Abstract
Pulmonary arterial hypertension (PAH) is a heterogenous clinical entity with poor prognosis, despite recent major pharmacological advances. To increase awareness about the pathophysiology, epidemiology, and management of the disease, large national registries are required. The Hellenic pulmOnary hyPertension rEgistry (HOPE) was launched in early 2015 and enrolls patients from all pulmonary hypertension subgroups in Greece. Baseline epidemiologic, diagnostic, and initial treatment data of consecutive patients with PAH are presented in this article. In total, 231 patients with PAH were enrolled from January 2015 until April 2018. At baseline, about half of patients with PAH were in World Health Organization functional class II. The majority of patients with PAH (56.7%) were at intermediate 1-year mortality risk, while more than one-third were low-risk patients, according to an abbreviated risk stratification score. Half of patients with PAH were on monotherapy, 38.9% received combination therapy, while prostanoids were used only in 12.1% of patients. In conclusion, baseline data of the Greek PAH population share common characteristics, but also have some differences with other registries, the most prominent being a better functional capacity. This may reflect earlier diagnosis of PAH that in conjunction with the increased proportion of patients with atypical PAH could partially explain the preference for monotherapy and the limited use of prostanoids in Greece. Nevertheless, early, advanced specific therapy is strongly recommended.
Keywords
Introduction
Pulmonary arterial hypertension (PAH) is a rare heterogenous disease of the pulmonary vasculature, defined by an increased mean pulmonary artery pressure (mPAP) ≥25 mmHg, a pulmonary artery wedge pressure (PAWP) <15 mmHg, and increased pulmonary vascular resistance (PVR) ≥3 Wood Units. 1 PAH is characterized by an adverse remodeling of the small pulmonary arteries with progressive increase in PVR that eventually leads to right heart failure and death, if not treated promptly. It could be idiopathic, familial, induced by drugs and toxins, or may be associated with a wide spectrum of diseases, such as connective tissue disease (CTD), congenital heart disease (CHD), portal hypertension, or schistosomiasis.2–4 A Task Force from the 6th World Symposium on Pulmonary Hypertension recently proposed a new definition of pre-capillary pulmonary hypertension (PH), with mPAP > 20 mmHg, PAWP < 15 mmHg, and PVR ≥ 3 Wood Units; however, prospective trials are required to determine whether this PH population might benefit from specific management. 5
Despite major advances in pharmacotherapy of PAH in the last 15 years, 6 we still lack thorough knowledge about the pathophysiology and the epidemiology of PAH. Although large national and international registries7–15 have so far provided useful information on the epidemiology, the diagnosis, and management of patients with PAH, regional differences certainly exist regarding patients' characteristics, diagnostic workup, and treatment availability. Moreover, since the registries include a mix of PAH patients with advanced age and various comorbidities, there is a scientific need for more epidemiological data from different countries, with different healthcare organization and financial background, in order to obtain a more comprehensive view of PAH worldwide.
Prompted by the increasing number of patients with PH who have been treated in expert centers in Greece so far and by the lack of epidemiological and management data for the Greek population, the Hellenic Society for the Study of Pulmonary Hypertension established a national network on PH. In this initial observational study, we present baseline data regarding the epidemiology, diagnosis, and management of patients with PAH in Greece.
Methods
The Hellenic Pulmonary Hypertension Registry (HOPE) is a PH registry that was launched in January 2015 and continues to enroll patients, mainly with PAH. The HOPE registry has been approved by the Institutional Review Board of each one of the nine participating PH expert centers in Greece according to the Declaration of Helsinki. All patients provided written informed consent for their inclusion in the study. Documentation has been Internet-based (PAH tool by Inovultus Lda, Portugal) and includes demographics, type of PH according to the European Guidelines, 1 comorbidities, clinical symptoms and signs, World Heart Organization (WHO) functional class (FC), 6-minute walk distance (6-MWD), N-terminal pro-brain natriuretic peptide (NT-proBNP) serum levels, echocardiographic parameters, hemodynamics, lung function tests, computed tomography data, ventilation/perfusion scintigraphy, and detailed information about medications for PH, including supportive measures, such as oxygen therapy and anticoagulation, as well as PAH-specific medical therapy. The participating centers enter all of their eligible patients on a consecutive basis. Data are collected at the time of first visit of patients at the PH centers and at least in 6-month intervals or whenever the patient has a predefined clinical event (death, transplantation, PAH-related hospitalization, deterioration in FC, any unscheduled change in PAH therapy, or other serious adverse events).
