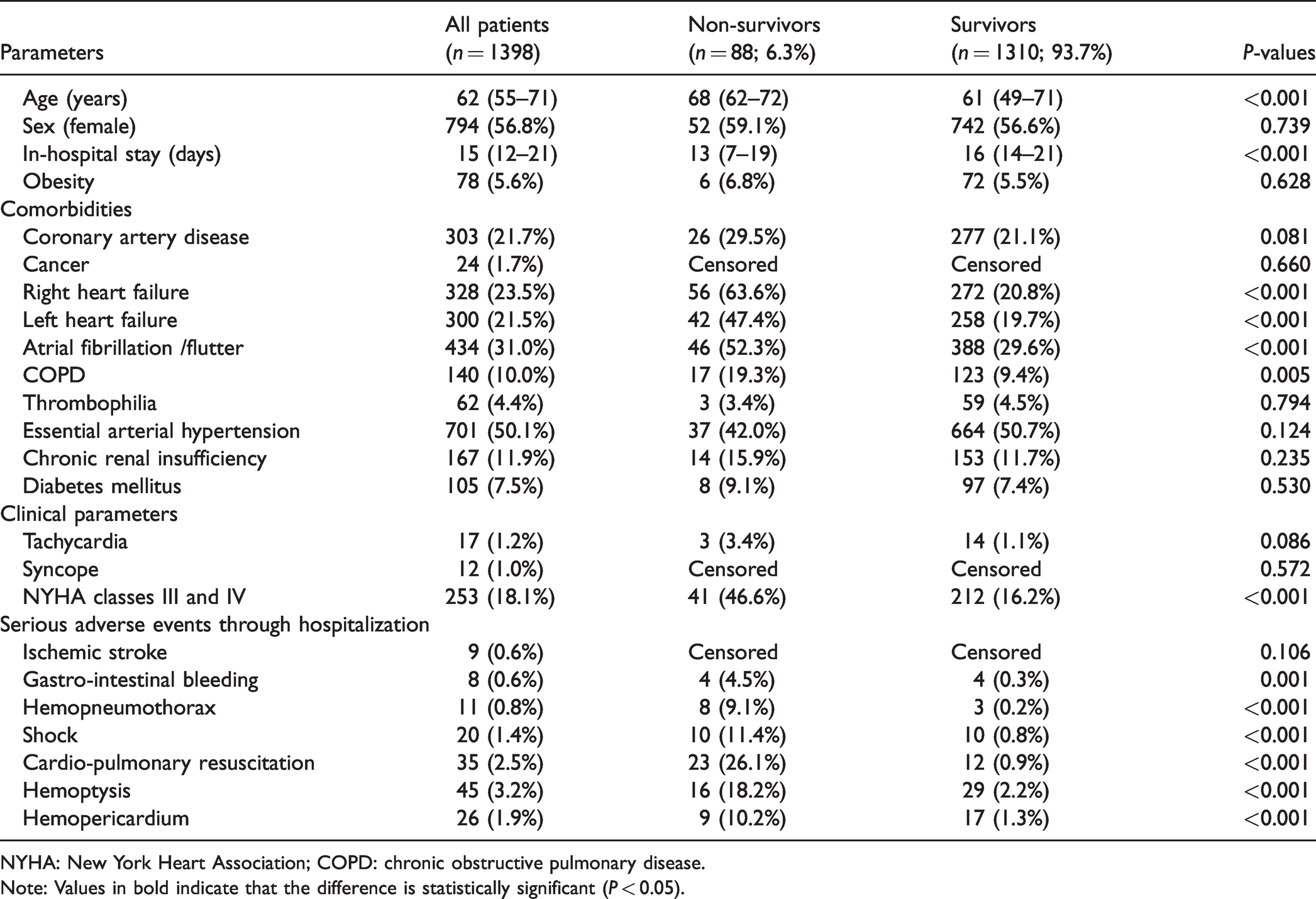
NYHA: New York Heart Association; COPD: chronic obstructive pulmonary disease.
