Abstract
Background
Adiponectin is a polypeptide hormone related to obesity, and a known modulator of pulmonary vascular remodeling. Association between plasma adiponectin levels and pulmonary hypertension (PH) has not been studied in African Americans (AAs) who are disproportionately affected by obesity. The relationship between adiponectin and heart failure (HF) and mortality, outcomes associated with PH, is unclear.
Methods
We performed cross-sectional and longitudinal analysis to examine if there is an association between plasma adiponectin and PH and associated clinical outcomes, in participants of Jackson Heart Study (JHS). JHS is a prospective observational cohort study of heart disease in AAs from Jackson, Mississippi.
Results
Of the 3161 participants included in the study, mean age (SD) was 56.38 (12.61) years, 1028 were men (32.5%), and mean (SD) BMI was 31.42 (7.05) kg/m2. Median (IQR) adiponectin was 4516.82 (2799.32–7065.85) ng/mL. After adjusting for potential confounders including BMI, higher adiponectin levels were associated with increased odds of PH (adjusted odds ratio per log increment in adiponectin, (1.81; 95% CI, 1.41–2.32). High adiponectin levels were also associated with associated HF admissions (adjusted hazard ratio [HR] per log increment in adiponectin, 1.63, 95% CI, 1.24–2.14) and mortality (adjusted HR per log increment in adiponectin, 1.20; 95% CI 1.02–1.41).
Conclusions
Elevated plasma adiponectin levels are associated with PH, HF admissions and mortality risk in AAs. High adiponectin levels may help identify an at-risk population that could be evaluated for targeted prevention and management strategies in future studies
Introduction
Adiponectin is a polypeptide hormone secreted by adipocytes that is involved in multiple pathways of inflammatory, hormonal, thrombotic and metabolic signaling and has been identified as a modulator of vascular remodeling.1–9 These pathophysiological mechanisms and pathways are important in the development of pulmonary vascular disease and suggest a possible relationship between the adipose tissue and pulmonary vascular health. Basic science and in vivo studies have shown beneficial effects of adiponectin on low grade inflammation, oxidative stress and apoptosis, underscoring an important role for adiponectin as a protective adipokine in cardiovascular and vascular function.10,11 However, in contrast to these observations, large prospective epidemiological studies have demonstrated higher adiponectin levels to be associated with adverse prognosis, with increased risk of mortality in several chronic disease states, including diabetes and cardiovascular disease.12,13 This concept of ‘adiponectin paradox’ remains to be understood, particularly in a complex disease process like pulmonary hypertension (PH).
PH is an important chronic illness in the African American (AA) community. 14 However, to our knowledge, the relationship between circulating adiponectin levels and PH has not been studied in a community-based cohort of AAs. Importantly, whether adiponectin can serve as a biomarker of PH in a vulnerable AA cohort and predict future HF admissions or mortality is unknown. In this study we sought to examine the relationship of plasma adiponectin with PH and clinical outcomes, namely mortality and HF admissions in a large community-based cohort of AAs, a population group at risk for obesity, PH and HF.
Methods
Study design and population
We conducted cross-sectional and longitudinal analyses using data from the Jackson Heart Study (JHS). The conduct of the JHS was approved by the University of Mississippi Medical Center Institutional Review Board. JHS is a longitudinal, population-based cohort study of cardiovascular disease that recruited noninstitutionalized AA adult participants (N = 5306) residing in Jackson, Mississippi. 15 Participants answered predefined questionnaires, underwent venipuncture, including plasma Adiponectin measurement, echocardiography, and spirometry at the time of first examination between 2000 and 2004.
The cohort used for this study included participants who had measureable tricuspid regurgitation (TR) jet velocity on echocardiography (allowing for estimation of the PASP, as described in Outcomes section in Supplement) and plasma adiponectin levels (n = 3161) at their baseline visit (Fig. 1). Excluded participants (n = 2141) had comparable baseline characteristics (Supplemental Table 1).