The cut-off date for the data analysis of the present study was April 2, 2018. Inclusion criteria for this study were a diagnosis of PAH according to the definitions of the 2015 guidelines, 1 age ≥14 years, and availability of data from right heart catheterization showing an mPAP ≥25 mmHg, a PAWP ≤ 15 mmHg, and PVR ≥ 3 Wood units. The exception was patients with Eisenmenger syndrome who could be diagnosed only by echocardiography. Classification of specific PAH subtype was assigned by the investigator reporting the case to the registry. The present study focused only on patients' baseline data, that is data at the time of first visit of patients at the PH centers, since follow-up data were not available yet for the majority of PAH patients.
An abbreviated version of the 2015 European Society of Cardiology (ESC)/European Respiratory Society (ERS) risk stratification strategy was used to categorize patients as low, intermediate, or high risk, as proposed by Hoeper et al. 16 WHO FC, 6-MWD, NT-proBNP, right atrial pressure (RAP), cardiac index (CI), and mixed venous oxygen saturation at baseline were used for the risk stratification, if available. 16 The cut-off values proposed in the European guidelines 1 were graded 1–3 (1: low risk, 2: intermediate risk, and 3: high risk). For each patient, the sum of all grades was divided by the number of available variables and rounded to the next integer to define the risk group.16,17
Statistical methods