Note: Values in bold indicate that the difference is statistically significant (P < 0.05).
Predictors of in-hospital mortality
Overall, 88 (6.3%) CTEPH patients with PEA died during the in-hospital stay. Non-survivors were older and had more frequently comorbidities including right heart failure, atrial fibrillation/flutter, and COPD. NYHA class III/IV (46.6% vs. 16.2%, P < 0.001) was also more prevalent in non-survivors as well as bleeding events such as gastrointestinal bleeding, hemoptysis, hemopericardium, and hemopneumothorax compared to survivors (Table 1).
Independent predictors of in-hospital mortality in the multivariate logistic regression model were age, right heart failure, atrial fibrillation/flutter, and in-hospital complications such as ischemic stroke, bleeding (e.g. hemopericardium or hemopneumothorax), shock, and cardio-pulmonary resuscitation (Table 2).
Impact of baseline characteristics, comorbidities, clinical presentation, and complications on in-hospital mortality.
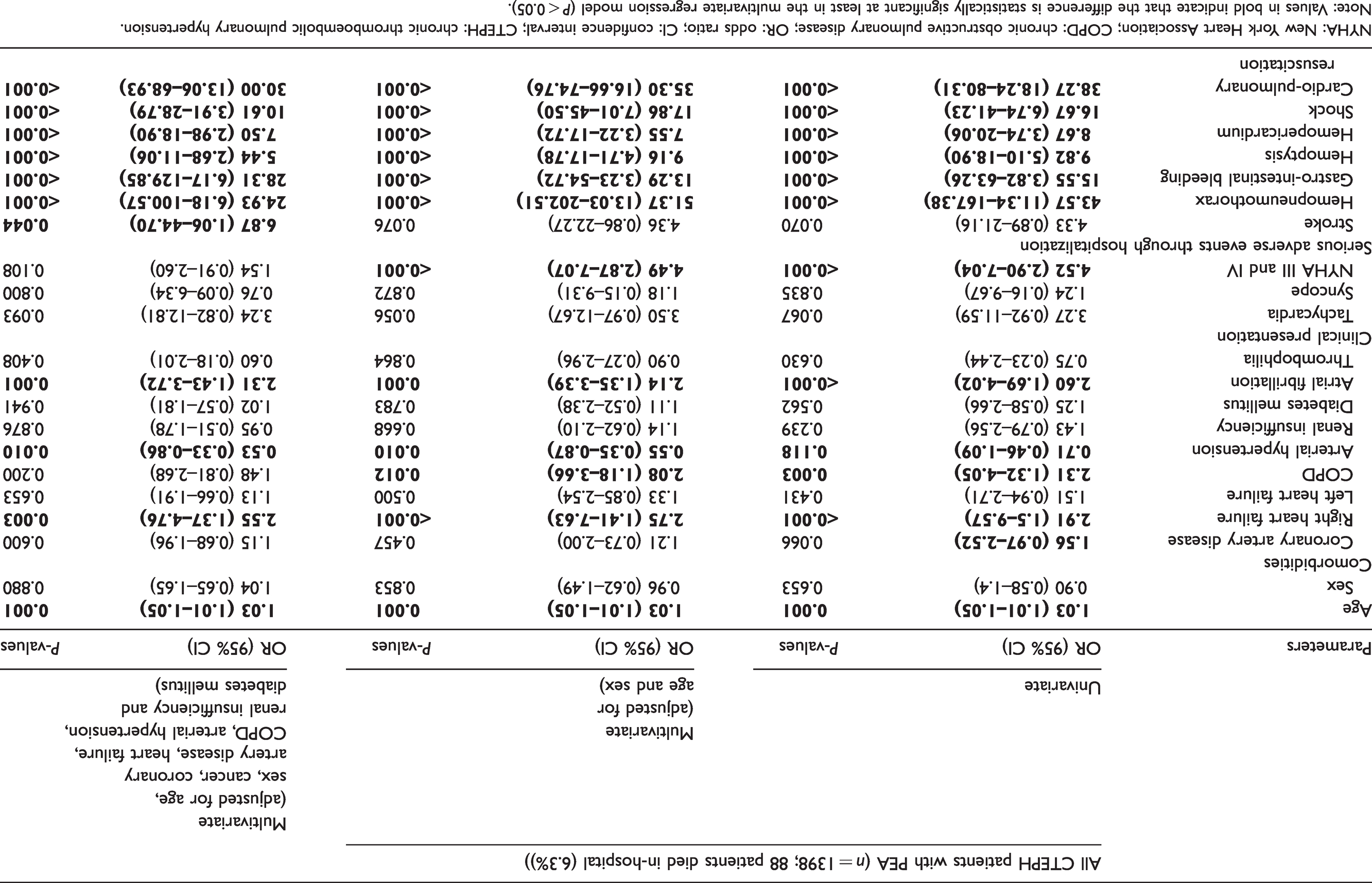
NYHA: New York Heart Association; COPD: chronic obstructive pulmonary disease; OR: odds ratio; CI: confidence interval; CTEPH: chronic thromboembolic pulmonary hypertension.
