Abstract
Objective:
This study evaluated the prognostic value of the multivariable risk assessment for systemic lupus erythematosus (SLE)-associated pulmonary arterial hypertension (PAH).
Methods:
A multicenter prospective cohort of SLE-associated PAH (CSTAR-PAH cohort) diagnosed based on right heart catheterization (RHC) was established. Baseline and follow-up records were collected. Three methods of risk assessment, including (1) the number of low-risk criteria, based on World Health Organization functional class (WHO FC), 6-min walking distance (6MWD), right atrial pressure (RAP), and cardiac index (CI); (2) the three-strata stratification based on the average risk score of four variables (WHO FC, 6MWD, RAP, and CI); and (3) the four-strata stratification based on COMPARE 2.0 model were applied. A risk-assessment method using three noninvasive low-risk criteria was applied at the first follow-up visit. Survival curves between patients with different risk groups were compared by Kaplan–Meier’s estimation and log-rank test.
Results:
Three-hundred and ten patients were enrolled from 14 PAH centers. All methods of stratification at baseline and first follow-up significantly discriminated long-term survival. Survival rates were also significantly different based on the noninvasive risk assessment in first follow-up visit. Survival deteriorated with the escalation of risk from baseline to first follow-up. Patients with baseline serositis had a higher rate of risk improvement in their follow-up.
Conclusion:
The risk assessment has a significant prognostic value at both the baseline and first follow-up assessment of SLE-associated PAH. A noninvasive risk assessment can also be useful when RHC is not available during follow-up. Baseline serositis may be a predictor of good treatment response in patients with SLE-associated PAH.
Keywords
Introduction
Pulmonary arterial hypertension (PAH) is one of the most serious consequences and the third highest cause of death in patients with systemic lupus erythematosus (SLE). 1 Due to the high prevalence of SLE in Asian nations, it has surpassed primary Sjogren’s syndrome (pSS) and systemic sclerosis (SSc) as the predominant cause of connective tissue disease (CTD)-associated PAH.2–5 We recently published a study using a right heart catheterization (RHC)-based multicenter cohort to show the prognostic value of clinical and hemodynamic variables, including World Health Organization functional class (WHC FC), 6-min walking distance (6MWD), N-terminal pro-brain natriuretic peptide (NT-proBNP), and cardiac index (CI). 6 However, risk assessment and its predictive relevance in patients with SLE-associated PAH have not been thoroughly investigated.
The European Society of Cardiology (ESC) and the European Respiratory Society (ERS) published pulmonary hypertension guidelines in 2015 that recommended a multidimensional stratification system to assess the mortality risk in patients with PAH, and the prognostic value of risk assessment at baseline and the first follow-up has been validated in PAH cohorts. 7 The Swedish PAH Registry investigated the discriminatory capacity of the average risk score that included WHO FC, 6MWD, NT-proBNP, right atrial area, mean RAP, pericardial effusion, CI, and SvO2. 8 COMPERA (Comparative, Prospective Registry of Newly Initiated Therapies for Pulmonary Hypertension) also found that an abbreviated version of the risk assessment, which included WHO FC, 6MWD, BNP or NT-proBNP, RAP, CI, and mixed venous oxygen saturation, had a predictive role. 9 Recently, a refined risk stratification model (COMPARE 2.0) using a four-strata method showed more sensitive to the original three-strata model. 10 The French Pulmonary Hypertension Network (FPHN) registry revealed that the number of four low-risk criteria (WHC FC I or II, 6MWD > 440 m, RAP < 8 mmHg, CI ⩾ 2.5 L min−1 m−2) could be utilized as a baseline risk assessment, and the follow-up score was likewise linked to long-term survival. 11 Among patients with CTD-associated PAH, the first validation of this new risk stratification strategy based on four parameters was in SSc-associated PAH. 12 In addition, the average score based on the WHO FC, 6MWD, RAP, and CI according to guideline cut-offs has been investigated in SSc-PAH. 13 However, neither of these approaches has been tested in SLE-associated PAH.
