Abstract
A 10-year-old boy, with multiple comorbidities presented with fever, exertional dyspnea, fatigue and an obliterated brachiocephalic and inferior caval vein. Chronic thromboembolic pulmonary hypertension (CTEPH) was diagnosed. Nadroparine, antibiotics and supplemental oxygen were successfully started. Aged 12 years, supplemental oxygen was permanently needed with progressive exertional dyspnea and fatigue. In the country of residence the patient was considered as inoperable. The right ventricle was severely dilated, hypocontractile and hypertrophic. Mean pulmonary artery pressure (mPAP) was 79 mmHg and cardiac output 2.2 L/min. Pulmonary endarterectomy was uneventful. Four days later, mPAP was 33 mmHg and cardiac output 6.4 L/min. Three months later the boy restarted his education without supplemental oxygen. Six months after surgery right ventricular size and function and mPAP (14 mmHg) were normal. We demonstrated that pulmonary endarterectomy in young aged children is feasible and well-tolerated, even in the presence of severe co-morbidities. CTEPH should be an important diagnostic consideration in symptomatic children with a known hypercoaguable state, a history of thrombo-embolism or venous catheter placement, and/or a diagnosis of pulmonary hypertension. Hesitating to refer children for surgical consideration, or attempting to treat them by medication, only postpones the single potentially curable treatment and may worsen their prognosis.
Case description
A 10-year-old boy presented with fever, exertional dyspnea and fatigue. His medical history included: surgically corrected spina bifida, paralyzed from L3; ventriculoperitoneal drainage for Chiari malformation and hydrocephalus; Monti urostoma with recurrent urinary infections for neurogenic bladder; bilateral hip dysplasia; complicated colon resections ending up with intestinal failure, ileostoma and permanent total parental nutrition; and regular exchanges of an infected port-a-cath. This intellectual normal developing boy played wheelchair basketball.
Transthoracic echocardiography (TTE) showed pulmonary hypertension (PH) with a tricuspid regurgitation peak systolic pressure of 65 mmHg and an estimated cardiac output (CO) of 5.3 L/min. Computed tomography (CT) scanning of the lungs revealed thrombotic occlusions of both lower lobe arteries (rather sub-acute) and an extensive amount of adherent wall material in both upper lobe arteries (rather indicating chronic disease). The left brachiocephalic vein was obstructed and showed collaterals towards the hemiazygos vein. Both azygos and hemiazygos veins were connected with very wide intraspinal veins. The inferior caval vein (ICV) was completely obliterated starting from both femoral veins. Liver veins drained into the right atrium and kidney veins into paravertebral veins. Blood and urine culture were positive for staphylococcus epidermidis and candida albicans, respectively. Positron emission tomography (PET)-CT showed an extensive infected ICV thrombus with bilateral involvement of renal veins. Nadroparine and oxygen therapy were started and both infections were successfully treated with antibiotics.
Aged 12 years, wheelchair basketball and scholar education had become impossible, and supplemental oxygen was permanently needed. A pediatric surgical center and subsequently a chronic thromboembolic pulmonary hypertension (CTEPH) center in his country of residence both had considered him as inoperable. No specific CTEPH treatment (e.g. riociguat) was attempted. Our center was visited for a third opinion.
TTE showed a severely dilated, hypocontractile and hypertrophic right ventricle (RV) with tricuspid insufficiency 2–3/4, pulmonary artery pressure (PAP) (systolic/diastolic (mean)) of 127/37(79) mmHg and a CO of 2.2 L/min. Calculated total pulmonary vascular resistance (PVR) was 2873 dynes.s.cm−5. Bilateral selective pulmonary angiography (Fig. 1(a) and (b)) confirmed CTEPH. Venous angiography confirmed ICV obliteration.
