Abstract
Pulmonary hypertension (PH) occurs in patients with acute respiratory distress syndrome (ARDS); the most severe form comprises acute cor pulmonale (ACP). Here, we investigated the prevalence of PH in patients with ARDS to evaluate its correlation with ACP risk, ARDS severity and central venous pressure (CVP). We conducted a secondary analysis using data from the MIMIC-III open-source clinical database. The prevalence of PH associated with new-onset ARDS during the first 72 h after intensive care unit admission was investigated; moreover, the association between ACP risk score and PH was validated. We also evaluated the association between elevated CVP (mean CVP > 10 mmHg) and PH and other clinical outcomes. Among 2434 patients who met the ARDS Berlin criteria and underwent echocardiography or pulmonary artery catheterization evaluation, a total of 583 (24.0%) patients were diagnosed with moderate or severe PH, of which 418 had low and 165 had high ACP risk. After adjustment for disease/ARDS severity, ACP risk score, and other demographic variables, elevated CVP was independently associated with the occurrence of PH (odds ratio, 2.239 (1.674, 2.993), p < 0.005). Among patients with PH, higher mean CVP was associated with prolonged hospital stay (13.4 vs. 15.2 days, p = 0.041) and duration of ventilation (116.5 vs. 150.5 h, p = 0.023). Incidence of PH was 24.0% in patients with new-onset ARDS in this retrospective study. Elevated CVP is relevant with higher incidence of PH and worse clinical outcome; these highlighted the importance of hemodynamic monitoring in the management of ARDS.
Keywords
Acute respiratory distress syndrome (ARDS) is frequently associated with pulmonary hypertension (PH), which is related to increased pulmonary vascular resistance and subsequent right ventricular dysfunction. 1 Previous echocardiography (ECHO) 2 or pulmonary artery catheter (PAC)3–5 studies have shown that mild to moderate elevation in mean pulmonary artery pressure (mPAP) occurs in most patients with ARDS. In particular, acute cor pulmonale (ACP), which is the most severe presentation of PH, is associated with a poor clinical outcome; this illustrates the importance of pulmonary hemodynamic assessment by intensivists. 6
Recently, Mekontso Dessap et al. proposed an ACP risk score that was validated in a prospective cohort of patients with ARDS, that study showed a 22% prevalence of ACP in 752 patients with moderate-severe ARDS. 7 Although the importance of ACP identification and prevention is widely accepted in the management of ARDS,6,8 the prevalence, progression, and prognosis of PH associated with ARDS, has not been fully investigated. 9 Additionally, scoring criteria have been limited to the field of mechanical ventilation (pneumonia as a cause of ARDS and driving pressure) and gas exchange (PaCO2 and PaO2/FiO2) and have not involved any hemodynamic monitoring parameters; notably, CVP has been associated with prognosis in critically ill patients10–12 and should be included during the evaluation and identification of PH.
Considering these limitations, we aimed to investigate the prevalence of PH in patients with new-onset ARDS, as well as in subgroups with varying levels of ACP risk and ARDS severity; moreover, we aimed to evaluate the association between PH and a basic hemodynamic parameter: central venous pressure (CVP). We hypothesized that elevated CVP (mean CVP > 10 mmHg) would be independently associated with the risk, severity, and clinical outcome of PH in patients with ARDS.
Methods
Data source
We conducted a secondary analysis using data collected from the MIMIC-III open-source clinical database (version 1.4, released on 2 September 2016), which was developed and maintained by the Massachusetts Institute of Technology (MIT), Philips Healthcare, and Beth Israel Deaconess Medical Center (BIDMC). 13 Information derived from the electronic medical records of 46,476 unique critical care patients admitted to the intensive care units (ICUs) at BIDMC between 2001 and 2012 was included in this free accessible database. 14 MIMIC-III data are Health Insurance Portability and Accountability Act of 1996 compliant. Use of the MIMIC-III database was approved by the Institutional Review Boards of BIDMC and MIT, and a waiver of informed consent was granted.