Note: Values in bold indicate that the difference is statistically significant at least in the multivariate regression model (P < 0.05).
Trends between 2006 and 2016
Although the total number of CTEPH patients who underwent PEA increased over the 11-year period (β: 0.69 (95% CI: 0.51–0.86), P < 0.001), the in-hospital mortality decreased significantly, from 10.9% in 2008 to 1.5% in 2016 (β: –1.85 (95% CI: –2.46 to 1.24), P < 0.001) (Table S1 and Fig. 1). Patients’ age at the time of procedure changed only slightly over time (β: 0.01 (95% CI: 0.01–0.02), P = 0.003). Comorbidities such as coronary artery disease, atrial fibrillation/flutter, arterial hypertension, chronic renal insufficiency, and diabetes mellitus remained roughly unchanged over time. Interestingly, markers of disease severity other than NYHA III/IV (β: –1.50 (95% CI: –1.89 to 1.12), P < 0.001) or right heart failure (β: –1.39 (–1.74 to –1.04), P < 0.001) were less often present in CTEPH patients over time during this 11-year analysis. The time of in-hospital stay decreased over time (β: –0.21 (95% CI: –0.12 to –0.31), P = 0.021). Serious adverse events during hospitalization remained unchanged (Table S1 and Fig. 2). In 2006, the majority (62.7%) of patients with a CTEPH diagnosis were treated in PEA centers with a volume performing on average < 25 procedures annually. In contrast, in 2016, the vast majority (88.2%) of patients were treated in high volume PEA centers with more than 25 PEA surgical procedures per year (Figure S1).
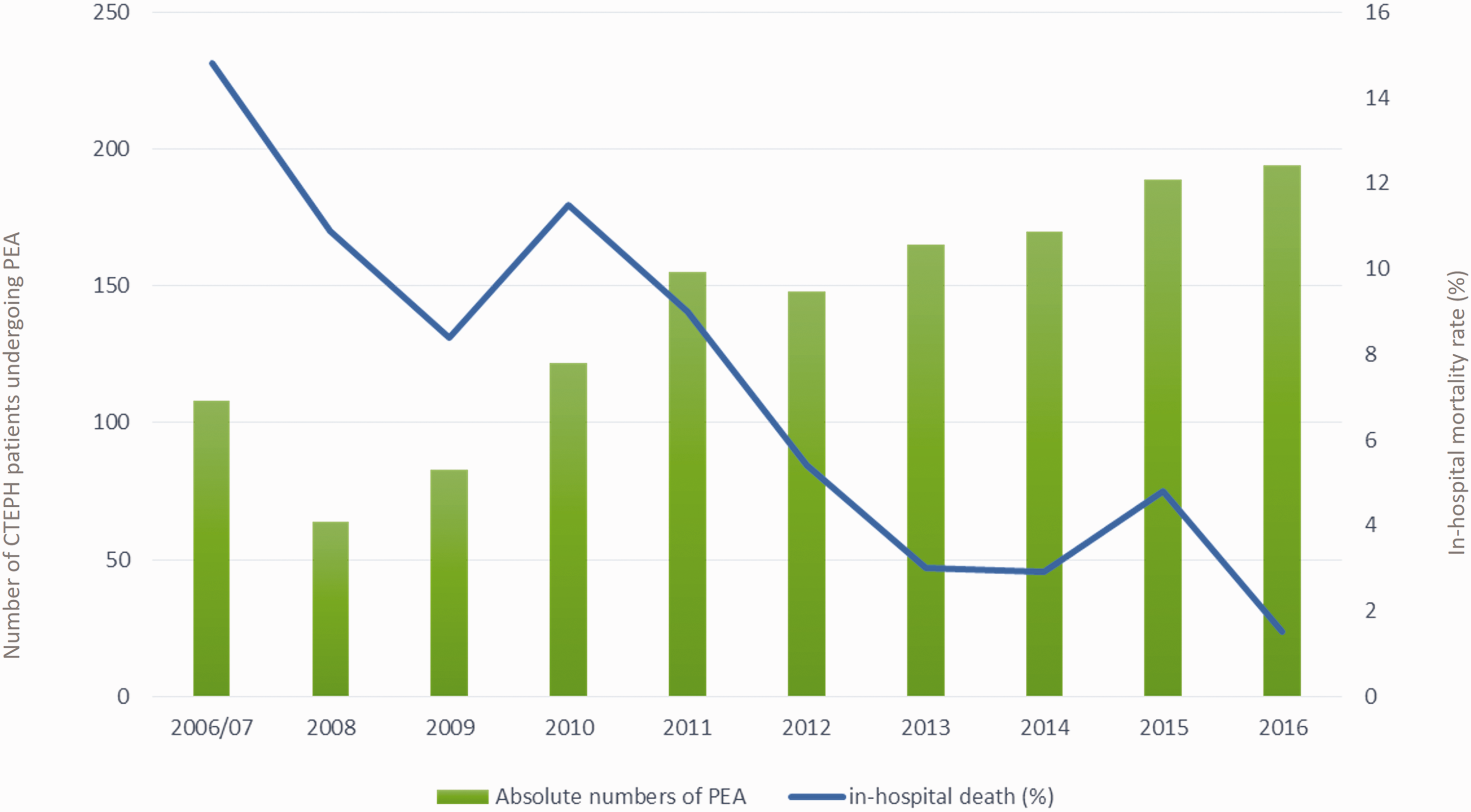
Annual numbers and in-hospital mortality rate of hospitalized CTEPH patients undergoing PEA between 2006 and 2016 in Germany.
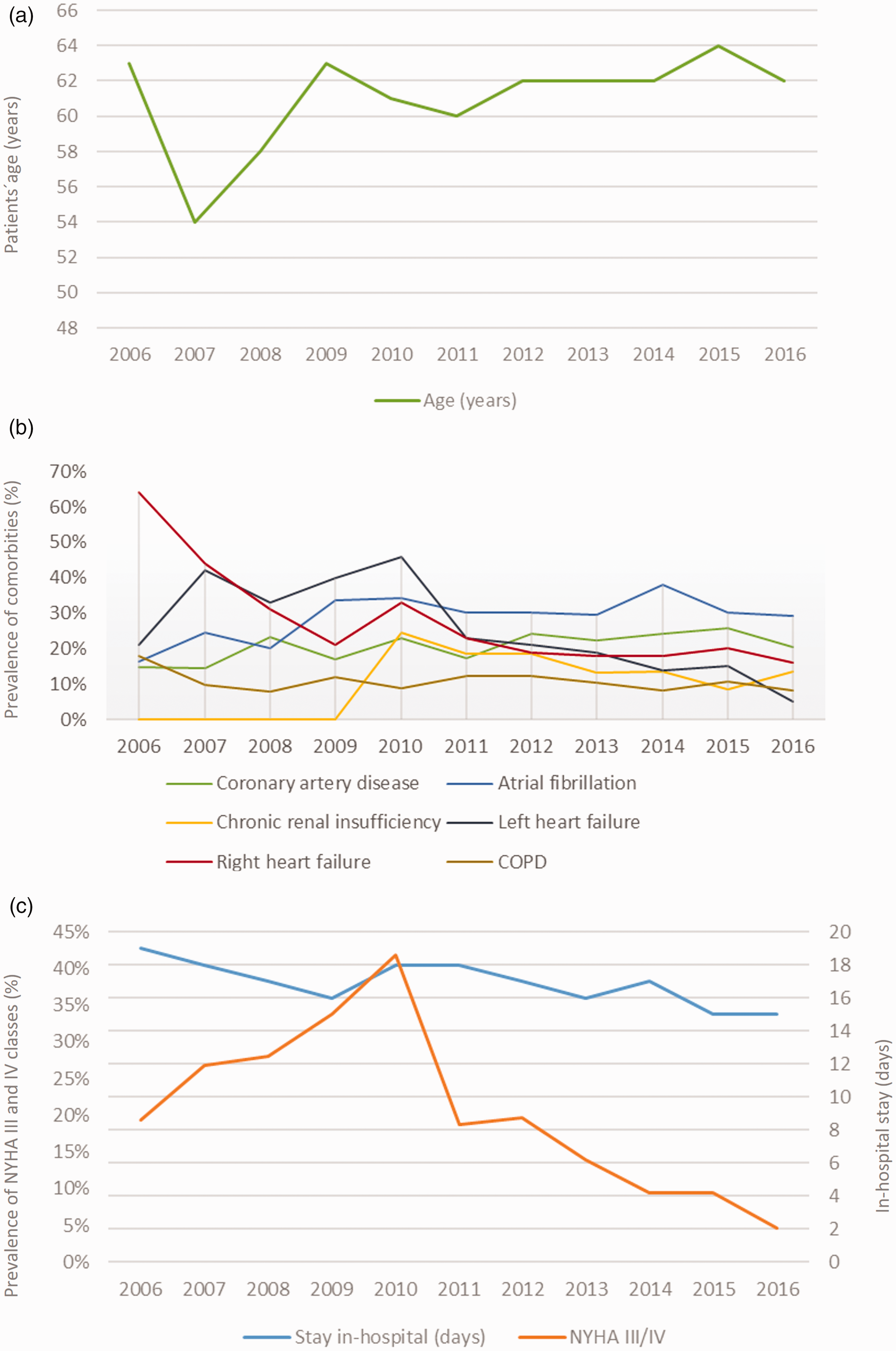
Trends regarding age, in-hospital stay (a) and comorbidities (b and c) in hospitalized CTEP patients undergoing PEA from 2006 and 2016 in Germany.
Procedural volume and in-hospital events, mortality, and complications
Patients treated with PEA in centers with a procedural volume of ≤10 annually showed a considerably higher in-hospital mortality compared to patients treated in centers with > 10 PEA procedures (14.2 vs. 5.6%, P < 0.001) (Figure S2).
Discussion
This nationwide inpatient sample (NIS) included almost 1400 CTEPH patients undergoing PEA in Germany. The key findings of our study can be summarized as follows: (1) over the 11-year period, the total annual numbers of CTEPH patients undergoing PEA increased considerably; (2) in parallel to a decrease of in-hospital mortality, patients’ characteristics shifted toward older age but with less severe symptoms or clinical findings; (3) predictors of in-hospital death were age and right heart failure, but also in-hospital complications such as ischemic stroke and bleeding; and (4) PEA volume per center below a critical number of at least 10 annual procedures was associated with higher rates of adverse in-hospital events.
