Abstract
The combination of bosentan and sildenafil is commonly used to treat patients with pulmonary arterial hypertension (PAH); however, there is evidence of a significant drug interaction between these two medications. We sought to evaluate the safety and efficacy of transitioning patients with PAH from the combination of bosentan and sildenafil to alternative therapy. A retrospective database review was performed on 16 patients with PAH who were treated with the combination of bosentan and sildenafil and transitioned to alternative treatment at our center. Invasive and non-invasive patient parameters were collected at baseline and after transition. 56.3% of patients were in World Health Organization functional class (WHO FC) III and a majority of patients (68.7%) were on background prostacyclin therapy. The most common reason for transition was concern for a drug interaction in seven patients (43.8%). The most common transition was bosentan to macitentan in eight patients (50%). Fifteen patients (93.8%) tolerated the transition after a median follow-up of 6.5 months with minor adverse events occurring in four patients (25%). In 11 patients, 6-min walk distance (6MWD) was unchanged comparing baseline to post transition measurements with a median change of +8 m (range: −50 to + 70; P = 0.39). Nine patients (81.8%) had stable (within 15% margin) or significant improvement (increase by ≥15%) in 6MWD after transition. All patients demonstrated stable or improved WHO FC after transition. There were no significant changes after transition in hemodynamics, N-terminal pro-brain natriuretic peptide (NT-proBNP) values, or Registry to Evaluate Early and Long-Term PAH Disease Management (REVEAL) risk scores. In our study, transitioning patients from bosentan and sildenafil to alternative therapy was safe and resulted in clinical stability.
Pulmonary arterial hypertension (PAH) is a rare and progressive disease characterized by increased pulmonary arterial pressures leading to right heart failure. 1 Recent advancements in the diagnosis and treatment of PAH have improved the survival of patients with this condition. 2 Available therapies to treat PAH target three different molecular pathways: the prostacyclin, endothelin, and nitric oxide pathways.3,4 Currently available endothelin receptor antagonists (ERA) include bosentan, macitentan, and ambrisentan. These agents work by blocking the pulmonary vasoconstrictive actions of endothelin-1. 5 Phosphodiesterase type 5 inhibitors (PDE5i), such as sildenafil and tadalafil, increase availability of nitric oxide by inhibiting PDE5i mediated cyclic guanosine monophosphate degradation. 6
Combination therapy with a PDE5i and ERA is recommended by clinical practice guidelines for the treatment of PAH.3,4 Bosentan and sildenafil have been commonly used as combination therapy; however, several pharmacokinetic studies have shown a significant drug–drug interaction between these two medications, causing reduced sildenafil drug concentrations.7–10 Recent clinical trials failed to show an improvement in clinical outcomes in patients who received combination therapy with bosentan and sildenafil.11,12 For patients already receiving the combination of bosentan and sildenafil, transitioning to alternative medications may be warranted to avoid this drug interaction and therefore improve efficacy of combination therapy. However, there is limited clinical experience with this transition, and the impact of transition on safety, tolerability, and efficacy is not clearly established. Therefore, we conducted a retrospective study to assess the safety and efficacy of transitioning patients with PAH from the combination of bosentan and sildenafil to alternative therapy.
Methods
A retrospective database review was performed on adult patients with PAH who were taking the combination of bosentan and sildenafil and transitioned to alternative therapy in the Allegheny General Hospital Pulmonary Hypertension Clinic between January 2010 and February 2019. Patients taking both bosentan and sildenafil were identified by review of our PAH database and electronic medical records. Patients were included if they had World Health Organization (WHO) Group 1 PAH confirmed with right heart catheterization and were transitioned from the combination of bosentan and sildenafil to a different ERA, PDE5i, or riociguat. Patients on background therapy with a prostacyclin analogue or prostacyclin receptor agonist at baseline were eligible for inclusion. Patients with pulmonary hypertension not from WHO Group 1 were excluded. The reason for transition and choice of alternative PAH medication was made at the discretion of the treating provider. All patients were evaluated for potential drug interactions with their PAH medications as a part of our routine practice. Transition success was defined as a patient successfully transitioned from bosentan and sildenafil to alternative therapy and remaining on alternative therapy without intolerable adverse events or clinical deterioration attributable to therapy transition. This study was approved by the institutional review board at our institution.
Registry to Evaluate Early and Long-Term PAH Disease Management (REVEAL) risk scores were calculated before and after transition using the updated REVEAL 2.0 risk calculator. 13 Briefly, the REVEAL risk score incorporates 13 variables including different etiologies of PAH, 6-min walk distance (6MWD), demographics, vital signs, N-terminal pro-brain natriuretic peptide (NT-proBNP) values, pulmonary function testing, and right heart catheterization information to provide risk assessment and predict survival in patients with PAH. For the REVEAL risk score, low risk is defined as a score of ≤6, intermediate risk is defined as a score of 7 or 8, and high risk is defined as a score of ≥9.
