Abstract
Adrenomedullin is a potent vasodilatory peptide, linked to pulmonary arterial hypertension pathology. Proximity extension assays were utilized to study plasma biomarkers related to vasoregulation, with focus on adrenomedullin peptides and precursor levels, collectively referred to as ADM. ADM was measured in 48 treatment-naïve pulmonary arterial hypertension patients at diagnosis, and in 31 of them at an early treatment follow-up. Plasma ADM was additionally assessed in patients with chronic thromboembolic pulmonary hypertension (n = 20) and pulmonary hypertension due to heart failure with preserved (HFpEF(PH)) (n = 33) or reduced (HFrEF(PH)) (n = 36) ejection fraction, as well as healthy controls (n = 16). ADM was studied in relation to pulmonary arterial hypertension hemodynamics, risk assessment, prognosis, treatment response, and differentiation. Plasma ADM levels in pulmonary arterial hypertension patients at diagnosis were higher than in healthy controls (p < 0.001), similar as in chronic thromboembolic pulmonary hypertension patients (p = ns), but lower compared to HFpEF(PH) (p < 0.03) and HFrEF(PH) (p < 0.001). In pulmonary arterial hypertension, specifically, plasma ADM at diagnosis correlated mainly to mean right atrial pressure (r = 0.73, p < 0.001), N-terminal prohormone of brain natriuretic peptide (r = 0.75, p < 0.001), six-minute walking distance (r = –0.57, p < 0.001), and venous oxygen saturation (r = –0.57, p < 0.001). ADM also correlated to the ECS/ERS- (r = 0.74, p < 0.001) and REVEAL risk scores (r = 0.54, p < 0.001) at pulmonary arterial hypertension diagnosis. Plasma ADM in pulmonary arterial hypertension patients was unaltered at early treatment follow-up compared to baseline (p = ns). Pulmonary arterial hypertension patients with supra-median ADM at diagnosis showed worse overall survival than those with infra-median levels (median survival 34 versus 66 months, p = 0.0077). In conclusion, the present results suggest that baseline plasma ADM levels mirror disease severity, correlating to both ECS/ERS- and the REVEAL risk scores.
Keywords
Introduction
In pulmonary arterial hypertension (PAH), small pulmonary arteries are subjected to vasoconstriction and vascular remodeling. PAH patients suffer from dyspnoea, fatigue, edema, and eventually syncope, as right ventricular failure ensues. 1 As such symptoms can be inherent to other cardiovascular and pulmonary diseases, the time between symptom onset and PAH diagnosis is often delayed. 2 This delay may contribute to the poor survival rates observed in PAH.
Assessing risk for early mortality is important in PAH management and can be estimated using the risk assessment instrument of the European Society of Cardiology/European Respiratory Society (ESC/ERS) PAH 2015 guidelines or the REVEAL risk score.1,3 Risk stratification according to the ESC/ERS guidelines has recently been validated for predicting one-year mortality.4–6 Notably, N-terminal prohormone of brain natriuretic peptide (NT-proBNP) is the only circulating biomarker used to assess risk in this model.
The endothelin, nitric oxide, and prostacyclin systems are the main targets for the drugs used in the treatment of PAH, reflecting the importance of disturbed vasoregulation in PAH pathology. 1 Adrenomedullin is a potent vasodilatory peptide that is overexpressed in many cardiovascular diseases, including pulmonary hypertension (PH).7,8 Previous research has provided evidence suggesting that adrenomedullin administration may attenuate PH in animal models, and might even improve hemodynamics in PH patients.9–13 Adrenomedullin has also been shown to inhibit proliferation in pulmonary artery smooth muscle cells and to alleviate pulmonary artery collagen accumulation in PH.13–15 Besides adrenomedullin, the processing of the adrenomedullin precursor also yields pro-adrenomedullin N-20 terminal peptide (PAMP), which also acts as a vasodilator, as well as the mid-regional pro-adrenomedullin (MR-proADM). 16 Reliable measurement of adrenomedullin and PAMP may be difficult.17,18 Although MR-proADM appears to lack a biological function, it seems to be stable in plasma, and may reflect adrenomedullin and PAMP activity.17,18 Notably, a previous study has shown that MR-proADM is correlated to exercise capacity and related to one-year mortality in PAH and chronic thromboembolic PH (CTEPH) patients. 19
In the present study, we investigated plasma biomarkers related to altered vasoregulation in incident treatment-naïve PAH patients compared to healthy subjects, CTEPH, and left heart failure with or without PH. This study focused specifically on adrenomedullin peptides and precursor levels in plasma, collectively referred to as ADM. Plasma biomarker levels were assessed in relation to PAH hemodynamics, risk stratification, and treatment response.
Method
Study population and blood sampling
The patients included in the study had PAH (n = 48), CTEPH (n = 20), PH due to left heart failure with reduced (HFrEF(PH)) (n = 36) or preserved (HFpEF(PH)) (n = 33) ejection fraction, as well as heart failure patients without PH (HF(no PH)) (n = 15). In the HF(no PH) group, eight patients had reduced and seven had preserved ejection fraction.
