Abstract
To describe the frequency with which pulmonary capillary wedge pressure measurements, obtained during right heart catheterization, are falsely elevated and to educate operators on techniques to improve accuracy of pulmonary capillary wedge pressure reporting. Failure to completely occlude pulmonary artery branch vessels during balloon inflation can lead to falsely elevated, “incomplete” pulmonary capillary wedge pressures. Balloon deflation prior to catheter retraction may result in catheter advancement into smaller branch vessels, yielding an inadvertent but more accurate alternative pulmonary capillary wedge pressure. We hypothesized that this phenomenon can be identified on retrospective review of right heart catheterization tracings, which occurs commonly and goes unrecognized by operators. We conducted a retrospective study of patients undergoing right heart catheterization or right heart catheterization and left heart catheterization with computer-generated pulmonary capillary wedge pressure ≥20 from January 2015 to June 2017. Alternative pulmonary capillary wedge pressures were defined as a pulmonary capillary wedge pressure trace during balloon deflation ≥3 mmHg lower than the reported pulmonary capillary wedge pressure. Inter-rater reliability of tracing reviewers was also evaluated. Results showed that, of the 182 tracings reviewed, an alternative pulmonary capillary wedge pressure was identified in 26 or 14.3% of cases. Eleven of these alternative pulmonary capillary wedge pressures were ≤15 mmHg with a calculated pulmonary vascular resistance ≥3 Wood units in 10 patients, re-classifying the etiology of pulmonary hypertension from post-capillary to pre-capillary in 38.5% of cases. For the eight patients for whom left heart catheterization data were available, left ventricular end-diastolic pressure aligned with the alternative pulmonary capillary wedge pressure. In conclusion, inadvertently obtained, but likely more accurate, alternative pulmonary capillary wedge pressures were identified in almost 15% of procedures reviewed from a busy academic institution. As wedge pressures often drive diagnosis and treatment decisions for patients with cardiac and pulmonary pathology, operators should be attuned to balloon deflation as a time when alternative pulmonary capillary wedge pressures may be identified as they are likely more reflective of left ventricular end-diastolic pressure. Additional tools to ensure accuracy of pulmonary capillary wedge pressure reporting are reviewed.
Introduction
Accurate pulmonary capillary wedge pressure (PCWP) measurements are critical in the evaluation of patients with pulmonary and cardiac pathology. Obtained during right heart catheterization (RHC), PCWPs are required to differentiate between patients with pre-capillary, group 1 pulmonary arterial hypertension (PAH), and post-capillary, group 2 pulmonary hypertension, due to left heart failure. As defined by the World Health Organization (WHO), both PCWP ≤ 15 mmHg and pulmonary vascular resistance (PVR) ≥ 3 Wood units are necessary to differentiate PAH from patients with group 2 disease. 1 This classification not only determines the therapeutic options available to patients, as most therapeutics are only approved for group 1 disease, but also determines feasibility of patient inclusion in clinical trials. Invasive monitoring with assessment of PCWP is also useful in patients with acute heart failure, particularly for those with refractory hypotension or in whom renal function worsens despite standard therapy. 2
It is estimated that at least 1–2 million RHCs are performed each year.3,4 Despite the facility of experienced operators at institutions specializing in the care of patients with pulmonary vascular disease, complications ranging from pneumothorax during vascular access to hypotension as a result of vasoreactivity testing to death as a result of pulmonary artery (PA) rupture do occur, albeit at low frequencies (1–5%). 5 Arguably most common, however, and of substantial consequence to patients, is the misinterpretation of RHC tracing data, specifically estimates of PCWP. 6 In fact, self-report of perceived inadequacies in ability to interpret RHC data are strikingly high with studies revealing surveys in which 50% of critical care trained attendings queried could not correctly identify PCWP from a clear chart recording. As a result, patients are misclassified within pulmonary hypertension groups. 7 In the critical care setting, some question whether the mortality associated with RHC utilization reflects data misinterpretation among critical care practitioners rather than lack of benefit from catheter use itself. 6 The provider impact on patient care between those who can safely insert a PA catheter versus those who then have the capacity to accurately interpret the resulting data cannot be overemphasized.
