Abstract
This study validated a novel virtual echocardiography screening tool (VEST), which utilized routinely reported echocardiography parameters to predict hemodynamic profiles in pulmonary hypertension (PH) and identify PH due to pulmonary vascular disease (PHPVD). Direct echocardiography imaging review has been shown to predict hemodynamic profiles in PH; however, routine use often overemphasizes Doppler-estimated pulmonary artery systolic pressure (PASPDE), which lacks discriminatory power among hemodynamically varied PH subgroups. In patients with PH of varying subtypes at a tertiary referral center, reported echocardiographic findings needed for VEST, including left atrial size, E:e’ and systolic interventricular septal flattening, were obtained. Receiver operating characteristic analyses assessed the predictive performance of VEST vs. PASPDE in identifying PHPVD, which was later confirmed by right heart catheterization. VEST demonstrated far superior discriminatory power than PASPDE in identifying PHPVD. A positive score was 80.0% sensitive and 75.6% specific for PHPVD with an area under the curve of 0.81. PASPDE exhibited poorer discriminatory power with an area under the curve of 0.56. VEST’s strong discriminatory ability remained unchanged when validated in a second cohort from another tertiary center. We demonstrated that this novel VEST using three routine parameters that can be easily extracted from standard echocardiographic reports can successfully capture PH patients with a high likelihood of PHPVD. During the Covid-19 pandemic, when right heart catheterization and timely access to experts at accredited PH centers may have limited widespread availability, this may assist physicians to rapidly and remotely evaluate PH patients to ensure timely and appropriate care.
Keywords
Pulmonary hypertension (PH) is broadly defined as an abnormal elevation in mean pulmonary artery pressures (mPAP) which can result from several distinct disease processes. 1 By far, the most common type of PH is World Health Organization (WHO) group II or PH caused by left heart disease (PHLHD).2,3 In contrast to PHLHD, PH due to pulmonary vascular disease (PHPVD; defined as the presence of PH with pulmonary capillary wedge pressure (PCWP) ≤15 mmHg and pulmonary vascular resistance (PVR) > 3 Wood Units (WU), which encompasses WHO group I or pulmonary arterial hypertension (PAH), is associated with poor survival rates and must be promptly diagnosed and treated with PH-specific medications.1,4,5 These poor survival rates in PAH are known to be associated with right ventricular (RV) dilatation and dysfunction.6–8 The gold standard to diagnose PH is right heart catheterization (RHC) which allows for hemodynamic “phenotyping” of the different PH subgroups, a critical first step in evaluation of a patient with newly suspected PH. 3
The coronavirus disease-19 or Covid-19 is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and was first reported in Wuhan, China in December 2019. 9 By March 2020, Covid-19 had evolved into a global pandemic with drastic implications on health care systems. 10 Evidence soon emerged that patients with cardiovascular disease, including those with RV dilatation during hospitalization, were at heightened risk of Covid-19 with poorer outcomes.11–13 This ultimately led to a shift in clinical practice where only patients deemed to be in urgent need of hospitalization or clinical assessment were to be evaluated in person, leaving other patients to be managed virtually and remotely. Specifically, cardiac catheterization for patients not experiencing ST-elevation myocardial infarction have been discouraged, without specific guidelines regarding utility of RHC. 14 When taken together with the limited numbers of expert accredited PH referral centers, widespread evaluation of newly suspected PH by RHC, the historically described diagnostic gold standard is significantly less readily available during the pandemic. This has serious implications in the PAH population where delays in accurate diagnosis and subsequent initiation of PH-specific therapies pose a significant risk on morbidity and mortality. 15
Echocardiography has traditionally been used as the initial diagnostic test to recognize and predict the presence of PH, initially based on Doppler estimated pulmonary artery systolic pressure (PASPDE). Neither the presence of elevated PASPDE, nor its magnitude of abnormality, however, can reliably predict hemodynamic profiles in isolation, despite the common temptation for this application in clinical practice. The 2015 European guidelines have suggested additional echocardiographic parameters to aid in assessing the probability of PH, including RV size, eccentricity index, PA size, right atrial size, acceleration time (AccT) or profile notching of the right ventricular outflow tract (RVOT). Applicability of these guidelines rely on expert echocardiographic assessment of direct imaging, and do not support making a distinction between PHPVD and PHLHD. The use of echocardiography to predict hemodynamic profiles in PH has otherwise been previously described.3,6,16–25 While RHC remains the gold standard for diagnosis of PH, we demonstrated previously that a scoring system based on left atrial (LA) size, transmitral and tissue Doppler E:e’ ratio, and the presence of at least one of two abnormal measurements (AccT or profile notching) from the pulsed wave (PW) Doppler of the RVOT had an area under the curve (AUC) of 0.904 for prediction of PHPVD. 23 In the same study, PASPDE, which is often used to screen for the presence of PH, had a much smaller AUC equal to 0.689. However, the score’s reliance on access to direct review of imaging data, less commonly performed Doppler interrogation of the RVOT, and expertise in echocardiography pose significant limitations to its widespread use.
In the current study, we propose that a new virtual echocardiography screening tool (VEST), using only variables available on a routine echocardiogram report, can be used to distinguish between PHLHD and PHPVD. This approach allows the clinician without expertise on echocardiographic interpretation or access to the primary images to gain rapid, noninvasive insight into the hemodynamic origin of PH. This tool will be of incremental value in the Covid-19 era, when patient access to travel to accredited PH specialty centers and RHC are limited, and the need to use telemedicine as a platform for timely and appropriate medical care for PH is paramount.
Methods
Study design
The study is a retrospective review of patients with PH who underwent RHC by the PH program at two tertiary referral centers. In the original cohort, data were collected on consecutive patients with PH who underwent RHC at a single center between January 2012 and December 2013 (Fig. 1). In cases where a patient had multiple RHCs within one year, the first RHC in which all study inclusion criteria were met was included. Patients were excluded if they did not undergo a transthoracic echocardiogram (TTE) within one year of RHC, underwent changes in PH-specific therapies, received diuretics or hemodialysis between RHC and TTE, underwent heart transplantation, or had congenital heart disease (Fig. 1). In cases where a patient had multiple TTEs within one year of the index RHC, the TTE closest to the time of RHC in which all inclusion criteria were met was selected. A total of 154 patients met inclusion criteria. Of these, 96 had data for all variables included in the screening tool, described in detail below.