Patients
All patients in the database were screened. The inclusion criteria in this study were as follows: (1) adults (≥18 years of age) at ICU admission, with complete medical records including available CVP measurement records during the first 72 h after ICU admission; (2) ICU stay ≥72 h; and (3) Berlin definition criteria for ARDS 15 were met at the time of ICU admission. The ARDS definition included: acute onset, PaO2/fraction of inspired oxygen (FiO2) of ≤300 mmHg, bilateral infiltrates on chest radiograph, and absence of heart failure. Mild, moderate, and severe ARDS were classified on the basis of the PaO2/FiO2 ratio. 15 For patients with multiple ICU stays, only data related to the first ICU admission were considered.
ACP risk score
According to the study reported by Mekontso Dessap et al., 7 we calculated the ACP risk score based on the medical records (average values) during the first day after ICU admission; this included four variables: pneumonia as a cause of ARDS, driving pressure ≥18 cmH2O, PaO2/FiO2 ratio <150 mmHg, and arterial carbon dioxide partial pressure ≥48 mmHg. For this study, driving pressure was defined as the difference between plateau pressure and positive end-expiratory pressure (PEEP).
Outcome
The primary outcome was moderate or severe PH, as defined by either diagnosis in the ECHO report or mPAP measured by PAC (moderate PH: mPAP between 30 and 45 mmHg; severe PH: mPAP > 45 mmHg).5,16 Pulmonary artery pressure (systolic and mean PAP) estimation by ECHO was based on the measurement of tricuspid regurgitation peak velocity, which is a classic and widely accepted method in PH estimation, and mean PAP by ECHO was calculated from the estimated systolic PAP. 16 Only ECHO or PAC evaluations performed during the first 72 h after ICU admission were considered when assessing evidence of PH. If both results were available and the grades were inconsistent, the PAC measurement was used as the decisive outcome. Enrolled patients without any available PAC or ECHO evaluations were considered to be non-PH subjects. Other clinical outcomes were recorded, including 28-day mortality, ICU stay, hospital stay, vasopressor duration, and ventilation duration.
Statistical analysis
Data are expressed as the mean ± standard deviation, median with interquartile range, and proportions (absolute and relative frequencies), as appropriate. Student's t test or the Mann–Whitney test was used to compare continuous variables, while the χ 2 test or Fisher's exact test was used to compare proportions. All statistical analyses were performed by using the IBM® SPSS® Statistics version 24 (SPSS Inc., Chicago, IL). A p value of <0.05 was considered to indicate a statistically significant difference.
Mean CVP level during the first 72 h after ICU admission was used to categorize the patients into two groups: ≤ 10 and >10 mmHg, which comprised patients of low and high CVP groups, respectively. 11 To assess the independent effect of high mean CVP level on outcome (moderate or severe PH) in patients with ARDS, we performed a multivariate logistic regression regarding the association between elevated mean CVP and occurrence of PH after adjustment on the basis of relevant illness severity variables, which comprised demographic characteristics; ICU type; admission type; Simplified Acute Physiology Score II (SAPS II); Sequential Organ Failure Assessment (SOFA); and ACP risk score at admission and whether lung protective ventilation (LPV) strategy was followed. To further investigate the association of the elevated mean CVP and severity of PH, we also compared the proportions of various degrees of PH among subgroups of patients with varying levels of ACP risk and ARDS severity.
Results
Basic characteristics
Among the 46,476 ICU patients and 61,532 ICU admissions in the MIMIC-III v1.4 databases, 7823 patients met the ARDS Berlin criteria at ICU admission and 2434 patients with ARDS stayed in the ICU for >72 h. Among these 2434 patients, 1824 underwent ECHO evaluation and 958 received PAC evaluation, as illustrated in Fig. 1. According to the reports of ECHO and average mPAP from PAC evaluation during the first 72 h after ICU admission, 583 patients with ARDS were diagnosed with moderate or severe PH (PH group), while 1851 patients exhibited no evidence, or had borderline/mild degree of PH (non-PH group). SAPS and SOFA scores at admission, as well as the proportion of patients for whom the LPV strategy was followed, were comparable between PH and non-PH groups. The severity of ARDS showed an opposite trend with incidence of PH.