Schematic diagram for our study cohort.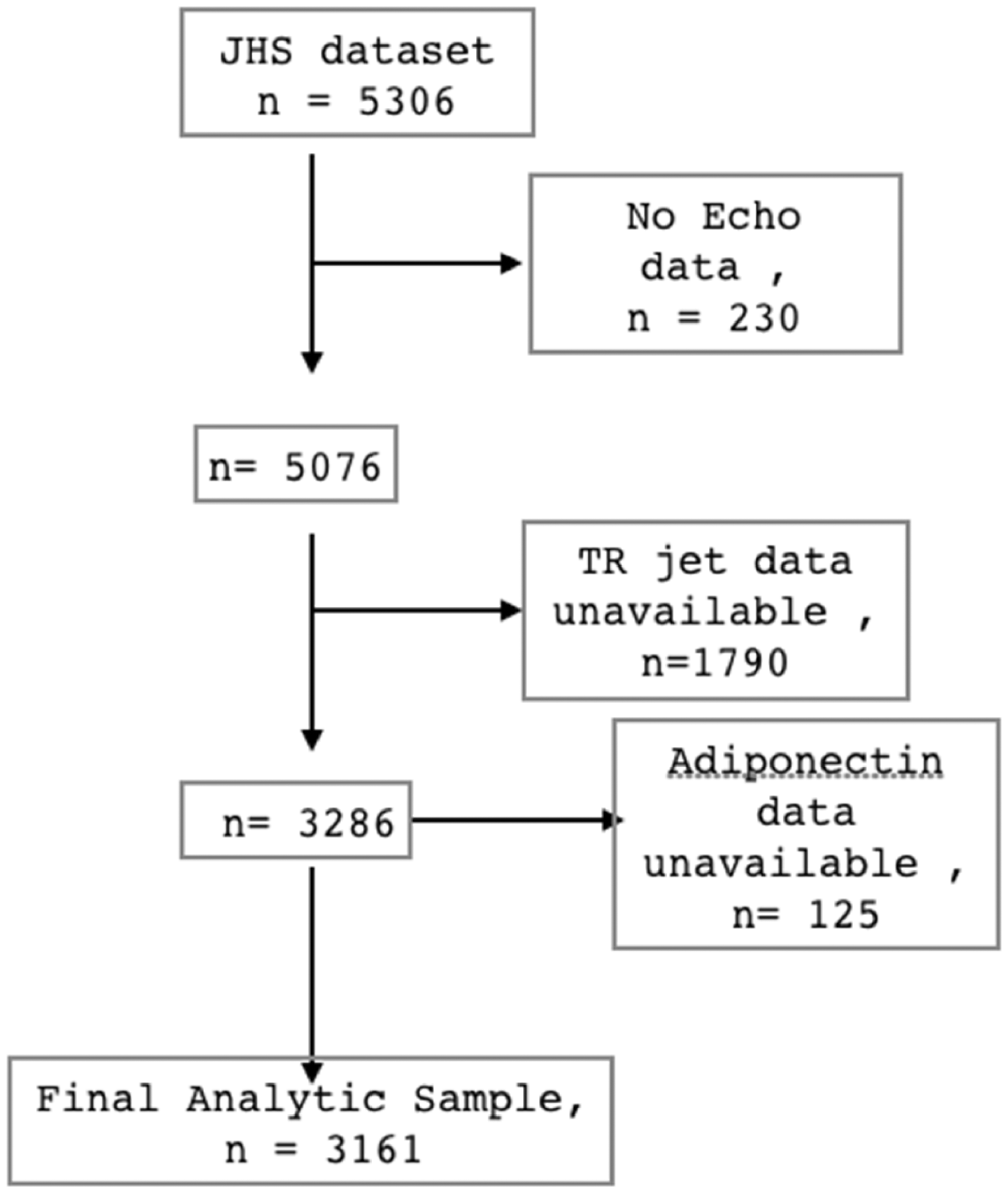
Exposure
The main exposure was plasma adiponectin level at their baseline visit. Plasma adiponectin was measured in nanograms per milliliter, by an ELISA system (R&D Systems; Minneapolis, MN, USA). 16
Outcome
The main outcome for the cross-sectional analysis was presence of PH, defined as PASP greater than 40 mm Hg on baseline echocardiography. We also analyzed PASP as a continuous variable.
For the longitudinal analysis, the outcomes were (1) decompensated HF requiring hospital admission, with event adjudication beginning on 1 January 2005, until censored by event, death or end of study date (December 31, 2012); and (2) all-cause mortality, with time to death calculated from the time of the index echocardiographic examination. Adjudication of heart failure was based on abstracted data on history, physical examination, diagnostic tests, biochemical analysis, and medication use as per procedures for event adjudication used in the Atherosclerosis Risk in Communities Study. 17 Participants with a self-reported HF hospitalization history prior to 1 January 2005 were excluded from the longitudinal analyses.
