Abstract
Globally, non-communicable diseases are increasing in people living with HIV. Pulmonary hypertension is a rare non-communicable disease in people living with HIV with a reported prevalence of <1%. However, data on pulmonary hypertension in people living with HIV from Africa are scarce and are non-existent from Ethiopia. This study aimed to examine the prevalence and severity of echocardiographic pulmonary hypertension and risk factors associated with pulmonary hypertension in people living with HIV in Ethiopia. A total of 315 consecutive adult people living with HIV followed at the Tikur Anbessa Specialized Hospital HIV Referral Clinic were enrolled from June 2018 to February 2019. Those with established pulmonary hypertension of known causes were excluded. A structured questionnaire was used to collect data on demographics, respiratory symptoms, physical findings, physician-diagnosed lung disease, and possible risk factors. Pulmonary hypertension was defined by a tricuspid regurgitant velocity of ≥2.9 m/sec on transthoracic echocardiography. A tricuspid regurgitant velocity ≥3.5, which translates into a pulmonary arterial pressure/right ventricular systolic pressure of ≥50 mmHg, was considered moderate-to-severe pulmonary hypertension. The mean age of the participants was 44.5 ± 9.8 years and 229 (72.7%) were females. Pulmonary hypertension was diagnosed in 44 (14.0%) of participants, of whom 9 (20.5%) had moderate-to-severe disease. In those with pulmonary hypertension, 17 (38.6%) were symptomatic: exertional dyspnea, cough, and leg swelling were seen in 12 (27.3%), 9 (20.5%), and 4 (9.1%), respectively. There was no significant difference in those with pulmonary hypertension compared to those without the disease by gender, cigarette smoking, previous history of pulmonary tuberculosis treatment, physician-diagnosed chronic obstructive pulmonary disease or bronchial asthma, duration of anti-retroviral therapy therapy or anti-retroviral regimen type. Pulmonary hypertension looks to be a frequent complication in people living with HIV in Ethiopia and is often associated with significant cardiopulmonary symptoms. Further studies using right heart catheterization are needed to better determine the etiology and prevalence of pulmonary hypertension in people living with HIV in Ethiopia compared to other countries.
Keywords
Introduction
The development of highly active antiretroviral therapy (HAART) has led to increased survival and a change in the epidemiology of diseases in people living with HIV (PLHIV).1,2 Non communicable diseases such as cardiovascular disease, metabolic syndrome, and some malignancies are replacing Acquired Immunodeficiency Syndrome.3–5 as PLHIV are living longer. Pulmonary hypertension (PH) is one such disease that contributes to morbidity and mortality in these individuals. Although the reported incidence of PH in PLHIV is low, it occurs much more frequently in HIV patients than in the general population.6,7
The association of HIV with PAH is well established 8 and the prevalence of pulmonary arterial hypertension (PAH) PAH in HIV-positive persons is about 0.5% which is nearly 2500 times greater than in HIV-negative people. 9 The cause of PAH in HIV is not well understood but is believed to be due to HIV-related proteins such as transcriptional trans-activator (Tat), Negative factor (nef), and Gp120 that target the pulmonary endothelium and promote inflammation, endothelin secretion, and down regulation of BMPR2. 10 However, various other factors contribute to an increasing prevalence of PH in PLHIV. These include extended survival after HIV diagnosis, female gender 11 , and in some populations increased prevalence of smoking and associated diseases such as chronic obstructive pulmonary disease (COPD), pulmonary tuberculosis treatment, uncontrolled viral infection (high viral load and low CD4), 11 and current use of protease inhibitors. 12 Current use of the antiretroviral drug tenofovir was reported to be less associated with PAH. 13
Although definitive diagnosis of PAH requires right heart catheterization, transthoracic echocardiography has become the standard screening tool for assessing the presence of PH in cohort studies of patients at increased risk of having the disease. 14 Numerous studies have used echocardiography to indirectly estimate systolic pulmonary arterial pressure by measuring the tricuspid regurgitant velocity (TRV) and the modified Bernouli formula to calculate right ventricular peak pressure.9,11,15–22
The prevalence of PH associated with HIV in the developed world varies from 0.5% in studies using right heart catheterization 23 to 6% in echocardiography-based studies. 15 Data on HIV-associated PH are scarce in Africa and only three large studies in adults have been published thus far.19–21 The three studies included symptomatic patients and only one screened for the presence or absence of associated risk factors. In a systematic review that included those three studies, the pooled prevalence of PH in HIV was 14%, more than double that of any study outside of Africa. 24 While this could be due to methodology, contributing risk factors of schistosomiasis and high altitude may have synergistic effect.