Flowchart showing step-by-step selection of patients included in the study. ICU: intensive care unit; CVP: central venous pressure; ARDS: acute respiratory distress syndrome; ACP: acute cor pulmonale; PAC: pulmonary artery catheter; PAP: pulmonary artery pressure.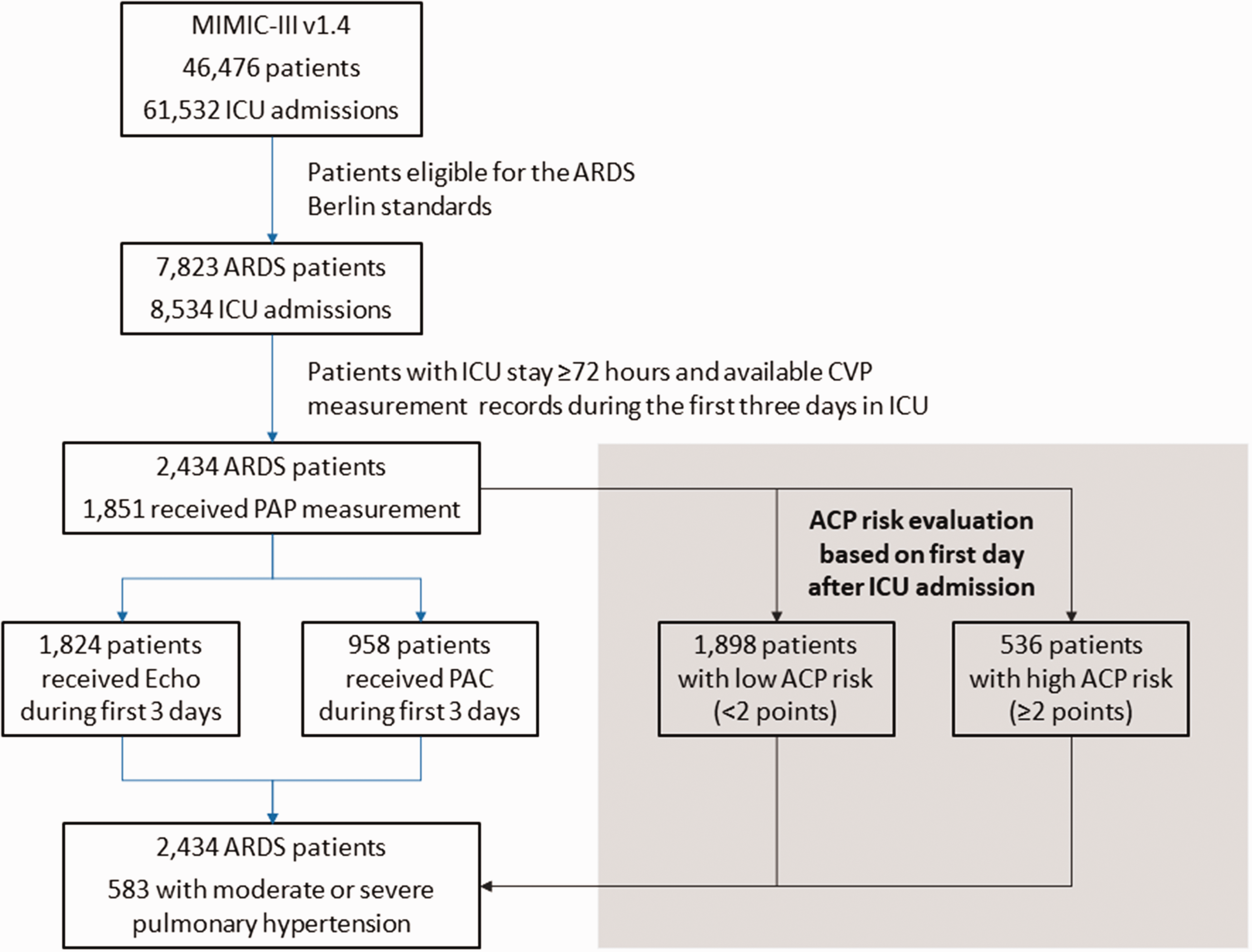
General patient characteristics and outcome of patients who exhibited acute respiratory distress syndrome with or without severe pulmonary arterial hypertension.