Variation in RHC tracing interpretation, particularly identification of PCWP, is in part due to differences in operator technique. The ability to interpret the data obtained during a RHC within the clinical context of the patient and the data expected a priori is critical (i.e. expectation of a low PCWP in a patient who has undergone diuresis and is now clinically euvolemic). If the data obtained diverges from that which is expected, can the operator troubleshoot and intuit where the discrepancy between the patient and the catheter data lies? One previously described pitfall which operators may confront is “partial wedging,” a phenomenon in which an incompletely occluded PA yields an overestimation of true PCWP due to reflection of both left atrial and PA pressures. 8 A study of patients with known pulmonary hypertension demonstrated a need for variable balloon volumes (often half normal) when measuring PA occlusion or wedge pressures, hypothesizing the impact of a proximally distorted pulmonary vascular bed preventing complete occlusion. 9
We hypothesized that experienced catheterization operators are at risk of reporting falsely elevated, “incomplete” wedge pressures as a result of failing to completely occlude PA branch vessels during balloon inflation. During balloon deflation, the catheter may then inadvertently and briefly move distally, fully occluding a PA branch leading to a more accurate and lower “alternative” wedge pressure. Drawing operator attention to the transition from PCWP to PA will increase scrutiny of this important procedural time point and therefore identification of lower and more accurate PCWPs. In addition to drawing attention to alternative PCWPs, educating clinicians on other techniques to ensure accurate reporting of pulmonary wedge pressures is critical to ensuring the appropriate use of RHCs in the diagnosis and management of patients with complex cardiopulmonary conditions. Thus, this paper serves as both a retrospective analysis, drawing attention to a common error during RHC, as well as an educational review of the steps providers should follow to ensure the accuracy of their PCWP reporting.
Methods
This retrospective cohort study queried the cardiac catheterization reporting database at the Massachusetts General Hospital (MGH) for all patients undergoing right and/or right and left heart catheterization (LHC) from January 2015 to June 2017. As a national referral center for patients with pulmonary hypertension, operators at MGH were experienced providers who, as a whole, do approximately 750 RHCs per year. The majority of RHCs in this cohort (85%) were performed by interventional or general cardiologists with experience performing diagnostic catheterizations, using a 7F Edwards Lifesciences catheter. The minority of the procedures were done by pulmonologists specializing in the care of patients with pulmonary vascular disease. Catheterization reports reviewed did not distinguish whether procedures were done by fellows or attendings, though attendings were present for all procedures.
Reports in which a computer-generated PCWP was greater than or equal to 20 mmHg were selected for tracing review. Given the delineation of group 1 versus group 2 pulmonary hypertension by WHO criteria of PCWP ≤ 15, patients with PCWP ≥ 20 were of interest as it was felt that those with an incomplete wedge of 20 would be more likely to be misclassified if the alternative PCWP was, in fact, lower. Our specific inclusion criteria of PCWP ≥20 mmHg, as compared to PCWP between 15–20 mmHg, ensured that reported PCWP actually supported a diagnosis of post-capillary PH, obviating the possibility of inappropriate computer or operator interpretation of respirophasic variation of PCWP. Thus, inclusion of those with PCWP ≥ 20 mmHg more assuredly identified patients in whom an alternative PCWP would actually change WHO group. In line with the hypothesis that balloon deflation prior to transition from PCWP to PA is a procedural time point during which operators should be attuned to the possibility of transiently identifying an alternate and true wedge, only reports in which a tracing of the transition from PCWP to PA was recorded were included for review. As such, 28 cases were excluded for lack of documented waveform transition from PCWP to PA. Selected tracings were then reviewed to determine whether the computer-generated PCWP matched that listed in the report. Nine tracings in which PCWP was edited by the operator to be < 20 mmHg were excluded. Of the tracings meeting inclusion criteria, 35 of the 182 PCWP reviewed remained above 20 but had been edited by the operator to at most 6 mmHg above and 9 mmHg below the computer-generated number. The revised, operator-determined value was used for inclusion and further analysis. Based on the described criteria, a total of 219 tracings were reviewed. Of the 182 tracings ultimately included, 95 were reviewed by an additional physician to obtain information on inter-rater reliability (Fig. 1).