Unlike SSc, inflammation is a major factor in SLE-associated PAH. Patients with SLE-associated PAH may benefit from extra immunosuppressive medications. 14 We also showed that patients with a higher inflammatory background had a better treatment response to immunosuppressants.6,15 In SLE, serositis has been identified as an inflammation indication. 16 SLE-associated PAH patients with baseline serositis benefit from intensive immunosuppressive medication, according to our findings. 6 The significance of serositis as an indicator of inflammation in the risk assessment of SLE-associated PAH needs to be investigated. Thus, the aim of this study was to evaluate the prognostic value of the multivariable PAH risk-assessment system in SLE-associated PAH and to explore the role of inflammation in the risk assessment.
Methods
CSTAR-PAH cohort
The Chinese SLE Treatment and Research Group (CSTAR) registry is a national registry that includes 104 rheumatology centers from 30 provinces in China. Patients with SLE-associated PAH visited the 14 qualified CTD-associated PAH referral centers (Supplementary) from November 2006 to May 2016 and met the following criteria were enrolled. The eligible criteria included the diagnosis of SLE confirmed by the 2012 Systemic Lupus International Collaborating Clinics classification criteria and the diagnosis of PAH based on RHC defined as the mean pulmonary arterial pressure (mPAP) ⩾ 25 mmHg at rest, pulmonary arterial wedge pressure (PAWP) ⩽15 mmHg, and pulmonary vascular resistance (PVR) >3 Wood units. Patients with other types of pulmonary hypertension revealed by a pulmonary function test showing total lung capacity < 60% and ventilation perfusion scintigraphy or computed tomographic pulmonary angiography showing pulmonary thromboembolism were excluded. Patients with overlapping CTD, such as SSc, or other CTDs were also excluded. The application of PAH guidelines and a census-based SLE-associated PAH treatment regimen were also required as inclusion criteria. Rheumatologists validated the diagnosis of SLE according to 2012 Systemic Lupus International Collaborating Clinics classification criteria. The Peking Union Medical College Hospital Institutional Review Board and Ethical Board approved this study (ethical number JS-2038), and each patient gave written informed permission.
Data collection, follow-up, and risk assessment
The study baseline was defined as the time of enrollment, when we gathered demographic information, medical history, physical examination, laboratory evaluations, transthoracic echocardiography (TTE) and RHC parameters were collected as we described previously. 6 Every patient had a full follow-up evaluation that was recorded and reported on at least once a year. WHO FC, 6MWD, serum BNP, serum NT-proBNP, and TTE were among the tests used. Treatment plans were also kept track of. The database also kept track of death, causes of death, hospitalization owing to disease deterioration, disease-related transplantation, and atrial septostomy.
Variables were assessed at the time of baseline and the first follow-up RHC. Three methods of risk assessment were applied at baseline and at the first follow-up visit. The three methods were as follows: (1) The number of the following low-risk criteria was calculated as 0, 1, 2, 3, or 4: WHO FC I or II, 6MWD > 440 m, RAP < 8 mmHg, and CI ⩾ 2.5 L min−1 m−2. (2) Three-strata stratification: the average risk score was calculated based on the following guideline cut-offs: WHO FC (1 if I–II, 2 if III, and 3 if IV), 6MWD (1 if > 440 m, 2 if 440–165 m, and 3 if < 165 m), RAP (1 if < 8 mmHg, 2 if 8–14 mmHg, and 3 if > 14 mmHg), and CI (1 if ⩾ 2.5 L min−1 m−2, 2 if 2.4–2.0 L min−1·m−2, and 3 if < 2 L min−1 m−2) and further stratified into three risk groups according to the score calculated by dividing the sum of all grades by the number of variables and rounding to the next integer (1 point = low-risk group, 2 points = intermediate-risk group, or 3 points = high-risk group). Only few patients were evaluated by RHC at their first follow-up visit due to socioeconomic reasons; therefore, a risk-assessment method using three noninvasive low-risk criteria (WHO FC I or II, 6MWD > 440 m, and BNP < 50 ng L−1 or NT-proBNP < 300 ng L−1) was applied at the first follow-up visit. (3) Four-strata stratification: the average risk score was calculated based on the following cut-offs: WHO FC (1 if I–II, 3 if III, and 4 if IV), 6MWD (1 if > 440 m, 2 if 440–320 m, 3 if 319–165 m, and 4 if < 165 m), BNP (1 if < 50 ng L−1, 2 if 50–199 ng L−1, 3 if 200–800 ng L−1, and 4 if > 800 ng L−1), or NT-proBNP (1 if < 300 ng L−1, 2 if 300–649 ng L−1, 3 if 650–1100 ng L−1, and 4 if >1100 ng L−1) and further stratified into four risk groups according to the score calculated by dividing the sum of all grades by the number of variables and rounding to the next integer (1 point = low-risk group, 2 points = intermediate-low-risk group, 3 points = intermediate high-risk group; 4 points = high-risk group). The end point was death from any cause.