All included patients were adults (≥18 years), and diagnosed with right heart catheterization (RHC), between September 2011 and March 2017, at the Hemodynamic Lab at Skåne University Hospital in Lund, Sweden. Patients were diagnosed by experienced cardiologists in a national PAH and transplantation reference center, where different PH causes were differentiated using techniques that include computer tomography, spirometry, pulmonary scintigraphy, echocardiography and/or magnetic resonance tomography, besides RHC. Venous blood samples were obtained from all patients during baseline RHCs, through the introducer in the internal jugular vein. Of the baseline PAH population, 33 patients also had venous plasma samples taken during RHCs at early follow-ups, 116 (90–127) days after diagnosis. Among them, two were excluded due to missing values, rendering 31 patients for follow-up analyses. Peripheral venous blood was moreover obtained from 16 healthy control subjects (median age = 46.5 years, 69% were female). Plasma extracted from blood samples was stored at –80 ℃ in the Lund Cardio Pulmonary Register cohort of Region Skånes biobank.
PAH and CTEPH patients had not received pulmonary vasodilatory treatment before diagnosis. PAH patients having idiopathic (n = 21) or familial (n = 2) PAH were pooled into one entity referred to as idiopathic/familial PAH (IPAH/FPAH). PAH patients with systemic sclerosis associated (n = 21) or other connective tissue disease associated (n = 4) PAH were regarded as another entity, referred to as connective tissue disease PAH (CTD-PAH).
At treatment follow-up, PAH patients were on monotherapy with either bosentan (n = 5), macitentan (n = 4), ambrisentan (n = 7), sildenafil (n = 4), or tadalafil (n = 2) or a combination therapy with bosentan and sildenafil (n = 1), macitentan and sildenafil (n = 2), macitentan and tadalafil (n = 1), macitentan, tadalafil, and treprostinil (n = 1), ambrisentan and sildenafil (n = 1), or ambrisentan and tadalafil (n = 3). Among them, some had nifedipine due to rheumatologic symptoms during both baseline and follow-up (n = 9) or only at baseline (n = 1). Two acute vasoreactive PAH patients were on either nifedipine alone (n = 1) or nifedipine and macitentan (n = 1).
Among the 48 PAH patients at diagnosis, 15% had ischemic heart disease, 35% systemic hypertension, 25% diabetes mellitus, 8% atrial fibrillation, 4% stroke, and 23% thyroid disease.
All participants were well-informed regarding the purpose of the blood sampling and gave their written consent to participate. The local ethics board in Lund approved the study (Dnr 2015/270, Dnr 2011/777, Dnr 2011/368, Dnr 2010/114, Dnr 2010/442).
Biomarker analyses
Proseek multiplex cardiovascular II and III 96-plex immunoassays (Olink Proteomics, Uppsala, Sweden) were used for the present biomarkers. Adrenomedullin precursor and peptides, collectively referred to as ADM, renin, angiotensin converting enzyme 2 (ACE2), as well as NT-proBNP, were assessed using proximity extension assays (PEA). The Olink ADM assay used polyclonal antibodies that can detect the following peptides and pro-peptides of ADM (UniProt accession number: P35318): PAMP that can be found in amino acid position 22–41, MR-proADM found in position 45–92, adrenomedullin in position 95–146, as well as the unprocessed ADM protein.
The PEA analysis has previously been thoroughly described. 20 In brief, oligonucleotide-labeled antibody pairs are used to detect targeted biomarkers, in order to avoid unspecific antibody binding and potential cross-reactivity events. As two related probes are brought in close proximity, the oligonucleotides hybridize in a pair-wise manner. DNA polymerase addition results in a proximity-dependent DNA polymerization event, creating a unique PCR target sequence. The DNA sequence is then detected and quantified using a microfluidic real-time PCR instrument (Biomark HD; Fluidigm, San Francisco, CA, USA). Data quality control and normalization are performed utilizing an internal extension control and an inter-plate control, in order to adjust for intra- and inter-run variation. Assay validation data and panel information are available at www.olink.com.
The biomarkers are presented as arbitrary units (AU) on a log2 scale. Since PEA does not report absolute concentration, NT-proBNP values, analyzed at the hospital laboratory by electro-chemiluminescence immunoassays, were retrieved from the patient journals and used to calculate patient risk scores.
Hemodynamics, six-minute walking distance, and WHO functional class
RHCs were performed using Swan-Ganz catheters. Thermodilution was used to measure cardiac output (CO). Hemodynamics, six-minute walking distance (6MWD), mixed venous oxygen saturation (SvO2), and WHO functional class (WHO-FC) were extracted from the patient records.