Data on PH in PLHIV in Ethiopia are non-existent. This study aimed to examine the prevalence and associated factors for echocardiographic PH in adult PLHIV cared for at the largest HIV referral center in Ethiopia.
Methods
We conducted a cross-sectional study in the HIV clinic of Tikur Anbessa Specialized Hospital (TASH) which is located in the capital of Ethiopia, Addis Ababa. TASH is the largest tertiary teaching hospital in the country with a total bed capacity of nearly 700 and provides specialized referral services throughout Ethiopia. It has an established HIV referral clinic run by HIV/anti-retroviral therapy (ART)-trained nurses, residents, and infectious diseases specialists. All consenting HIV participants ≥18 years old who were seen at the HIV referral clinic from June 2018 to February 2019 were included. Patients with evidence of Group 2 PH on echocardiography and those with already known Group 3 PH from interstitial lung disease (ILD), COPD, or post-tuberculosis structural parenchymal pathologies were excluded. The study was approved by the institutional review board of our University.
Sample size was calculated using Statcalc in EPI info version 7.1 software. Based on the assumption of 14% expected proportion of PH in the HIV patients, 12 with a confidence level of 95%, and margin of error 3.8, a total of 320 patients were needed.
A structured questionnaire was employed to collect data on the clinical manifestations, demographics, risk factors, and echocardiography findings of all the participants irrespective of the presence or absence of symptoms. All echocardiography examinations were performed by cardiologists on the same echocardiography machine (GE-Vivid E 9) with an S3MHz transducer. It included M-mode, bi-dimensional imaging, pulsed and continuous Doppler. Color Doppler was used to help identify the presence of tricuspid regurgitation and to achieve a parallel position to the ultrasound beam. The noninvasive assessment of the pulmonary artery pressure was estimated by the addition of the trans-tricuspid gradient (obtained by the peak systolic velocity of the tricuspid regurgitation and applying the Bernouilli formula) to the right atrium pressure according to the American Society of Echocardiography recommendations. 18 PH was defined as a tricuspid regurgitant (TR) velocity ≥2.9 m/sec which translated to a right ventricular pressure above 35 mmHg. Bubble study was not done during the examination.
Statistical analysis was conducted using SPSS version 20. Descriptive analysis was performed for socio-demographic variables, respiratory symptoms, PH risk factors, and echocardiography values. Chi-square tests were performed to look for association between HIV status, ART duration, age of the patient, recent viral load/CD4 level, and PH status. Bivariate analysis was conducted to see associations between associated factors and PH. Values are reported as mean ± standard deviation.
Results
A total of 315 patients were enrolled. The mean age of the participants was 44.5 ± 9.8 years and 229 (72.7%) were females (Table 1). Echocardiographic evidence of PH was seen in 44 patients (14.0%). Of these 44 patients, one-fifth (20.5%) had moderate-to-severe disease defined as pulmonary arterial systolic pressure (PASP) ≥50mmHg or TRV of ≥3.5 m/sec and over one-third (38.6%) were symptomatic. The prevalence of PH was no greater in women (12.2%) than it was in men (18.6%).
Sociodemographic characteristics and other health-related behaviors of adult patients visiting HIV-clinic at Tikur Anbessa Hospital, 2019.