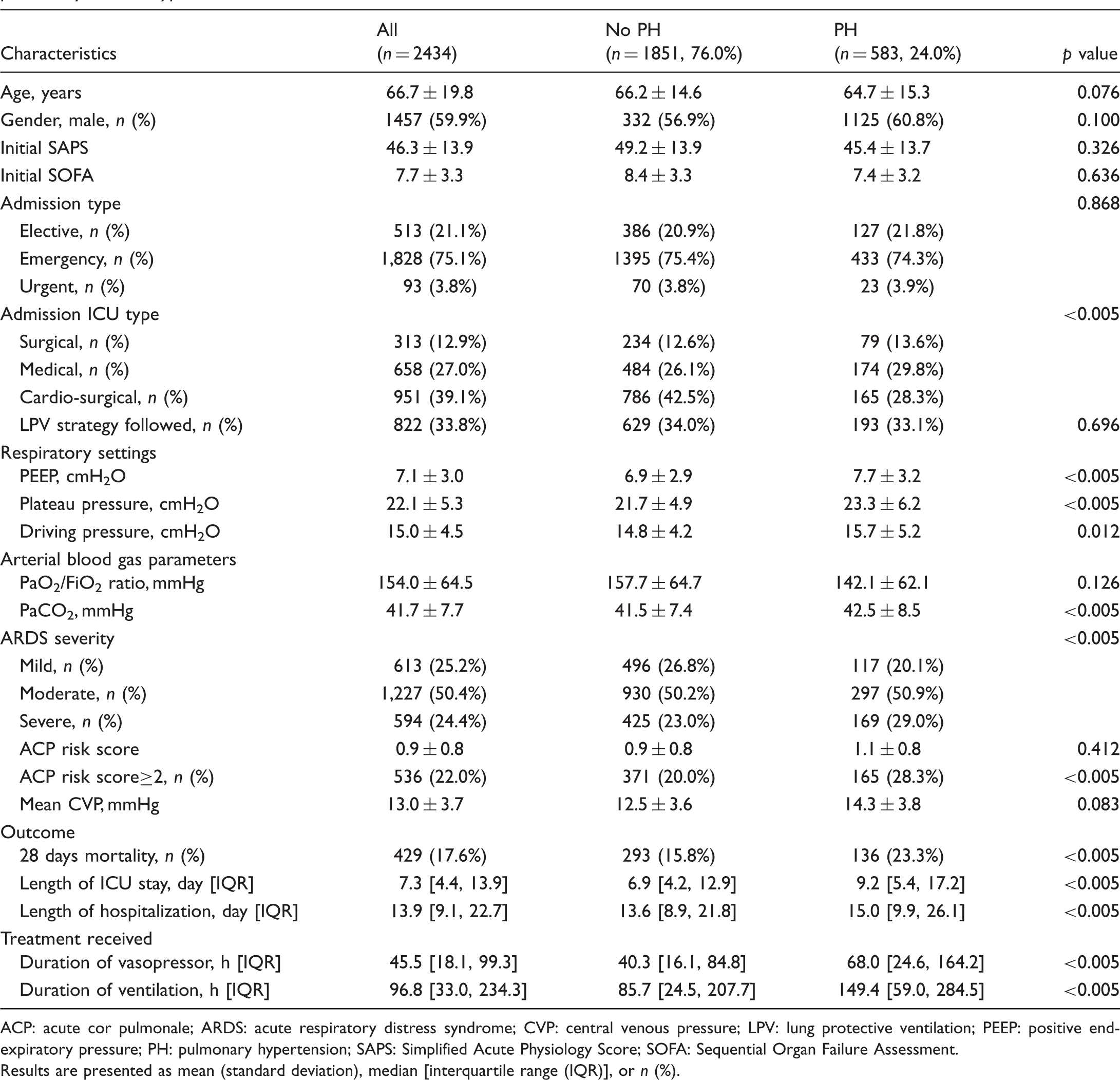
ACP: acute cor pulmonale; ARDS: acute respiratory distress syndrome; CVP: central venous pressure; LPV: lung protective ventilation; PEEP: positive end-expiratory pressure; PH: pulmonary hypertension; SAPS: Simplified Acute Physiology Score; SOFA: Sequential Organ Failure Assessment.
Results are presented as mean (standard deviation), median [interquartile range (IQR)], or n (%).
Mean CVP level and outcome
Multivariate analysis between patients with and without pulmonary hypertension in patients with adult respiratory distress syndrome.