Baseline demographics and characteristics were collected from chart review. Hemodynamic assessments by right heart catheterization and clinical evaluations, including WHO functional class (FC) and 6MWD, were conducted according to our center's routine practice and recorded before and after transition when available. Cardiac index (CI) values were calculated by the Fick method. A stable 6MWD was defined as a post transition change less than 15% compared to the baseline value.14,15 A clinically meaningful change in 6MWD was defined as an increase or decrease in 6MWD after transition by greater than or equal to 15%. Tricuspid annular plane systolic excursion (TAPSE) values were assessed before and after transition by echocardiography. NT-proBNP values were assessed at baseline and after transition. After three months from the time of transition, the most proximate value for 6MWD, NT-proBNP, TAPSE, WHO FC, and hemodynamic parameters was utilized for the post-transition assessment. The timing for follow-up was at the discretion of the treating provider. The alternative medication or medications used for the transition was recorded. Adverse events during transition were assessed by review of the follow-up visit after transition and nursing phone call documentation. Any change to background PAH therapy after the transition was recorded. Patients who were newly started on a prostacyclin analogue or prostacyclin receptor agonist after transition were included for evaluation of adverse events and success of transition but were excluded from post-transition assessment of 6MWD, NT-proBNP values, TAPSE, WHO FC, REVEAL risk score, and hemodynamic parameters.
Statistical analysis