Statistical analyses
The means and standard deviations are used to describe quantitative data. Counts and percentages are used to describe nonquantitative data. The Kaplan–Meier estimation was used to compute cumulative survival probabilities. A log-rank test was used to make comparisons between different risk groups. In the survival analysis, all-cause mortality was employed. From the date of the initial diagnostic RHC, the follow-up time was determined. The values of p < 0.05 were deemed significant. SPSS 19.0 was used to analyze the data (SPSS Inc., Chicago, IL, USA). Power Analysis and Sample Size (PASS) software version 15.0.5 was performed to calculate the power of each comparison (see Supplementary Table 1).
Results
Overall, 310 patients with SLE-associated PAH were enrolled in this CSTAR-PAH cohort. Baseline characteristics are shown in Table 1. The majority of patients were female (99.4%), with a mean age at recruitment of 35.0 years. A total of 44.6% and 3.7% of patients were in WHO FC III and IV, respectively, at baseline. The mPAP was 46.5 ± 12.1 mmHg, and CI was 2.8 ± 0.9 L min−1 m−2. A total of 99.4% and 92.6% patients were treated with glucocorticoids and immunosuppressants, respectively. A total of 69.4% patients received initial PAH target medications, and among them, 19.5% received combination therapy.
Baseline characteristics of the CSTAR-PAH cohort.
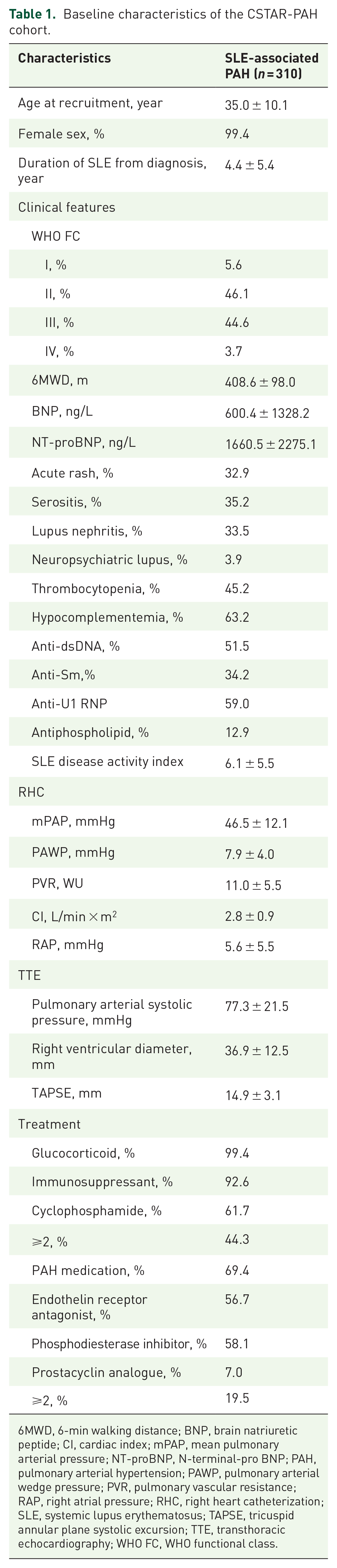
6MWD, 6-min walking distance; BNP, brain natriuretic peptide; CI, cardiac index; mPAP, mean pulmonary arterial pressure; NT-proBNP, N-terminal-pro BNP; PAH, pulmonary arterial hypertension; PAWP, pulmonary arterial wedge pressure; PVR, pulmonary vascular resistance; RAP, right atrial pressure; RHC, right heart catheterization; SLE, systemic lupus erythematosus; TAPSE, tricuspid annular plane systolic excursion; TTE, transthoracic echocardiography; WHO FC, WHO functional class.