Enrollment criteria. One hundred seventy-nine subjects with pulmonary hypertension underwent right heart catheterization in pulmonary hypertension clinic between January 2012 and December 2013. One hundred fifty-four subjects were eligible for study inclusion. Of these, 96 had full echocardiographic parameters included in the virtual echocardiography screening tool. TTE: transthoracic echocardiogram; RHC: right heart catheterization.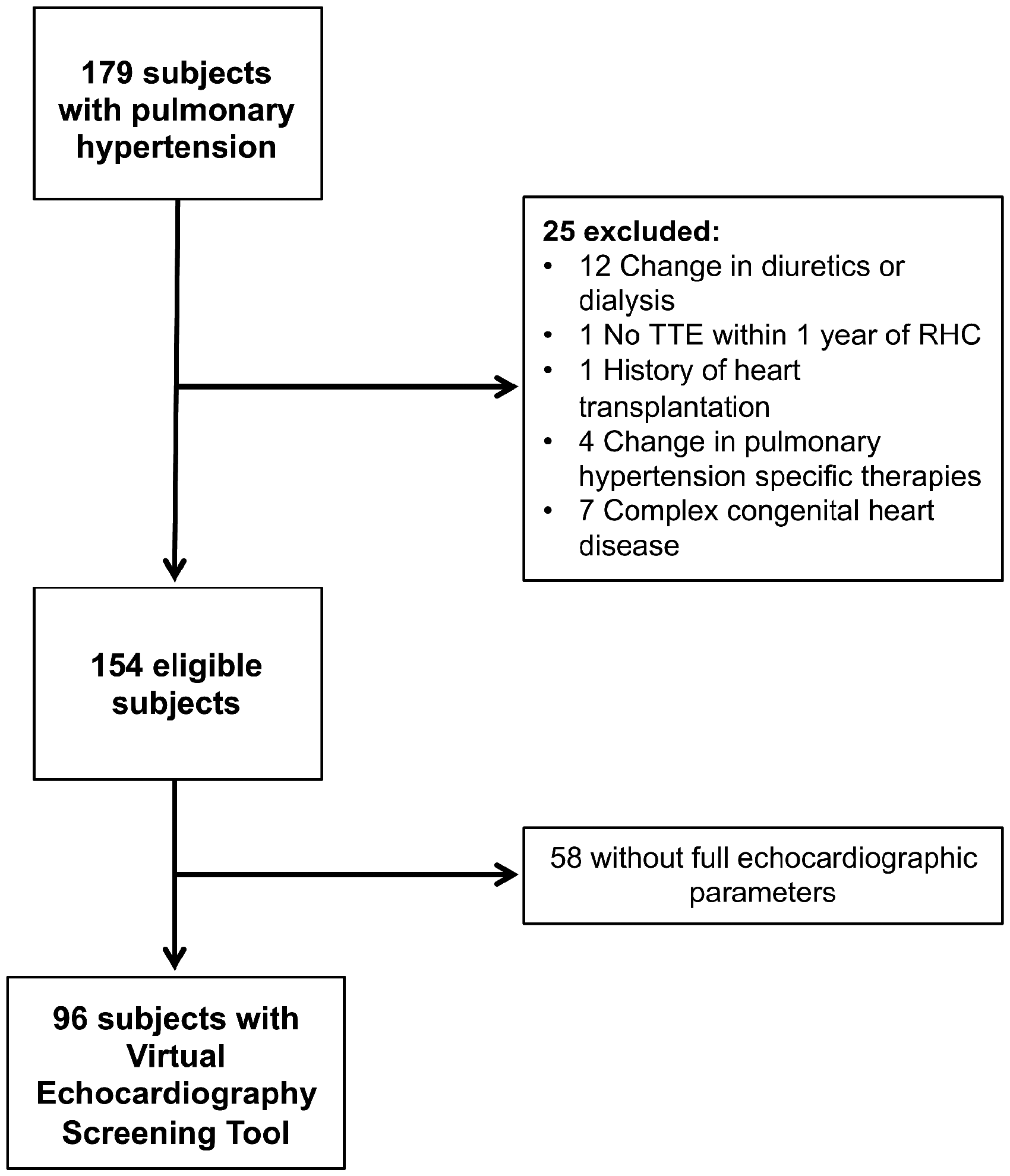
In the validation cohort, data were collected from 30 patients with incident PH diagnosed between June 2013 and June 2014. Similar to the original cohort, the validation cohort consisted of different PH physiologies: PHPVD (n = 10) and non-PHPVD (n = 20). The study was approved by the Institutional Review Boards (IRB) at the Hospital of the University of Pennsylvania and at Temple University School of Medicine. IRB approved the request to waive informed consent at both institutions based on the retrospective nature of the study.
Patient variables
Descriptive statistics and hemodynamics of the original cohort.