Body surface area (BSA), mean right atrial pressure (MRAP), diastolic pulmonary artery pressure (DPAP), mean arterial pressure (MAP), mean pulmonary artery pressure (MPAP), pulmonary artery wedge pressure (PAWP), heart rate (HR), and CO were used to calculate cardiac index (CI), transpulmonary gradient (TPG), diastolic pulmonary gradient (DPG), pulmonary vascular resistance (PVR), systemic vascular resistance (SVR), right (RVSWI), and left (LVSWI) ventricular stroke work indexes, according to the formulae: CI = CO/BSA, SVI = (CO/HR)/BSA, TPG = MPAP – PAWP, DPG = DPAP – PAWP, PVR = TPG/CO, SVR = (MAP – MRAP)/CO, RVSWI = (MPAP – MRAP) × SVI, and LVSWI = (MAP – PAWP) × SVI.
Risk score calculations
An ESC/ERS guideline risk score, based on the Kylhammar et al. Swedish PAH Registry (SPAHR) model, was calculated using MRAP, CI, WHO-FC, 6MWD, NT-proBNP, and SvO2. 5 Each parameter was graded with a score from 1 to 3, according to the cut-offs provided in the risk assessment instrument from the 2015 ESC/ERS PAH guidelines, 1 where 1 corresponded to “low”, 2 to “intermediate”, and 3 to “high” risk. 5 A mean of the parameter scores was then calculated, which defined a patient's risk score in the present study. This risk score is referred to as “ESC/ERS guideline risk score”. Ten patients had one missing parameter, and one patient had two missing parameters.
REVEAL risk score (1.0) was calculated for all PAH patients. 3 As some patients had a few missing values, we calculated a modified score using a mean of available points and did not include the always-assigned six points in the original algorithm. Renal insufficiency was defined as plasma creatinine > 120 µmol/L. Additional data related to the REVEAL risk score parameters used in the present study population have previously been presented as supplemental material. 21
Statistics
Statistics were performed using GraphPad Prism (version 8, GraphPad Software, La Jolla, CA, USA) or R 3.5.1 (R Foundation for Statistical Computing, Vienna, Austria).
Kruskal–Wallis test was used to compare plasma biomarkers in PAH patients to controls and disease groups. The Benjamini–Hochberg procedure was used to assess false discovery rate (FDR) for the statistical tests comparing PAH patients to controls and disease groups. FDR was set to 5%. FDR according to the Benjamini–Hochberg method was, for each biomarker separately, also used to identify the differences between PAH and other studied groups.
Mann–Whitney U test was used to evaluate differences between IPAH/FPAH versus CTD-PAH subgroups. To study how plasma ADM levels at baseline relate to treatment response, patients with follow-up data were dichotomized based on supra- and infra-median baseline ADM levels. The change in 6MWD, MRAP, CI, PVR, SvO2, and NT-proBNP, between diagnosis and follow-up, were compared between the dichotomized groups using a Mann–Whitney U test. In PAH patients, biomarker levels were compared between baseline and follow-up with a Wilcoxon signed-rank test.
Baseline MRAP, CI, PVR, SvO2, 6MWD, and NT-proBNP in PAH patients were tested for correlations against baseline biomarker levels using Spearman's method. Plasma ADM at PAH diagnosis was, moreover, selected for correlation analyses against REVEAL and ESC/ERS guideline risk scores.
Event-free survival in PAH patients was analyzed using the Kaplan–Meier method, where the difference between patients with supra- versus infra-median baseline ADM was assessed using the log-rank test. An event was defined as death or lung transplantation. The area under the curve (AUC) of the receiver operating characteristics was calculated to assess whether ADM can differentiate PAH patients that meet versus those that do not meet the defined event within one year. Similar analysis was performed to study patients meeting and not meeting the event within three years.
Statistical significance was defined as p < 0.05. All values are presented as medians (lower–upper quartiles), unless otherwise stated.
Results
Baseline characteristics.

Notes: The patient characteristics in this table are presented for descriptive purposes, with no statistical testing performed. Values are presented as median (lower–upper quartiles), unless otherwise stated.
6MWD: six minute walk distance; BSA: body surface area; CI: cardiac index; DPG: diastolic pulmonary gradient; LVSWI: left ventricular stroke work index; MPAP: mean pulmonary arterial pressure; MRAP: mean right atrial pressure; PAWP: pulmonary artery wedge pressure; PVR: pulmonary vascular resistance; RVSWI: right ventricular stroke work index; SVR: systemic vascular resistance; TPG: transpulmonary gradient; WU: wood units; PAH: pulmonary arterial hypertension; CTEPH: chronic thromboembolic pulmonary hypertension; HFrEF(PH): pulmonary hypertension due to left heart failure with reduced ejection fraction; HFpEF(PH): pulmonary hypertension due to left heart failure with preserved ejection fraction; HF(no PH): heart failure without pulmonary hypertension; SvO2: mixed venous oxygen saturation.