Among the 310 recruited patients, five patients were lost to follow-up and excluded with an unconfirmed mortality status. A total of 305 patients were included in the survival analysis (Supplementary Figure 1). The median follow-up was 59.2 months. A total of 67 (22.0%) deaths occurred, and there was no PAH-related transplantation or atrial septostomy. The 1-, 3-, and 5-year survival rates for patients with SLE-associated PAH were 92.1%, 84.9%, and 79.8%, respectively (Supplementary Figure 2). The mean survival time was 9.45 years.
Among the 305 patients with SLE-associated PAH at baseline, 20.0%, 24.6%, 26.2%, 20.3%, and 8.9% of patients had none, one, two, three, and four low-risk criteria, respectively. The 1-year survival rates of patients with 0, 1, 2, 3, and 4 low-risk criteria were 83.6%, 88.0%, 95.0%, 98.4%, and 100%, respectively. The 3-year survival rates of patients with 0, 1, 2, 3, and 4 low-risk criteria were 70.5%, 77.3%, 92.5%, 93.5%, and 96.3%, respectively. The 5-year survival rates of patients with 0, 1, 2, 3, and 4 low-risk criteria were 59.2%, 69.7%, 90.9% 93.5%, and 92.4%, respectively. The survival rates between groups with different numbers of low-risk criteria were significantly different (log-rank test, p < 0.001) and are shown in Figure 1(a) and Table 2.

Survival according to the number of low-risk criteria: (a) at baseline and (b) at first follow-up visit.
Parameters in risk assessment and survival according to the three methods.

6MWD, 6-min walking distance; BNP, brain natriuretic peptide; CI, cardiac index; NT-proBNP, N-terminal proBNP; RAP, right atrial pressure; WHO FC, WHO functional class.
When categorized according to the three-strata stratification at baseline, 42.0%, 51.8%, and 6.2% patients were in the low-risk, intermediate-risk, and high-risk groups, respectively. The 1-year survival rates of patients in the low-, intermediate-, and high- groups were 98.4%, 88.6%, and 78.9%, respectively. The 3-year survival rates of patients in the low-, intermediate-, and high-risk groups were 93.8%, 79.1%, and 73.7%, respectively. The 5-year survival rates of patients in the low-, intermediate-, and high-risk groups were 92.9%, 73.0%, and 47.5%, respectively. The survival rates between different risk groups at baseline were significantly different (log-rank test, p < 0.001) and are shown in Figure 2(a) and Table 2.

Survival according to the three-strata stratification (a) at baseline and (b) at first follow-up visit.
When categorized according to the four-strata stratification at baseline, 15.3%, 33.9%, 35.9%, and 14.9% patients were in the low-, intermediate-low-, intermediate-high-, and high-risk groups, respectively. The 1-year survival rates of patients in the low-, intermediate-low-, intermediate-high-, and high-risk groups were 100%, 96.0%, 88.7%, and 81.8%, respectively. The 3-year survival rates of patients in the low-, intermediate-low-, intermediate-high-, and high-risk groups were 93.3%, 91.0%, 83.1%, and 63.6%, respectively. The 5-year survival rates of patients in the low-, intermediate-low-risk, intermediate-high-, and high-risk groups were 93.3%, 86.1%, 75.4%, and 58.4%, respectively. The survival rates between different risk groups at baseline were significantly different (log-rank test, p < 0.001) and are shown in Figure 3(a) and Table 2.