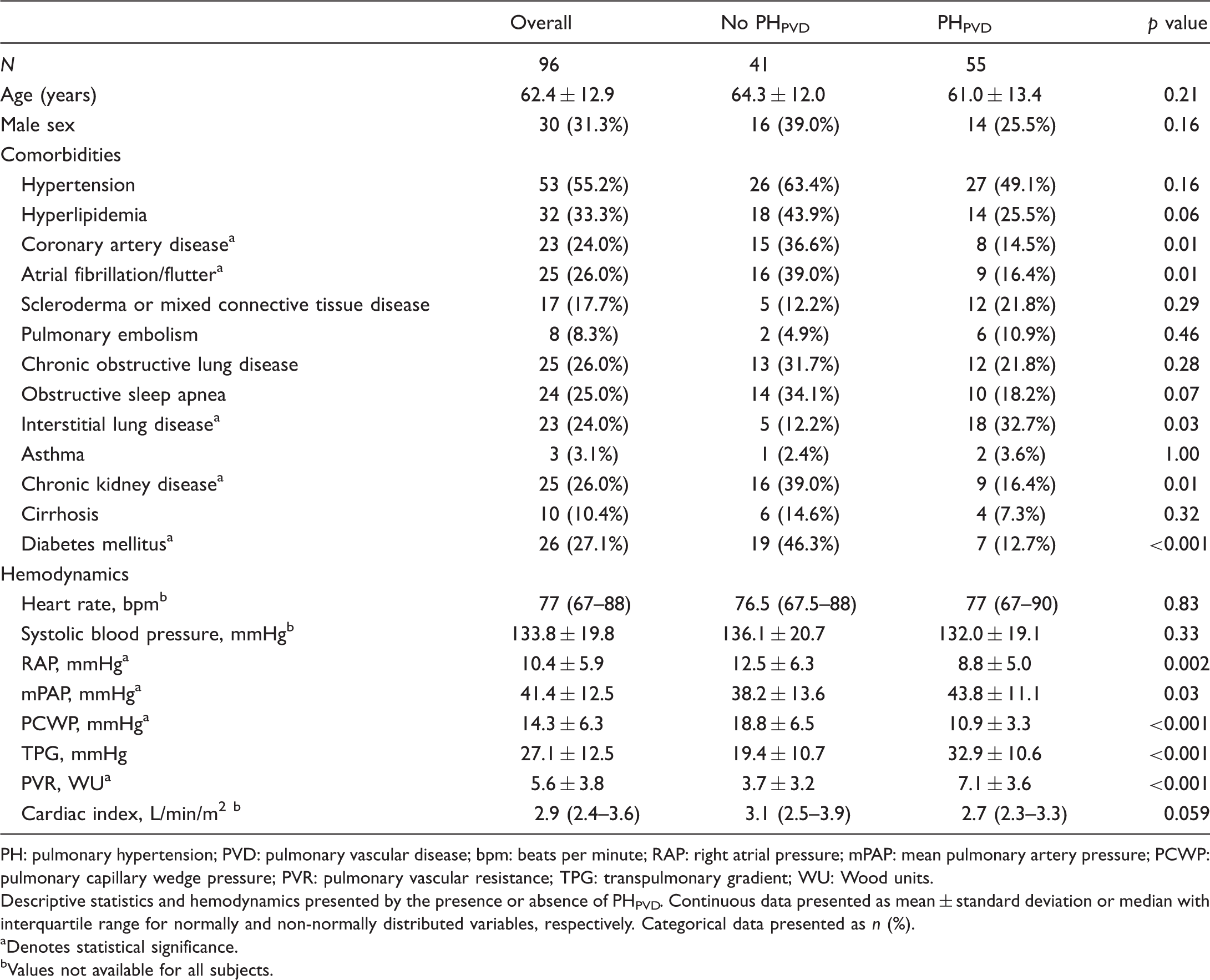
PH: pulmonary hypertension; PVD: pulmonary vascular disease; bpm: beats per minute; RAP: right atrial pressure; mPAP: mean pulmonary artery pressure; PCWP: pulmonary capillary wedge pressure; PVR: pulmonary vascular resistance; TPG: transpulmonary gradient; WU: Wood units.
Descriptive statistics and hemodynamics presented by the presence or absence of PHPVD. Continuous data presented as mean ± standard deviation or median with interquartile range for normally and non-normally distributed variables, respectively. Categorical data presented as n (%).
Denotes statistical significance.
Values not available for all subjects.
The virtual echocardiography screening tool
Virtual echocardiography screening tool (VEST).

Subjects received either +1 or −1 point for the presence or absence of each of three echocardiographic parameters. Scores range from −3 to +3.
Individuals with a qualitative description of elevated left-sided filling pressures without a quantitative measurement of mitral E:e’ lateral received −1 point (n = 12).
Simplified versions of VEST were also analyzed, including replacing E:e’ with E-velocity alone to include a greater sample size, and including only patients with RHC and TTE performed within a 30-day interval. Scoring was performed by two authors (JRG, FR) using echocardiogram reports generated by providers without knowledge of the patients’ clinical characteristics or hemodynamic measurements.
Analysis
The primary outcome was the ability of VEST to predict the presence or absence of PHPVD as defined by a mPAP ≥ 25 mmHg, PCWP ≤ 15 mmHg, and PVR > 3 WU. Categorical data are expressed as the number of patients (%) and continuous variables as mean ± standard deviation or median (interquartile range; IQR) as appropriate. Unpaired T-test and Wilcoxon rank-sum were used to compare means for normally and non-normally distributed continuous variables, respectively, whereas chi-squared or Fisher exact test were used to compare proportions for categorical variables. An ANOVA and Kruskal–Wallis test were used to compare the differences among group means for normally and non-normally distributed variables, respectively. Optimal cutoffs for continuous variables were estimated from receiver operating characteristic (ROC) curves. Analyses were performed using Stata v13.1 (StataCorp LP, College Station, TX).
Results
Patient characteristics
Demographic, clinical, and hemodynamic data for patients in the original cohort are listed in Table 1. Average age was 62.4 ± 12.9 years and 68.8% were female. The median interval between RHC and TTE was 27.5 days (IQR 8.5 to 67.5). Patients with PHPVD had, on average, a higher mPAP, transpulmonary gradient (TPG), and PVR but lower right atrial pressure (RAP), PCWP, and cardiac index. PH was classified as WHO group I in 42 (43.8%), II in 32 (33.3%), III in 12 (12.5%), IV in 6 (6.3%), and V in 4 (4.2%) of the cases.