Plasma ADM in PAH differentiation and disease severity
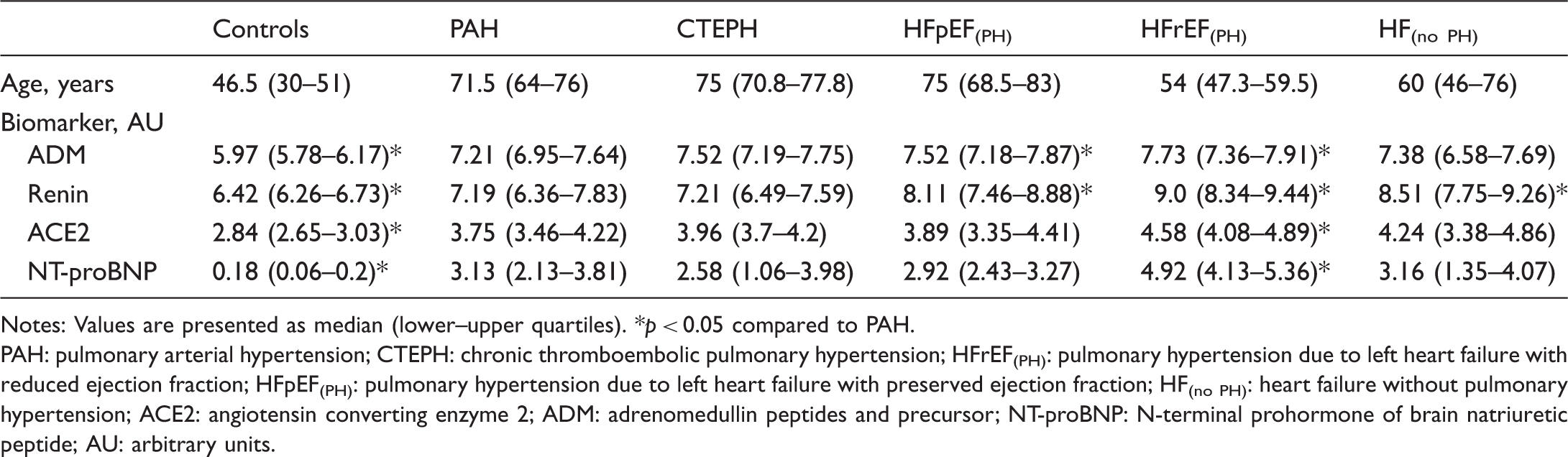
In PAH patients at diagnosis, plasma ADM levels were higher compared to healthy controls (p < 0.001), similar as in CTEPH and HF(no PH) (p = ns), but lower compared to HFpEF(PH) (p < 0.03) and HFrEF(PH) (p < 0.001) (Table 2 and Fig. 1). There was no difference in baseline ADM plasma levels between IPAH/FPAH and CTD-PAH (p = ns).
Plasma adrenomedullin peptides and precursor (ADM) (a), renin (b), and angiotensin converting enzyme 2 (ACE2) (c) levels across investigated study groups. Plasma biomarker levels at baseline. Notes: Values are presented as median (lower–upper quartiles). *p < 0.05 compared to PAH. PAH: pulmonary arterial hypertension; CTEPH: chronic thromboembolic pulmonary hypertension; HFrEF(PH): pulmonary hypertension due to left heart failure with reduced ejection fraction; HFpEF(PH): pulmonary hypertension due to left heart failure with preserved ejection fraction; HF(no PH): heart failure without pulmonary hypertension; ACE2: angiotensin converting enzyme 2; ADM: adrenomedullin peptides and precursor; NT-proBNP: N-terminal prohormone of brain natriuretic peptide; AU: arbitrary units.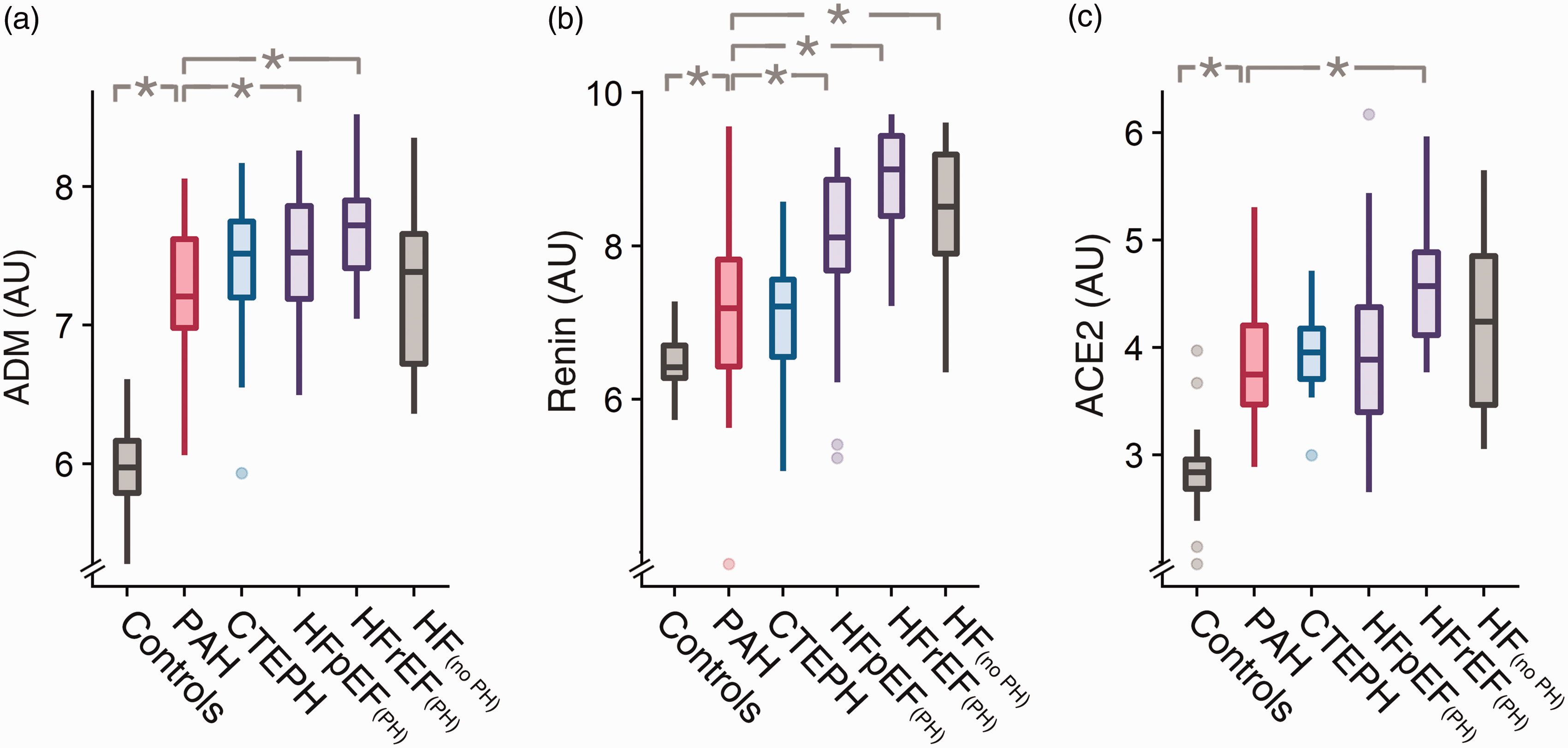
Baseline plasma ADM in patients with PAH correlated to MRAP (r = 0.73, p < 0.001), NT-proBNP (r = 0.75, p < 0.001), 6MWD (r = –0.57, p < 0.001), SvO2 (r = –0.57, p < 0.001), CI (r = –0.43, p < 0.003), and PVR (r = 0.33, p < 0.03). Since ADM, among the investigated biomarkers, showed the strongest correlations to the studied clinical parameters, it was chosen for further analyses.