(a) and (b) Pulmonary angiography with perfusion deficits suggestive for CTEPH. (a) Right lung. Amputation of apical upper lobe artery (white arrow). Stricture in the middle lobe artery (light gray arrow). Amputation of apicolateral (dark grey arrow) and dorsobasal (black arrow) branches of lower lobe. Large right pulmonary artery. (b) Left lung. Amputation basomedial segmental branch left lower lobe (white arrow). Perfusion deficit dorsobasolateral subsegmental branch of left lower lobe (black arrow). Large left pulmonary artery. (c) Endarterectomy specimen right lung. (d) Endarterectomy specimen left lung.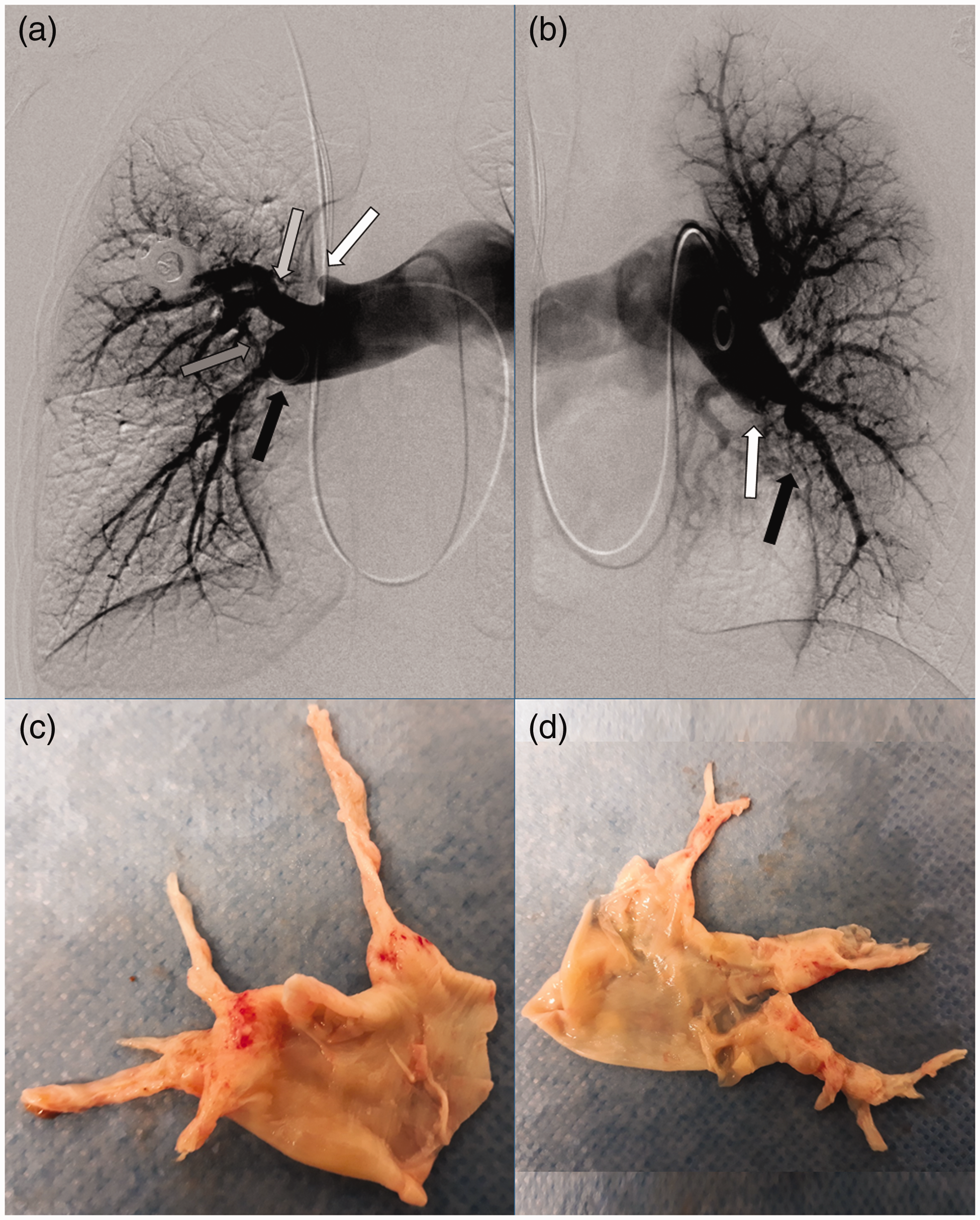
Pulmonary endarterectomy (PEA) was uneventful. A thin-flex single stage cannula of 24Fr was bended for 90°, 3 cm proximal of its tip and this tip was positioned in the ICV to drain the liver veins. A similar second cannula of 20 Fr was bended the same way and positioned in the superior caval vein in order not to obstruct and to adequately drain the azygos system (both Edwards Lifesciences, Irvine, CA). Methylprednisolone of 10 mg/kg added to the priming of the cardiopulmonary bypass system and topical head cooling were used to protect the brain. The patient was cooled to a rectal measured temperature of 20℃ (esophageal temperature 18℃). As the value given by the Bispectral Index™ (BIS™) brain monitoring system at these temperatures was 0, we did not administer thiopental. Circulation was stopped 20 and 25 min on the right and left side, respectively. As in adults, we used the Madani PTE set (Wexler Surgical, Houston, TX). The pulmonary trunk and the right and left pulmonary artery had diameters of 40.6 mm, 23.1 mm, and 27.2 mm, respectively (CT-scan). Surgery was experienced to be slightly easier compared to adults due to more medially localized splits of pulmonary arteries. Resection was considered complete.
Bilaterally, UCSD level II disease (Madani et al.) was found (Fig. 1(c) and (d)): the most proximal fibrous tissue was encountered in lobar arteries expanding distally(1). No disease was encountered in the right middle lobe or lingula.