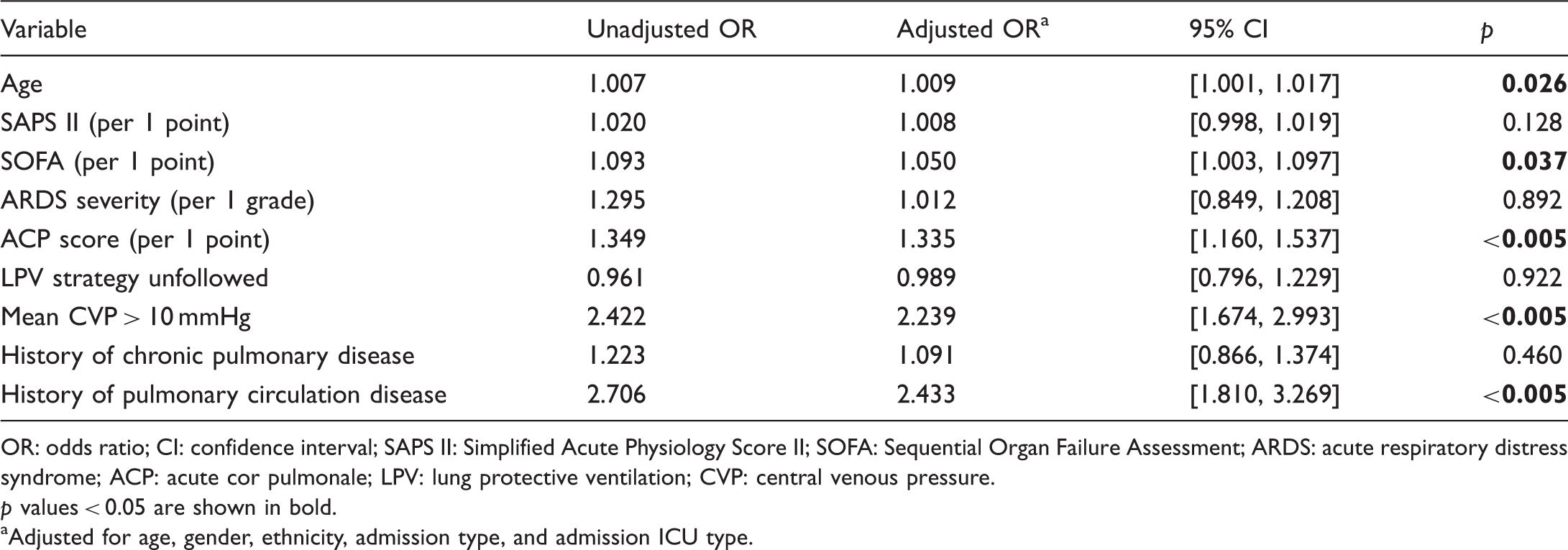
OR: odds ratio; CI: confidence interval; SAPS II: Simplified Acute Physiology Score II; SOFA: Sequential Organ Failure Assessment; ARDS: acute respiratory distress syndrome; ACP: acute cor pulmonale; LPV: lung protective ventilation; CVP: central venous pressure.
p values < 0.05 are shown in bold.
Adjusted for age, gender, ethnicity, admission type, and admission ICU type.
Main clinical outcomes in subgroups with mean central venous pressure ≤10 mmHg and mean central venous pressure >10 mmHg in patients with moderate or severe pulmonary hypertension.
