Abstract
Relative to healthy subjects, patients with pulmonary arterial hypertension often present with decreased respiratory muscle strength, resulting in decreased maximum inspiratory pressure. Little is known about the impact of reduced respiratory muscle strength on the ability to achieve the peak inspiratory pressures needed for effective drug delivery when using portable dry powder inhalers (≥1.0 kPa). The objective of this study was to assess the impact of inhaler resistance and patient instruction on the inspiratory flow profiles of pulmonary arterial hypertension patients when using breath-actuated dry powder inhalers. The inspiratory flow profiles of 35 patients with pulmonary arterial hypertension were measured with variants of the RS01 dry powder inhaler. Profiles were determined with a custom inspiratory flow profile recorder. Results showed that going from the low resistance RS01 dry powder inhaler to the high resistance AOS® dry powder inhaler led to increases in mean peak inspiratory pressures for pulmonary arterial hypertension subjects from 3.7 kPa to 6.5 kPa. Instructions that ask pulmonary arterial hypertension subjects to inhale with maximal effort until their lungs are full led to a mean peak inspiratory pressures of 6.0 kPa versus 2.1 kPa when the same subjects are asked to inhale comfortably. Significant decreases in mean peak inspiratory pressures are also observed with decreases in lung function, with a mean peak inspiratory pressures of 7.2 kPa for subjects with FEV1 > 60% predicted, versus 3.3 kPa for those subjects with FEV1 < 50% predicted. In conclusion, despite having reduced respiratory muscle strength, subjects with pulmonary arterial hypertension can effectively use a breath-actuated dry powder inhaler. The probability of achieving effective dose delivery may be increased by using dry powder inhalers with increased device resistance, particularly when subjects do not follow the prescribed instructions and inhale comfortably.
Keywords
Introduction
Pulmonary arterial hypertension (PAH) is a chronically progressive disease, which causes ongoing remodeling of pulmonary vasculature, leading to worsening right heart failure and ultimately death. 1 While prognosis in the PAH population continues to be disconcerting, there has been tremendous progress in the development of PAH treatments over the last two decades. 2
Current drug therapies aim to alleviate vasoconstriction, vascular endothelial cell proliferation, smooth muscle cell proliferation, and endothelial dysfunction within the pulmonary vasculature.3,4 These drugs are administered via multiple routes including oral, parenteral, and inhalation. Relative to oral therapeutics, inhaled therapies provide improved targeting to the pulmonary vasculature with a lower nominal dose and decreased off-target effects. 5 Moreover, inhalation provides a more rapid onset of action and improved dose consistency.
The first inhaled therapeutic approved for the treatment of PAH was Ventavis® (nebulized iloprost). 6 Iloprost has a very short duration of action (≈30 min), necessitating frequent administration (6–9 times daily). 6 The daily treatment burden including the time required to gather the supplies, prepare the nebulizer, administer the dose, and clean the device is approximately two hours per day. 7 A second inhaled prostacyclin (Tyvaso®, nebulized treprostinil) was later developed. Tyvaso has an extended duration of action (≈4 h), decreasing the number of treatments to four times daily and significantly reducing daily treatment burden to approximately 39 min per day. 7
More recently, dry powder formulations of treprostinil that enable rapid administration with a portable dry powder inhaler (DPI) have been advanced into late stage development (NCT03399604). 8 A dry powder formulation of vardenafil hydrochloride, a phosphodiesterase type-5 inhibitor, has also advanced into Phase 2 (ACTRN12619001178134). 9 DPIs offer improved convenience (i.e. portable delivery system with no power source requirements), and a significantly reduced daily treatment burden (administration time ≈ 1 min) relative to nebulizers.
Nebulizers are considered “active” devices because the device supplies the energy required to create the aerosolized droplets that are subsequently inhaled. In contrast, with breath-actuated DPIs, a patient’s inspiratory maneuver provides the energy for powder fluidization and dispersion. As such, respiratory muscle function may be an important factor for treatment efficacy with a DPI.
Many patients with PAH exhibit respiratory muscle dysfunction and weakness,10–13 which can negatively impact their maximum inspiratory pressure (MIP). Meyer et al. 10 assessed the MIP values achieved by male and female subjects with PAH in comparison to healthy controls. Male controls achieved a mean MIP of 9.5 ± 2.1 kPa, consistent with previous studies. 14 In contrast, male subjects with PAH had a significantly decreased mean MIP of 6.2 ± 2.6 kPa. Consistent with findings in males, female controls had a mean MIP of 8.2 ± 2.0 kPa, while female subjects with PAH had a mean MIP value of 5.3 ± 2.0 kPa. The low MIP values observed in female subjects with PAH (mean age of 55 years) are comparable to MIP values observed in healthy seven-year-old children. 14
Despite the increased use of portable DPIs in the PAH population, there have been no systematic “breathing studies” published to date. Most subjects inhale with sub-maximal effort when using DPIs (i.e. about 40–80% of their MIP). 14 This coupled with the low MIP values highlight the importance of assessing the ability of subjects with PAH to effectively use portable, breath-actuated DPIs. The objective of the present study was to assess the impact of device resistance and patient instruction on the inspiratory flow profiles of subjects with PAH when using variants of the RS01 DPI (Plastiape S.p.A., Osnago, Italy).
Materials and methods
Ethics
This was a non interventional study conducted at a single large PH center. Institutional Review Board approval was obtained prior to initiation of the study. No active drug or placebo was administered to subjects in the conduct of the study. Written informed consent was obtained from each subject prior to performing any study-related assessment.