Data were presented as mean ± standard deviation for continuous variables with normal distribution, and as median and interquartile range for non-normally distributed variables. Categorical variables were presented as frequencies and percentages (%). Continuous variables were compared using the t-test for independent samples or the Mann–Whitney U test, while the chi-square test or the Fisher exact test was used to assess categorical variables. For multiple comparisons, one-way ANOVA or the Kruskal–Wallis test with post hoc analysis was used as appropriate. A p-value < 0.05 was considered statistically significant in this study. Data were analyzed using the SPSS version 23.0.
Results
Baseline characteristics
Between January 2015 and April 2018, 231 patients with PAH from nine PH centers from all over Greece were enrolled. Figure 1 shows the distribution of 231 patients across the various types of PAH. The most frequent PAH subtypes were idiopathic PAH (IPAH) (82, 35%), PAH due to CTD (71, 31%), and PAH associated with CHD (60, 26%). Four patients (2%) had heritable PAH (HPAH), three patients (1%) had PAH induced by drugs and toxins, six patients (3%) had PAH due to portal hypertension, and five patients (2%) had PAH due to more than one etiology. The most frequent cause of PAH-CTD was systemic sclerosis in 63% of patients, while Eisenmenger syndrome was present in 36% of patients with PAH-CHD. In PAH-CHD, simple type defects were the most frequent etiology (76%).
Classification of patients with PAH. The distribution of 231 patients across the various types of PAH in Greece (absolute number; %) is shown.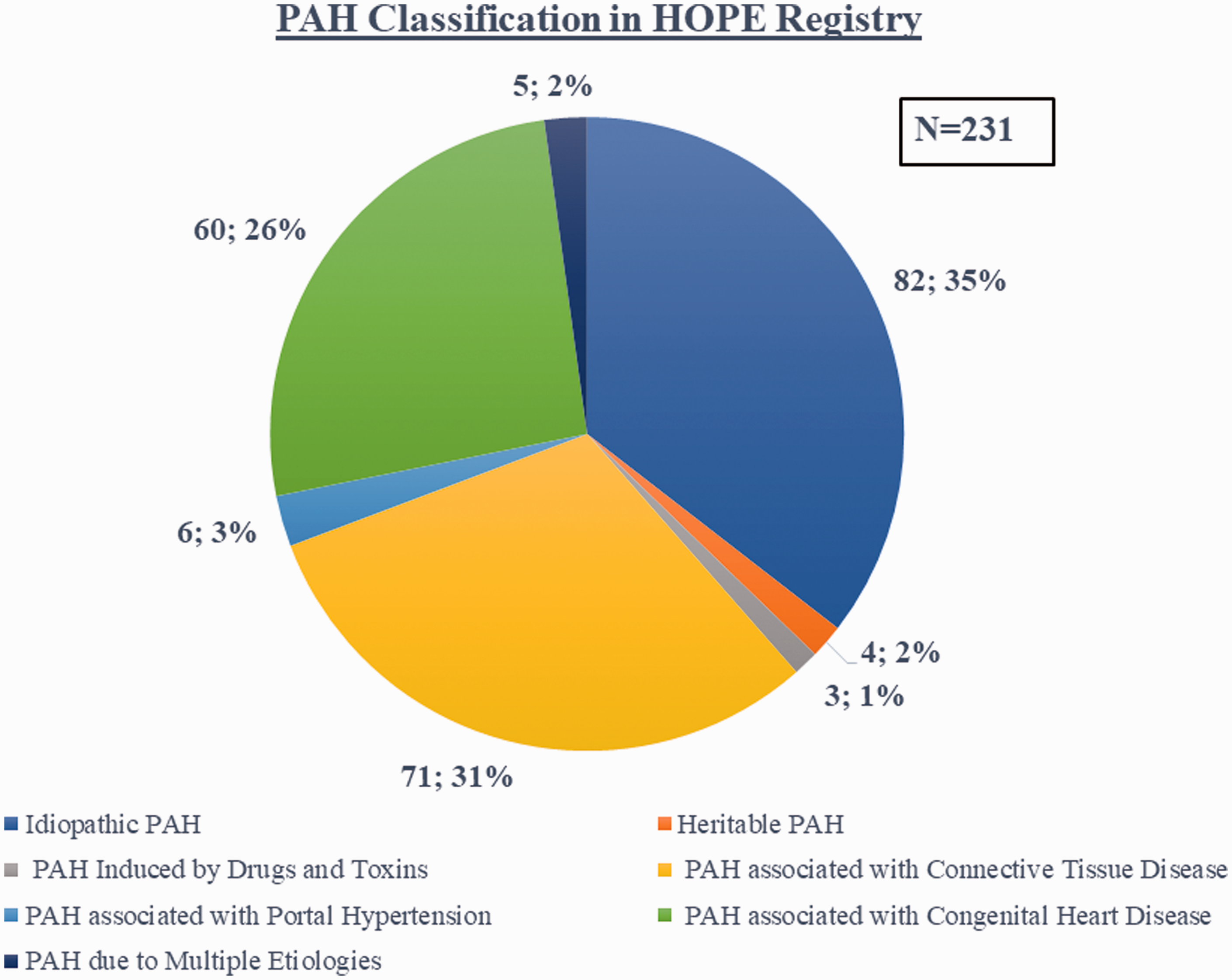
Baseline demographics and clinical characteristics of patients with PAH and differences among subgroups.
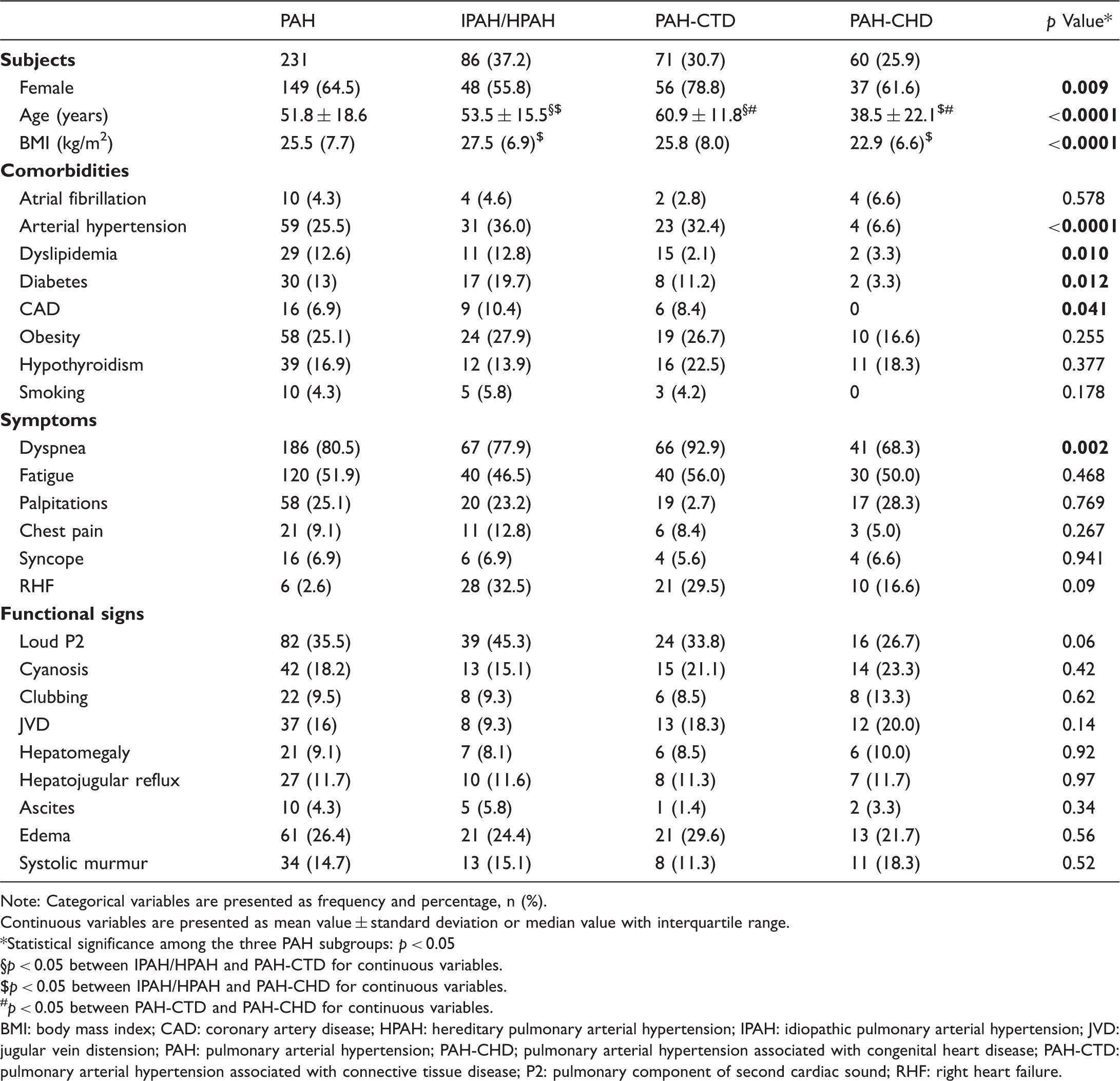
Note: Categorical variables are presented as frequency and percentage, n (%).