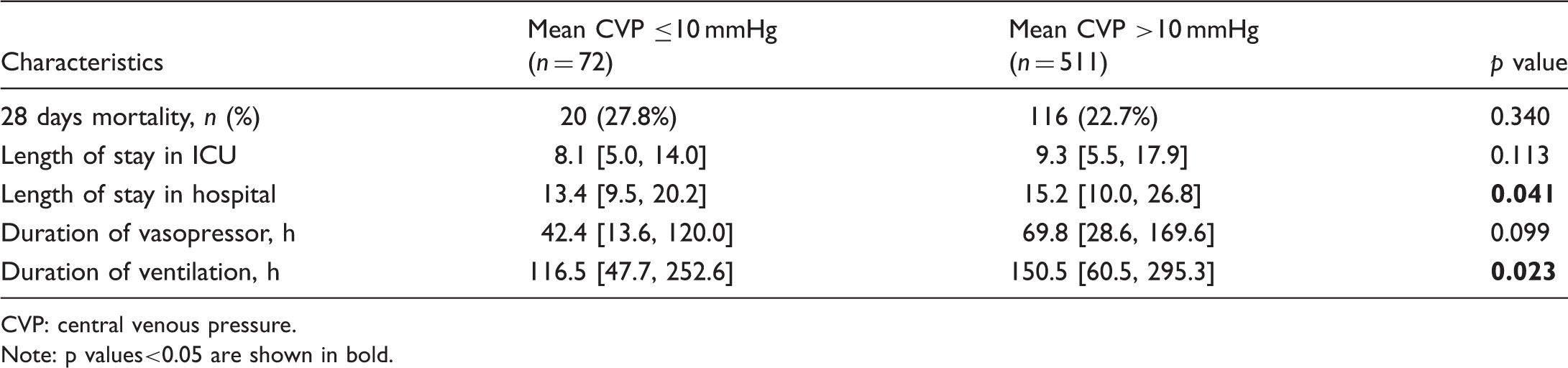
CVP: central venous pressure. Note: p values<0.05 are shown in bold.
Subgroups and sensitivity analysis
Because the ACP risk score and ARDS severity may reflect the potential risk of PH in patients with ARDS, we investigated the associations between mean CVP level and PH prevalence among different subgroups after stratification by ACP risk score and ARDS severity, as shown in Fig. 2. The results showed that the incidence and severity of PH were lower in patients with ARDS who exhibit mean CVP ≤10 mmHg than in patients with ARDS who exhibit mean CVP >10 mmHg among most subgroups stratified by ACP risk (0 points: 29 (11.6%) vs. 125 (22.2%), p < 0.005; 1 point: 28 (14.2%) vs. 236 (26.6%), p < 0.005; 2 points: 11 (14.1%) vs. 121 (32.4%), p = 0.001; ≥3 points: 4 (23.5%) vs. 29 (42.6%), p = 0.175) and ARDS severity (mild: 25 (13.4%) vs. 92 (12.7%), p = 0.019; moderate: 30 (12.0%) vs. 267 (14.0%), p < 0.005; severe: 17 (16.0%) vs. 152 (31.1%), p = 0.001).
Subgroups and sensitivity analysis of prevalence of pulmonary hypertension in patients with acute respiratory distress syndrome. (a and b) Subgroup analysis of prevalence of PH according to the ACP risk score, ARDS severity, and mean CVP level during the first three days after intensive care unit admission. (c) Forest plot for adjusted odds ratio values of higher mean CVP level (≥10 mmHg) and moderate or severe pulmonary hypertension per subgroup. Adjusted for age, gender, ethnicity, admission type, admission ICU type, SAPS II and SOFA score at ICU admission, ARDS severity, ACP score, and whether the LPV strategy was followed.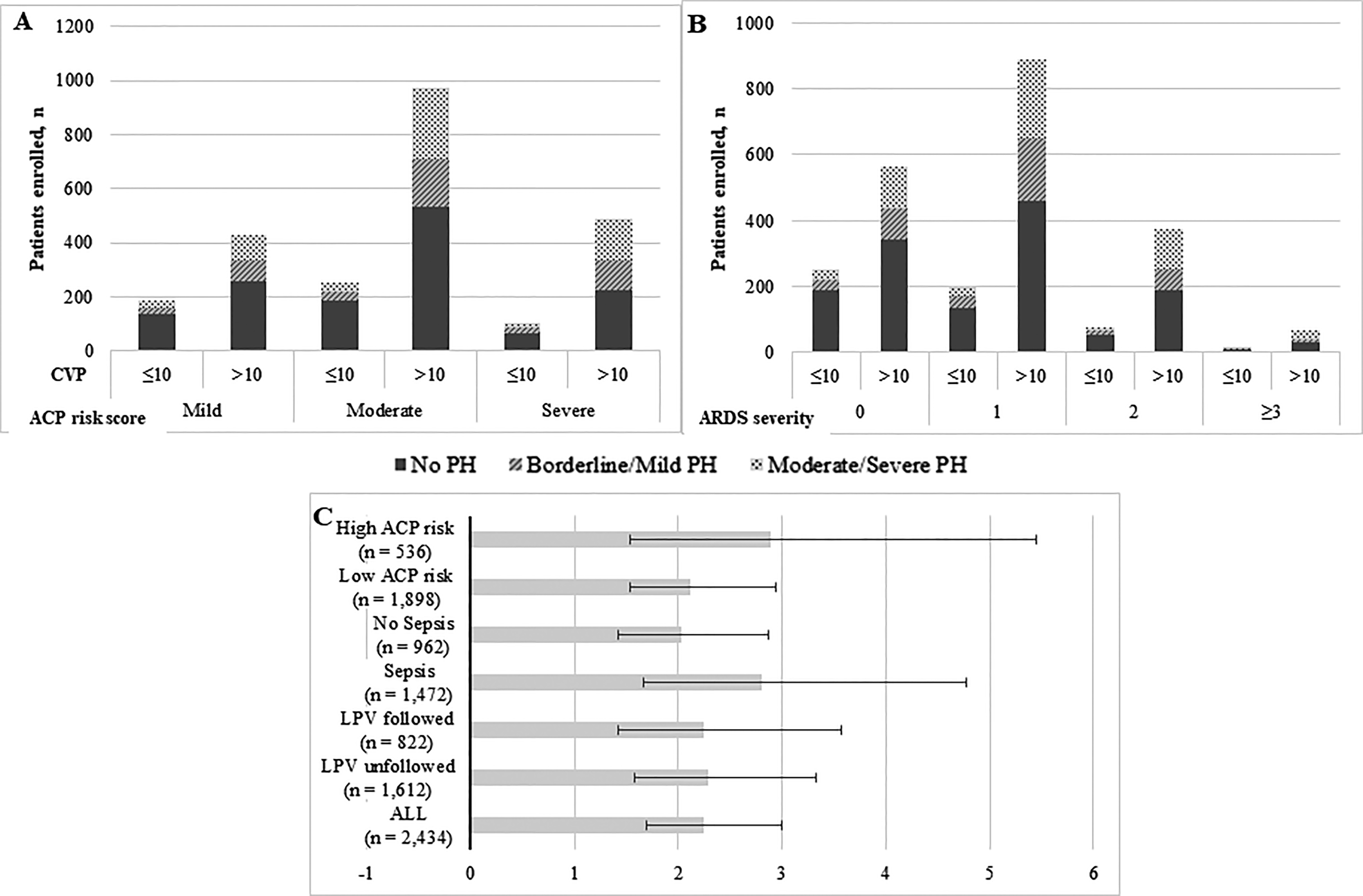