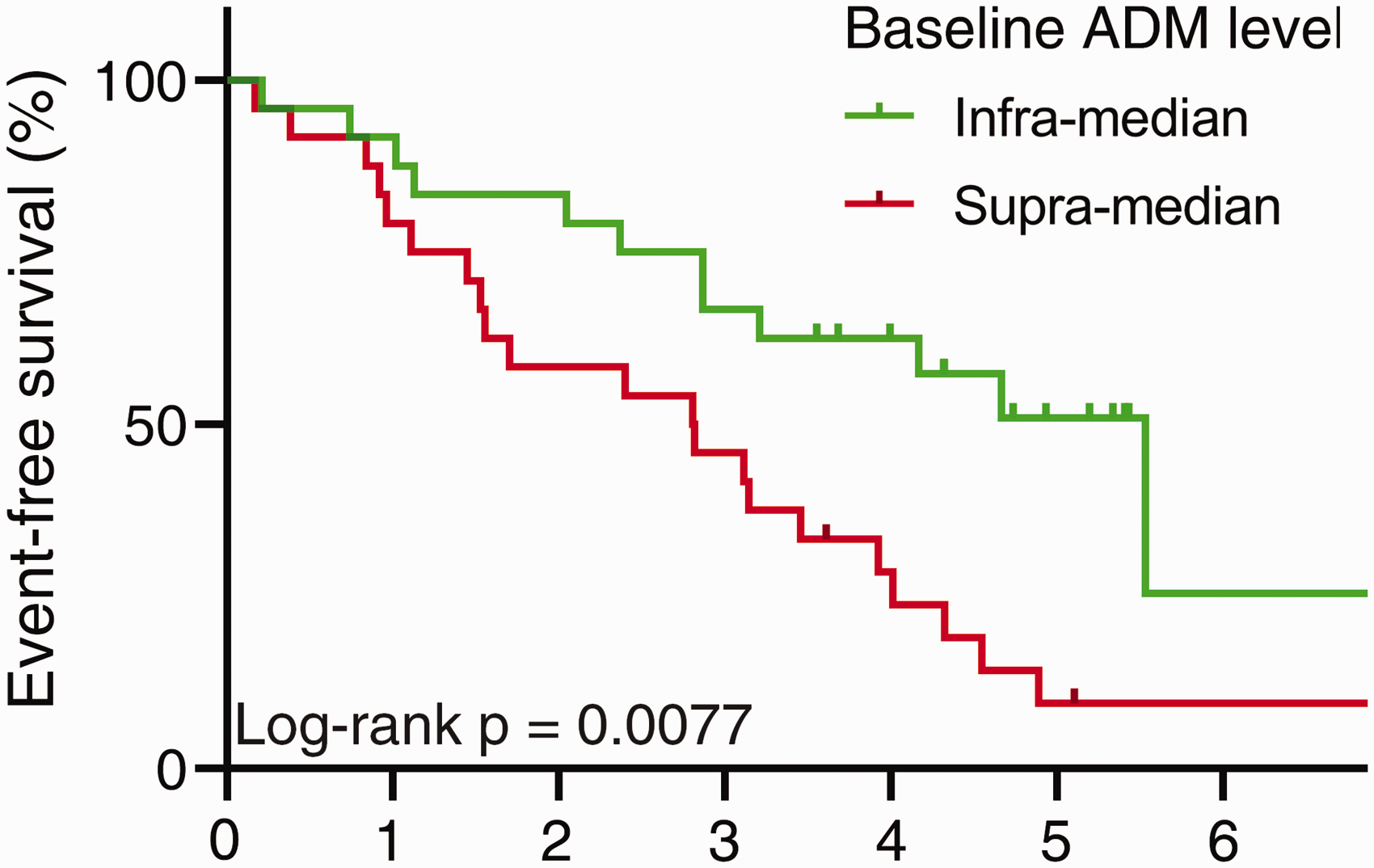
In PAH patients at diagnosis, plasma ADM levels correlated to the ECS/ERS- (r = 0.74, p < 0.001) and REVEAL risk scores (r = 0.54, p < 0.001) (Fig. 2). PAH patients with supra-median plasma ADM levels at diagnosis showed worse overall event-free survival than those with infra-median levels (median survival 34 versus 66 months, p = 0.0077) (Fig. 3). However, ADM plasma levels did not differentiate PAH patients that died within a year from diagnosis (7 of 48) and those that survived past one year (AUC = 0.6, p = ns). No patients were lung transplanted within one year from diagnosis. Also, patients that died or received a lung transplant (18 and 3 patients, respectively) within three years from diagnosis did not show different ADM levels compared to those that survived past three years (AUC = 0.58, p = ns).
In pulmonary arterial hypertension patients, plasma adrenomedullin peptides and precursor (ADM) correlated to mean right atrial pressure (MRAP) and N-terminal prohormone of brain natriuretic peptide (NT-proBNP) (a) as well as ESC/ERS guideline- (b) and REVEAL risk scores (c). Pulmonary arterial hypertension (PAH) patients with supra-median adrenomedullin peptides and precursor (ADM) levels at diagnosis exhibit worse overall event-free survival than those with infra-median levels.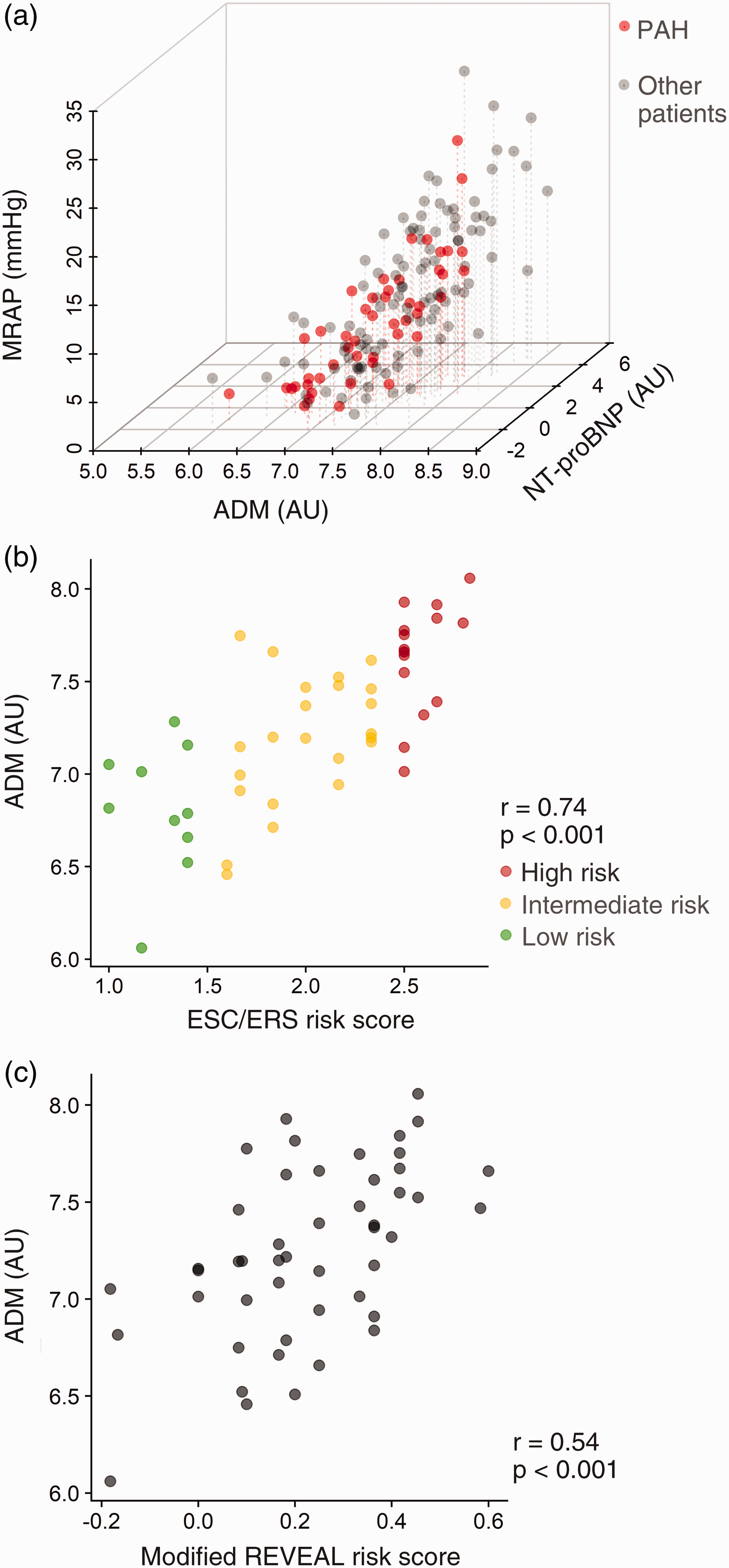
Plasma ADM levels in PAH patients were unaltered at early treatment follow-up compared to baseline (7.2 (6.99–7.63) versus 7.24 (6.98–7.48) AU) (p = ns). PAH patients with supra-median (and median) baseline ADM plasma levels showed better treatment response in MRAP (–1.5 (−3 to –1) versus 2 (–1 to 5) change in mmHg, p < 0.02) and 6MWD (84 (19 to 97) versus 3 (–20 to 70) change in meters, p < 0.02) compared to those with infra-median levels.