Baseline demographic and characteristic data are summarized using descriptive statistics. Continuous variables are represented as mean ± standard deviation (SD) for parametrically distributed values or median (range) for non-parametrically distributed values. Categorical variables are displayed as number and percentage. A paired t-test was used to compare differences before and after transition for parametrically distributed continuous data. The Wilcoxon signed-rank test was used to compare differences for ordinal or nonparametric continuous data. A P value < 0.05 was considered statistically significant. Statistical analyses were performed using Microsoft Office Excel 2010 (Microsoft, Redmond, WA).
Results
We identified a total of 22 patients receiving the combination of bosentan and sildenafil for WHO Group 1 PAH during the inclusion period. Five patients were excluded as they were not transitioned to alternative therapy. Lastly, one patient was transitioned from the combination of bosentan and sildenafil at another institution and was not included in this study. The remaining 16 patients were included for this analysis.
Baseline characteristics
Patient demographics and baseline characteristics.
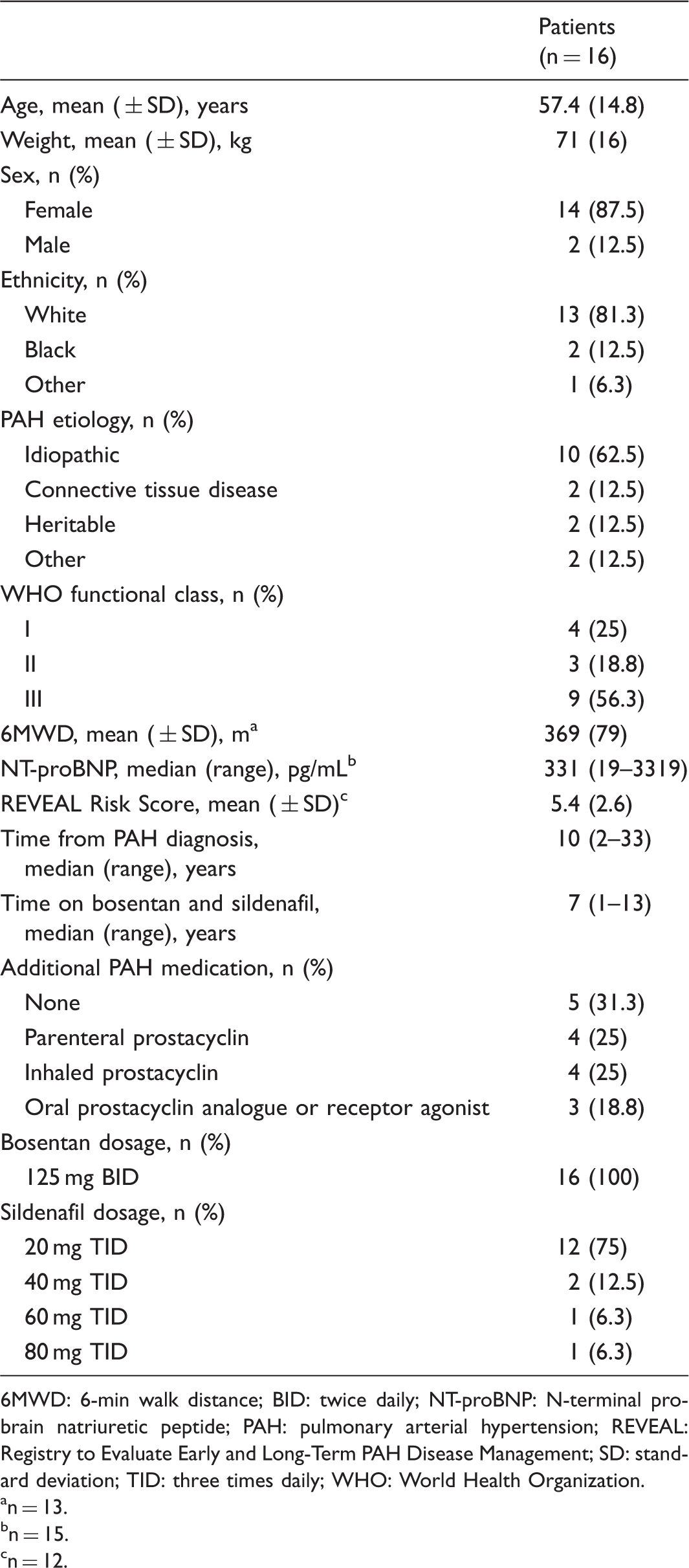
6MWD: 6-min walk distance; BID: twice daily; NT-proBNP: N-terminal pro-brain natriuretic peptide; PAH: pulmonary arterial hypertension; REVEAL: Registry to Evaluate Early and Long-Term PAH Disease Management; SD: standard deviation; TID: three times daily; WHO: World Health Organization.
n = 13.
n = 15.
n = 12.
Details on the transition of therapy from bosentan and sildenafil to alternative therapy
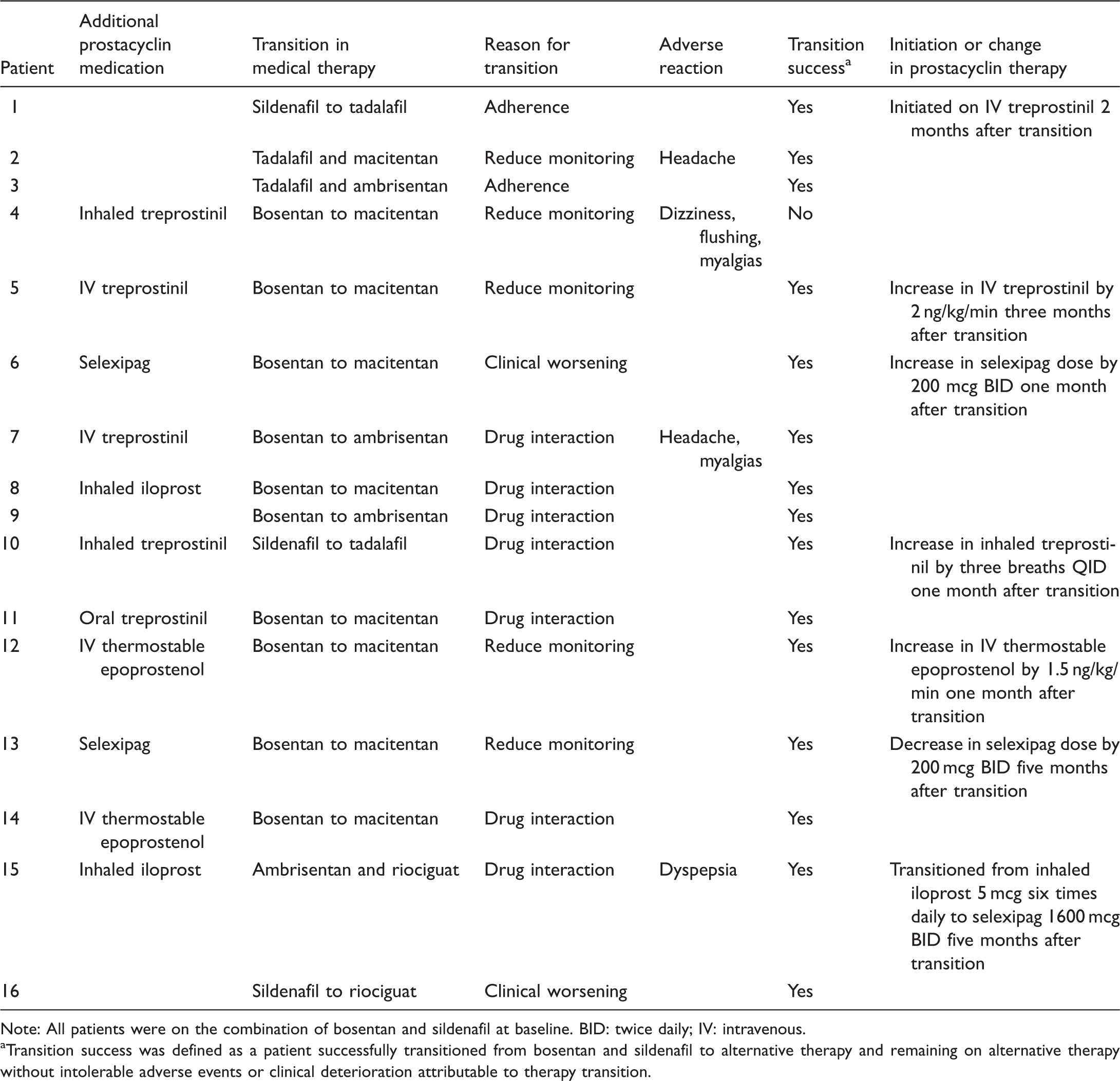
The most common reason for transition of therapy from bosentan and sildenafil to alternative therapy in our cohort was concern for a drug interaction between the two medications in 43.8% of patients. The other reasons for transition are displayed in Table 2. In most instances, only one medication was changed for the transition with a change from bosentan to macitentan occurring in 50% of patients (Fig. 1). In three patients, both sildenafil and bosentan were simultaneously changed to alternative therapy (Fig. 1).