Continuous variables are presented as mean value ± standard deviation or median value with interquartile range.
Statistical significance among the three PAH subgroups: p < 0.05
p < 0.05 between IPAH/HPAH and PAH-CTD for continuous variables.
p < 0.05 between IPAH/HPAH and PAH-CHD for continuous variables.
p < 0.05 between PAH-CTD and PAH-CHD for continuous variables.
BMI: body mass index; CAD: coronary artery disease; HPAH: hereditary pulmonary arterial hypertension; IPAH: idiopathic pulmonary arterial hypertension; JVD: jugular vein distension; PAH: pulmonary arterial hypertension; PAH-CHD; pulmonary arterial hypertension associated with congenital heart disease; PAH-CTD: pulmonary arterial hypertension associated with connective tissue disease; P2: pulmonary component of second cardiac sound; RHF: right heart failure.
Baseline clinical assessment and hemodynamics of patients with PAH and differences among subgroups.
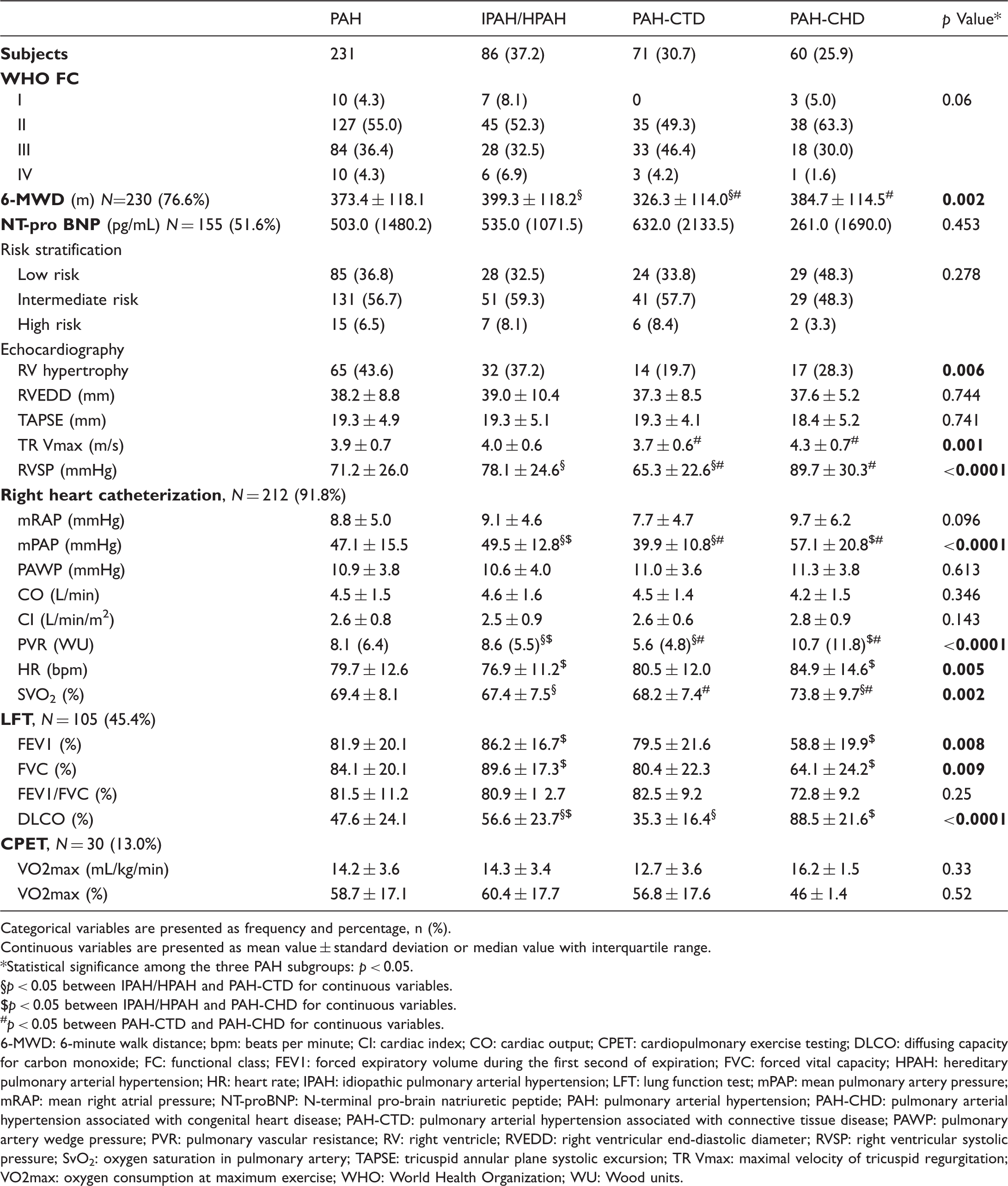
Categorical variables are presented as frequency and percentage, n (%).