Selection and review of right heart catheterization tracings.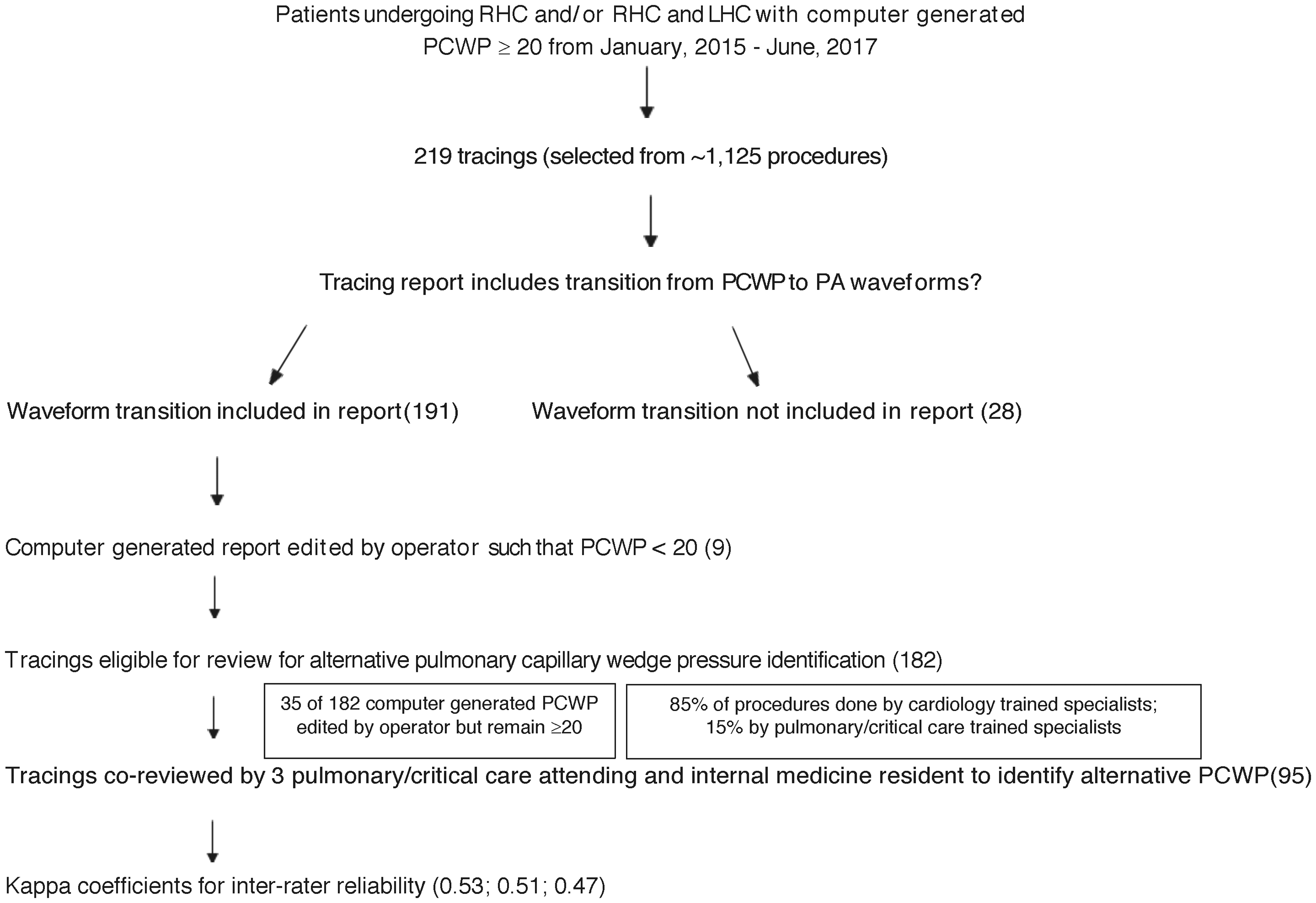
Within the group of 182 tracings meeting inclusion criteria, reviewers looked to identify an alternative PCWP, defined as a PCWP tracing at least 3 mmHg lower than the initial PCWP, during balloon deflation prior to transition from PCWP to PA. The alternative PCWP was identified at the lowest point in the tracing. Given the inadvertent nature of these alternative tracings (i.e. operators did not notice these alternative pressures), recordings were transient and not maintained over several cardiac or respiratory cycles. As a result, tracings could not be examined for respirophasic variation. However, in all cases, the identified alternative wedge was below the inspiratory variation of the reported PCWP.
Prevalence with which an alternative PCWP was incidentally captured during balloon deflation, prior to PCWP to PA transition, was then defined. The frequency with which the alternative PCWP re-defined patients' pulmonary hypertension group was also defined based on an alternative PCWP ≤ 15 as well as a calculated alternative PVR ≥ 3 Wood units. The alternative PVR was specifically calculated using the reported mean pulmonary artery pressure (mPAP) and cardiac output (CO) as determined by thermodilution as follows: (reported mPAP – alternative PCWP)/CO). For the single case in which thermodilution was not available, CO by Fick was used to calculate the alternative PVR. For cases in which an alternative PCWP was defined, concordance between reported PCWP and diastolic pulmonary artery pressure (DPAP) was examined as a DPAP lower than PCWP could be indicative of an incomplete PCWP. For cases in which left ventricular end-diastolic pressure (LVEDP) was available from LHC, computer-generated LVEDP was extracted for comparison to PCWP.