Study population
This study enrolled clinically stable subjects between the ages of 18 and 75 years with a confirmed diagnosis of PAH. Relevant and clinically significant medical and social histories were collected by subject interview prior to testing. This included a complete history of their PAH, including data and details of diagnosis, results of pulmonary function tests, and associated illnesses and diseases. All prescription and over-the-counter medications taken at the time of study entry were documented. Based on data extracted from clinical records, subjects were classified in terms of their WHO/NYHA functional class.
Vital signs were measured prior to testing. This included assessments of blood pressure, heart rate, respiratory rate, height, and weight. At the completion of inspiratory flow profile testing, subjects completed a six-minute walk test.
Study design
This investigation assessed the inspiratory flow profiles of subjects with PAH in two randomized crossover studies, A and B, each containing three assessments. All inspiratory flow profile tests were administered by a single individual (S.L.).
Subjects in Group A (N = 18) were tested with three unmarked/unspecified variants of the RS01 DPI (Plastiape S.p.A., Osnago, Italy), which differed in their resistance to airflow. The standard RS01 DPI is available in low resistance (R = 0.019 kPa0.5 L–1 min) and medium resistance (R = 0.031 kPa0.5 L–1 min) designs. The AOS® DPI (supplementary FIGURE E1), a variant designed by variant designed by Respira Therapeutics, Inc. (Albuquerque, NM), has a high resistance to airflow (R = 0.050 kPa0.5 L–1 min). 15
Each device was loaded with an empty capsule that was pierced prior to conducting the inhalation maneuver. Subjects were instructed to exhale to empty their lungs, then inhale with maximal effort until their lungs were full. Subjects performed three replicate inhalation maneuvers with each resistance-blinded device. The inspiratory flow profile with the highest peak inspiratory pressure (PIP) was utilized for the analysis.
Subjects in Group B (N = 17) utilized the AOS DPI and tested three different sets of inhalation instructions: (1) exhale to empty lungs, then inhale with maximal effort through inhaler until your lungs are full (i.e. the instruction utilized in Part A); (2) exhale to empty lungs, then inhale comfortably through the inhaler until your lungs are full; and (3) inhale with maximal effort through inhaler until your lungs are full (no exhalation step before inhalation).
Measurement of inspiratory flow profiles
Patient inspiratory flow profiles were recorded with a custom data acquisition device developed by iPharma Limited (Union City, CA). The setup consists of three main parts: an Inhalation Recorder Box containing a model 8510B-2 pressure transducer (National Instruments Corporation, Austin, TX), a custom-built DPI adaptor, and a laptop computer with custom LabView software (supplementary FIGURE E2).
The test DPI is connected into the adaptor. The adaptor contains a tap that allows connection with the pressure transducer. The adaptor is connected on the distal end to a Respirgard filter (Airlife™ model 303EU, Carefusion, San Diego, CA), which is then connected to a disposable 22 mm mouthpiece (model 56013, Qosina Corp, Ronkonkoma, NY). The filter and mouthpiece were replaced for each subject.
The calibrated pressure transducer measures pressures up to 13.79 kPa (2 psi), with a sampling rate of 10,000 Hz. The LabView software tabulates data from the pressure transducer and smooths the pressure profiles to limit the impact of bead oscillation in the AOS DPI on quantitation of the measured pressures. The software also calculates the various inspiratory flow parameters detailed in Fig. 1.
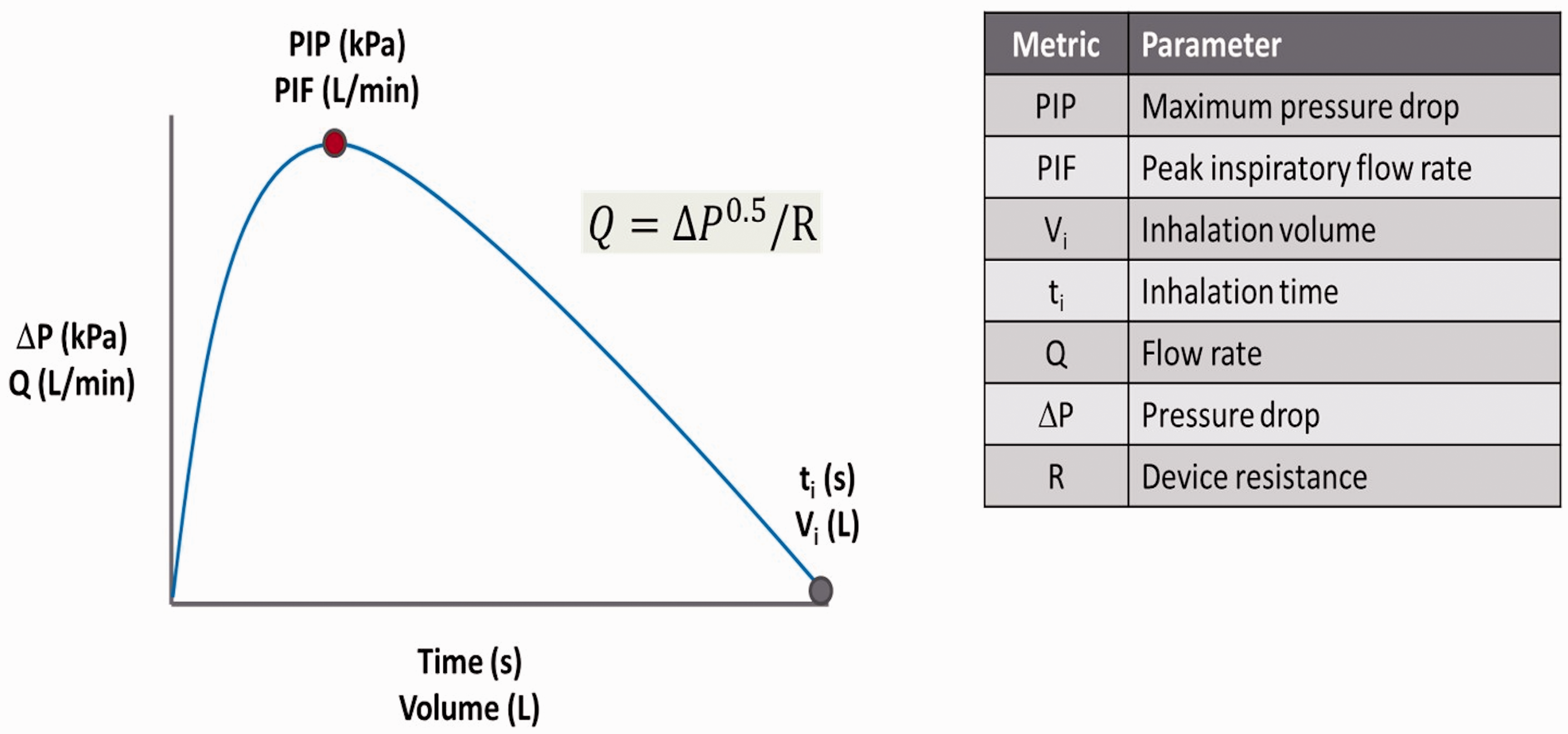
Parameters used to define an inspiratory flow profile.