Continuous variables are presented as mean value ± standard deviation or median value with interquartile range.
Statistical significance among the three PAH subgroups: p < 0.05.
p < 0.05 between IPAH/HPAH and PAH-CTD for continuous variables.
p < 0.05 between IPAH/HPAH and PAH-CHD for continuous variables.
p < 0.05 between PAH-CTD and PAH-CHD for continuous variables.
6-MWD: 6-minute walk distance; bpm: beats per minute; CI: cardiac index; CO: cardiac output; CPET: cardiopulmonary exercise testing; DLCO: diffusing capacity for carbon monoxide; FC: functional class; FEV1: forced expiratory volume during the first second of expiration; FVC: forced vital capacity; HPAH: hereditary pulmonary arterial hypertension; HR: heart rate; IPAH: idiopathic pulmonary arterial hypertension; LFT: lung function test; mPAP: mean pulmonary artery pressure; mRAP: mean right atrial pressure; NT-proBNP: N-terminal pro-brain natriuretic peptide; PAH: pulmonary arterial hypertension; PAH-CHD: pulmonary arterial hypertension associated with congenital heart disease; PAH-CTD: pulmonary arterial hypertension associated with connective tissue disease; PAWP: pulmonary artery wedge pressure; PVR: pulmonary vascular resistance; RV: right ventricle; RVEDD: right ventricular end-diastolic diameter; RVSP: right ventricular systolic pressure; SvO2: oxygen saturation in pulmonary artery; TAPSE: tricuspid annular plane systolic excursion; TR Vmax: maximal velocity of tricuspid regurgitation; VO2max: oxygen consumption at maximum exercise; WHO: World Health Organization; WU: Wood units.
Medical therapy
Baseline medical therapy in patients with PAH and differences among subgroups.
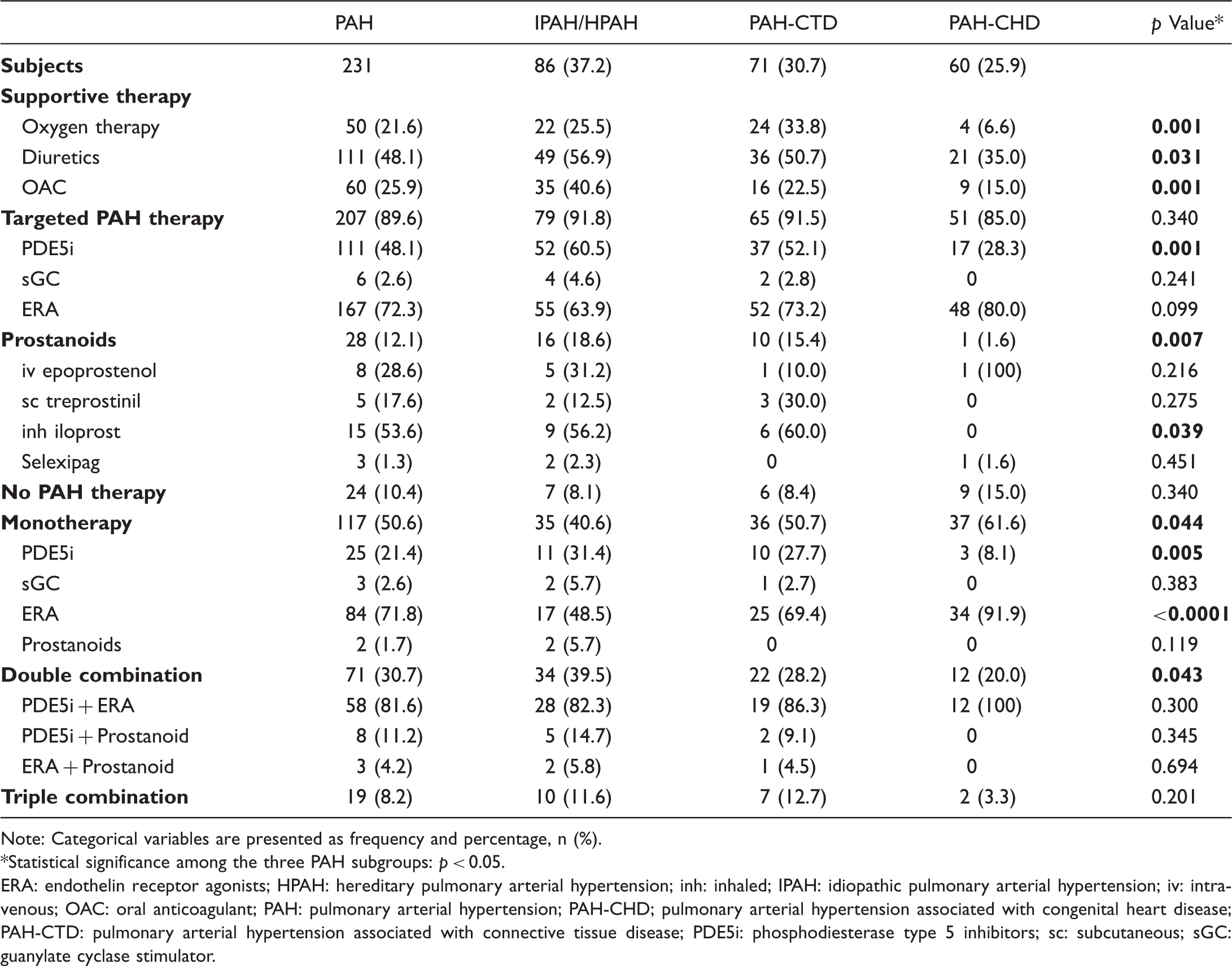
Note: Categorical variables are presented as frequency and percentage, n (%).
Statistical significance among the three PAH subgroups: p < 0.05.
ERA: endothelin receptor agonists; HPAH: hereditary pulmonary arterial hypertension; inh: inhaled; IPAH: idiopathic pulmonary arterial hypertension; iv: intravenous; OAC: oral anticoagulant; PAH: pulmonary arterial hypertension; PAH-CHD; pulmonary arterial hypertension associated with congenital heart disease; PAH-CTD: pulmonary arterial hypertension associated with connective tissue disease; PDE5i: phosphodiesterase type 5 inhibitors; sc: subcutaneous; sGC: guanylate cyclase stimulator.