Survival according to the four-strata stratification (a) at baseline and (b) at first follow-up visit.
Twenty-eight patients died before the first follow-up visit, and 32 patients were excluded without follow-up evaluations. A total of 245 patients were included in the follow-up risk-assessment study (Supplementary Figure 2). The baseline characteristics between the overall study population and the study population included in the follow-up risk assessment were compared (Supplementary Table 2). The median interval between baseline and the first follow-up visit was 9.4 months. Since only 18 patients were evaluated by RHC at their first follow-up visit, three noninvasive parameters, including WHO FC, 6WMD, and BNP/NT-proBNP, were applied for the risk assessment. At the first follow-up visit, 18.1%, 27.2%, 25.1%, and 29.6% of patients had 0, 1, 2, and 3 noninvasive low-risk criteria, respectively. The survival rates between different risk groups at the first follow-up visit were significantly different (log-rank test, p < 0.001) and are shown in Figure 1(b).
When categorized according to the three-strata stratification at the first follow-up visit, 228 patients with at least two parameters (mainly 6MWD and WHO FC) were included. A total of 39.5% of patients were stable in the low-risk group, 28.1% of patients improved into the lower risk group, 21.9% of patients were stable in the medium-risk group, 8.8% of patients worsened into the higher risk group, and 1.8% of patients remained in the high-risk group. There were significant differences between each group, and the survival deteriorated after the escalation of risk group [Figure 2(b)].
When categorized according to the four-strata stratification at the first follow-up visit. A total of 43.2%, 26.7%, 27.5%, and 2.6% patients were in the low-, intermediate-low-, intermediate-high-, and high-risk groups, respectively. The 1-year survival rates of patients in the low-, intermediate-low-, intermediate-high-, and high-risk groups were 98.3%, 98.6%, 80.0%, and 57.1%, respectively. The 3-year survival rates of patients in the low-, intermediate-low-, intermediate-high-, and high-risk groups were 94.0%, 95.8%, 68.2%, and 42.9%, respectively. The 5-year survival rates of patients in the low-, intermediate-low-, intermediate-high-, and high-risk groups were 90.1%, 87.0%, 58.8%, and 21.4%, respectively. The survival rates between different risk groups at baseline were significantly different (log-rank test, p < 0.001) and are shown in Figure 3(b) and Table 2.
To identify the relationship between SLE activity and risk group change, we compared the risk assessment change during baseline and the first follow-up visit in patients with or without baseline serositis. There was significantly more risk group improvement in patients with serositis at baseline than in those without baseline serositis (p = 0.008) (Figure 4). No significant difference was identified in parameters regarding the severity of PAH (data not shown).