Extensive reperfusion edema was successfully treated with diuretics and positive pressure ventilation. Extubation occurred on postoperative day 4. PAP (Swan-Ganz) was 44/18 (33) mmHg and CO 6.4 L/min, with a total PVR of 413 dynes.s.cm−5. The patient was discharged from hospital on postoperative day 8. To prevent recurrent CTEPH, lifelong anticoagulation is mandatory. This remains a concern as our patient's short-bowl syndrome makes oral warfarin administration impossible.
Three months later the boy had restarted daily scholar education without supplemental oxygen and considered restarting wheelchair basketball. Hypercoaguable workup revealed increased levels of factor V Leiden and of antiphospholipid antibodies. TTE six months after surgery showed normalized RV size and function and normal PAP (20/7 (14) mmHg).
Discussion
CTEPH results from incomplete resolution of pulmonary thromboemboli and formation of chronic, fibrotic, flow-limiting organized thrombi within the pulmonary vascular bed and occurs in 3.2% of acute pulmonary embolism survivors.2,3 Left untreated, it inevitably results in right ventricular failure and death.
During PEA, the diseased intima, affected by cellular proliferation and inflammatory infiltration, and the fibrotic remodeled thrombi are removed from the pulmonary arteries.4,5 This complex and technically challenging surgical procedure uses cardiopulmonary bypass to cool the patient to 20℃. Subsequently, (repeated) periods of circulatory arrest for a maximum of 20 min each are required. Once completed, circulation is restarted and the patient is brought to normothermia. 6
The previous 10 years, our in-hospital PEA-mortality was 4.8% (international CTEPH registry: 4.4% 7 ) and our five-year survival was 86% (79–82% in larger series8,9) (own unpublished data). Our patient's high PVR increased the surgical risk. However, his young age, high pulmonary pulse pressure, and the proximal anatomic obstruction argued in his favor. 10 Moreover, PEA has been shown to be the strongest independent predictor of survival in CTEPH patients. 11 Therefore, after interdisciplinary operability judgment, we did not hesitate to proceed to surgery.