Only 1 out of 10 patients with PAH did not receive a targeted PAH medical therapy at baseline. About half of patients received a phosphodiesterase type 5 inhibitor (PDE5i), while nearly three-fourth of them received an endothelin receptor agonist (ERA). Prostanoid use was limited in 12.1% of patients with PAH. Half of patients with PAH were on monotherapy at baseline, mainly with an ERA (71.8%), while 38.9% of them received a combination therapy (mostly an ERA with a PDE5i) (Table 3).
Among PAH subgroups, oral combination therapy was most frequently used in IPAH/HPAH (51.1%). PDE5i and prostanoid use were also higher in this subgroup (60.5% and 18.6%, respectively). The majority of patients with PAH-CHD received an ERA (80%), either as monotherapy (56.6%) or combination therapy (23.3%) (Table 3). Calcium channel blockers (CCBs) were most frequently prescribed in PAH-CTD (19.7%), mainly due to digital ulcers and comorbidities, but also in five patients with IPAH due to positive vasoreactivity test.
Risk stratification in PAH
Differences in baseline clinical assessment, hemodynamics, and therapeutic management of patients with PAH according to their 1-year mortality risk.

Note: Categorical variables are presented as frequency and percentage, n (%).
Continuous variables are presented as mean value ± standard deviation or median value with interquartile range.
Statistical significance among low-, intermediate- and high-risk patients with pulmonary arterial hypertension: p < 0.05.
p < 0.05 between low- and intermediate- risk patients for continuous variables.
p < 0.05 between low- and high-risk patients for continuous variables.
p < 0.05 between intermediate- and high-risk patients for continuous variables.
6-MWD: 6-minute walk distance; BMI: body mass index; bpm: beats per minute; CI: cardiac index; CO: cardiac output; ERA: endothelin receptor agonists; FC: functional class; HR: heart rate; inh: inhaled; iv: intravenous; mPAP: mean pulmonary artery pressure; mRAP: mean right atrial pressure; NT-proBNP: N-terminal pro-brain natriuretic peptide; OAC: oral anticoagulant; PAH: pulmonary arterial hypertension; PAWP: pulmonary artery wedge pressure; PDE5i: phosphodiesterase type 5 inhibitors; PVR: pulmonary vascular resistance; RVEDD: right ventricular end-diastolic diameter; RVSP: right ventricular systolic pressure; sc: subcutaneous; sGC: guanylate cyclase stimulator; SvO2; oxygen saturation in pulmonary artery; TAPSE: tricuspid annular plane systolic excursion; TR Vmax: maximal velocity of tricuspid regurgitation; WHO: World Health Organization; WU: wood units.