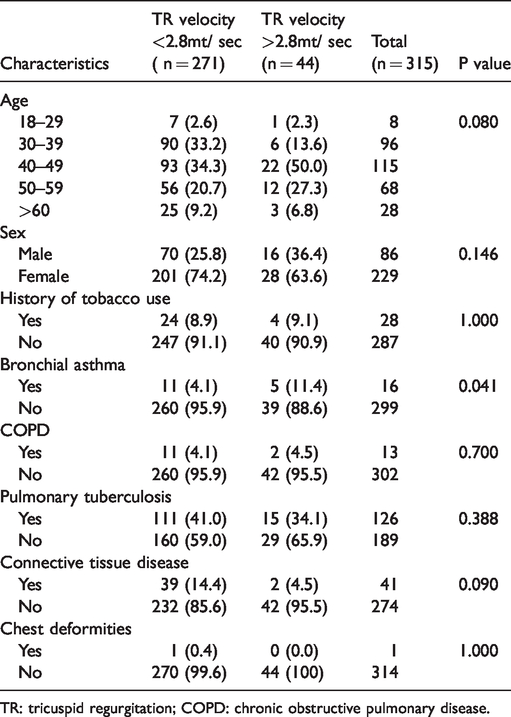
TR: tricuspid regurgitation; COPD: chronic obstructive pulmonary disease.
Among patients with PH, the most common symptoms were exertional dyspnea (27%) cough (21%), and chest pain (18%; Table 2). Chest pain and shortness of breath were more common in patients with PH than in those without.
Clinical manifestations of patients with pulmonary hypertension among adult patients visiting HIV-clinic at Tikur Anbessa Hospital, 2019.
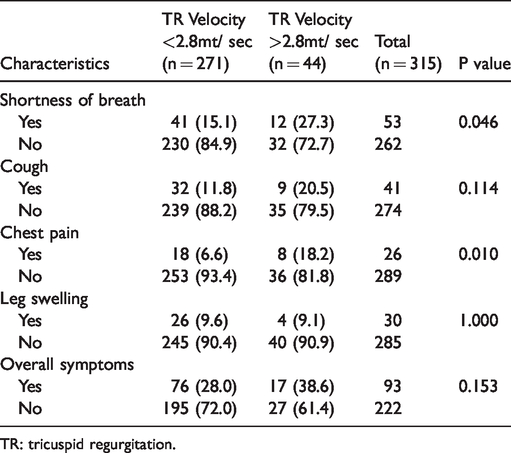
TR: tricuspid regurgitation.
Although patients with PH associated with previously diagnosed chronic heart or lung disease were not included in this study, a small number of the patients we enrolled were found to have these conditions. Approximately 3% were found to have COPD, but all of these patients were felt to have PH out of proportion to the underlying lung disease. There were also a small number of patients who self-reported an asthma diagnosis but never had pulmonary function tests (PFTs) testing to confirm the diagnosis. This was found to be significantly associated with PH in our study population (p = 0.041). However, no significant association was seen between the presence of PH and sex, smoking history, COPD, connective tissue diseases, chronic heart diseases, or chest deformities. The prevalence of PH did not appear to be related to the severity of HIV infection or treatment history. We saw no association between the presence of PH and current CD4 levels, HIV viral load, duration of HIV infection, or ART regimen used (see Tables 1–3).
Immunologic, virologic, and treatment status of adult patients visiting HIV-clinic at Tikur Anbessa, Hospital 2019.
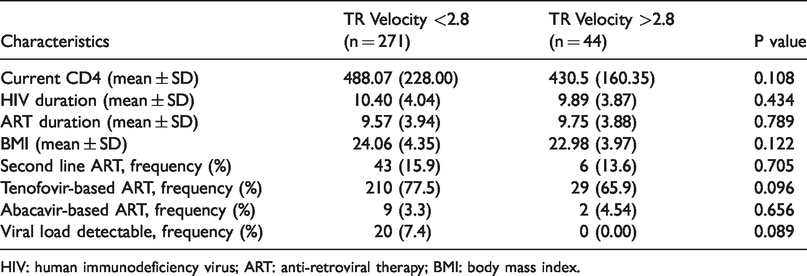
HIV: human immunodeficiency virus; ART: anti-retroviral therapy; BMI: body mass index.