PEA remains the only potential curative treatment, significantly improving hemodynamics, gas exchange, functional status, and survival. 11 No medical therapy is effective for operable disease. Only Riociguat is currently approved for patients deemed inoperable or with persistent PH after PEA. 2 Balloon Pulmonary Angioplasty (BPA) for inoperable CTEPH is promising; however, long-term results are scarce. In principle, BPA is only used for (sub)segmental originating disease. 2 However, for more proximal disease, palliative BPA procedures can be more appropriate in patients with severe comorbidities and limited expected lifespan. But we aimed to offer a curable treatment to this young patient, despite his impressive co-morbidities.
The two-year-old ICV thrombus was considered organized and fibrotic, probably very adherent to the vessel wall. In order not to damage the ICV and given the well-developed collateral venous drainage, no attempt was made to free up the ICV.
Case-reports of PEA in children are rare.12,13 Two case series have been published. Madani et al. described PEA in 17 patients aged <18 years with minimal postoperative complications and low perioperative mortality, resulting in improved hemodynamics and functional status. 14 Only three were aged ≤12 years. Kumbasar et al. reported three successful pediatric PEA procedures in patients aged 4–13 years. 15 Common presenting symptoms were (exertional) dyspnea, chest pain and syncope. Compared to adults, higher rates of thrombophilic states, higher incidences of unilateral main pulmonary arterial disease, and a higher risk of rethrombosis are notable.12–15
Risk factors for developing CTEPH are, besides previous acute pulmonary embolism, hemostatic disorders and several associated medical conditions. 2 Factor V Leiden is no risk factor, but antiphospholipid antibodies are, with a prevalence of 20% in CTEPH. 2 Also the wheelchair bounded condition and repetitive port-a-cath infections undoubtedly made our patient more susceptible to develop an (infected) ICV thrombus that probably served as source for pulmonary emboli. To the best of our knowledge, we are the first to report a successful PEA in a child with such extensive comorbidities.
As venous thromboembolism and pulmonary embolism are increasingly recognized in pediatric patients, CTEPH should be an important diagnostic consideration in children with a known hypercoaguable state, a history of thrombo-embolism or venous catheter placement, and/or a diagnosis of PH who have exercise intolerance, exertional dyspnea and/or (pre)syncope. 14 A normal ventilation-perfusion scan then rules out CTEPH.
Ongoing CTEPH leads to progressive secondary distal arteriopathy. This condition increases the risk on potential lethal residual PH and reperfusion edema after PEA. 1 In selective cases of disabling residual postoperative PH or inoperable CTEPH, a (double) lung or heart-lung transplantation can be offered to the patient. Long-term results of double lung vs heart-lung transplantation for precapillary PH are comparable and given the donor organ shortage, that is even more pronounced in children, our center's preference for CTEPH goes to a double lung transplantation. 16 However, one should realize that PEA significantly outperforms the results of double lung transplantation for CTEPH.7,11,16 Therefore PEA remains the first and only curative option and transplantation can only be considered as a rescue therapy. Hesitating to refer children for surgical consideration, or attempting to treat them by medication, only postpones the single potentially curable treatment and may worsen their prognosis post PEA. Therefore, the authors want to plea for prompt and early referral to a CTEPH center experienced with PEA in cases CTEPH is suspected or diagnosed in children. Hence, young age itself is not an impediment to surgery.
Footnotes
Authors' note
The boy and his mother delivered written consent to publish patient information and images.
Author contributions
Tom Verbelen and Bart Meyns performed the surgery, and reviewed and revised the manuscript. Tom Verbelen drafted the initial manuscript. Bjorn Cools coordinated the perioperative diagnosis and care of the patient, and reviewed and revised the manuscript. Zina Fejzic diagnosed the patient with CTEPH and referred the patient to our center, and reviewed and revised the manuscript. Raf Van Den Eynde performed anesthesia and immediate postoperative care of the patient, and reviewed and revised the manuscript. Geert Maleux conducted all the imaging modalities, and reviewed and revised the manuscript. Marion Delcroix coordinated the monthly CTEPH meeting, discussing this patients' case and was involved in the postoperative care, and reviewed and revised the manuscript. All authors contributed to the multidisciplinary CTEPH meeting where this case was discussed.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