Comparison of the German NIS with other study cohorts worldwide
Up to now, the European CTEPH registry 2 and a single-center experience of 1500 CTEPH patients with PEA4 were the largest CTEPH cohorts, but reports from a large nationwide sample are missing. Patients in the German nationwide sample were slightly younger (62 vs. 66 years) compared to the European CTEPH registry and comparable to previous PEA studies. 4 , 13 , 14 A single-center CTEPH registry reported a median in-hospital stay of 15 days (IQR: 13–18) from 2014 to 2015, 6 which is comparable with the present results. Of note, the median in-hospital stay decreased over the 11-year observational period from 19 days (IQR: 15--25) in 2006 to 15 days (IQR: 13–18) in 2016. In line with previous reports, 2 , 4 , 5 CTEPH was almost equally frequent in men and women in our study cohort. CTEPH patients were characterized by numerous severe comorbidities15– 17 including right heart failure, atrial fibrillation/flutter, and coronary artery disease, whereas obesity or history of cancer were less frequent described in comparison to the International CTEPH registry. 18 Interestingly, Germany has a similar proportion of CTEPH patients referred to surgery (per one million inhabitants per year) to the UK (each 2.7 PEAs) but more compared to the rest of Europe (1.7 PEAs per one million inhabitants per year) or substantially more compared to the US (0.9 PEAs per one million inhabitants per year). 7 , 8 The in-hospital mortality rate was 6.8% over the entire 11-year time period. This is in line with previously reported short-term mortality rates, which ranged between 2.2 and 16.0%.4– 6
Predictors of in-hospital mortality in the German NIS
Regarding predictors of mortality, the UK-PH registry demonstrated in 239 CTEPH patients that a preoperative pulmonary resistance above 1000 dyne·s·cm−5 was associated with increased perioperative mortality, whereas higher cardiac index and longer six-minute walk distance were associated with a better perioperative survival. 16 Similarly, an elevated PVR as well as NYHA class IV were identified as a predictor of 30-day mortality in 21419 and 10620 CTEPH patients who underwent PEA in previous studies. In the present study, NYHA class III/IV was also associated with increased in-hospital mortality in an univariate, but not in a multivariate logistic regression model. Neurologic and bleeding complications are an important group of complications after PEA. The rate of ischemic stroke in the German NIS was coded with 0.6% during hospitalization for PEA. The stroke incidence during cardiac surgery has been reported higher from 0.8 to 4.5% in retrospective studies.21– 23 Next to reperfusion lung edema, peri- and postoperative bleeding events are feared complications. 7 Our findings demonstrated less often bleeding complications such as hemopneumothorax or hemopericardium with a rate of 0.8 and 1.9%, respectively, compared to previous studies. 24 , 25 However, despite those bleeding events occurred rarely in the Germany NIS, the present study identified hemopneumothorax and hemopericardium independently predicting in-hospital mortality.
Time trends in CTEPH patients undergoing PEA
Two previous studies demonstrated trends in patients’ characteristics between two time segments for PEA surgery. 4 , 26 Madani et al. compared 1000 patients operated between March 1999 and October 2006 with 500 patients operated between October 2006 and December 2010. 4 As a result, they established a lower PVR and a higher cardiac output as patient characteristics’ in the more recent time frame (2006–2010). 4 Total number of CTEPH patients with severe dyspnoea and right heart failure, who were operated with PEA, decreased over time in the German NIS, and are in line with findings of Madani et al. 4 Interestingly, while the number of PEA per month remains stable between both groups, 4 the German nationwide sample observed an increased number of PEAs over the observation period of 11 years. In earlier studies, which included CTEPH patients from the late 1919s to 2006, 5 , 27 , 28 the in-hospital mortality ranged between 7.8 and 16.0% and decreased over time in recent study cohorts (including patients from 2006 to 2014) with a mortality rate from 2.2 to 2.5%. 2 , 4 , 6 Those results were confirmed in our nationwide sample with a considerable drop in-hospital mortality rate from 2006 with 10.9% to 2016 with 1.5%. Additionally, the fact that, in recent years, patients were predominantly treated in centers with more than 25 PEA surgical procedures per year, might explain the decrease in mortality (Figure S1).