Multivariable regression analysis also confirmed that in the low or high ACP risk subgroups, sepsis or non-sepsis subgroups, and LPV followed or not subgroups, the significant effect of mean CVP >10 mmHg on PH morbidity remained in patients with ARDS, as illustrated in Fig. 2.
Discussion
ARDS is a clinical condition characterized by acute hypoxemic respiratory failure. In recent years, intensivists have progressively recognized the significance of circulatory failure in ARDS, which is caused by pulmonary vascular dysfunction (PVD) and may eventually cause ACP. 1 Several studies have indicated that ACP is common in patients with moderate-to-severe ARDS; moreover associated with increased mortality in patients with ARDS.4,6,7,17,18 As noted in the Introduction of the present article, Mekontso Dessap et al. proposed a risk prediction model to identify high risk factors of developing ACP; notably, they observed a 22% prevalence rate of ACP within three days from the diagnosis of ARDS in the group with high ACP risk. 7
Nevertheless, further studies have enhanced the understanding of pulmonary hemodynamics in ARDS; thus, ACP should not be regarded as the sole clinical outcome of PVD evaluation. 9 Instead, moderate to severe PH has been designated for early recognition and intervention before the onset of ACP. In the present study, we investigated the prevalence of PH associated with new-onset ARDS during the first 72 h after ICU admission. The ACP risk model was validated, and the prevalence of PH was found to be significantly higher in the group of patients with ARDS who exhibited high ACP risk than in those with low ACP risk (30.8% vs. 22.0%). Our results indicate an increased incidence of PVD associated with ARDS and support the use of ACP risk score as a sign of more generalized PH, rather than for the extreme manifestation of ACP.
