Abstract
Pulmonary capillary hemangiomatosis is a rare form of pulmonary artery hypertension; to date, only few descriptions of myocardial pathology in pulmonary capillary hemangiomatosis have been reported in the literature. We report the case of a Japanese female patient who was diagnosed with pulmonary capillary hemangiomatosis combined with acute myocardial inflammation on performing autopsy. She was admitted to our hospital because of acute pneumonia and subsequently suddenly developed severe hypoxemia with breathing difficulty and died 13 days after admission. At autopsy, the histology of the lung was consistent with pulmonary capillary hemangiomatosis. Additionally, a diffuse severe infiltration of inflammatory cells was associated with edema in the myocardium. Myocytolysis was limited and fibrosis was absent. To the best of our knowledge, pulmonary capillary hemangiomatosis with acute myocarditis-like histological findings has been described for the first time through our case.
Case description
The patient who visited our hospital owing to exertional breathlessness was a Japanese woman in her 40 s with no major history of illnesses. Her symptoms were classified under class IV according to World Health Organization Function Classification, and her blood test results were as follows: total protein, 5.6 g/dl; hemoglobin, 15.4 mg/dl; total bilirubin, 3.4 mg/dl; creatinine, 0.87 mg/dl, and brain natriuretic peptide, 64 pg/ml. The serum antinuclear antibody was below the minimum detectable sensitivity level. Her electrocardiogram showed evidence of right heart strain, and her chest X-ray scan revealed dilation of the bilateral pulmonary arteries without heart enlargement. With regard to the respiratory function test, though the forced vital capacity and forced expiration volume for 1 s/forced vital capacity were normal, the diffusion capacity of the lungs for carbon monoxide was 36.4%.
Echocardiography findings were consistent with pulmonary hypertension (tricuspid regurgitation with peak velocity of 4.2 m/s and distortion of the left ventricular cavity at end systole owing to flattening of the septum). Right heart catheterization (RHC) demonstrated a pulmonary capillary wedge pressure of 14 mmHg, mean pulmonary artery pressure of 52 mmHg, right atrium pressure of 4 mmHg, pulmonary vascular resistance of 10.1 Wood units, and cardiac index of 2.4 l/min/m2. Computed tomography findings revealed smooth, thick interlobular septa (Fig. 1a), suggesting pulmonary veno-occlusive disease or pulmonary capillary hemangiomatosis (PCH). Because her symptoms, namely the non-invasive imaging and RHC findings, did not improve after the initiation of standard and careful medical treatment with sildenafil, ambrisentan, and epoprostenol, we clinically diagnosed her with pulmonary veno-occlusive disease or PCH and considered lung transplantation to be essential for improving her condition and prognosis. While waiting for the lung transplantation date, she visited our hospital with fever and cough for three days. Physical examination showed bilateral moist crackles. Laboratory tests were as follows: white blood cell count, 3.81 × 103/µl; C reactive protein, 1.54 mg/l; brain natriuretic peptide, 537 pg/ml. Chest X-ray revealed no bilateral pulmonary vascular congestion and computed tomography showed infiltrative shadows in bilateral lower lobe of lungs. She was diagnosed with acute pneumonia and was admitted to our hospital. On the 13th day after hospital admission, she suddenly developed severe hypoxemia with breathing difficulty, which resulted in cardiopulmonary arrest.