Discussion
Comparative baseline demographic, clinical, hemodynamic, and treatment data between patients with PAH in Greece and other PAH Registries.
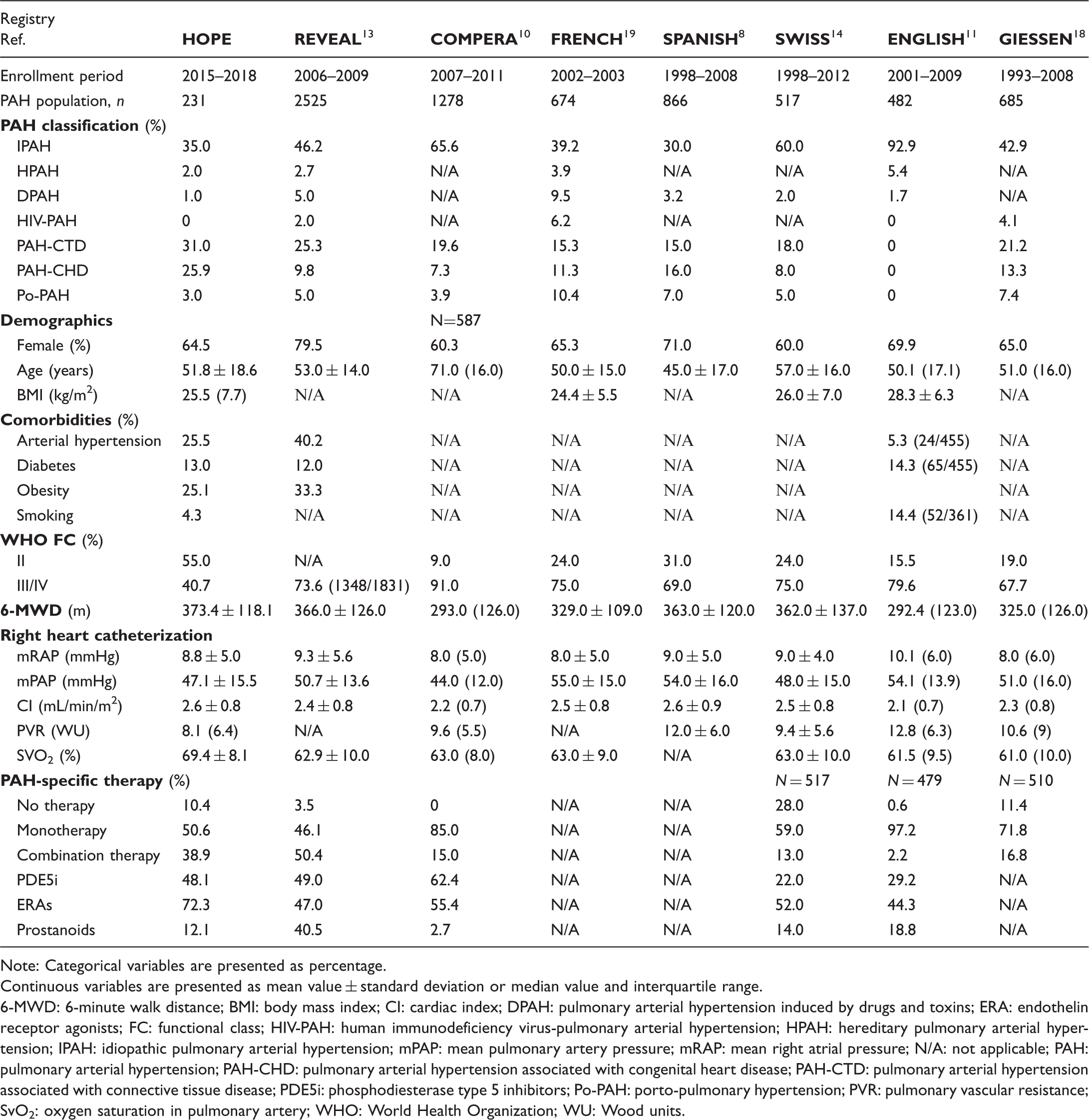
Note: Categorical variables are presented as percentage.
Continuous variables are presented as mean value ± standard deviation or median value and interquartile range.