The MGH institutional review board approved review of prior RHC tracings without need for informed consent (protocol # 2017P001130).
Results
For all 26 cases in which an alternative PCWP was identified, an alternative PCWP ≤ 15 along with a calculated alternative PVR ≥ 3 resulted in reclassification of patients from a post-capillary to a pre-capillary pathology (highlighted in light gray).
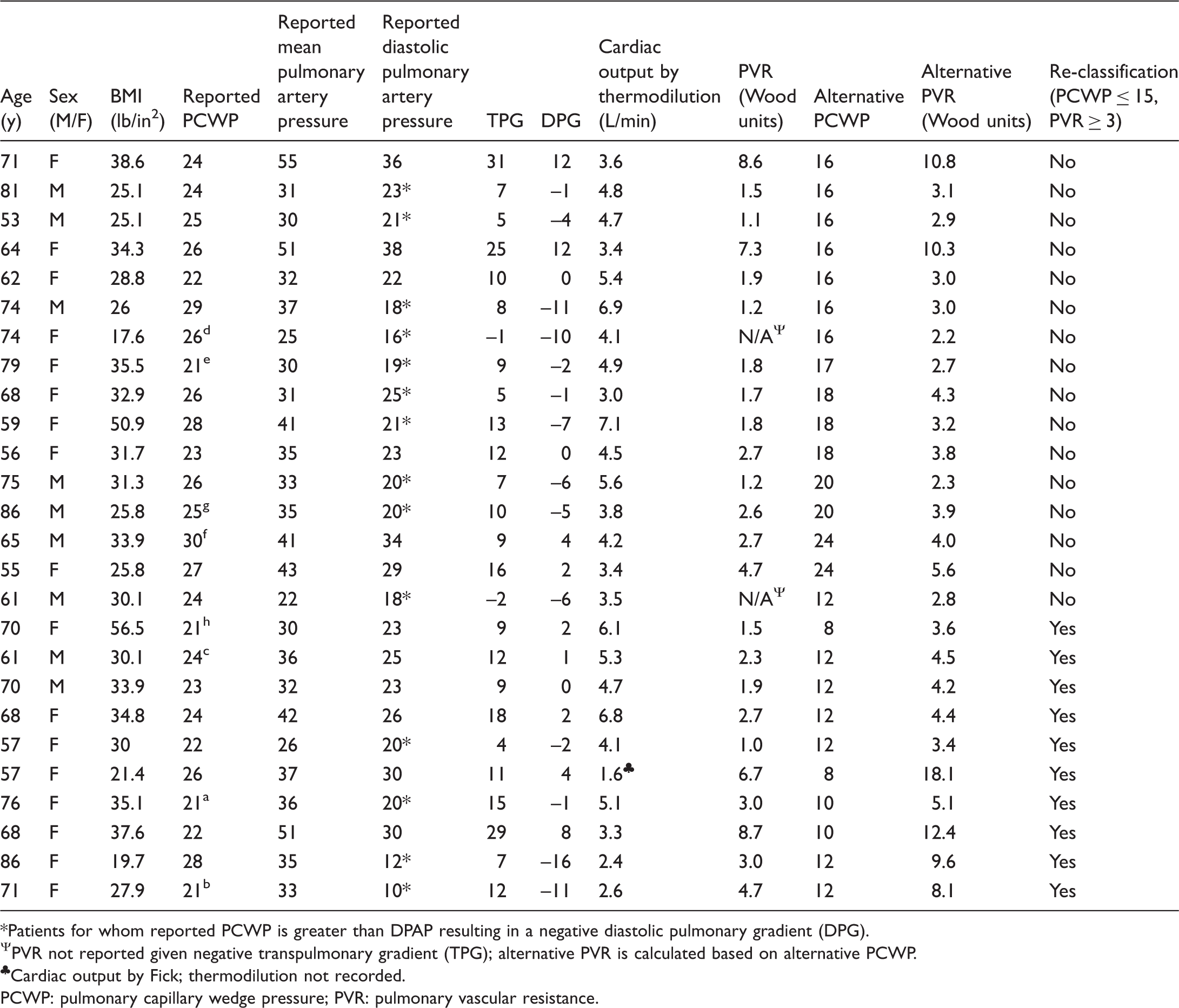
Patients for whom reported PCWP is greater than DPAP resulting in a negative diastolic pulmonary gradient (DPG).