Prediction of PHPVD by VEST versus by Doppler-estimated PASP
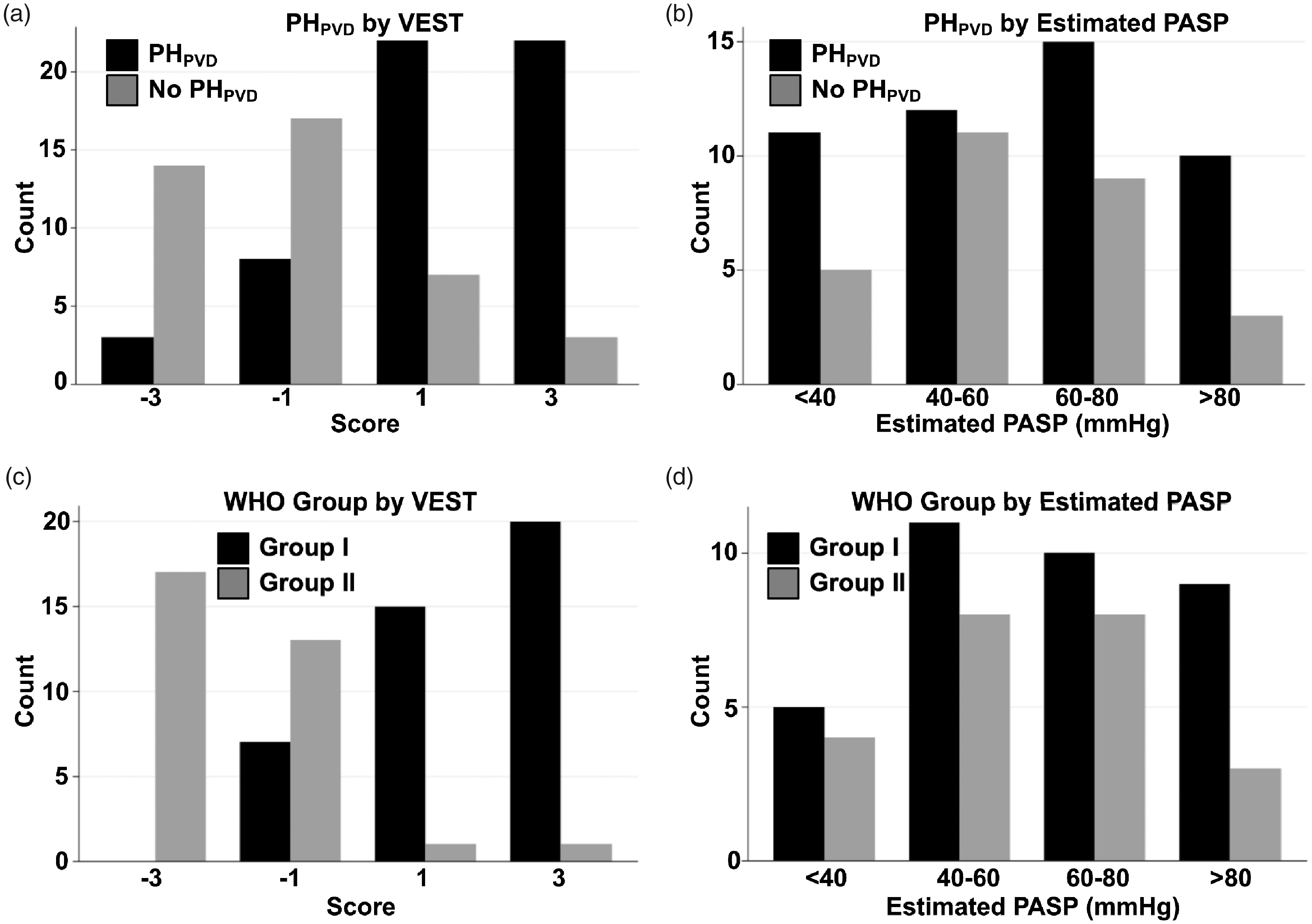
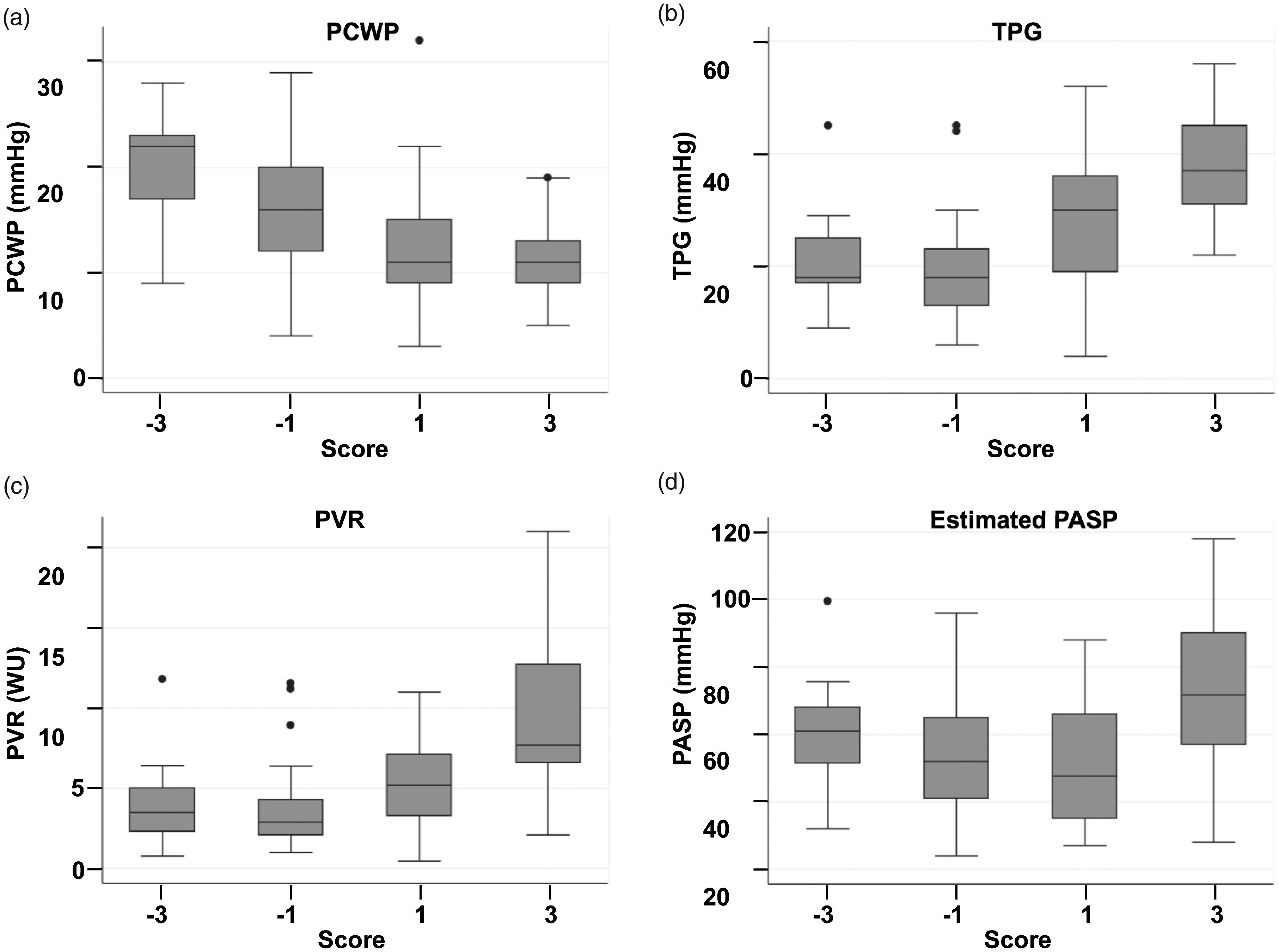
Estimated PASPDE as a continuous variable displayed poor discriminatory power (AUC = 0.56) in the prediction of PHPVD (Fig. 2). In fact, 16 of 96 individuals had a reported PASPDE < 40 mmHg of which 11 had PHPVD (Fig. 3). In these 16 subjects, the average PASP by RHC was 58 mmHg. A VEST score greater than 0, in contrast, was 80.0% sensitive and 75.6% specific for the presence of PHPVD (AUC = 0.81; Fig. 2). A VEST score of +3 was 92.7% specific for the presence of PHPVD with a positive predictive value (PPV) of 88.0%. PCWP decreased significantly with increasing VEST score (−3, −1, +1, +3; 20.4 ± 4.8, 15.9 ± 6.6, 12.6 ± 5.7, 11.2 ± 3.6 mmHg; p < 0.0001) while PVR (4.1 ± 2.6, 3.9 ± 2.8, 5.1 ± 2.6, 9.1 ± 4.3 WU; p < 0.0001) and TPG (20.3 ± 8.2, 19.8 ± 9.5, 28.2 ± 12.6, 37.9 ± 9.6 mmHg; p < 0.0001) increased significantly with increasing VEST score (Fig. 4). There was no clear trend in the PASPDE by VEST score.