The evaluation of right heart dysfunction plays a critical role in the development of PH associated with ARDS. 18 Therefore, in this study, CVP, which is the most convenient and direct method for right-heart monitoring, was used for the evaluation and prognosis of PH associated with ARDS. After adjustment for disease/ARDS severity, ACP risk score, and other demographic variables, we showed that elevated mean CVP (>10 mmHg) was independently associated with the occurrence of PH in patients with ARDS during the first three days after ICU admission. Based on the data-driven analysis on MIMIC III v1.4, our result highlights the critical role of CVP assessment in the evaluation and treatment of ARDS. Analysis of patients with PH showed that patients with lower mean CVP had better clinical outcomes. Subgroup analysis of patients with varying degrees of ACP risk score and ARDS severity also showed that the incidence and severity of PH was lower in patients with ARDS who exhibited mean CVP of ≤10 mmHg.
Elevated CVP (ECVP) occurs frequently in critical care settings 19 and may be caused by several conditions, such as heart failure, pericardial disease, and especially as a surrogate of intravascular volume. However, recent studies have challenged the validity of ECVP in critical care settings. An ECVP might indicate an impediment to the venous return and microcirculatory blood flow as well as accompanying lung edema. ECVP may work as a preliminary indicator of increased pulmonary vascular resistance or right ventricular dysfunction, which was crucial during the development of PH.
It is noteworthy the initial SAPS and SOFA scores were comparable between PH and non-PH groups and the proportions of patients employing LPV were both <40%. The use of LPV was introduced in the 2000s and was widely accepted, because it specifically lowered the incidence of ventilation-induced lung injury and mortality in ARDS. Multiple studies, however, showed the reality in clinical settings was not satisfactory, considering the obvious underuse of the LPV strategy.20–22 Therefore, in this real-world study, we did not restrict subjects into strict LPV strategy, in order to more clearly evaluate the current status of PH and the use of ACP risk score. Our results did not indicate whether following LPV was independently associated with PH after adjustment; this may partially be because the PaO2/FiO2 ratio and driving pressure were already included in the ACP risk score. Therefore, we suggest that ACP risk evaluation should not be limited to patients with ARDS who followed the LPV strategy.
Our study has several limitations. First, this study is limited by its retrospective nature and the source of data used. Therefore, no causal relationships could be established among CVP, PH, and mortality. Additionally, CVP records, ECHO reports, and PAC results could only be assessed in patients who underwent the corresponding evaluation after ICU admission. Thus, the associations of CVP (or other variables) with PH may be overemphasized in such situations. Additionally, further investigation is needed regarding whether early CVP-targeting intervention, which consists of fluid management and cardiac optimization, may affect the incidence of PH and other clinical outcomes in patients with ARDS. Second, prone position ventilation has been proven to be associated with an improved oxygenation level and lowered risk of ACP in patients with ARDS. Nevertheless, considering the lack of uniform standards of such therapy, we did not include this indicator in the study. Thus, further investigation is needed to determine whether prone position ventilation may lower the risk of PH in patients with ARDS who exhibit a high risk of ACP.
In conclusion, in this retrospective study using a large intensive care database, we investigated the prevalence of PH in patients with new-onset ARDS and validated the use of ACP risk score to predict the incidence of PH. We also confirmed independent associations between elevated CVP and both higher incidence of PH and worse clinical outcome; these findings highlight the importance of hemodynamic evaluation in the identification and management of ARDS.
Footnotes
Authors’ contributions
LDW designed the experiment. LDK drafted and revise the manuscript. MJY assembled input data, participated in the design of the study, and helped to draft the manuscript. LY assembled input data, participated in the design of the study, and helped to revise the manuscript. WXT conceived of the study, participated in its design and coordination, All authors have read the manuscript and approved of the version to be published.
Acknowledgments
The authors acknowledge all of those involved in the design, construction, and maintenance of the open access MIMIC-III database at the MIT Laboratory of Computational Physiology as well as the online community at Open Data Stack Exchange (http://opendata.stackexchange.com) and GitHub (![]() ). This manuscript has been edited and proofread by Ryan Chastain-Gross, PhD, from Liwen Bianji, Edanz Group China.
). This manuscript has been edited and proofread by Ryan Chastain-Gross, PhD, from Liwen Bianji, Edanz Group China.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Natural Science Foundation of Beijing Municipality (No. 7194306).