Clinical covariates
Definitions for clinical covariates, such as diabetes, hypertension, hyperlipidemia, coronary heart disease, stroke, heart failure, lung disease, smoking history, among others are outlined in the Supplement.
Echocardiographic parameters
Details regarding echocardiographic data and procedures, used in this study are outlined in the Supplement.
Statistical analysis
Adiponectin levels were log-transformed to approximate normality. Adiponectin levels were also classified as tertiles; low tertile (≤3260.04 ng/ml), middle tertile (>3260.04–5981.60 ng/ml) and upper tertile (>5981.6 ng/ml). Differences in baseline characteristics amongst the tertiles of adiponectin were compared using one-way ANOVA or Wilcoxon rank sum test for continuous variables and χ 2 analysis for categorical variables. The association between log adiponectin or tertiles of adiponectin and presence of PH was assessed using logistic regression. The model was adjusted for age, sex, body mass index as a continuous variable (BMI), brachial pulse pressure (mm Hg), hypertension, diabetes, coronary heart disease, severe mitral/aortic valvular heart disease, history of chronic lung disease, spirometry profile (normal, obstruction, and restriction), and a left ventricular ejection fraction less than 50%, adapted from Choudhary et al. 18
Cox proportional hazards modeling was used to determine the association of HF events with log-adiponectin in a univariate analysis, followed by an adjusted model for prediction of HF (Atherosclerosis Risk in Communities model) from Agarwal et al., 19 adjusting for age, sex, coronary heart disease, diabetes, systolic blood pressure, blood pressure medication use, heart rate, smoking status, and BMI. Subsequently, this model was also adjusted for presence of PH on baseline echocardiogram. Participants who died before a HF event were censored.