PVR not reported given negative transpulmonary gradient (TPG); alternative PVR is calculated based on alternative PCWP.
Cardiac output by Fick; thermodilution not recorded.
PCWP: pulmonary capillary wedge pressure; PVR: pulmonary vascular resistance.
For cases in which LVEDP was available (
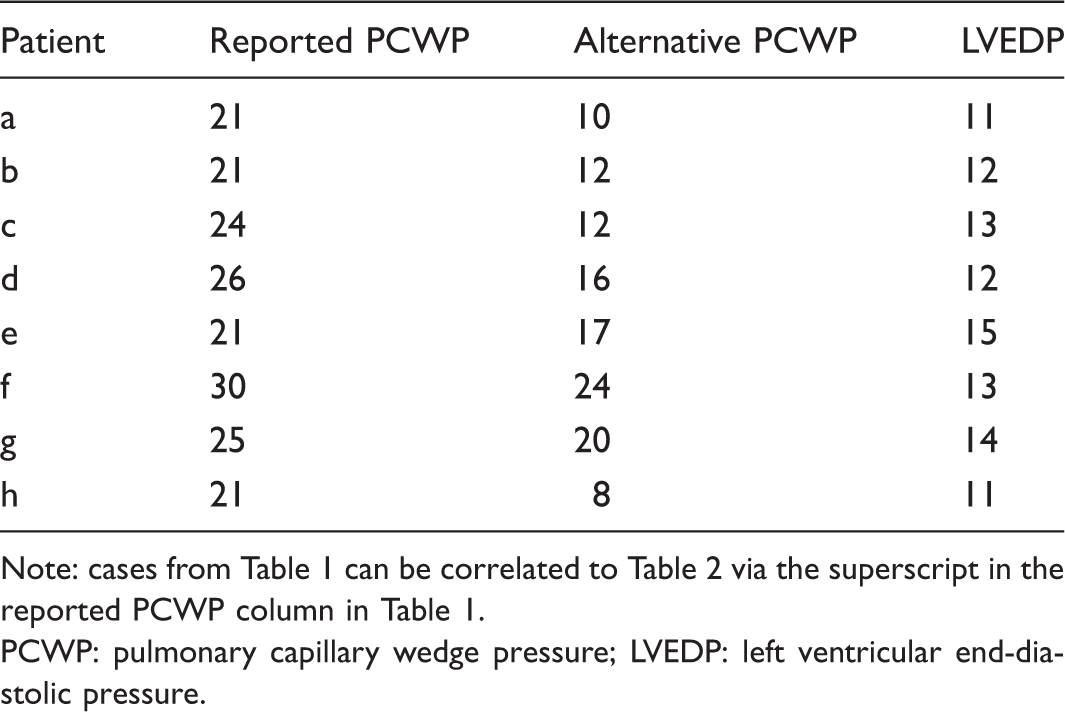
Note: cases from Table 1 can be correlated to Table 2 via the superscript in the reported PCWP column in Table 1.
PCWP: pulmonary capillary wedge pressure; LVEDP: left ventricular end-diastolic pressure.