The inhalation recorder measures the negative pressure drop (ΔP) created by the inhalation maneuver. This can be converted into a flow rate (Q) using equation (1)
The constant of proportionality is the device resistance (
Statistical methods
Continuous data are reported as mean ± SD. Statistical comparisons between various parameters were made using an ANOVA with post-hoc analysis using a two-sided t-test for two independent means. All statistical tests with a p-value < 0.05 was considered statistically significant.
Results
Table 1 lists the demographics of the 35 subjects enrolled in the study. Consistent with the PAH patient population, a large percentage of the subjects (92%) were female. 16 Most of the subjects were functional class II or III, and on a PAH disease-specific drug regimen comprising two or three maintenance medications (94.4%).
Patient demographics.
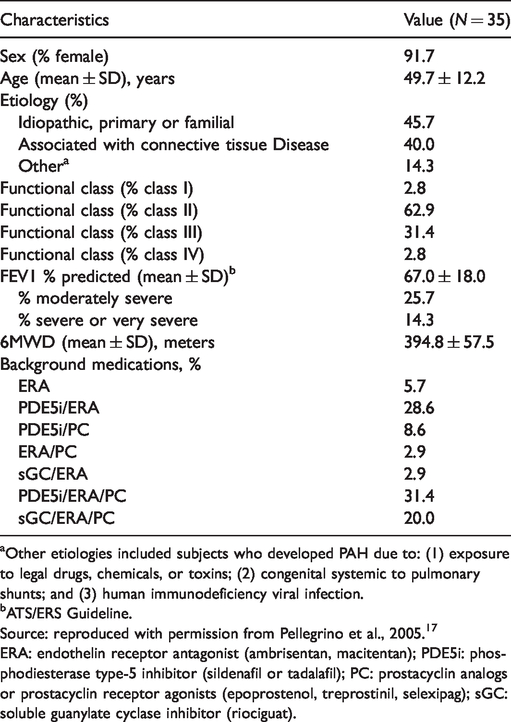
aOther etiologies included subjects who developed PAH due to: (1) exposure to legal drugs, chemicals, or toxins; (2) congenital systemic to pulmonary shunts; and (3) human immunodeficiency viral infection.
bATS/ERS Guideline.
Source: reproduced with permission from Pellegrino et al., 2005. 17
ERA: endothelin receptor antagonist (ambrisentan, macitentan); PDE5i: phosphodiesterase type-5 inhibitor (sildenafil or tadalafil); PC: prostacyclin analogs or prostacyclin receptor agonists (epoprostenol, treprostinil, selexipag); sGC: soluble guanylate cyclase inhibitor (riociguat).
Impact of device resistance on inspiratory flow profiles (Group A)
The inspiratory flow profiles of representative PAH subjects following inhalation with three variants of the RS01 DPI are presented in Fig. 2a and b. The profiles demonstrate that subjects provide greater effort (i.e. a higher PIP) when inhaling through a higher device resistance for the same inhalation instruction. The quantitative inspiratory flow parameters for all 18 subjects in the crossover study are presented in Table 2, with the mean PIP values plotted in Fig. 2c.