Cox proportional hazards modeling was used to assess the relationship of adiponectin levels to mortality in a univariate analysis. The relationship of all-cause mortality and log-transformed adiponectin level was then adjusted for potential confounders using a mortality model adapted from Gu et al., 20 controlling for age, sex, BMI, physical activity, smoking status, high cholesterol, diabetes, history of HF, history of coronary heart disease, hypertension, estimated glomerular filtration rate, and history of stroke. Subsequently, the model was also adjusted for presence of PH. Interaction testing was performed to assess possible effect modification of BMI on the association of log adiponectin and outcomes. Missing data for clinical covariates were imputed using multiple imputation method by STATA (Supplemental Table 2). All analysis was performed using Stata/SE, version 15.1 software (StataCorp LP). A two-sided P-value of less than .05 was considered significant.
Results
Baseline characteristics
Pulmonary hypertension sample characteristics by adiponectin tertiles.
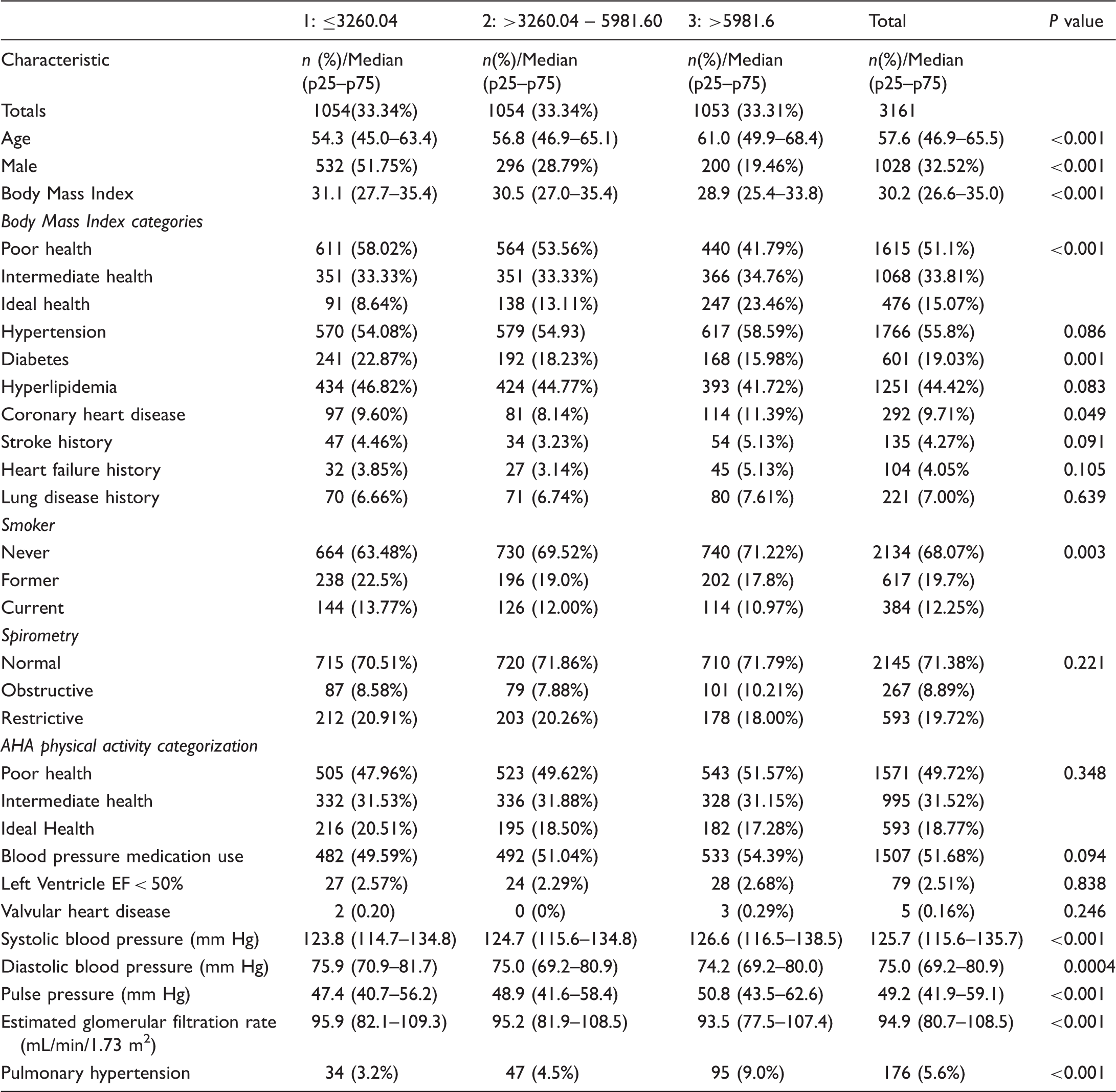
Table 1 also includes a comparison of the baseline characteristics of participants across tertiles of adiponectin. Participants in the highest adiponectin tertile group were more likely to be women and had a higher mean age. They were less likely to be active smokers, obese, or have diabetes. Systolic and brachial pulse pressures were highest in the highest adiponectin tertile group, but frequency of abnormal lung function or reduced LV ejection fraction was similar across adiponectin tertiles.
Association of plasma adiponectin with PH
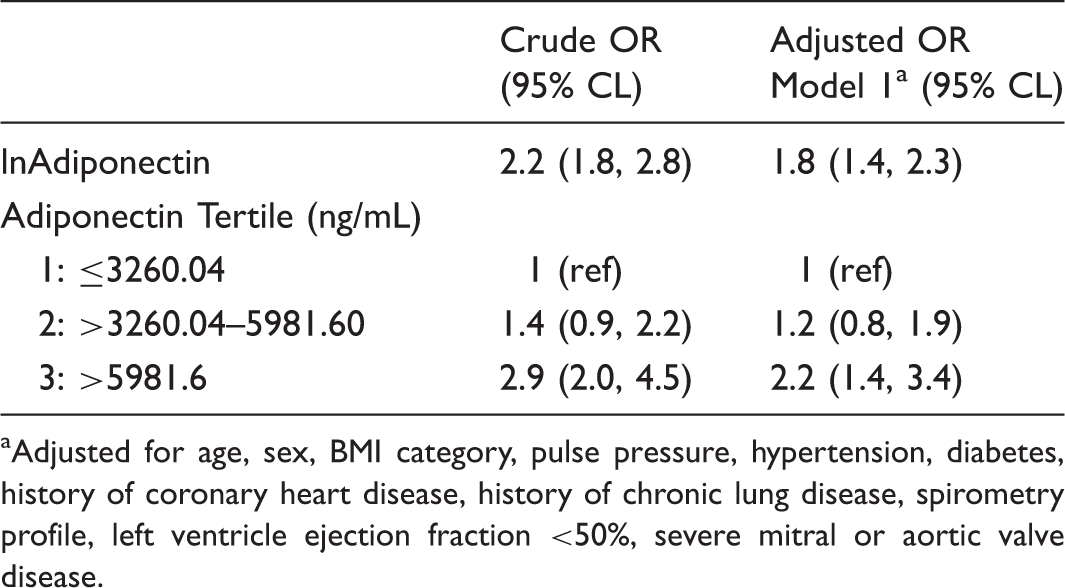