Axial computed tomographic image showed (a) smoothly thickened interlobular septa (yellow arrow). Hematoxylin–eosin staining sections of lung showed (b) extensive proliferation of pulmonary capillaries within alveolar septa and capillary congestion and (c) expansion of the interlobular septum that contains lymphatic dilation. Hematoxylin–eosin staining sections of right ventricle showed (d, e) numerous inflammatory cell infiltration and interstitial edema without myocytolysis. Picrosirius-red staining section of right ventricle myocardium showed (f) little amount of fibrosis.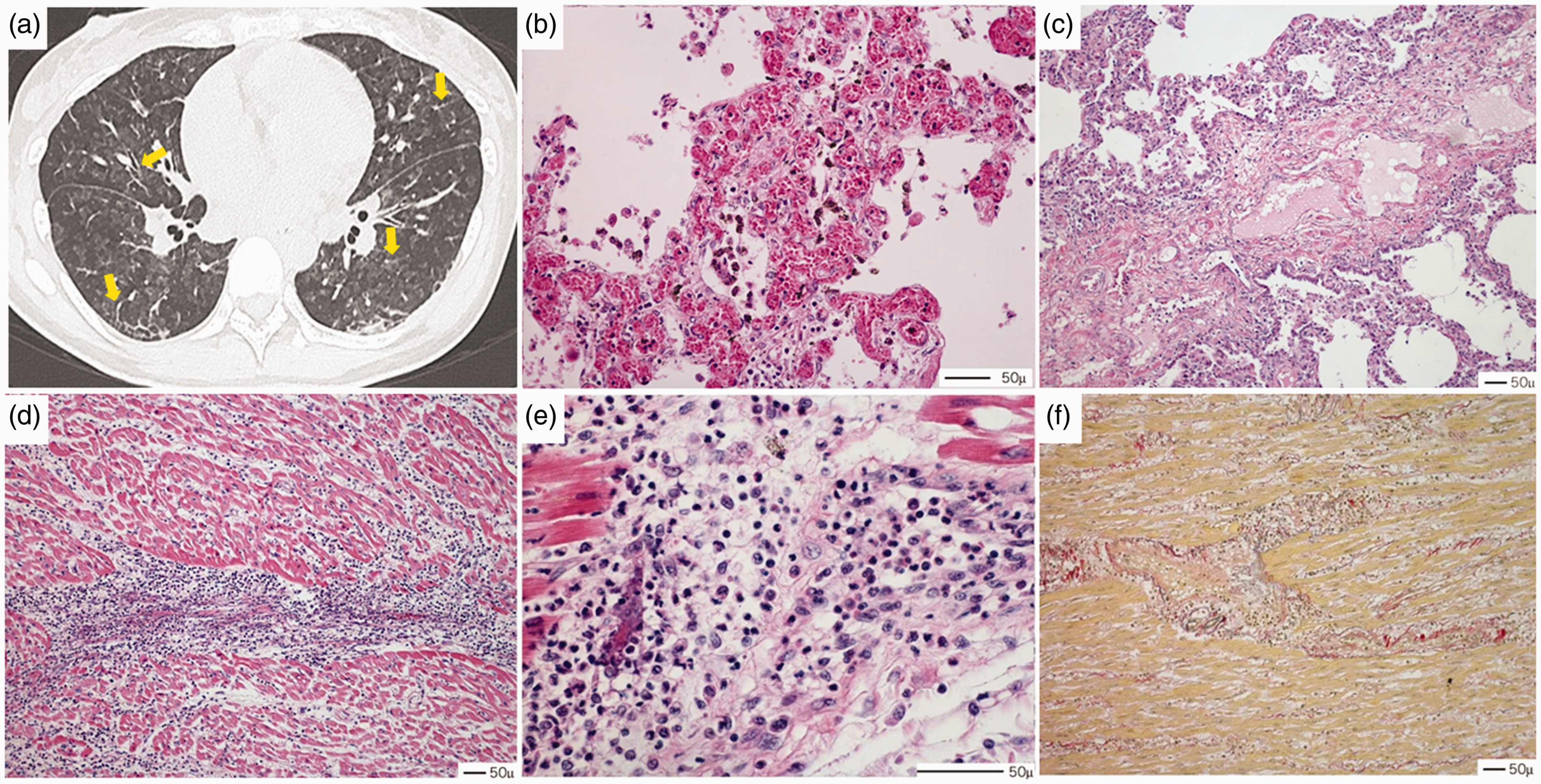
Autopsy findings revealed that there was no massive thrombus in the pulmonary artery and no severe edema or congestion in the lungs. Histologically, there were thickened alveolar septa expanded by capillary proliferation and capillary congestion (Fig. 1b), expansion of the interlobular septum (Fig. 1c), and small muscular pulmonary artery and interlobular vein occlusion owing to capillary proliferation. The heart weighed 625 g and showed marked dilatation, with hypertrophy of the right ventricle. No infarcts or fibroses were seen in the myocardium. None of the coronary arteries showed any abnormalities. Histological findings showed diffuse and severe infiltration of inflammatory cells associated with edema in both the left and right ventricles of the myocardium, particularly prominent in the vicinity of vessels (Fig. 1d and e). Myocytolysis was limited and fibrosis was not observed (Fig. 1f). Immunostaining findings revealed an extensive T-cell infiltration and only a moderate amount of eosinophilic infiltration.
Discussion
PCH was first described by Wagenvoort et al. 1 in 1978. PCH is a rare type of pulmonary artery hypertension, which is histologically characterized by pulmonary capillary proliferation within the alveolar septa 2 and is associated with a poor prognosis.3,4 It is difficult to diagnose PCH because the only reliable method for its diagnosis is confirmation of histological findings via pathological examination of the lung; 2 therefore, until recently, only a limited number of reports on PCH have been available. Furthermore, few descriptions about myocardial pathology in PCH have been reported to date. The patient in our case exhibited characteristic lung pathology consistent with PCH accompanied by the infiltration of inflammatory cells into the myocardium. To the best of our knowledge, this is the first description of PCH with acute myocarditis-like histological findings.
In the current case, histological myocardial changes were considered to have developed rapidly based on the lack of granulation tissues or fibrosis. Therefore, further deterioration of cardiac function because of acute myocardial pathological changes may have caused acute cardiopulmonary decompensation in this patient with severe pulmonary hypertension and right ventricular hypertrophy.
Severe inflammatory infiltration into the myocardium without fibrosis suggested onset of acute myocarditis; however, myocytolysis and tissue destruction were not prominent, which are atypical histological changes associated with myocarditis.5,6 The etiology of inflammatory cell infiltration into the myocardium was unclear. Some reports described that PCH was associated with myocarditis, which is likely caused by viral infection,7,8 and that catecholamines released owing to acute physical stress may secondarily result in catecholamine-induced myocarditis. 9 A possible explanation is that acute myocardial pathological changes occur owing to some viral infections, such as respiratory infection, or catecholamine release induced by acute physical stress, thereby suggesting that patients with PCH have a higher risk of developing adverse outcomes associated with the abovementioned factors.
Footnotes
Author contributions
Drafting the manuscript: Taku Omori, Shiro Nakamori Revising the manuscript critically for important intellectual content: Keiko Ohta-Ogo, Akimasa Matsuda, Yoshito Ogihara, Norikazu Yamada, Kyoko Imanaka-Yoshida, Masaaki Ito, Kaoru Dohi
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