Performance of the virtual echocardiography screening tool for prediction of PHPVD in the original and validation cohorts. Estimated PASP (a) was inferior to the virtual echocardiography screening tool (VEST) (b) for prediction of PHPVD in the original cohort. VEST was 80.0% sensitive and 75.6% specific for PHPVD with an AUC of 0.81. This significantly improved prediction of PHPVD when compared to PASPDE which had an AUC of only 0.56. Estimated PASP (c) was inferior to VEST (d) for prediction of PHPVD in the validation cohort. VEST was 100.0% sensitive and 75.0% specific for PHPVD with an AUC of 0.94. This significantly improved prediction of PHPVD when compared to PASPDE which had an AUC of only 0.61. PH: pulmonary hypertension; PVD: pulmonary vascular disease; PASP: pulmonary artery systolic pressure; AUC: area under the curve. Physiologic and clinical PH classifications by the virtual echocardiography screening tool (a, c) versus estimated PASP (b, d). PASP: pulmonary artery systolic pressure; PH: pulmonary hypertension; PVD: pulmonary vascular disease. Box plots of hemodynamics by the virtual echocardiography screening tool versus estimated PASP. Pulmonary capillary wedge pressure (PCWP) (a); transpulmonary gradient (TPG) (b); pulmonary vascular resistance (PVR) (c); estimated pulmonary artery systolic pressure (PASP) by echocardiography (d). Line through the box plot denotes the median.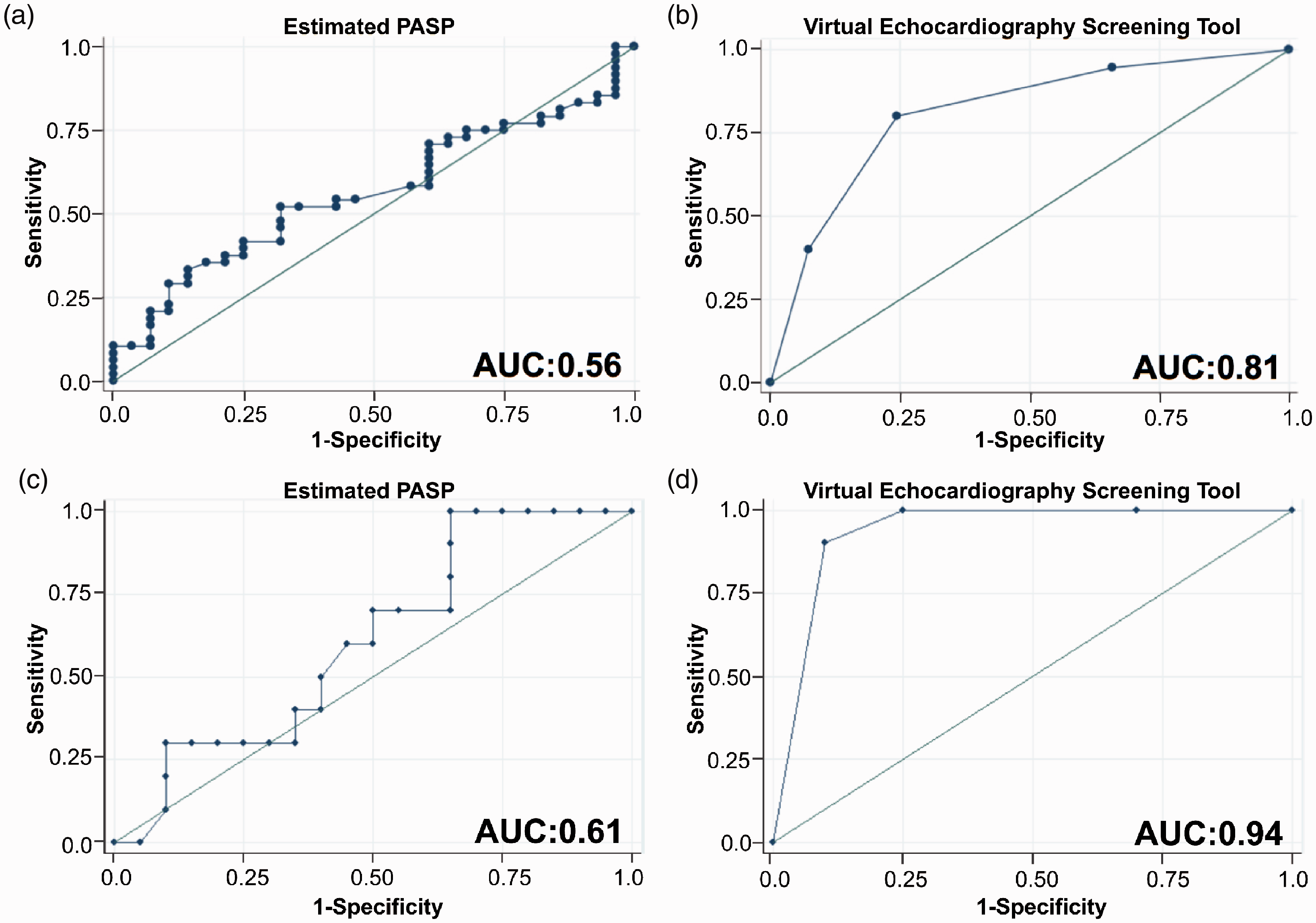
VEST and WHO group classification
A positive VEST was 83.7% sensitive and 66.0% specific for WHO Group I PH (AUC = 0.83). The distribution of patients with WHO Group I PH versus WHO Group II PH using both the VEST and PASPDE was compared (Fig. 3). PASPDE poorly differentiated between the two categories as demonstrated by an AUC equal to 0.61. In fact, five individuals with a PASPDE <40 mmHg had WHO group I PH. Four of these five individuals had a positive VEST.