On univariate analysis, higher adiponectin levels were significantly associated with the presence of PH. Following adjustment for relevant clinical characteristics, the adjusted odds ratio (OR) for PH per log increment of adiponectin was 1.8 (95% CI, 1.4–2.3; P < .05) (Table 2). By tertiles of adiponectin, PH prevalence was 9.0% in the high group compared with 4.5% in the middle and 3.2% in the low adiponectin group (P < .001). In adjusted analysis, odds of having PH were significantly higher in the highest compared with the lowest tertile group (adjusted OR, 2.2; 95% CI, 1.4–3.4). The odds of having PH did not significantly differ between the middle and low groups. In sensitivity analyses, adjusting for moderate to severe aortic or mitral valvular heart dysfunction, as opposed to just severe aortic or mitral valvular dysfunction, did not alter the relationship between PH and adiponectin. Higher adiponectin levels were also significantly associated with higher PASP and results are outlined in Supplement Table 3. A scatterplot of (log-transformed) values of adiponectin versus (log-transformed) pulmonary artery systolic pressure are illustrated in Supplemental Fig. 1. Supplemental Fig. 2 illustrates levels of adiponectin in the PH and non-PH groups, while Supplemental Table 4 details the clinical characteristics of the PH and non-PH groups.
Hazard Ratios and 95% confidence interval for heart failure and mortality associated with log transformed adiponectin. The adjustment model for heart failure includes age, gender, history of coronary heart disease, diabetes, systolic blood pressure, heart rate, blood pressure medication use, smoking status and BMI. The adjustment model for mortality includes age, sex, BMI, AHA categories of physical activity, smoking status, history of heart failure, diabetes, hypertension, total cholesterol, stroke history, coronary heart disease, and estimated GFR. Odds ratios and 95% confidence limits for the association of plasma adiponectin and pulmonary hypertension. Adjusted for age, sex, BMI category, pulse pressure, hypertension, diabetes, history of coronary heart disease, history of chronic lung disease, spirometry profile, left ventricle ejection fraction <50%, severe mitral or aortic valve disease.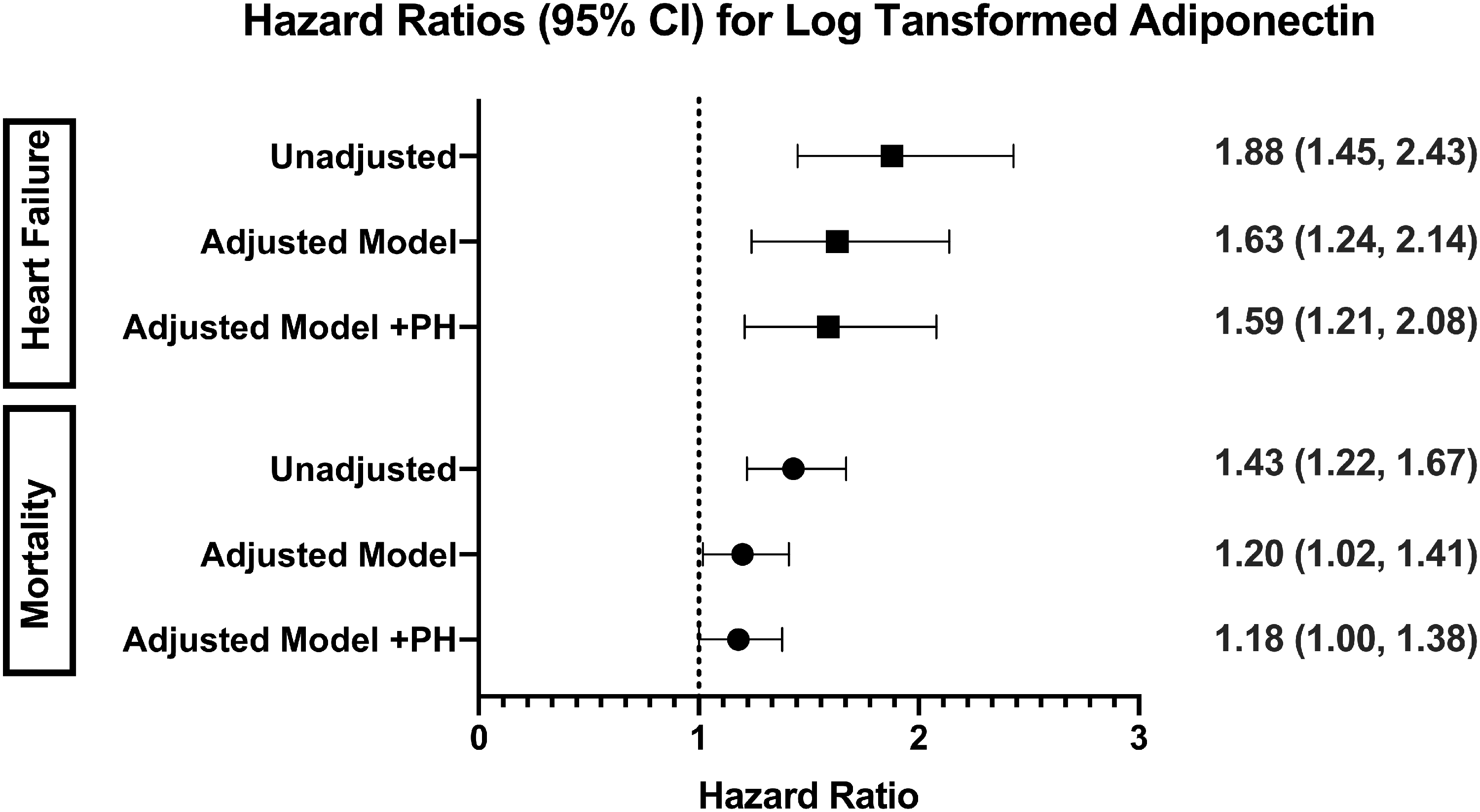