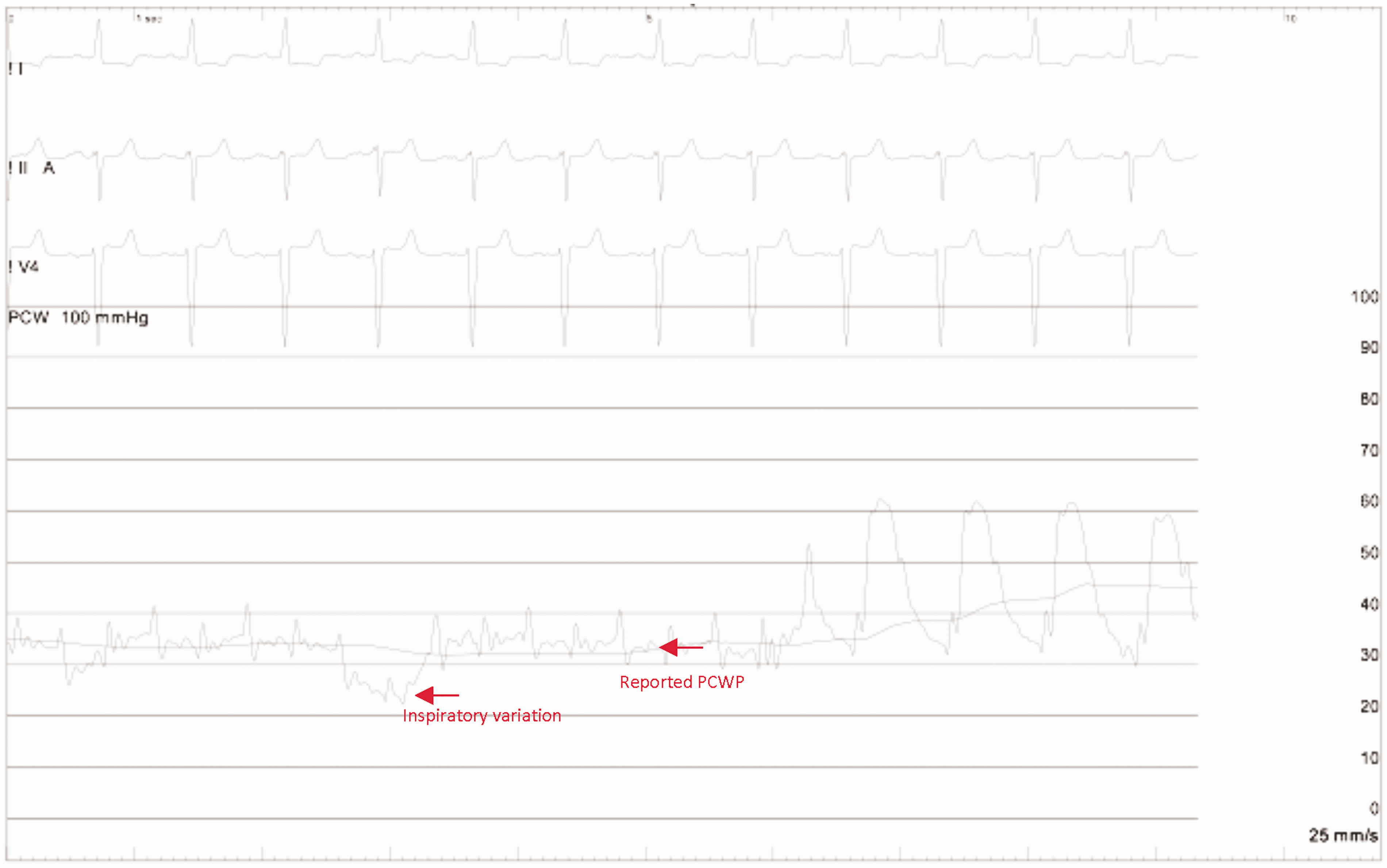
Brief isolation of an alternative PCWP is demonstrated in Fig. 2. Given the inadvertent nature of this alternative pressure tracing, it is not observed over multiple cardiac cycles or respiratory cycles though the pressure is definitively below the inspiratory variation of the reported PCWP tracing. A comparative figure, in which wedge is accurately reported, demonstrates transition from PCWP to PA without isolation of an alternative PCWP (Fig. 3).
Example of reported pulmonary capillary wedge pressure (28 mmHg), identified on balloon deflation prior to catheter pull back from PCWP to pulmonary artery, compared to alternative wedge pressure (12 mmHg). Example of accurate reporting of PCWP (34 mmHg); during transition from PCWP to pulmonary artery, no alternative PCWP is identified.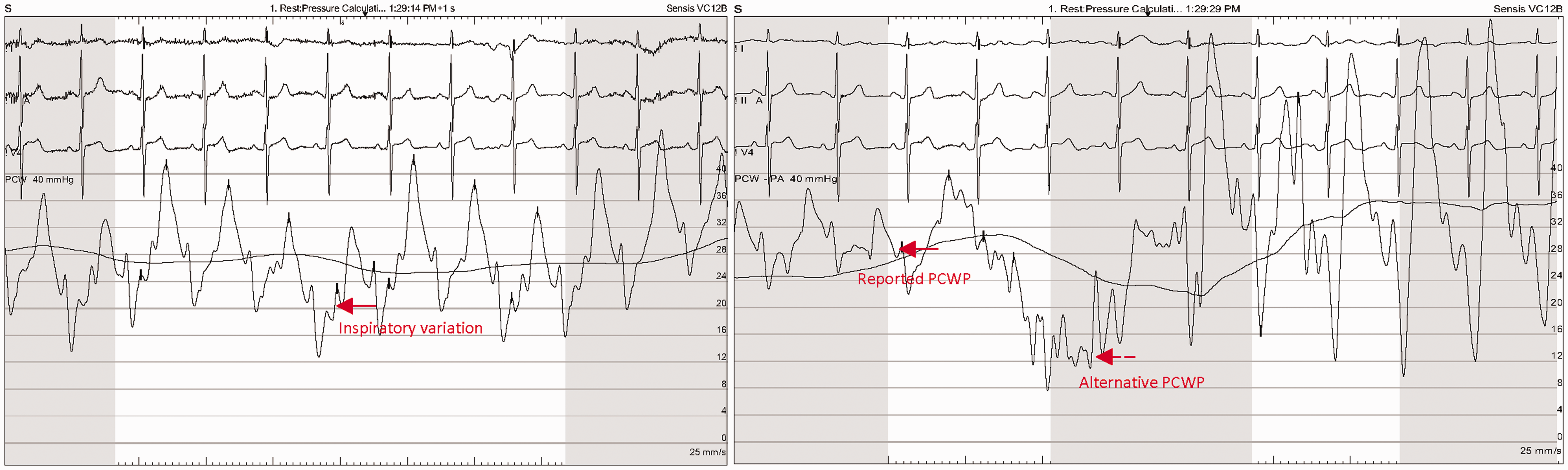
With respect to the validity of the tracing reviews, four experienced clinicians were involved in the evaluation of 95 of the 182 tracings (52%). Kappa coefficients for inter-rater reliability across experienced clinicians of 0.53, 0.51, and 0.47 supported good reliability allowing for a single reviewer to analyze the additional 87 cases.
Discussion
Inadvertent alternative wedge pressures were identified in almost 15% of RHCs reviewed from a large cardiac catheterization lab at a busy academic institution. The concordance between alternative PCWP and LVEDP in all cases in which LVEDP was available supports the accuracy of the alternative PCWP over the reported PCWP, which likely represents an incomplete and falsely elevated wedge. Negative DPGs (reported PCWP higher than DPAP) were common suggesting that understanding of the anticipated relationship between these variables may not be sufficient