Alternative therapy used during transition from bosentan and sildenafil. Reason for therapy transition, adverse reactions, and changes to background prostacyclin therapy. Note: All patients were on the combination of bosentan and sildenafil at baseline. BID: twice daily; IV: intravenous. Transition success was defined as a patient successfully transitioned from bosentan and sildenafil to alternative therapy and remaining on alternative therapy without intolerable adverse events or clinical deterioration attributable to therapy transition.
Safety of transition from bosentan and sildenafil to alternative therapy
Out of 16 total patients, 15 (93.8%) tolerated the transition from bosentan and sildenafil to alternative therapy. One patient who underwent a transition from bosentan to macitentan experienced intolerable adverse events including dizziness, flushing, and myalgias, which required bosentan to be resumed. Another patient was started on intravenous (IV) treprostinil two months after transition from bosentan to macitentan that was deemed unrelated to therapy transition. In this patient, the IV treprostinil was planned to be initiated several months prior to transition of therapy due to clinical worsening, but a delay in insurance approval ultimately caused the prostacyclin to be initiated after bosentan was transitioned to macitentan. A total of four patients (25%) experienced at least one adverse event. Most adverse events that were reported during the transition were mild and are displayed in Table 2.
Effect of transition on clinical data and pulmonary hemodynamics
Fourteen patients were included for evaluation of clinical data and hemodynamics after transition from bosentan and sildenafil to alternative therapy. The patient who did not tolerate the transition and the patient who was newly started on IV treprostinil two months after transition from bosentan to macitentan were excluded from this analysis. The median time after therapy transition to follow-up was 6.5 months (range: 3–13 months).
Six patients on background prostacyclin therapy had a change in prostacyclin therapy during the period from baseline to clinical follow-up (Table 2). One patient had a reduction in oral selexipag dose by 200 mcg twice daily. One patient was transitioned from inhaled iloprost 5 mcg six times daily to selexipag 1600 mcg twice daily. Four patients had an increase in effective prostacyclin dose during the period from baseline to after follow-up (Table 2).
Clinical data at baseline and after transition from bosentan and sildenafil to alternative therapy.