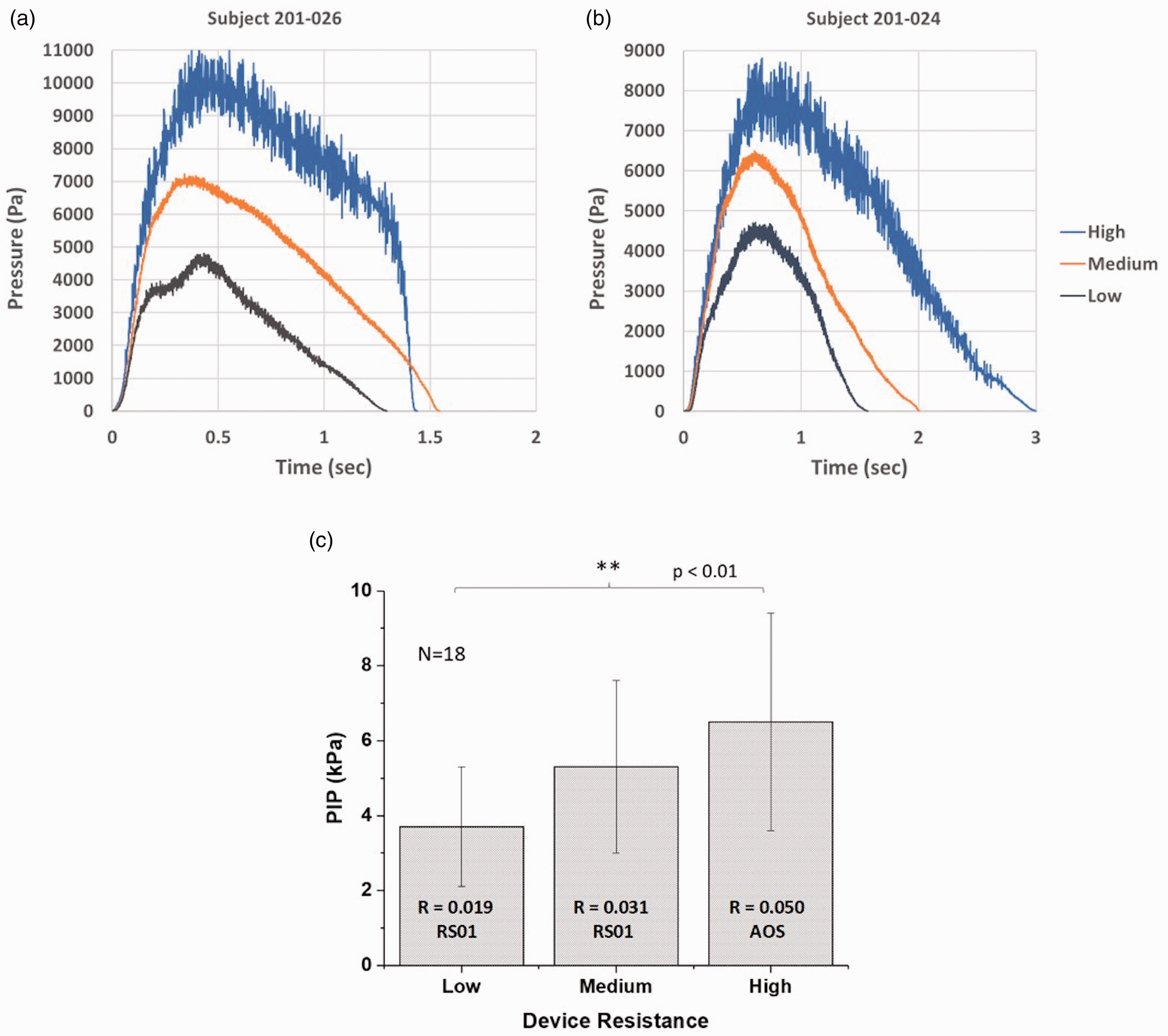
Impact of device resistance on inspiratory flow profiles of PAH patients instructed to inhale with maximal effort until their lungs were full with variants of the RS01 DPI (N = 18); (a) inspiratory flow profiles for representative subject, 201-026; (b) inspiratory flow profiles for representative subject 201-024; and (c) plot of PIP vs. device resistance (**p < 0.01).
Impact of variations in device resistance, instruction on inspiratory flow parameters, and lung function of PAH patients with various portable dry powder inhalers.
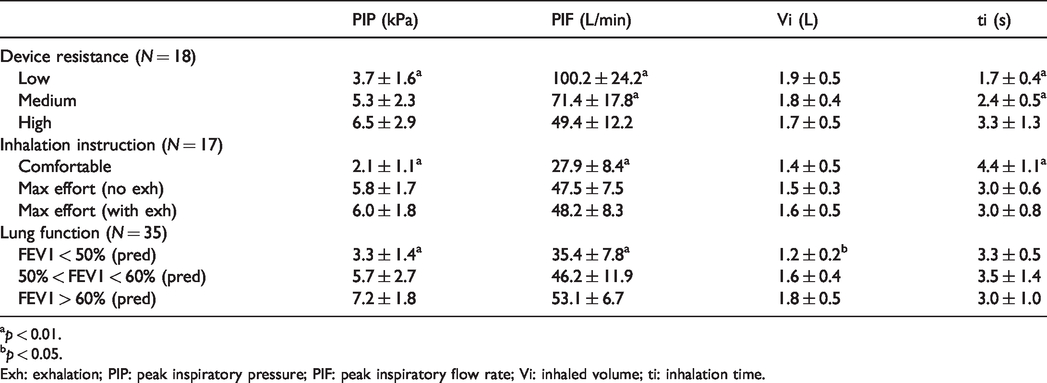
ap < 0.01.
bp < 0.05.
Exh: exhalation; PIP: peak inspiratory pressure; PIF: peak inspiratory flow rate; Vi: inhaled volume; ti: inhalation time.
PAH subjects inhaling through the low resistance RS01 DPI achieved a mean PIP of 3.7 ± 1.6 kPa (N = 18). A significant increase in PIP was observed for subjects when using the higher resistance DPIs, with the high resistance AOS™ DPI (Fig. 2c), having a mean PIP of 6.5 ± 2.9 kPa (p < 0.01).
Increases in device resistance also lead to statistically significant decreases in mean PIF (p < 0.01), as predicted by equation (1) (Table 2). Mean inhalation time, ti, also increased significantly (p < 0.01), as it takes longer to achieve a comparable Vi with a lower inspiratory flow rate (Table 2).