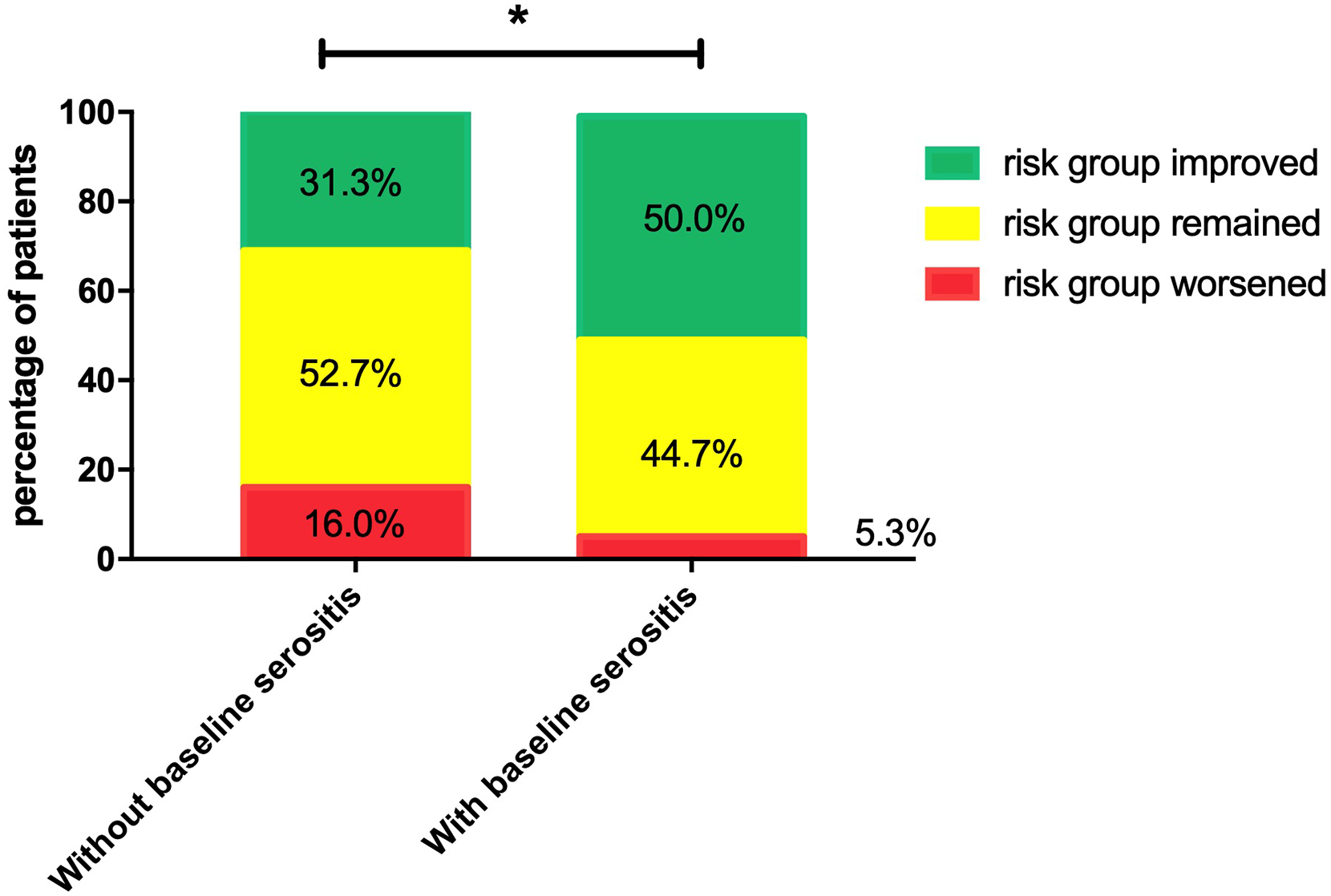
Risk group changes in patients with and without baseline serositis.
Discussion
This study validated the prognostic value of three methods of risk assessment using the number of low-risk criteria, the three- and four-strata average risk in patients with SLE-associated PAH at baseline and at first follow-up. We reported that the 1-year survival rates in patients with 0, 1, 2, 3, and 4 low-risk criteria at baseline were 83.6%, 88.0%, 95.0%, 98.4%, and 100%, respectively. The 1-year survival rates of patients in the low-, intermediate-, and high-risk groups at baseline were 98.4%, 88.6%, and 78.9%, respectively. Compared with other studies (Table 3), the 1-year survival rates are consistent and mostly within the predicted range. However, the 1-year survival of patients with intermediate risk is slightly lower than the predicted survival (95–90%) according to the ESC/ERS guidelines, indicating that the predicted survival may be different based on different underlying causes of PAH and may need to be adjusted in future studies.
Comparison of the 1-year survival in different risk groups between studies.