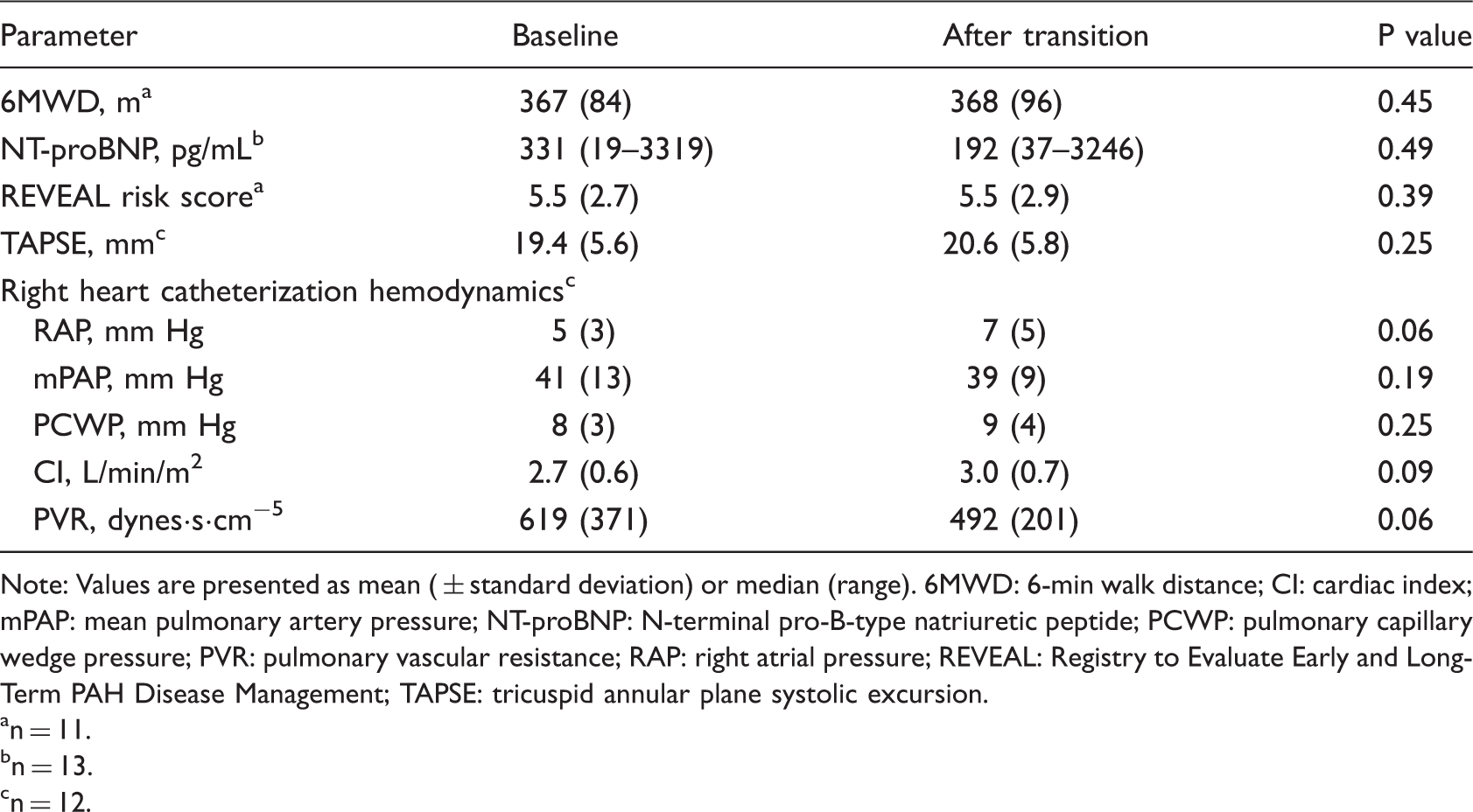
Note: Values are presented as mean ( ± standard deviation) or median (range). 6MWD: 6-min walk distance; CI: cardiac index; mPAP: mean pulmonary artery pressure; NT-proBNP: N-terminal pro-B-type natriuretic peptide; PCWP: pulmonary capillary wedge pressure; PVR: pulmonary vascular resistance; RAP: right atrial pressure; REVEAL: Registry to Evaluate Early and Long-Term PAH Disease Management; TAPSE: tricuspid annular plane systolic excursion.
n = 11.
n = 13.
n = 12.
Twelve patients demonstrated stable WHO FC symptoms after transition while two patients improved from WHO FC III to II (Fig. 2). REVEAL risk scores (n = 11) were similar in patients comparing baseline scores to scores after transition (Table 3). All patients remained stable in their REVEAL risk category (i.e. low, intermediate, or high) after transition. Additionally, there were no significant changes in right ventricular function as assessed by TAPSE (n = 12) or NT-proBNP (n = 13) values after transition (Table 3).