to suggest inadequacy of the PCWP to the operator. Of the patients in whom an alternative PCWP was identified, all of whom had pulmonary hypertension based on a mPAP ≥ 20 mmHg, 11 patients had an alternative PCWP ≤ 15 mmHg with a calculated alternative PVR ≥ 3 Wood units in 10 of these 11 patients, subsequently re-classifying pathology group in 38.5% of cases. While one might hypothesize alternative PCWP to be more likely in patients with chronic thromboembolic disease pulmonary hypertension (CTEPH), potentially due to webs or bands interfering with accurate occlusion, none of the patients in whom an alternative PCWP was identified in this cohort had CTEPH. The implication of these findings is relevant not only with regards to characterizing patients as having post-capillary pulmonary hypertension who in fact have pulmonary arterial pathology but also when considering more general decisions regarding patient volume status and need for diuresis.
While we did not do a retrospective analysis of each patient in whom an alternate PCWP was identified to assess whether they incurred a poor outcome as a result of treatment directed toward an elevated PCWP (i.e. ≥20 mmHg), the uncovering of this alternative, more accurate PCWP during balloon deflation supports previously published concerns that partial occlusion of the PA can result in falsely elevated wedge pressures.8,9 As the balloon is being deflated prior to transition from PCWP to PA, we suspect that the catheter is able to briefly move in to a smaller PA branch, creating a more complete occlusion and therefore more accurate reflection of left atrial pressure across a static column of blood. It is also possible that when the balloon is completely deflated, the catheter itself is lurching forward to wedge.