No significant differences in mean Vi were observed for the three RS01 variants (Table 2). Nonetheless, all the measured Vi values are significantly greater than the ∼ 0.5 L volume needed to effectively empty a 25 mg fill mass of powder from the AOS DPI (supplementary FIGURE E4).
Impact of inhalation instruction on inspiratory flow profiles (Group B)
A second randomized crossover study was conducted in 17 PAH subjects to assess the impact of inhalation instruction on PIP and other inspiratory flow parameters with the high resistance AOS DPI (Table 2, Fig. 3). The measured inspiratory flow profiles for representative subjects are presented in Fig. 3a and b.
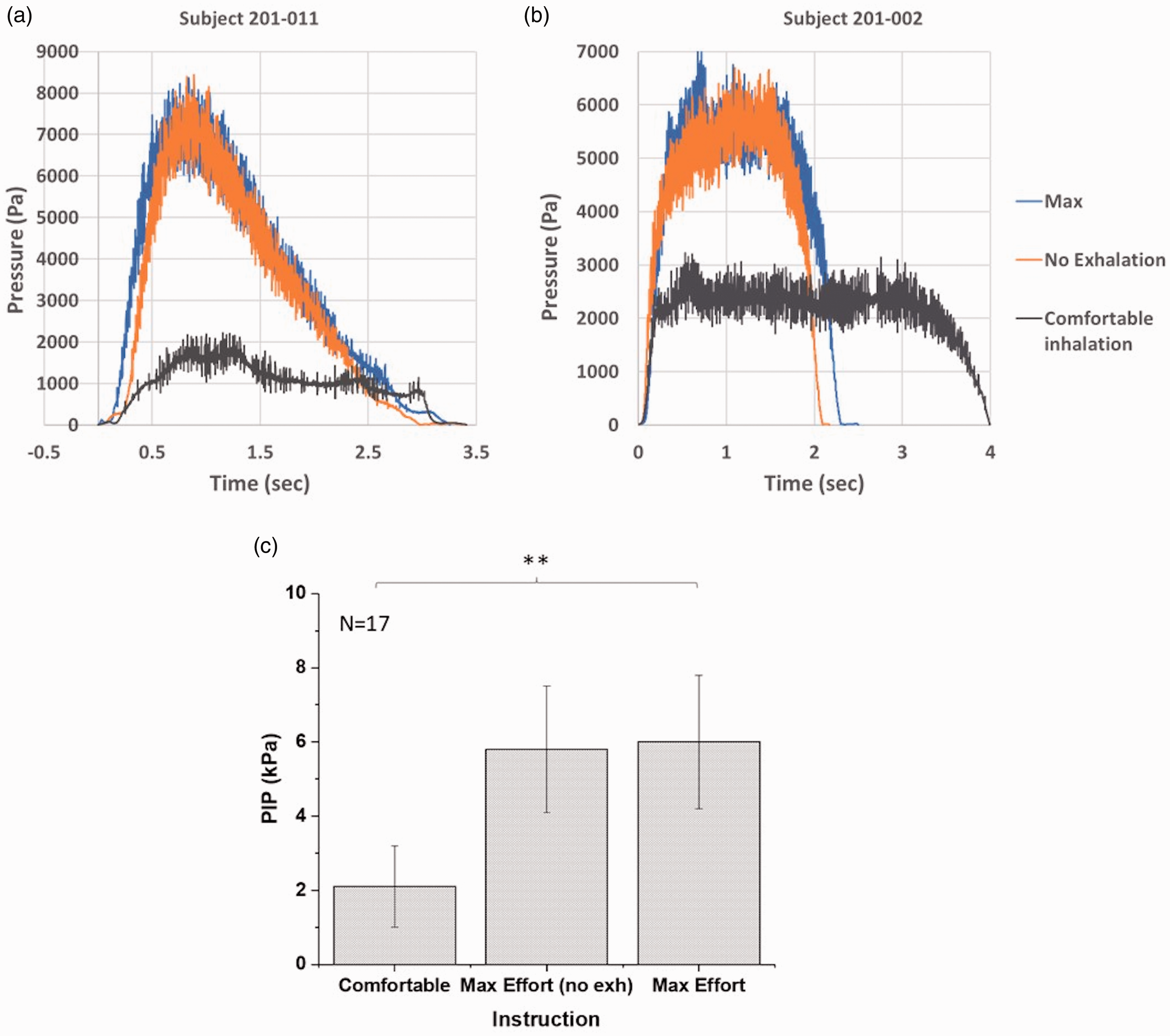
Impact of instruction on inspiratory flow profiles of PAH patients with the high resistance AOS DPI (N = 17); (a) inspiratory flow profiles for representative subject, 201-011; (b) inspiratory flow profiles for representative subject 201-002; and (c) plot of PIP vs. instruction (**p < 0.01).
PAH subjects inhaling with maximal effort until their lungs are full (with a prior exhalation step) had a mean PIP of 6.0 ± 1.8 kPa. Comparable results were observed when subjects inhaled with maximal effort in the absence of a prescribed exhalation step (mean PIP = 5.8 ± 1.7 kPa, p = 0.32). In contrast, a significant decrease in mean PIP was observed when the same PAH subjects were instructed to inhale comfortably until their lungs were full, with a mean PIP of 2.1 ± 1.1 kPa (p < 0.00001) (Fig. 3c). The coefficient of variation on the mean PIP values also increased from 30% for subjects inhaling with maximal effort to 52% for comfortable effort.
Consistent with the decreases in PIP observed, significant decreases in PIF were noted between maximal and comfortable inhalation (48.2 ± 8.2 L/min vs. 27.9 ± 8.4 L/min, p < 0.00001) (Table 2). No significant differences were observed in Vi (Table 2).