Impact of center volumes regarding number of PEA
Our study results and previous studies suggest that the surgical technique, and also general patient care, have improved over time, and that PEA can be performed safely in experienced centers. 2 However, data regarding the impact of institutional experience on the patient outcomes are sparse. The results of the present study indicate that mortality was more than two-fold higher in centers performing < 10 PEAs per year, compared to those with a larger volume of surgical procedures. Those results are in line with results from an international prospective CTEPH registry. 2 A previous trial suggested that an “experienced” surgeon be defined as one who performs > 20 PEAs per year. 29 Although the current study cannot distinguish individual surgeon volume and experience, our data suggest that the procedural volume of a center should exceed a critical number of at least 10 PEA per year to ensure low perioperative mortality and low rates of adverse in-hospital events.
Limitations
There are limitations of our study that require consideration: the analysis is based on ICD discharge codes used for re-imbursement of costs, which might lead to incomplete data due to under-/overreporting and under-/overcoding. Therefore, we focused on hard endpoints such as in-hospital death and in-hospital complications, which are very unlikely to be miscoded or not coded. This study includes in-hospital results only and cannot address the long-term course. Moreover, we were able to study the association between variables registered during hospitalization, but had no information on their temporal or causal relationship. It also needs to be mentioned that the available data do not include individual surgeon volume or the EUROSCORE II of each patients. Finally, due to national data protection regulations, we were not allowed to show the results of analysis for complications or procedures involving in two or fewer patients for each subgroup.
Conclusion
The data from this nationwide patient sample highlight a marked increase in the number of CTEPH patients treated with PEA in Germany between 2006 and 2016. In parallel, in-hospital mortality decreased substantially. Independent predictors of in-hospital mortality were age and right heart failure, and in-hospital complications such as ischemic stroke and bleeding events. Our findings may suggest that perioperative management of PEA, institutional experience, and patient selection have improved over time and might draw more attention to further predictors of in-hospital mortality in patients with CTEPH undergoing PEA.
Supplemental Material
sj-pdf-1-pul-10.1177_20458940211008069 - Supplemental material for Time trends of pulmonary endarterectomy in patients with chronic thromboembolic pulmonary hypertension
Supplemental material, sj-pdf-1-pul-10.1177_20458940211008069 for Time trends of pulmonary endarterectomy in patients with chronic thromboembolic pulmonary hypertension by Lukas Hobohm, Karsten Keller, Thomas Münzel, Stavros V. Konstantinides and Mareike Lankeit in Pulmonary Circulation
Footnotes
Author contributions
L.H. and K.K. had full access to all of the data in the study, take responsibility for the integrity of the data and the accuracy of the data analysis, including and especially any adverse effects. L.H., K.K., T.M., S.V.K., and M.L. contributed substantially to the study design, data analysis, and interpretation. L.H., K.K., and M.L. drafted the manuscript. L.H., K.K., T.M., S.V.K., and M.L. critically revised the manuscript and approved the final version for submission.
Conflict of interest
None of the authors report a relationship with industry and other relevant entities—financial or otherwise—that might pose a conflict of interest in connection with the submitted article. The following authors report financial activities within the past two years outside the submitted work: Lukas Hobohm reports having received lecture honoraria from MSD and Actelion. Karsten Keller reports no conflict of interest. Thomas Münzel reports no conflict of interest. Stavros V. Konstantinides reports having received consultancy and lecture honoraria from Bayer AG, Daiichi-Sankyo, MSD, and Pfizer—Bristol-Myers Squibb; and institutional grants from Actelion, Bayer AG, Boehringer Ingelheim, Daiichi-Sankyo, and Biocompatibles Group UK—Boston Scientific Group. Mareike Lankeit reports having received consultancy and lecture honoraria from Actelion, Bayer, Daiichi-Sankyo, MSD, Pfizer—Bristol-Myers Squibb, and research funding from BRAHMS—Thermo Fisher scientific.
Funding
The author(s) disclosed receipt of the following financial support: This study was supported by the German Federal Ministry of Education and Research (BMBF 01EO1503; institutional grant to the Centre for Thrombosis and Hemostasis of the University Medical Center Mainz, Germany).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