Discussion
In the present study, we used transthoracic echocardiography to screen for the presence of PH in consecutive HIV patients presenting for evaluation or follow-up at the HIV Clinic in TASH. Echocardiographic evidence of PH was seen in nearly 1 out of 7 patients. The great majority of patients (>86%) were between 40 and 59 years of age and men were affected at least as often as women. HIV patients with PH were more likely to have symptoms of shortness of breath and chest pain than those without PH.
Previous studies evaluating the prevalence of PH in PLHIV produced varied results depending on the availability of HAART and the method of screening for PH. Prior to the availability of HAART, prevalence of PH in PLHIV was found to be about 0.5% using transthoracic echocardiography 9 and a similar prevalence was reported during the HAART era in a large prospective study in France that use right heart catheterization. 23 . However, subsequent studies using transthoracic echocardiography have found a higher prevalence of PH in PLHIV.11,15–18,22 Studies done in Africa have also reported higher prevalence rates,19–21,24 although some of these studies used a cut-off level of systolic pulmonary arterial pressure >30 mmHg or TR velocity greater than 2.5 m/sec to define PH and likely overestimated the true prevalence of elevated PA pressure. In studies like ours, that use the more acceptable echocardiographic estimate of PH as right ventricular systolic pressure >36 or TR velocity >2.8, the prevalence of PH has been <10%. In the present study, using the more stringent PH screening criteria, we found a prevalence higher than that reported in studies from the western,11,15,17,18 African,19–21 and Asian 22 countries (see Table 4), but similar to the pooled prevalence reported from Africa. 24 However, unlike the other African studies which focused on symptomatic patients, our study included patients visiting the HIV referral clinic irrespective of the presence or absence of symptoms. The reason for what may be a higher prevalence of PH in HIV patients living in Sub Saharan Africa is unclear, but could be due to the presence of delayed diagnosis of HIV resulting in a greater period of time in which HIV was present and untreated. Finally, it should be noted that Addis Ababa is at an altitude of 7800 feet where reduced oxygen tension could contribute to elevation of PA pressure. However, population-based studies of Ethiopians living in the Simien plateau in Northern Ethiopia suggest that native Ethiopians living at high altitude have evolved to maintain near normal oxygen saturation. 25 The impact of Schistosomiasis might not be undermined here.
Comparison of studies that used echocardiography to define Pulmonary Hypertension, 2019.
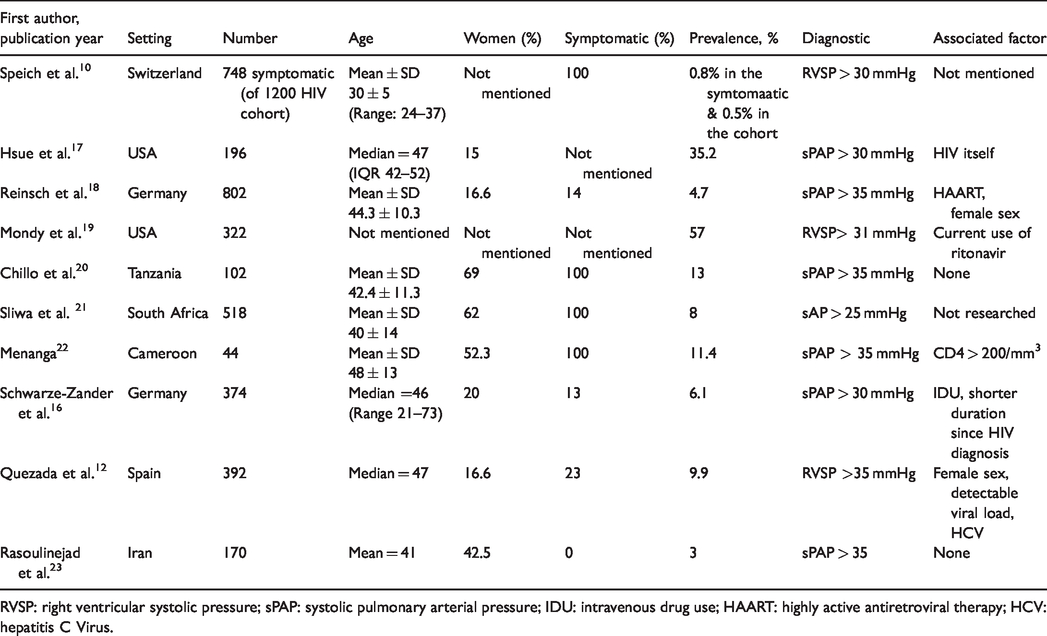
RVSP: right ventricular systolic pressure; sPAP: systolic pulmonary arterial pressure; IDU: intravenous drug use; HAART: highly active antiretroviral therapy; HCV: hepatitis C Virus.