Elevated plasma adiponectin and incident HF events
The median (range) follow-up time for incident HF analysis was 8.0 (0–8.0) years. During the follow-up period, 130 patients were admitted with decompensated HF. Higher adiponectin levels were significantly associated with HF events (adjusted HR per log increment of adiponectin, 1.63; 95% CI, 1.24–2.14) (Fig. 2). This association remained significant when the model was further adjusted for PH (HR, 1.59; 95% CI, 1.21–2.08). No significant relationship was observed between tertiles of adiponectin and incident HF hospitalization.
Elevated plasma adiponectin and all-cause mortality
The median (range) follow-up time for mortality was 9.91 (0–12.3) years. During the follow-up period, 338 deaths occurred. After risk adjustment, higher adiponectin levels were significantly associated with mortality (adjusted HR 1.20 per log increment of adiponectin; 95% CI 1.02–1.41) (Fig. 2). However, this association was no longer significant (p = 0.05) after additional adjustment for presence of PH. No significant relationship was observed between tertiles of adiponectin and all-cause mortality.
Effect modification of BMI on association of plasma adiponectin and outcomes
There was no significant interaction between log Adiponectin levels and BMI with any of the main outcomes including PH, mortality and HF events. Results are detailed in Supplement Table 5. In addition, we did not observe any significant interaction between adiponectin and hs-CRP or adiponectin and insulin resistance, as measured by the Homeostatic Assessment of Insulin Resistance (HOMA-IR), with regards to PH.