Simplified versions of the VEST
E-velocity. Noninvasive assessment of left-sided filling pressures is challenging in the setting of arrhythmias and requires tissue Doppler imaging to calculate the gold standard E:e’.
26
In order to increase the applicability of our prediction tool, we used a simplified VEST in which E:e’ was replaced with E-velocity alone, allowing for inclusion of 119/154 (77.3%) eligible subjects that did not have e’ reported. In this simplified VEST, those with an E-velocity ≤90 cm/s received +1 point. The simplified score had similar test characteristics to the original VEST (AUC = 0.81; Fig. 5). A positive simplified VEST was 81.4% sensitive and 69.4% specific for PHPVD. As with the original VEST, this simplification also significantly improved discrimination of PHPVD when compared to the PASPDE (AUC = 0.56).
Performance of simplified virtual echocardiography screening tools. PASPDE (a) was inferior to multiple simplifications of the virtual echocardiography screening tool. This included scores in which E-velocity was used instead of the mitral E:e’ lateral (n = 119) (b) and in which only individuals with a TTE and RHC within 30 days were included (n = 52) (c). PASP: pulmonary artery systolic pressure; RHC: right heart catheterization; TTE: transthoracic echocardiogram.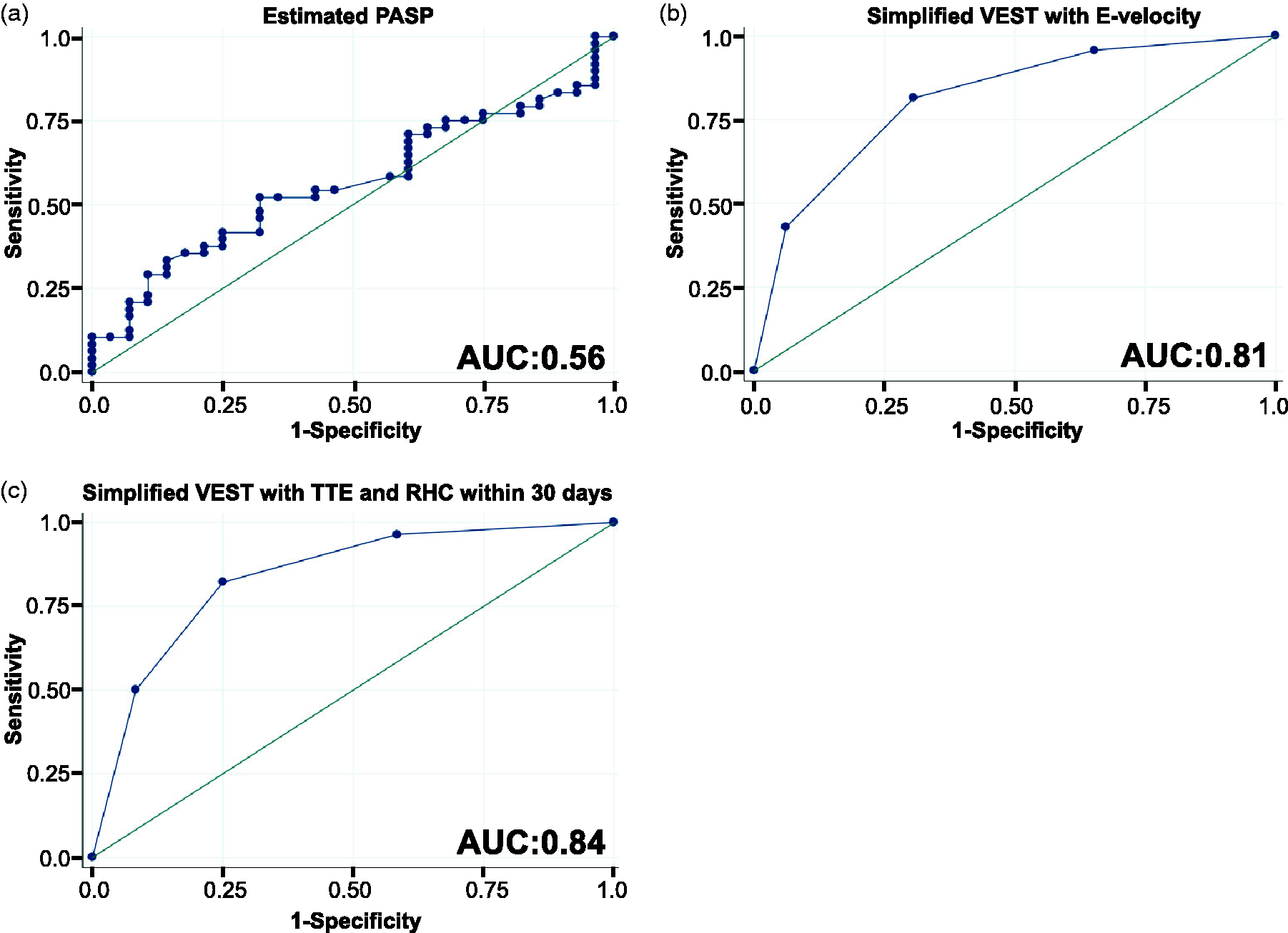
Delay between TTE and RHC. The VEST has similar test characteristics when applied only to the subset of subjects whose TTE and RHC were performed within 30 days of one another (n = 52; AUC = 0.84; Fig. 5). A positive score was 82.1% sensitive and 75.0% specific for PHPVD. These findings were very similar when applied only to the subjects whose TTE and RHC were separated by >30 days (n = 44, AUC = 0.80).