None of the previously known factors such as age, gender, CD4 level, and viral load that are associated with increased risk of PH were associated with the presence of PH in this study. Therefore, PH in this patient population may be related to HIV infection itself. Alternatively, the prevalence of PH in Ethiopia may be higher than other parts of the world. A history of asthma was found to be associated with PH in this study. However, asthma was self-reported and not confirmed by outside records or pulmonary function testing. Thus, it is possible that some of these patients had been misdiagnosed with “bronchial asthma” while they were symptomatic from PH. Asthma is a common diagnosis given to patients with unexplained dyspnea and PH can rarely masquerade as asthma. 26
The strength of the present study includes the large number of patients enrolled compared to previous studies conducted in Sub-Saharan Africa19,21 and that it screened for PH irrespective of the presence or absence of symptoms. Its limitations include the cross-sectional design of the study, the methodology of transthoracic echocardiography (TTE) compared to right heart catheterization (RHC) that may account for the higher estimated prevalence, the lack of additional diagnostic tests such as chest imaging, pulmonary function tests, or spirometry to exclude PH associated with lung disease, and the lack of right heart catheterization to confirm the diagnosis. However, right heart catheterization is not generally available in Ethiopia. In addition, details of the recruitment procedure were not documented (e.g., the number of patients who were excluded because of refusal to participate or inconvenience or those with established Group 2 or 3 PH).
PH as suspected by transthoracic echocardiography appears to be common in PLHIV in Ethiopia. Over a quarter of PLHIV patients with PH have symptomatic dyspnea and/or chest pain. While further studies will be needed to confirm the prevalence of HIV-associated PH in Ethiopia, the present study adds considerably to the limited data available regarding this condition in Sub-Saharan Africa and suggests that PH may be an important comorbid condition of PLHIV. The high prevalence of possible PH in this population suggests that routine screening by echocardiography may be warranted. In patients with HIV-associated PH, further testing to exclude hypoxemia, chronic heart and lung disease, and sleep-disordered breathing should be considered.
Footnotes
Acknowledgements
The authors would also like to thank the East African Training Initiative (Addis Ababa University, Vital Strategies and the Swiss Lung Foundation.
Author contributions
Dawit Kebede Huluka contributed to inception of the research question, protocol preparation, organization of the work, data analysis and write of the manuscript. Desalew Mekonnen contributed to data collection and manuscript reviewing. Sintayehu Abebe contributed to data collection, organization of data collection, and write up of the manuscript. Amha Meshesha and Dufera Mekonnen contributed to data collection and manuscript reviewing. Negussie Deyessa contributed to protocol draft, data analysis, and write up. James R. Klinger and Corey E. Ventetuolo contributed to write up of the manuscript. Neil W. Schluger and Charles B. Sherman contributed to protocol draft and write up. Wondwossen Amogne contributed to protocol draft and write up.
Conflict of interest
The authors whose names are listed above certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript. This case was presented at the American Thoracic Society International Conference in Dallas, Texas, May 2019.
Funding
This article is financially supported by an NIH funded project, Medical Education Partnership Initiative—Junior Faculty (D43W010143). The authors would like to pass our gratitude to MEPI-JF program at Addis Ababa University for financial support of the project.
Supplemental material
Supplemental material for this article is available online.