Discussion
In this study, we demonstrated that in a community-based population of AAs with a high burden of cardiopulmonary risk factors and related causes of non-group 1 PH, such as left heart disease and chronic lung diseases, increasing levels of plasma adiponectin are associated with echocardiographic-measured PH, future HF admissions and mortality. To the best of our knowledge, this study examines for the first time the association between adiponectin and PH in a vulnerable cohort of AAs, with high prevalence of PH and associated morbidity.14,18 Prior studies that have reported similar associations between adiponectin and PH have been limited to patients with pulmonary arterial hypertension 21 (PAH) and thromboembolic disease, which are relatively uncommon conditions. 22
In animal studies, the development of PH is associated with adiponectin deficiency23,24 and increases in adiponectin levels are associated with reduced PASP.25,26 Also, low levels of adiponectin have been previously demonstrated in conditions that are commonly associated with PH such as diabetes, 27 systemic hypertension, 28 obesity and metabolic syndrome. 29 Yet, contrary relationships with high adiponectin levels and complex pathological states such as CVD, 30 chronic heart failure, 31 group 1 and 4 PH21,22 and associated mortality have been observed in large-scale epidemiological studies. 32 Mendelian randomization studies 10 found no evidence of a causal relationship between adiponectin and Type 2 diabetes or insulin sensitivity, 33 or adiponectin and coronary heart disease risk. 34 Furthermore, study of an elderly cohort found that higher adiponectin levels were associated with mortality, but found evidence that this relationship was apparently explained by alterations in body composition and physical functionality, suggesting adiponectin was more a biomarker of frailty and catabolism than a pathogenic factor in older age. 35 Therefore, the exact role of adiponectin, whether protective, pathogenic, or incidental, to vascular disease and mortality remains in doubt.
Our study adds to the observations from prior epidemiological studies and the “adiponectin paradox”. 10 While we observed somewhat older age, lower BMI and slightly lower reported physical activity levels in the high adiponectin group in our study, it is not clear that adiponectin served as a marker of frailty or catabolism per se in this mostly middle-aged, non-institutionalized, community-based cohort. Furthermore, the relationship of PH with adiponectin was independent of age or body mass index and did not seem to be explained by interaction with systemic inflammation. Further studies are needed to ascertain if the observed association between adiponectin and PH in our study can be explained by the phenomenon of ‘functional adiponectin resistance’ whereby increased levels of adiponectin occur in response to decreased expression/activity of adiponectin receptors 36 as observed in the setting of chronic heart failure. 37 Certain factors that are commonly implicated with PH development such as hypoxemia and elevated insulin levels/insulin resistance, can induce deactivation of adiponectin receptors by PPAR-α/AdipoR1 pathway, 38 subsequently leading to functional adiponectin resistance. 39 While we could not assess adiponectin resistance directly, we found no evidence of an interaction between insulin resistance, as measured by HOMA-IR, and the adiponectin PH association. Furthermore, adiponectin levels have previously been shown to be elevated in females compared to males, a difference attributed to androgen sex hormones 40 ; we observed a greater female predominance amongst those with high adiponectin levels in our study, with a greater prevalence of PH in this high adiponectin group—the female predominance in PH has been well described and also may relate to sex hormone effects.41,42 While we cannot ascertain mechanisms underlying the observed relationship between adiponectin, female sex, and PH in this study, we show that adiponectin may be an important biomarker to identify those at risk for PH.
We also observed that higher levels of adiponectin were associated with future HF hospitalizations. Previously, higher adiponectin levels have been associated with worsening HF severity43,44 and were more likely to be observed in acute episodes of decompensated HF. 45 Studies have also evaluated the validity of adiponectin as a prognostic biomarker for mortality in HF46,47; however, data regarding its role in predicting incident HF admissions has been inconsistent. 48 George et al. demonstrated that patients with a known history of CHF and high serum adiponectin were at an increased risk of future HF hospitalizations. 46 However, cohort studies of patients with no prior history of HF 49 including a longitudinal analysis from the Framingham Heart Study 50 did not show adiponectin to be associated with future HF admissions. These studies, however, did not include AA participants. In our study, we demonstrate that in an AA cohort with no prior history of HF, the risk of HF admissions increased by 63% with each log increase in adiponectin levels, after adjustment for a validated HF risk prediction model. These results are particularly significant for AAs who have the highest age-adjusted incidence rates of HF and associated case fatality rates compared to other ethnicities.51–53 Further understanding of the exact mechanisms of this association can help with identification of potential prognostication and therapeutic targets in HF in this at-risk AA cohort.
In our study, higher levels of adiponectin were also strongly associated with an increased risk of all-cause mortality, after adjustment for relevant comorbidities. Elevated adiponectin levels have been described as a marker of adverse prognosis including cardiac and all-cause mortality in multiple conditions including advanced cardiac disease,54,55 kidney dysfunction 56 and malignancy. 57 Given that AAs have highest baseline risk for coronary artery disease, PH and associated cardiac mortality 58 , our study findings highlight the importance of further understanding the role of adiponectin as a prognostic marker for mortality in high-risk populations.