World Health Organization functional class at baseline and after transition in patients successfully transitioned from bosentan and sildenafil to alternative therapy. P-value = 0.08 for comparison by Wilcoxon signed-rank test.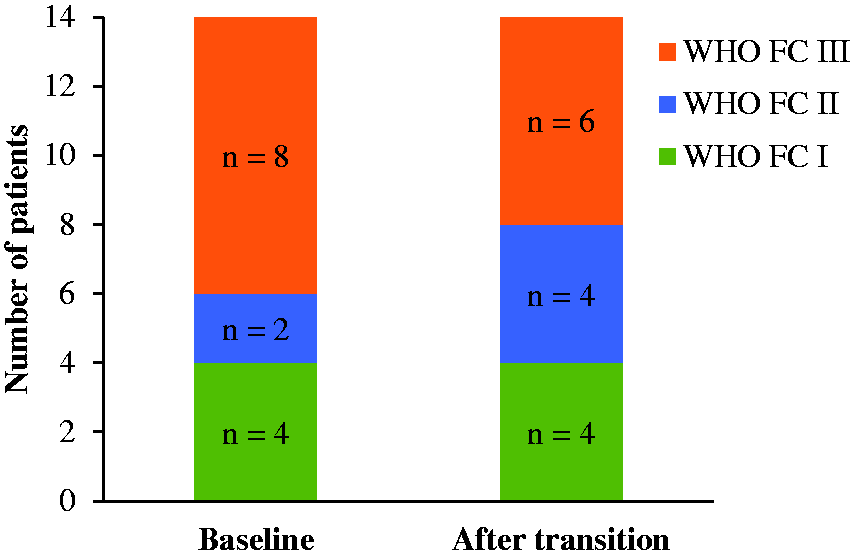
Complete hemodynamic parameters from baseline to after transition were available in 12 patients and are displayed in Table 3. Overall, there was no significant difference observed after transition with right atrial pressure (RAP), mean pulmonary artery pressure (mPAP), pulmonary capillary wedge pressure (PCWP), CI, or pulmonary vascular resistance (PVR). There were numerical changes with higher RAP (5 ± 3 vs. 7 ± 5 mm Hg; P = 0.06), higher CI (2.7 ± 0.6 vs. 3.0 ± 0.7 L/min/m2; P = 0.09), and lower PVR (619 ± 371 vs. 492 ± 201 dynes·s·cm−5; P = 0.06) after transition compared to baseline values. With regard to PVR, there was a trend for lower PVR after transition in those patients with higher PVR values at baseline compared to no observable difference in PVR after transition in those patients with lower PVR values at baseline (Fig. 3). The median change in PVR for those patients transitioned with a baseline PVR above the median value (417 dynes·s·cm−5) was −190 dynes·s·cm−5 (range: −53 to −757 dynes·s·cm−5, P = 0.03). In contrast, the median change was +33 dynes·s·cm−5 (range: −92 to + 211 dynes·s·cm−5, P = NS) in those patients transitioned with a baseline PVR below the median value of 417 dynes·s·cm−5.
Change in pulmonary vascular resistance in patients successfully transitioned from bosentan and sildenafil to alternative therapy. Each line represents an individual patient. Total n = 12.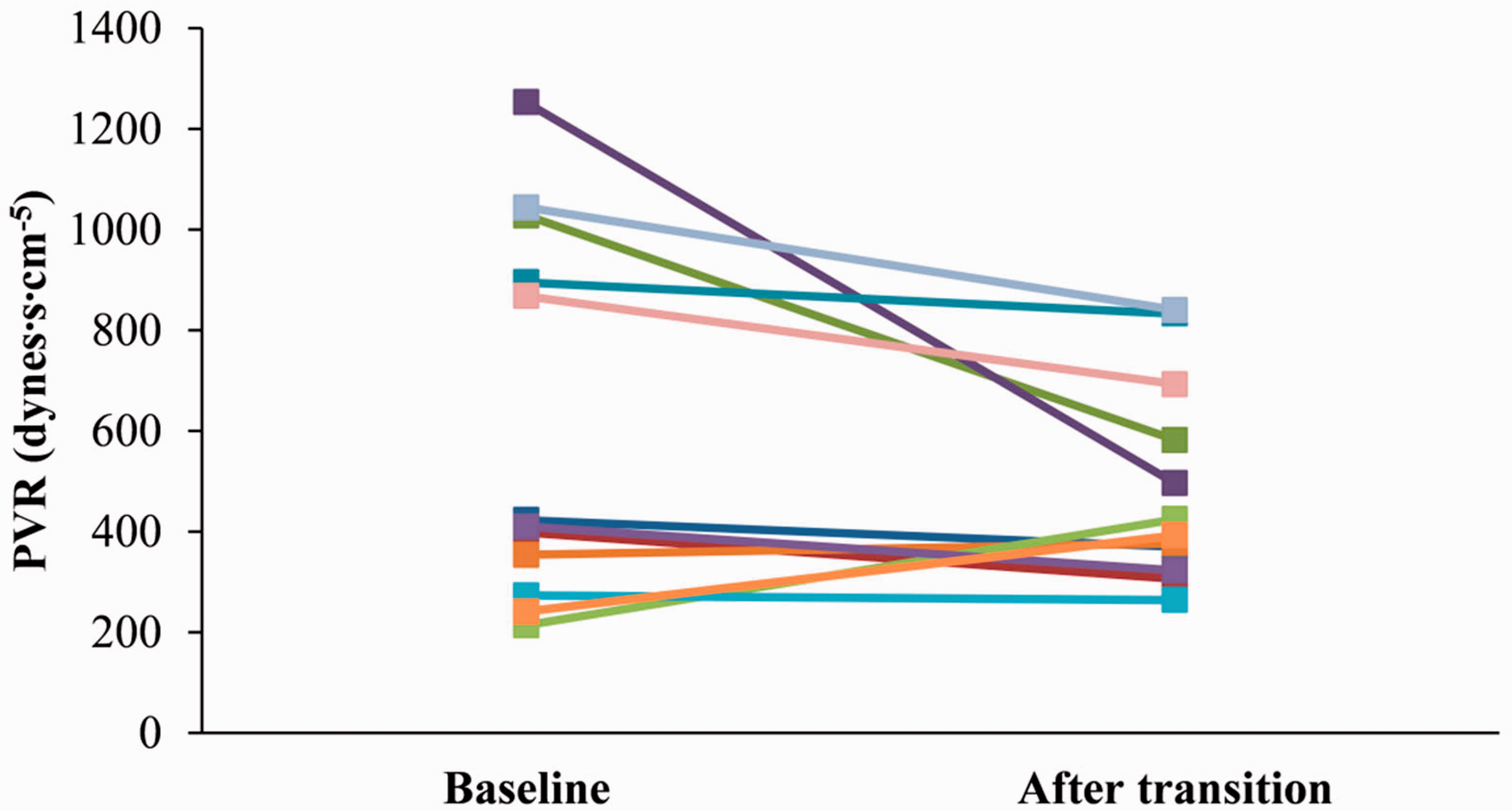
Discussion
This study is the first to evaluate the safety and feasibility of transitioning patients with PAH from the combination of bosentan and sildenafil to alternative therapy. Our study demonstrates that transitioning patients from the combination of bosentan and sildenafil to alternative therapy is safe and well tolerated. Fifteen out of 16 patients (93.8%) in our study successfully completed the transition. Adverse events during the transition period were minor in nature and occurred in 25% of our patients.
Previous studies looking at transitioning patients from bosentan and sildenafil to alternative medications have primarily been pharmacokinetic in nature and have not included an assessment of safety, tolerability, and hemodynamics after transition of therapy. Two studies have evaluated clinical parameters with the transition patients from bosentan and sildenafil to alternative therapy. Hakamata et al. compared seven total patients on combination therapy with sildenafil and bosentan to the same patients on sildenafil and ambrisentan in an open-label, crossover study. 9 There was no significant difference in 6MWD between comparing treatment with sildenafil and bosentan to sildenafil and ambrisentan, but they did find a significant improvement in an externally paced shuttle walking distance by a median of 35.0 meters (P = 0.042) in favor of the sildenafil and ambrisentan treatment. In another study, 20 patients with PAH who were already taking bosentan and sildenafil were transitioned to macitentan and sildenafil. 10 The authors found no significant change in WHO FC or TAPSE after transition, but there was a significant improvement in NT-proBNP values from 2204 ± 3823 pg/mL at baseline to 1181 ± 2899 pg/mL after transition (P = 0.003). Our study similarly found no significant change in 6MWD, WHO FC, or TAPSE after transition of therapy. We did not find a significant improvement in NT-proBNP values in our study, possibly due to the fact that the median NT-proBNP value (331 pg/mL) in our cohort was low at baseline.