Renin plasma levels are lower in PAH compared to left heart failure
Plasma renin levels in PAH patients at diagnosis were higher compared to controls (p < 0.03), not different to the levels in CTEPH (p = ns), but lower compared to HFpEF(PH) (p < 0.003), HFrEF(PH) (p < 0.001), and HF(no PH) (p < 0.001). In PAH patients at diagnosis, ACE2 plasma levels were higher compared to controls, lower compared to HFrEF(PH) (p < 0.001), but not different from HFpEF(PH), HF(no PH), or CTEPH (p = ns) (Table 2 and Fig. 1). Baseline plasma ACE2 correlated to MRAP (r = 0.49, p < 0.001), NT-proBNP (r = 0.61, p < 0.001), 6MWD (r = –0.5, p < 0.001), and SvO2 (r = –0.36, p < 0.02) in PAH. Baseline plasma renin did, however, not correlate to any of the studied clinical parameters in PAH.
Baseline plasma renin and ACE2 levels showed no difference between IPAH/FPAH and CTD-PAH (p = ns). Renin did not change at early follow-up after treatment initiation in PAH patients (7.14 (6.26–7.75) versus 7.17 (6.66–7.9) AU) (p = ns). Likewise, ACE2 was unaltered between baseline and early treatment follow-up (3.79 (3.5–4.37) versus 3.76 (3.28–4.19) AU) (p = ns).
Discussion
The present study investigated plasma biomarkers connected to altered vasoregulation in relation to PAH hemodynamics, risk stratification and treatment response, as well as differentiation from CTEPH and left heart failure with or without PH. The main results showed that plasma ADM levels were elevated in the present PAH cohort compared to healthy controls, and were correlated to key clinical parameters, including MRAP, 6MWD, and NT-proBNP. Furthermore, plasma ADM levels correlated to ESC/ERS- as well as REVEAL risk scores, and may reflect overall survival.
Adrenomedullin has previously been reported to be overexpressed in PH. 7 Plasma adrenomedullin levels have been shown to correlate to hemodynamics, most notably MRAP, in a pooled group of PAH and CTEPH patients. 7 This is in accordance with our present findings. In a sample encompassing both PAH and CTEPH patients, Kolditz et al. showed that plasma MR-proADM correlated to exercise capacity (6MWD and peak oxygen consumption) and was also related to one-year mortality, which remained valid when investigating each subgroup alone. 19
In the present study, PAH patients with supra-median baseline ADM plasma levels showed worse overall event-free survival than those with infra-median levels, where an event was defined as death or lung transplantation. Baseline plasma ADM levels did, however, not differ between PAH patients that died within one year from diagnosis and those that survived past one year. This observation differs from that observed in the study from Kolditz et al., who, however, mainly studied prevalent cases along with a few incident patients. 19 In the present study, only incident treatment-naïve PAH patients were included. Assessing plasma adrenomedullin peptides other than MR-proADM in the present study may also explain the difference in the findings between the two studies. This observation can, however, also be related to our relatively small sample (7 of 48 died within a year).