Limitations
There are a number of limitations to our study. Our analysis was cross-sectional; therefore, we cannot comment on causal relationships between adiponectin and PH.
Given the observational nature of the study, residual confounding may be present, despite using robust regression models. Studies have reported disparities in baseline adiponectin levels based on ethnicities and disease phenotypes.59,60 Since our population was exclusively African-American, future research in more ethnically diverse cohorts can help understand if the associations of adiponectin with PH and clinical outcomes is modified by race/ethnicity. We used total adiponectin levels in our analysis rather than high molecular weight adiponectin which is considered the more biologically potent form. This could potentially affect our findings since studies have reported differential associations between the various biological isoforms of adiponectin and diseases.61,62 Our definition of PH was based on available echocardiography data on estimated pulmonary artery systolic pressure, the only feasible assessment in a large community-based population, rather than right heart catheterization data on mean pulmonary artery pressure, the optimal clinical definition; this is an approach we and others have used previously and have shown to relate to RV remodeling and important clinical outcomes.17,63 Furthermore, in this epidemiologic study, we cannot reliably categorize those study participants with evidence of pulmonary hypertension into WHO clinical groups. HF hospitalization adjudication began on 1 January 2005 14 so interval HF events after baseline visits from 2000 to 2004 may have been missed. However, despite this, we were able to find a significant association between adiponectin and HF events.
Conclusion
In summary, our study demonstrates a strong association between high circulating plasma adiponectin levels and PH by echocardiogram in a large cohort of AAs in the JHS. Plasma adiponectin levels were also associated with risk of heart failure admission. Elevated plasma adiponectin can identify certain patients at elevated risk for PH and potentially serve as a prognostic marker in this subgroup. Further studies are needed to investigate the potential role of adiponectin in PH pathogenesis and how circulating levels of adiponectin may be used to better sub-phenotype and impact specific therapeutic strategies the PH population.
Supplemental Material
sj-pdf-1-pul-10.1177_2045894020961242 - Supplemental material for Association of plasma adiponectin with pulmonary hypertension, mortality and heart failure in African Americans: Jackson Heart Study
Supplemental material, sj-pdf-1-pul-10.1177_2045894020961242 for Association of plasma adiponectin with pulmonary hypertension, mortality and heart failure in African Americans: Jackson Heart Study by Suvasini Lakshmanan, Matthew Jankowich, Wen-Chih Wu, Siddique Abbasi, Alan R Morrison and Gaurav Choudhary in Pulmonary Circulation
Supplemental Material
sj-pdf-2-pul-10.1177_2045894020961242 - Supplemental material for Association of plasma adiponectin with pulmonary hypertension, mortality and heart failure in African Americans: Jackson Heart Study
Supplemental material, sj-pdf-2-pul-10.1177_2045894020961242 for Association of plasma adiponectin with pulmonary hypertension, mortality and heart failure in African Americans: Jackson Heart Study by Suvasini Lakshmanan, Matthew Jankowich, Wen-Chih Wu, Siddique Abbasi, Alan R Morrison and Gaurav Choudhary in Pulmonary Circulation
Footnotes
Acknowledgements
The Jackson Heart Study (JHS) is supported and conducted in collaboration with Jackson State University (HHSN268201800013I), Tougaloo College (HHSN268201800014I), the Mississippi State Department of Health (HHSN268201800015I) and the University of Mississippi Medical Center (HHSN268201800010I, HHSN268201800011I and HHSN268201800012I) contracts from the National Heart, Lung, and Blood Institute (NHLBI) and the National Institute on Minority Health and Health Disparities (NIMHD). The authors also wish to thank the staffs and participants of the JHS.
Authors’ contributions
All authors assisted in analysis or interpretation of data; drafting the article or revising it critically for important intellectual content, and approved the version to be published.
Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that there is no conflict of interest. Dr. Abbasi is currently employed by Amgen Inc.
Disclaimer
The views expressed in this manuscript are those of the authors and do not necessarily represent the views of the Department of Veterans Affairs; the United States government; National Heart, Lung, and Blood Institute; the National Institutes of Health; or the U.S. Department of Health and Human Services.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: GC was supported by Department of Veterans Affairs MERIT award I01CX001892, NIH/NHLBI R01HL148727.
Guarantor
GC is the guarantor.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