“Misclassified” subjects
There were 10 subjects in the original cohort with a positive VEST who did not meet hemodynamic criteria for PHPVD. The majority (n = 8) had a score of +1. These subjects had mixed physiology PH, defined as a PVR > 3 WU and a PCWP > 15 mmHg (mean PVR = 4.3 ± 4.5 WU and PCWP = 15.2 ± 8.4 mmHg). Three had a markedly elevated PVR of ≥7 WU, but did not meet criteria for PHPVD due to a mild elevation in their mean PCWP (17.6 mmHg). There were 11 subjects in the original cohort with a negative VEST who met hemodynamic criteria for PHPVD. The majority (n = 8) had a VEST score of −1, with PVR ranging from 3.4 to 4.7 WU. The average PVR in the 11 subjects was only 5.1 ± 2.2 WU with a PCWP of 11.5 ± 3.6 mmHg. Notably, those with VEST scores of −3 or +3 had more distinctive hemodynamic separation between pre-capillary and post-capillary mechanisms. A VEST score of +3, for example, was 92.7% specific for the presence of PHPVD with a PPV of 88.0%. In contrast, a VEST score of −3 had a negative predictive value of 82.4%.
Validation of the VEST
Demographic, clinical, and hemodynamic data for patients in the validation cohort are listed in Supplemental Table 1. The median interval between RHC and TTE was 12.0 days (IQR 6–12). Similar to the original cohort, patients with PHPVD had a higher mPAP, TPG, and PVR but lower RAP, PCWP, and cardiac index. A VEST score greater than 0 was 100.0% sensitive and 75.0% specific for the presence of PHPVD (AUC = 0.94; Fig. 2). A score of +3 was 90.0% specific for the presence of PHPVD. In contrast, PASPDE displayed poor discriminatory power (AUC = 0.61). Five patients without PHPVD had positive VEST. All of these patients had mixed physiology PH with an average PVR of 8.4 ± 3.9 WU.
Discussion
Herein we present a VEST that differentiates the hemodynamic basis of PH in a diverse referral cohort with much greater reliability than PASPDE. The distinction of pre- and post-capillary PH is critical in both diagnostic and therapeutic approaches. Unlike previous prediction rules for PH, our VEST was derived from information within the standard clinical echocardiography report and does not require direct analysis of echo-Doppler images. Thus, the VEST can also be applied by providers without access to or formal training in echocardiography, lending to its practical and timely widespread utility, particularly in the pandemic era of Covid-19.
This is the first study to describe a novel screening tool that captures patients with a high likelihood of PHPVD using findings on a standard echocardiogram report. Similarly, this approach identified patients with PH purely and largely related to passive left heart congestion in the relative absence of pulmonary vascular disease. By differentiating these two disparate patient phenotypes, additional PH workup and invasive hemodynamic assessment may be applied more precisely to the patients with PHPVD, while those with PHLHD may be considered to prioritize targeted treatment towards alleviating left heart disease as an initial strategy before additional invasive testing. We propose that implementation of the VEST can be conducted by physicians remotely and will allow for continued evaluation, minimizing delay, of patients with newly suspected PH during the Covid-19 era when extensive and invasive diagnostic testing such as RHC is less readily available.
An abnormal elevation in echo-derived PASP is often the primary clue used to determine the presence of PH on a standard echocardiogram report. However, it performed only slightly better than a coin flip in predicting which patients had PHLHD versus PHPVD (AUC = 0.56 in the original cohort and AUC = 0.61 in the validation cohort). In contrast, the VEST described in the present study demonstrated far superior discriminatory power with an AUC equal to 0.81 in the original cohort (Fig. 2). Approximately 57% of the original cohort had PHPVD while the remainder had non-PHPVD due to various etiologies, demonstrating the rigor of the screening tool when applied in a diverse population of PH patients (Table 1). In fact, we observed an increased AUC equal to 0.94 when the screening tool was performed in a second cohort of PH patients from a different institution.
We observed a considerable increase in score sensitivity for PHPVD when a patient had a score of +3 compared to +1 (92.7% compared to 75.6%, respectively). In other words, patients who receive scores of +3 are highly likely to have hemodynamic profiles consistent with PHPVD and should urgently be referred to a PH specialty center. In contrast, VEST also accurately identified patients with PHLHD with a score of −3. Opotowsky et al. observed a similar increase in hemodynamic discrimination at the extremes of a prior echocardiographic score that depended on direct analysis of RVOT Doppler profiles and imaging review. 23 Taken together, patients with a VEST score of −3 or +3 had more distinctive hemodynamic separation between pre-capillary and post-capillary mechanisms, allowing for earlier identification of the more severe PH phenotype as well as providing the necessary insight to triage consideration for RHC based on clinical response to initial treatment targeting left heart disease.
VEST outperformed PASPDE in prediction of patients with PHPVD confirmed by RHC. However, VEST tended to misclassify patients who had mixed physiology PH and therefore, scores of −1 or +1 should be interpreted with caution. By design of the VEST, patients with features of both subgroups will have absolute scores with smaller magnitudes due to a “cancelling out” effect. These patients, depending on the degree of cardiopulmonary clinical stability, may be triaged accordingly to RHC for definitive hemodynamic assessment. Upon closer examination of eight patients in the original cohort assigned a score of −1 but were found to have PHPVD by RHC, the hemodynamic profiles were suggestive of milder disease with a mean RAP and cardiac index of 5 mmHg and 3.4 L/min/m2, respectively. With a relatively compensated right heart, which may be supported by RV function description on the echocardiogram report, a later triage of referral of these patients may be appropriate, with close ongoing clinical follow-up.
As part of a secondary analysis, we replaced E:e’ at the lateral mitral annulus with E-velocity which increased the number of eligible patients in the original cohort from 96 (62.3%) to 119 (77.3%). Although E-velocity may also be influenced by factors other than LV diastolic function and left heart filling pressures, the simplified VEST shared similar test characteristics to that of the original VEST (AUC = 0.81). 26 We also hypothesized that in a subset of patients where the time elapsed between echocardiogram and RHC was less than 30 days, the predictive performance of the VEST would be enhanced. However, the AUC for this subset of patients was equal to 0.84, similar to that of the original cohort, indicating that temporal proximity between echocardiogram and RHC is not necessary to maintain the utility of the VEST, excluding those with significant interim medical interventions as described previously. This observation is consistent with prior work showing that the hemodynamic basis of PH in an individual patient does not change significantly within the typical time frame of clinical assessment.20,23 The durability of our approach lends to its practical applicability, given time delays are typical between noninvasive and invasive assessment before or after the Covid-19 era.