The goal of this descriptive study is to highlight the transition from PCWP to PA as a procedural time point at which the operator should be attuned to the possibility of their initial PCWP representing a partial or incomplete wedge. It is also to remind operators of additional techniques that can be utilized to ensure accuracy of PCWP reporting. We advocate for catheter advancement with a fully inflated balloon. The balloon should then be deflated to allow the catheter to fall further forward. The balloon can then be re-inflated at which time an alternate wedge may appear. In fact, intentional, slow balloon deflation, with attention toward the possibility of identifying an alternative, more accurate, PCWP may be revealing to the operator. Additional steps operators may take to ensure appropriate positioning for an attainment of a precise wedge include (1) assurance of waveform atrialization, (2) small balloon volume utilization (i.e. < 1.5 mL), (3) assessment of wedge saturation and wedge angiography, (4) consideration of the DPAP and PCWP relationship (i.e. DPAP shoulder be higher than PCWP in pre-capillary PH), and (5) correlation of PCWP to LVEDP if and when available.9–12
With variable data on the relationship between PCWP and the gold standard, LVEDP, the question of whether the alternative wedge identified in our study is the true wedge requires further exploration. Recent data exploring the relationship between PCWP and LVEDP suggests that PCWP can both over and underestimate LVEDP with mean differences ranging from –18 to 30 mmHg. Importantly, these data suggest that as PCWP and LVEDP increase (i.e. to > 20 and 20 respectively), the variables have especially poor agreement. 13 Regression analyses have identified atrial fibrillation, rheumatic valvular disease, and large atrial diameter as clinical characteristics associated with PCWP overestimation of LVEDP. Given that the mean reported PCWP in our study is 26 mmHg, it is likely that our data are reflective of and correlate with this previously described relationship in which PCWP overestimates LVEDP. The question raised by our study, however, is whether these are overestimates or just false wedge pressures. Hemnes et al. note that the measurements in their study were extracted from medical records and not through manual review. 13 The manual review of tracings in our study is a strength and raises the question of whether previously reported data, such as that in this study, might find an alternative wedge that minimizes PCWP and LVEDP discordance.
Limitations of this study are largely due to the inadvertent nature in which these alternative wedge tracings were obtained. Because the operator was unaware that they had localized a more accurate PCWP during balloon deflation, tracings are only transiently available and documented on retrospective review. As a result, the tracing of partial balloon deflation is not maintained over several cardiac and/or respiratory cycles, potentially limiting the precision of our alternative PCWP reporting. We are additionally unable to evaluate for respiratory variation within the alternative PCWP tracings. While it is possible that the alternative PCWP is representative of greater inspiratory effort, comparison of the alternative PCWP to variation across the respiratory cycle of the reported PCWP tracing supports the presumption that the alternative PCWP is representative of a new and more accurate occlusive pressure (Fig. 2). Additional limitations include lack of fluoroscopy to visualize the difference in catheter and/or balloon positioning between reported and alternative PCWP and lack of PA saturation reporting. While recent guidelines support wedge angiography or PA saturations if the accuracy of an elevated PCWP is in question, it is unlikely that operators questioned the accuracy of their reported PCWP or they would have edited their reports. 14
In response to these limitations, a study prospectively focused on isolating alternative PCWP to characterize frequency and confirm accuracy based on the additional techniques reviewed in this paper is warranted. Such a study would allow for description of respiratory variation seen within an alternative PCWP and potentially allow for further characterization of catheter positioning and balloon volume necessary to obtain a more accurate PCWP. It would additionally allow for analysis of provider level evaluation of frequency of identification of alternative PCWP and appropriate catheter adjustment among cardiology versus pulmonary trained operators. Inclusion criteria for a prospective analysis may also include patients with computer-generated PCWP ≥15 mmHg, as opposed to 20 mmHg, to fully describe the frequency of inaccurate classification, understanding these alternative PCWP, if and when identified, may not ultimately change classification group. Results of such a study could help direct future education efforts to ensure appropriate technique during RHC and interpretation of the data obtained by providers.
While RHC is deemed to be a low risk procedure when done at an institution with high volumes and subspecialty expertise, tracing misinterpretation is common. To mitigate error, operators should focus on slow and intentional balloon deflation prior to transitioning from PCWP to PA as they may identify an alternative PCWP that is likely a more accurate reflection of LVEDP. Additional techniques such as utilization of smaller balloon volumes, wedge saturation, or wedge angiography should also be considered if the PCWP obtained is incongruous with the clinical scenario, the tracing does not demonstrate the anticipated atrialization or the relationship between DPAP and PCWP is discordant from what is expected. Given clinical utilization of wedge pressure to drive decisions regarding the diagnosis and management of patients with pulmonary hypertension, accuracy of PCWP has significant implications on patient care. If concomitant LHC data are available to operators, discordance in PCWP and LVEDP should precipitate consideration of re-wedging to ensure appropriate classification of patients with pulmonary hypertension.
Footnotes
Acknowledgements
Eugene Pomerantsev, MD, for providing access to the cardiac catheterization database and organizing data for use. Joseph Locascio, PhD, for his statistical support.
Author contributions
All authors participated in the conception, design, data review, and interpretation of the data for this manuscript. Shelsey W. Johnson drafted the manuscript with revisions contributed by all authors. The final version was approved by all authors.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Ethical approval
The MGH institutional review board approved review of RHC tracings without need for informed consent (protocol # 2017P001130).
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Research institution
This research project was conducted at the Massachusetts General Hospital.