The most common reason for transitioning therapy in our study was concern about a drug interaction between bosentan and sildenafil. The interaction between bosentan and sildenafil has been well characterized in the literature. Chronic administration of bosentan results in a 40%–60% decrease in overall sildenafil plasma concentrations.7–10 The reduction in sildenafil plasma concentrations with concomitant bosentan administration is thought to be due to increased sildenafil metabolism. Bosentan is known to induce expression of cytochrome P450 3A4 (CYP3A4), and sildenafil is predominantly metabolized by CYP3A4.7,16,17 Furthermore, the addition of bosentan to patients on long-term sildenafil leads to a 50% increase in plasma bosentan concentrations, although the clinical significance of this remains uncertain.8,10 In contrast, there is no appreciable drug interaction between other ERA medications such as ambrisentan or macitentan with sildenafil.10,18,19
As the first drugs in their respective classes to be commercially available, bosentan and sildenafil have been used to treat patients with PAH despite conflicting data on the efficacy of this combination. A large observational study demonstrated an improvement in hemodynamics and 6MWD with this combination therapy. 20 However, this study was retrospective and lacked a comparator group. In contrast, recent placebo-controlled trials have failed to show a significant clinical benefit with this combination. In the COMPASS-2 trial, combination therapy with sildenafil and bosentan compared to sildenafil and placebo did not significantly improve a composite morbidity/mortality end-point. 11 Additionally, a study by Vizza et al. compared patients on bosentan and sildenafil to bosentan and placebo and failed to show a significant improvement in 6MWD between treatment groups at 12 weeks. 12
In regards to efficacy, our study demonstrated that patients remained stable after transition from bosentan and sildenafil to alternative therapy. Most patients demonstrated stable or improved 6MWD and all patients had stable or improved WHO FC symptoms. We utilized a criterion of an increase or decrease in 6MWD by 15% or more to determine a clinically meaningful improvement or worsening after transition. While this may appear to be a wide margin, previous randomized trials have used a similar criterion to define their end-points for clinical worsening.14,15 A small proportion of our cohort (18.2%) demonstrated a significant decrease in 6MWD after transition; however, both patients prematurely stopped their post transition 6-min walk test due to musculoskeletal issues. In addition to 6MWD and WHO FC symptoms, clinical stability was demonstrated with consistent post transition NT-proBNP measurements, TAPSE, and REVEAL risk scores.