In the present study, we did not observe a difference in plasma ADM levels between diagnosis and early treatment follow-up. This may be due to the short follow-up time in the present analyses. However, it may also be related to the progressive nature of the PAH disease. Also, we observed that PAH patients with supra-median baseline ADM levels actually showed better treatment response (between baseline and follow-up) in MRAP and 6MWD compared to those with infra-median levels. Although this might be attributed to chance and initially seem counter-intuitive, it might reflect that the patients with a higher plasma ADM at baseline may have a larger capacity to respond in MRAP and 6MWD, since they may have a more severe disease, and start at higher MRAP levels and lower 6MWD values.
When observing experimental studies, adrenomedullin has been suggested to inhibit proliferation in pulmonary artery smooth muscle cells and to alleviate pulmonary artery collagen accumulation in PH.13–15 Many reports have also shown that adrenomedullin administration attenuates PH in animal models, and that adrenomedullin may improve hemodynamics in human PH.9–13
In the present study, we also report that plasma renin protein levels in PAH are elevated compared to controls and unaltered versus CTEPH patients, but lower compared to left heart failure regardless of PH presence. This may merely mirror the real-life situation where patients with left heart disease receive more of medications that might affect the renin–angiotensin–aldosterone system (RAAS) than patients with PAH or CTEPH. 22 An increased RAAS response is present in patients with PAH. 23 Pulmonary arteries and endothelial cells from PAH patients have been shown to have increased angiotensin II receptor type 1 (AT1 receptor) expression and increased angiotensin II production, respectively. 23 Also, angiotensin II promotes proliferation in pulmonary artery smooth muscle cells via AT1 receptor signaling in PAH patients. 23 In accordance to these findings, PAH patients show increased renin activity and angiotensin II levels in blood samples, 23 which, moreover, are associated with an increased risk of death or lung transplantation. 23 We, however, found no associations between plasma renin protein levels and hemodynamics in our cohort of PAH patients.
Angiotensin II can, through ACE2, also be cleaved into angiotensin (1–7), which exerts vasodilatory effects. 24 In line with its physiological role, ACE2 activation has been suggested to improve PH in the monocrotaline model. 25 Circulating soluble ACE2 activity levels increase along with increased disease severity in heart failure patients, 26 likely due to a counter-mechanism to balance the effects of angiotensin II. In the present PAH cohort, we observed increased plasma ACE2 levels compared to healthy controls, which furthermore correlated to MRAP, NT-proBNP, and 6MWD.
When interpreting the present results, some limitations should be kept in mind. The PAH patients were older than controls and what is considered as the typical age for PAH, but had a similar age to the PAH patients in SPAHR, who have a median age of 67 years. 27 The patient and the control groups were also relatively small. Nonetheless, the present study only included PAH and CTEPH patients at baseline who had not received pulmonary vasodilatory treatment before diagnosis, resulting in fewer patients for analyses. For PAH patients, the time period from diagnosis to follow-up was short, which might not have been enough time to observe potential changes in plasma ADM levels.
Conclusion
Plasma ADM levels in PAH patients at diagnosis are elevated compared to healthy controls, similar to the levels in CTEPH, but lower than in PH due to left heart failure. Plasma ADM levels at PAH diagnosis correlated to hemodynamics, 6MWD, and NT-proBNP, as well as to the ECS/ERS- and the REVEAL risk scores. Plasma ADM levels were unaltered between baseline and an early treatment follow-up, which may reflect the progressive nature of PAH. The present results, together with previous evidence, suggest a relationship between plasma ADM and PAH disease severity, patient risk scores, and prognosis.
Footnotes
Acknowledgments
We acknowledge the support of the staff at the Hemodynamic lab, The section for heart failure and valvular disease, Skåne University Hospital, and at the Department of Cardiology, Clinical Sciences Lund, Lund University, Lund, Sweden. We specifically thank Anneli Ahlqvist for the support in assembling plasma samples for LCPR. We acknowledge the biobank services and retrieval of blood samples from LCPR performed at Labmedicin Skåne, University and Regional Laboratories, Region Skåne, Sweden.
Contributorship
HB and GR contributed to the concept and design of the work, and/or acquisition, analysis, and interpretation of data. HB drafted the article. HB and GR revised the manuscript critically for intellectual content and approved the version to be published.
Conflict of interest
Mr Bouzina reports personal lecture fees from Actelion Pharmaceuticals Sweden AB. Mr Bouzina reports unrestricted research grants from the Swedish Society of Pulmonary Hypertension on behalf of GlaxoSmithKline. Dr Rådegran reports personal lecture fees from Actelion Pharmaceuticals Sweden AB, GlaxoSmithKline, Bayer HealthCare AB, NordicInfu Care and Sandoz/Novatris, as well as unrestricted research grants from Actelion Pharmaceuticals Sweden AB and GlaxoSmithKline. Dr. Rådegran has been primary or co-investigator for PAH trials for Actelion Pharmaceuticals AB, GlaxoSmithKline, Pfizer, Bayer HealthCare AB, and United Therapeutics.
Funding
The present work was supported by an unrestricted research grant from ALF and the Swedish Society of Pulmonary Hypertension. The foundation had no role in analysis, interpretation, or publication of the manuscript.