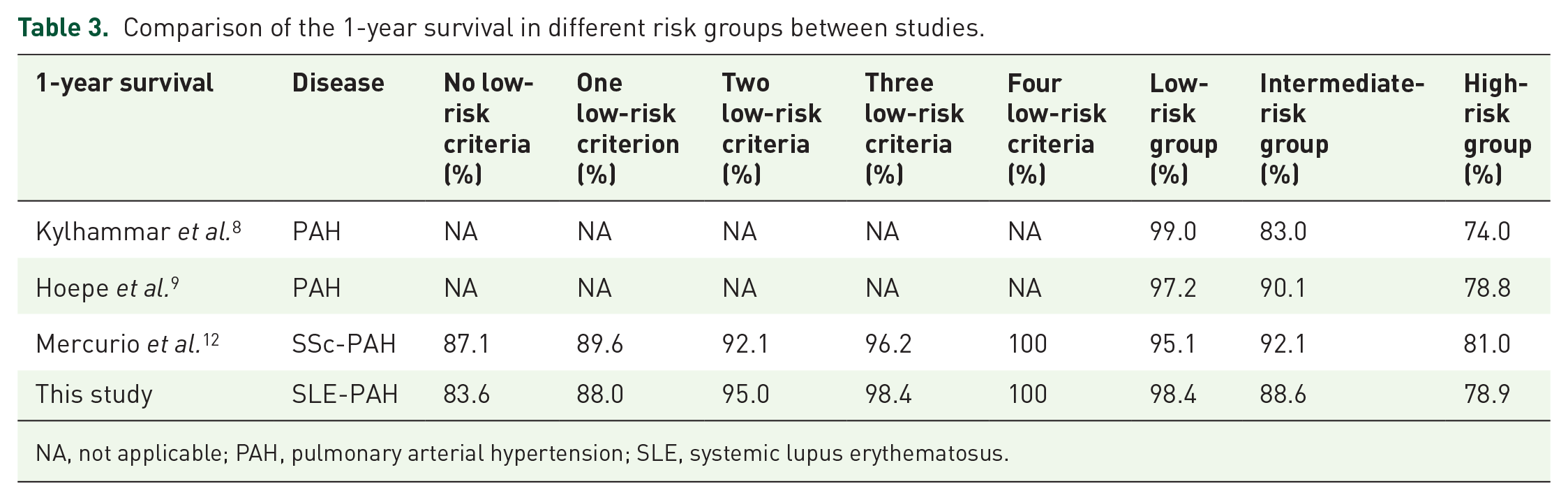
NA, not applicable; PAH, pulmonary arterial hypertension; SLE, systemic lupus erythematosus.
This study also showed that survival of SLE-associated PAH patients differed significantly among risk strata at baseline and at first follow-up assessment, regardless of the method of stratification. Moreover, improvements in risk category from baseline to first follow-up assessment may be of more value for prognostic purposes. In our study, patients who remained in the high-risk group had the worst survival. Those who remained in the low-risk group had the best survival. Patients with an improved risk category, that is, from high to intermediate risks, from high to low risks, or from intermediate to low risks, had a better survival than those who remained in the intermediate risk group, which shows that the dynamic improvement or deterioration after medication may have more value than the static risk state at baseline or follow-up assessment.
This study also showed the prognostic value of a noninvasive approach based on the WHO FC, 6MWD, BNP, or NT-proBNP. Ideally, hemodynamic assessment should be part of the follow-up assessment. However, due to reasons, including the invasive nature, cost, and availability, RHC was not routinely conducted for every patient. Here, we showed that the risk assessment based on the noninvasive low-risk criteria number significantly discriminated the survival of patients, indicating that the noninvasive approach to assessment can be acceptable when the hemodynamic assessment is not available. The fundamental role of RHC in the follow-up assessment has long been supported.17–19 Patients with clinical deterioration and poor treatment response may be benefit from prioritizing follow-up RHC to evaluate more precisely. Besides, the prognostic differences between thorough assessments, including RHC and noninvasive assessments, in patients with SLE-associated PAH need to be further studied.