Fig. 4 presents a plot of Vi versus PIP for the various inhalation maneuvers with the AOS DPI. The dotted lines represent the target Vi (> 0.5 L) and PIP (> 1.0 kPa) values needed for effective drug delivery 18 (supplemental FIGURE E4). The differences in inhalation instruction are clearly evident. Of the 69 inhalation profiles from the two crossover studies that make up the graph, 67 subjects met the target inhalation criteria (i.e. 97%).
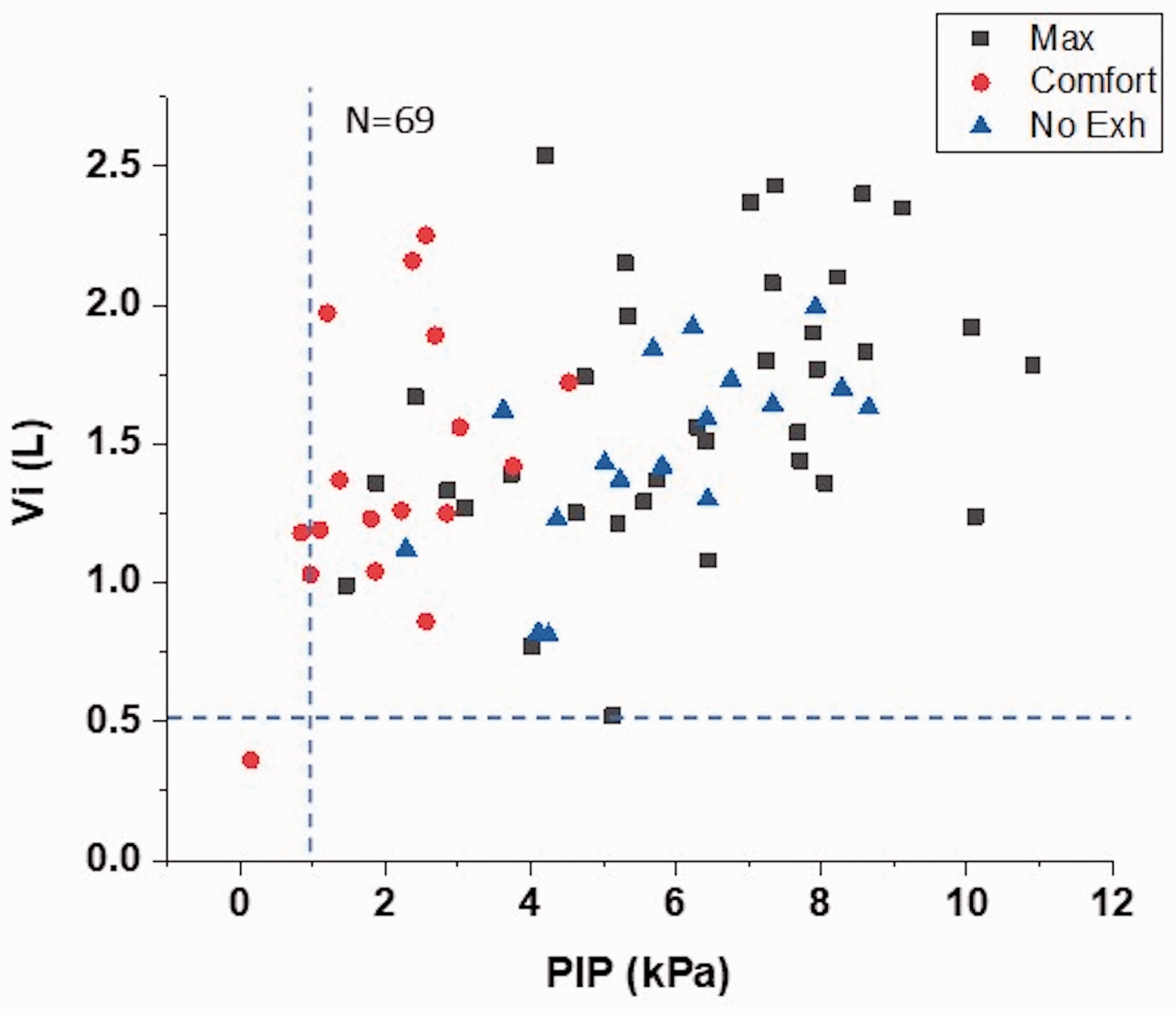
Plot of inhaled volume (Vi) versus PIP for PAH subjects with the AOS DPI and various inhalation instructions (N = 69). The dotted lines represent target values of Vi > 0.5 L and PIP > 1 kPa for effective dose delivery. The squares represent patients who exhale followed by inhalation with maximal effort until their lungs are full. The triangles represent the same maximal effort inhalation with no prescribed exhalation step. The circles represent an instruction to inhale comfortably until your lungs are full, with an exhalation step.
Subgroup analysis
No trends in PIP were observed with variations in age, disease etiology, smoking history, and background PAH medications when using the AOS DPI. Mean PIP values remained constant within these subgroups at ∼6 kPa. Trends that were not statistically significant were observed with variations in PAH functional class and six-minute walk test distance (6MWTD). The detailed results are presented in supplemental TABLE E2 and supplemental FIGURE E3.
Marked differences in PIP were observed for PAH subjects with decreased lung function (Table 2, Fig. 5). PIP decreased from 7.2 ± 1.8 kPa for subjects with an FEV1 > 60% predicted to 3.3 ± 1.4 kPa for subjects with an FEV1 < 50% predicted (p < 0.01). Significant decreases were also observed in Vi, with reductions from 1.8 ± 0.5 L to 1.2 ± 0.2 L as lung function decreased (p < 0.05).