Although our overall study did not show any significant difference with regards to hemodynamics after transition, we did observe non-significant, numerical changes with an increase in RAP, increase in CI, and decrease in PVR comparing baseline values on bosentan and sildenafil to those after transition. Previous studies with ERA medications have shown small increases in RAP after initiation or during a transition of therapy between different ERA medications.21,22 The ERA class is known to cause increased fluid retention, and the mild increase in RAP may represent worsened fluid retention with the alternative therapy used in this study. Alternatively, this change in RAP may merely represent underlying volume changes related to diuretic adjustments or dietary changes between the baseline and post transition measurements. The non-significant, numerically greater CI in our study is likely due to the reduction in PVR. The reduction in PVR comparing baseline values on bosentan and sildenafil to after transition values was not statistically significant in our overall cohort. Our study included five patients with baseline PVR values less than 400 dynes·s·cm−5 while on bosentan and sildenafil in whom it would be unlikely to detect any meaningful reduction in PVR with any therapy change. However, we observed a significant reduction in PVR for those patients with higher baseline PVR values with a median reduction of 190 dynes·s·cm−5 in this subgroup. The reduction in PVR after transition may be attributable to optimized drug exposure by eliminating the drug interaction of bosentan and sildenafil. Nonetheless, this finding should be considered exploratory in nature and requires further validation with additional studies.
There are several limitations with this study. First, this study was small and retrospective in nature. Therefore, our study was likely underpowered to detect differences in clinical parameters and susceptible to residual confounding. Some clinical and laboratory data had missing or incomplete information which may have impacted our results. Additionally, we did not have a comparator group to compare an expected clinical course with continued bosentan and sildenafil therapy. Changes in background medications such as diuretics or prostacyclin dosing adjustments could have affected hemodynamic measurements and clinical data. We did exclude one patient from evaluation of clinical data and hemodynamics who was started on IV treprostinil after transitioning therapy, and other changes to background prostacyclin therapy were minor in our study. The alternative therapy used for the transition was left to prescriber discretion, and we are unable to make any determination about which transition was most optimal. We did not measure plasma concentrations of bosentan and sildenafil in this study, and therefore, are not able to specifically determine any contribution of a drug interaction with our results. Lastly, our study did not assess long-term follow-up or clinical endpoints. Despite these limitations, this study provides meaningful information on the safety and efficacy of transitioning therapy from bosentan and sildenafil to alternative therapy in a real-world clinical setting.
In conclusion, transitioning patients from the combination of bosentan and sildenafil to alternative therapy is safe and generally well tolerated. The transition in our cohort of patients resulted in clinical stability after transition as evidenced by stable 6MWD in most patients, improved or stable WHO FC, and preserved REVEAL risk scores. Additional studies are needed to determine if this transition is associated with any improvement in clinical outcomes or hemodynamics. The transition from bosentan and sildenafil to alternative therapy appears to be a viable therapeutic option for adult patients with PAH in order to avoid the drug interaction between those two medications.
Footnotes
Acknowledgements
The material in this article was presented as an abstract at the Pulmonary Hypertension Association’s 2019 Pulmonary Hypertension Professional Network Symposium, Washington, DC, 5–7 September 2019. The authors would like to acknowledge Mark Doyle, PhD, for his statistical assistance and Brooke Stanton, PharmD, for her assistance with review of the preliminary study protocol.
Contributorship
N. J. V. contributed to the research design, data collection, analysis, and writing of the manuscript. All authors contributed to the research design and critical revision of the manuscript. All authors read and approved the final manuscript.
Conflicts of interest
N. J. V. reports no conflict of interest. R. L. B. has received grant support from Actelion, United Therapeutics, Liquidia, Abbott, and Bayer. A. R. has received honoraria and is a principle investigator for Actelion; is a consultant and principle investigator for United Therapeutics; is a principle investigator for Liquidia, Bellerophon, and Complexa; and has received speaking honoraria from Bayer.
Ethical approval
The institutional review board at Allegheny General Hospital approved the protocol and data collection.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor
All authors take responsibility for the validity of the work and the study results reported.