It was striking to note that in the original cohort 11 of the 16 subjects with a reported PASPDE <40 mmHg had PHPVD, with an average PASP by catheterization of 61 mmHg. Using the VEST, 9 of 11 false negative patients (by PASPDE) would have been identified correctly. In keeping with the hemodynamic data, ROC analysis demonstrated that the VEST was far superior to PASPDE in identifying WHO Group I PAH. Moreover, four of five PAH subjects with reported ‘normal’ PASPDE values had a score consistent with PHPVD. In the context of a progressive disease such as PAH, the lack of sensitivity observed with PASPDE is particularly worrisome. Indeed, delayed diagnosis of PAH risks postponement of PH-specific therapies and thus may negatively impact survival. 27 The implications of false negative PAH diagnoses may be further amplified in the Covid-19 era. Taken together, these data indicate that the VEST was far more sensitive for detecting serious, potentially life-threatening forms of PH than PASPDE alone. This is the first study to our knowledge that demonstrates that selected information taken from the echocardiography report was superior to directly measured PASPDE in differentiating the pathophysiology of PH.
We offer our findings in the spirit of contribution to the already complex clinical realm of PH, only further complicated in the Covid-19 era. Our data are in line with a growing body of literature that integration of selected, often easily obtainable echocardiographic variables can provide powerful insight into the hemodynamic basis of PH.6,20,22,23 Our data extend these observations further, given that the VEST utilizes routinely reported echocardiographic variables, thus obviating the need for direct image analysis or echocardiographic expertise to apply. As such, VEST enhances the prompt diagnostic abilities of physicians engaged in the growing paradigm of telemedicine and virtual patient assessment in the Covid-19 era.
We recognize that our study has some limitations that must be considered. The patients who contributed to the study were treated at two tertiary referral centers which potentially limit the generalizability of our findings. However, we demonstrated that within PH, there was a diversity of subgroups represented. Further, the VEST uses simple echocardiographic measurements that are consistent with the American Society of Echocardiography guidelines and should therefore be applicable across all institutions, regardless of PH expertise or academic environment.26,28,29 One advantage of using a small number of centers is the increased likelihood of uniform data abstraction which may increase the reliability of our findings.
We acknowledge that we did not include significant left heart valvular disease as an additional parameter for inclusion in this VEST, despite the prevalence and significance of these patients. Our intention was to provide a simple tool with high performance characteristics to aid in the diagnosis of dyspnea as it relates to PH, using as few variables as possible to maximize ease of utility for the user. Patients with moderate–severe left heart valvular disease pose less of a diagnostic conundrum regarding cause of dyspnea and appropriate next steps for evaluation and management, and are therefore less dependent on additional application of this VEST for diagnostic clarity. By including LA size and E/e’, we feel we maintained the simplicity of the VEST while also simultaneously capturing patients with PH on the basis of significant left sided valvular disease. We also acknowledge our study’s relatively small sample size. However, the original cohort captured all patients who met eligibility criteria and underwent RHC from a tertiary referral center in a consecutive two-year period. Even when we increased our sample size by 24% in our secondary analysis by replacing E:e’ with E-velocity, the simplified VEST’s predictive performance remained relatively unchanged. We note that the ROC performance metrics were increased in the validation cohort compared to the original cohort. We speculate that these differences were likely driven by a smaller sample size in the validation cohort, and a predominant inclusion of new, incident PH, which would accentuate baseline differences between the two patient populations. Importantly, this difference may strengthen the applicability of VEST in evaluating new PH in the Covid-19 pandemic.
Finally, we acknowledge that in our study, we used a PH definition to include mPAP ≥ 25 mmHg, rather than 20 mmHg, which has been proposed, although not broadly adapted or implemented in clinical trials or guidelines, since the 2018 World Symposium. 1 This historically accepted definition is supported by previously published referral cohorts and continues to better represent current-day referral patterns for PH, which do not largely represent those with mPAP of 20–24 mmHg.19,20,22,23,25,30–33 This should not limit applicability of the VEST to rapidly identify and hemodynamically characterize patients with PH as determined by echocardiography during the Covid-19 era.
In conclusion, we present a virtual screening tool (VEST) based on findings in a standard echocardiogram report that can be used to predict hemodynamic profiles in patients with PH. We propose a novel use of this tool in the Covid-19 era, when access to travel for in-person assessment by PH specialty centers and additional diagnostic testing including RHC are limited. Because of its reliance on an already performed echocardiogram and easy-to-perform screening tool, referring physicians and PH physicians can readily identify patients at the highest risk for PHPVD that warrant early invasive assessment and consideration for PH medical therapy. While we cannot endorse that this score should replace the gold standard of RHC in patients with PH, our data support that this tool may assist in rapidly and appropriately triaging patients. Patients with a +3 VEST score and clinical characteristics to support WHO Group I PAH should be triaged to a rapid and complete PH assessment, including RHC. It is important that these patients be clinically evaluated for WHO Group III PH associated with lung disease and WHO Group IV PH associated with chronic thromboembolic disease. In contrast, patients with a −3 score may forego extensive PH workup and instead have their care expeditiously directed at alleviating left heart failure. Further study should be done to assess long-term outcomes of applicability of the VEST during the Covid-19 era in patients with PH.
Supplemental Material
sj-pdf-1-pul-10.1177_2045894020950225 - Supplemental material for Virtual echocardiography screening tool to differentiate hemodynamic profiles in pulmonary hypertension
Supplemental material, sj-pdf-1-pul-10.1177_2045894020950225 for Virtual echocardiography screening tool to differentiate hemodynamic profiles in pulmonary hypertension by Anjali Vaidya, Jessica R. Golbus, Natasha A. Vedage, Jeremy Mazurek, Farhan Raza and Paul R. Forfia in Pulmonary Circulation
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Authors’ contributions
AV was responsible for entirety of project. AV & PF responsible for conception of the project. AV, JG, JM, and FR participated in data acquisition. AV, JG, FR, JM, PF contributed to data analysis, data interpretation. AV, JG, PF, NV contributed to writing the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