Our study also showed the ability of the four-strata stratification to distinguish patients with intermediate-risk into intermediate-high- and intermediate-low-risk groups with different prognosis. Notably, the four-strata stratification during first follow-up showed patients with intermediate-low risk had relatively good survival which was similar to patients with low risk. Compared with the three-strata stratification, four-strata method of stratification further refined the risk-assessment tools and may help predict patients’ survival more preciously. Further studies are needed.
In contrast to SSc-associated PAH and idiopathic PAH, SLE-associated PAH has a unique inflammatory background. In our previous study, baseline serositis, as the indicator of SLE disease activity, was identified as one of the independent prognostic factors of ‘treatment goal achievement’. 6 Here, we showed a significant increase in risk group improvement in patients with serositis at baseline compared with those without baseline serositis, which was consistent with our previous study. We also previously showed that patients with baseline serositis (inflammation) may benefit from immunosuppressive therapy and achieve better clinical outcomes. 6 Here, a new question has been raised, as even in patients with baseline serositis, there were nearly 50% of patients without risk group improvement. Are there different subtypes of SLE-associated PAH affecting therapeutic response? Two distinct clinical phenotypes, vasculitis and vasculopathy, of SLE-associated PAH have been proposed. 20 However, the prognosis was reported differently in other studies. 15 The definition, treatment response, and prognosis of the subtypes of SLE-associated PAH need to be further investigated.
The study has several limitations. First, this was a registry-based study, and follow-up assessments were not standardized, including the interval between baseline and the first follow-up assessment and the availability of hemodynamic assessment at first follow-up. Thus, further studies are warranted. Second, there were missing follow-up data, especially hemodynamic variables, which make the comparison between a thorough assessment and noninvasive assessment impossible. The low rate of follow-up RHC may have a potential impact in the risk stratification. Further study with complete follow-up RHC is needed. In addition, we reported that 69.4% of patients in our cohort initially received PAH target medications, and only 19.5% were treated with combination therapy at baseline due to socioeconomic reasons. Thus, the effects of different treatment regimens could not be avoided and need to be further studied. As a rare disease, our study is by far the largest cohort study of SLE-associated PAH based on RHC diagnosis. However, due to the relatively small sample size, the power may limit the significance in intra-group comparisons indicating that large cohort studies, including global-wide cohort study of SLE-associated PAH are called in the future.
Conclusion
In conclusion, this study is the first to validate the prognostic value of an abbreviated risk assessment based on three approaches according to the 2015 ESC/ERS guidelines in patients with SLE-associated PAH. The risk assessment has significant value in discriminating prognosis at both study baseline and the first follow-up assessment, and the improvement in risk category may be a more powerful prognostic tool. We also showed the prognostic value of the noninvasive approach during follow-up. A significant increase in risk group improvement in patients with serositis at baseline compared with those without baseline serositis suggests that SLE disease activity may affect the risk assessment due to differences in therapeutic responses. Further investigation into the combination of SLE disease activity and PAH assessment for patients with SLE-associated PAH is urgently needed.
Supplemental Material
sj-doc-1-taj-10.1177_20406223221112528 – Supplemental material for Risk assessment in systemic lupus erythematosus-associated pulmonary arterial hypertension: CSTAR-PAH cohort study
Supplemental material, sj-doc-1-taj-10.1177_20406223221112528 for Risk assessment in systemic lupus erythematosus-associated pulmonary arterial hypertension: CSTAR-PAH cohort study by Qian Wang, Junyan Qian, Mengtao Li, Xiao Zhang, Wei Wei, Xiaoxia Zuo, Ping Zhu, Shuang Ye, Wei Zhang, Yi Zheng, Wufang Qi, Yang Li, Zhuoli Zhang, Feng Ding, Jieruo Gu, Yi Liu, Can Huang, Jiuliang Zhao, Yongtai Liu, Zhuang Tian, Yanhong Wang, Miaojia Zhang and Xiaofeng Zeng in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