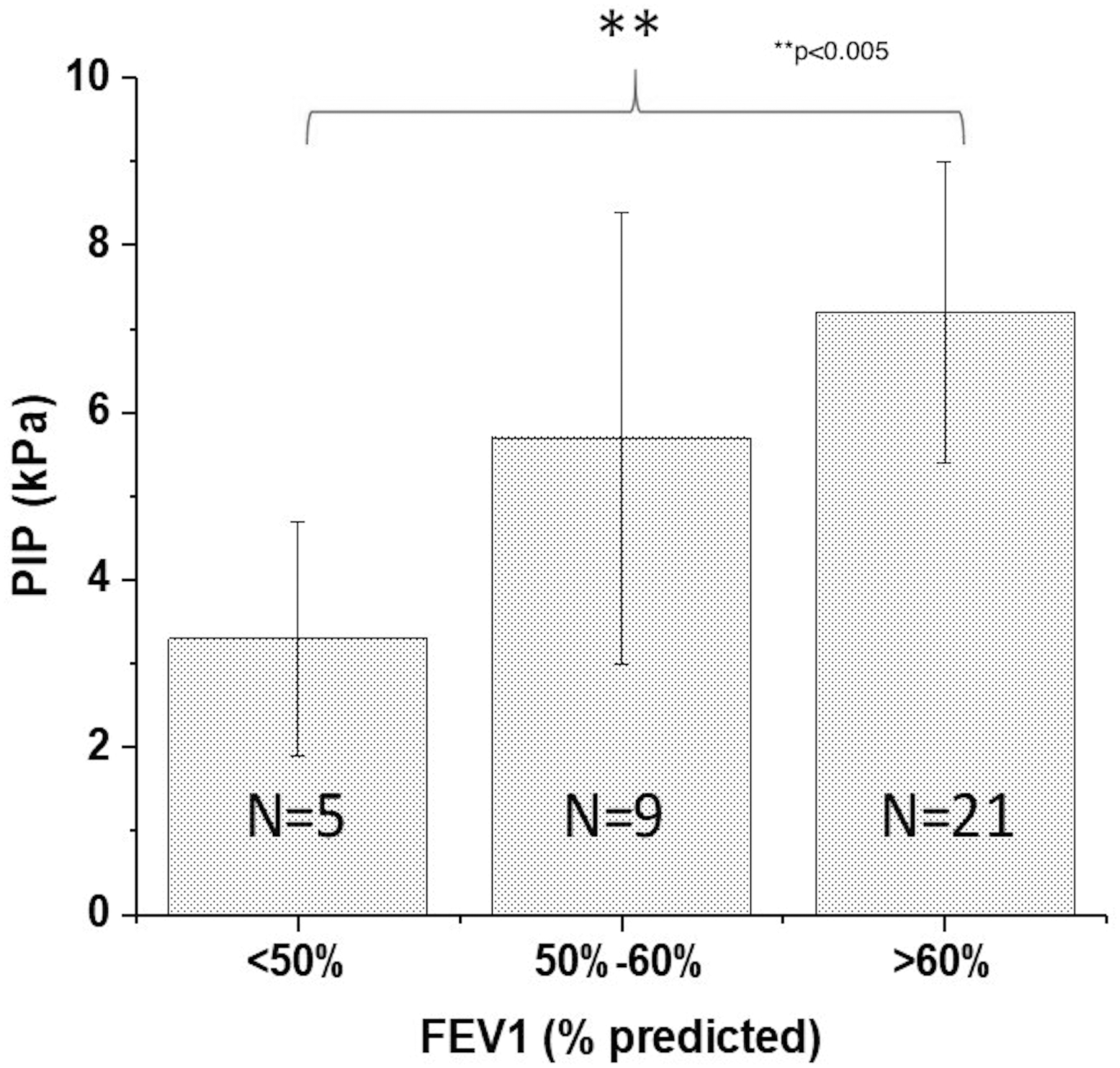
Plot of PIP as a function of variations in lung function with the AOS DPI.
Safety and tolerability
No adverse events were reported in this observational study. None of the 35 subjects had difficulty in performing multiple inhalations over a short period of time with the AOS DPI. Moreover, there were no complaints of discomfort when using the high resistance inhaler.
Discussion
Compared to other respiratory diseases, comparatively little is known about the inspiratory flow profiles of PAH patients when using DPIs. Their low mean MIP values highlights the importance of directing them to inhale with effort, either through instruction/training, or via incorporation of specific design features within the DPI that lead them to provide greater effort.
Impact of device resistance and inhalation instruction
In the present study, PAH subjects achieved a mean PIP of 3.7 ± 1.6 kPa when inhaling with maximal effort through the low resistance RS01 DPI. This is about 60% of their mean MIP value,10,11 and consistent with previous studies where subjects typically inhale at about 40–80% of their MIP when using portable DPIs. 14 It is important that subjects exceed the minimum PIP needed to provide effective drug delivery to the lungs. Clark et al. 18 argue that for most DPIs, this occurs when PIP ≥1 kPa. Hence, when compliant with the preferred instruction to inhale with maximal effort, most subjects are expected to achieve a PIP sufficient to achieve effective drug delivery.
When inhaling with maximal effort with the high resistance AOS DPI, PAH subjects achieved a mean PIP value of 6.5 ± 2.9 kPa. This mean PIP is comparable to the mean MIP values observed in previous studies.10,11 Hence, inhaling against a higher resistance leads patients to provide greater inspiratory effort naturally, for the same inhalation instruction (Fig. 2). This observation is not unique to PAH subjects, and has likewise been observed in subjects with asthma, chronic obstructive pulmonary disease (COPD), and cystic fibrosis (CF).19–22 The increased PIP observed with the high resistance AOS DPI increases the probability that most or all PAH subjects will achieve an acceptable inhalation profile.
Inhalation instructions can have a large impact on patient inspiratory flow profiles (Fig. 3). Instructing subjects to inhale comfortably through the AOS DPI leads to significantly lower mean PIP values (2.1 ± 1.1 kPa) than when subjects are instructed to inhale with maximal effort (6.0 ± 1.8 kPa) (Table 2). Unfortunately, while patients may follow inhalation instructions during training or while being instructed in a clinical trial, they often revert to inhaling more comfortably in real-world situations. This may be problematic for low resistance DPIs, where the PIP values are nearly 3 kPa lower than is observed with the AOS DPI. If comparable drops in PIP were observed with the low resistance RS01 DPI when inhaling comfortably, this could negatively impact drug delivery for some patients. The use of the high resistance AOS DPI in subjects with PAH helps to ensure that most subjects receive a therapeutic dose, independent of whether they inhale with maximal effort or comfortably through their inhaler. In the context of the present study, 97% of the inhalation maneuvers studied with the AOS DPI (N = 69) resulted in effective dose delivery as indicated by a PIP > 1.0 kPa and a Vi > 0.5 L (Fig. 4). The one subject who achieved a PIP of just 0.15 kPa when inhaling comfortably achieved a PIP of 7.3 kPa when inhaling with maximal effort. Clearly the subject was capable of achieving the target PIP but failed to do so with the comfortable inhalation instruction.
Exhalation before inhalation
The strength of the respiratory muscles decreases with lung inflation, with maximum muscle strength at residual volume and almost zero muscle strength when approaching total lung capacity when the respiratory muscles are fully extended. 23 Thus, if a subject begins inhalation from a larger lung volume, they will produce a lower mouth pressure.
Nonetheless, dropping the exhalation before inhalation instruction seems to have little impact on the pressure drop and Vi that subjects with PAH achieve with the AOS DPI (Table 2). Similar results were obtained in elderly COPD subjects with the Simoon™ DPI (Novartis, San Carlos, CA). 24 From a human factors’ perspective, it is an unnatural behavior for subjects to inhale through a DPI when their lungs are near total lung capacity, and hence the instruction may not be critical for effective drug delivery with the AOS DPI. Given that this instruction is deemed to be one of the most prevalent errors in using breath-actuated DPIs, 25 this observation is important in assessing the usability of the AOS DPI. Indeed, asking patients to exhale before they inhale may actually increase the probability of a critical error if patients exhale into the mouthpiece of the device.
Impact of lung function on inspiratory flow rates
Decreases in PIP, PIF, and Vi are observed for FEV1 values < 60% predicted (Table 2). In contrast to patients with obstructive lung disease where decreases in PIP are on the order of 20%, 14 reductions by more than 50% are observed in subjects with PAH.
Study limitations
This study had certain limitations. The inspiratory flow profiles measured in this controlled study may not reflect the inhalation profiles that subjects with PAH achieve in real-world situations (e.g. when they are at home or when performing an activity of daily living that leaves them breathless). This study also did not assess the impact of the differences in inspiratory flow profiles on drug delivery and pharmacological effects for a target drug product. The effectiveness of an inspiratory flow profile in dispersing a dry powder formulation will be product specific. Finally, the study would have benefited from having subjects inhale through the low resistance RS01 with a comfortable inhalation instruction. When we designed the study, we were concerned with the low muscle strength of subjects with PAH and how many inhalations they could reasonably complete over a short period of time.
Conclusion
In summary, despite having reduced muscle strength, PAH patients can effectively use breath-actuated DPIs. The probability of achieving effective drug delivery, especially when patients do not follow the preferred inhalation instructions or have FEV1 < 60% predicted, may be increased by using DPIs with increased device resistance.
IRB approval
Yes.
Patient consents
All patients provided informed consent.
Supplemental Material
sj-pdf-1-pul-10.1177_2045894020985345 - Supplemental material for Impact of human behavior on inspiratory flow profiles in patients with pulmonary arterial hypertension using AOS™ dry powder inhaler device
Supplemental material, sj-pdf-1-pul-10.1177_2045894020985345 for Impact of human behavior on inspiratory flow profiles in patients with pulmonary arterial hypertension using AOS™ dry powder inhaler device by Sandeep Sahay, Royanne Holy, Shirley Lyons, Edwin Parsley, Mari Maurer and Jeffry Weers in Pulmonary Circulation
Footnotes
Acknowledgements
The authors acknowledge Jennifer Lee CCRP and Ann Saulino RN.
Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.S. serves as a speaker and consultant for Actelion, United Therapeutics, and Bayer. He is also an Advisor to Liquidia Technologies and Altavant Sciences. R.H. has nothing to disclose. S.L., M.M., and J.W. are employees of the sponsor Respira Therapeutics, Inc. E.P. is a consultant of the sponsor Respira Therapeutics, Inc.
Author contributions
S.S.: responsible for patient enrollment, conduct of the study, consenting, oversight, scientific input in manuscript, and revision of the manuscript; R.H.: enrolling patient, consenting, and reviewing inclusion/exclusion criteria; S.L.: performing guiding patients in inspiratory flow profile measurements on patients; M.M.: protocol preparation; E.P.: study design and protocol preparation; and J.W.: concept design, protocol preparation, and initial version of protocol and manuscript.
Guarantor
Jeffry Weers, Respira Therapeutics.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Respira Therapeutics, Inc., Albuquerque, NM, USA.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