6-MWD: 6-minute walk distance; BMI: body mass index; CI: cardiac index; DPAH: pulmonary arterial hypertension induced by drugs and toxins; ERA: endothelin receptor agonists; FC: functional class; HIV-PAH: human immunodeficiency virus-pulmonary arterial hypertension; HPAH: hereditary pulmonary arterial hypertension; IPAH: idiopathic pulmonary arterial hypertension; mPAP: mean pulmonary artery pressure; mRAP: mean right atrial pressure; N/A: not applicable; PAH: pulmonary arterial hypertension; PAH-CHD: pulmonary arterial hypertension associated with congenital heart disease; PAH-CTD: pulmonary arterial hypertension associated with connective tissue disease; PDE5i: phosphodiesterase type 5 inhibitors; Po-PAH: porto-pulmonary hypertension; PVR: pulmonary vascular resistance; SvO2: oxygen saturation in pulmonary artery; WHO: World Health Organization; WU: Wood units.
Furthermore, in accordance with other PAH cohorts, dyspnea and fatigue were the most common symptoms at baseline. However, our population had more favorable clinical characteristics at diagnosis, since half of patients were in WHO II FC (Table 5). The 6-MWD distance was slightly better compared with other registries, while hemodynamics (lower mPAP and PVR, higher SVO2%) were slightly more favorable in our population.8,10,11,13–15,18,19 Similar to other registries, the majority of the PAH population was at intermediate 1-year mortality risk, according to the modified PAH risk assessment score.13,16,17 This observation implies that PAH patients in our cohort may have underestimated their symptoms and their FC, quite common in the PAH-CHD cohort, 25 which could have led the physician to opt for monotherapy. It should be taken into consideration that echocardiography is widely available in Greece. Thus, the fact that most Greek patients have easy access to this, inexpensive in our country, first-line screening tool may be associated with earlier diagnosis in the course of the disease.
In terms of supportive therapy, diuretics were used by approximately half of patients with PAH. Oral anticoagulants were also frequently used in IPAH/HPAH subgroup, despite the fact that the recommendation for their administration in patients with IPAH has been downgraded to IIb according to the latest ESC Guidelines. 1
Almost half of PAH patients received monotherapy at baseline assessment. ERAs and PDE5i, the former more frequently, were used as monotherapy. Within PAH group, ERAs were more frequently given as monotherapy in PAH-CHD subgroup, while PDE5i in PAH-CTD subgroup. These results are in agreement with previous reports demonstrating ERAs and PDE5i being the most widely used treatment-specific drugs.9–11,13,14 ERA and PDE5i combination was also the most widely used combination in PAH group, although the rate of dual therapy was lower than previously reported. 13 This is possibly related to the better WHO FC of the majority of our population, the large proportion of patients with Eisenmenger syndrome, 1 and the high prevalence of patients with PAH and comorbidities 6 that could explain why most patients received monotherapy. However, there were also registries that reported a much lower use of combination therapy at baseline compared with our cohort.10,11,14,18 Furthermore, the use of prostanoids as monotherapy or combination therapy was lower in our cohort when compared with other registries.11,13,14 In general, treatment management was in line with the ESC/ERS treatment guidelines.1,2 However, the satisfactory functional status (WHO I/II) of the majority of patients in our cohort, in contrast to other European registries, may partially explain the low percentage of prostanoid use in this population, albeit a more aggressive approach could also be considered. 26 Additionally, the intravenous route of administration along with the known side effects might have contributed to the low percentage of use of parenteral prostanoids as monotherapy or combination therapy in our cohort.
A limitation of this study is the relatively small sample size which does not yet permit the application of more sophisticated statistical methods that would provide reliable prognostic information. Additionally, in this paper, information regarding only the initial assessment is presented. Thus, data regarding the clinical course, survival, and evolution of treatment management over time are still pending.
In conclusion, this study indicates that the demographic, clinical, and management data of patients with PAH in Greece did not differ significantly when compared with other countries. However, the initial clinical assessment shows that although most PAH patients are of intermediate risk, their functional capacity is better than previously reported in similar populations. This may reflect an improvement in the timely identification of patients at risk for developing PAH and lead to early diagnosis of the disease; yet, adaptation of a contemporary aggressive targeted strategy should be encouraged.
Footnotes
Acknowledgements
We would like to thank the following co-investigators for their contribution in the HOPE Registry:
AHEPA University Hospital, Aristotle University of Thessaloniki, Thessaloniki: Vasilios Grosomanidis, Pavlos Rouskas, Κonstantinos Kouskouras, Irene Nikolaou, Despoina Parcharidou, and Stavros Hadjimiltiades
Cardiology Department, Mediterraneo Hospital, Glyfada, Athens: Lilika Papa and Antonios Marsonis
Multidisciplinary Pulmonary Hypertension Center, Attikon University General Hospital; National and Kapodistrian University of Athens Medical School, Athens: Apostolos Armaganidis, Dimitrios Konstantonis, Frantzeska Frantzeskaki, Michalis Rizos, Olympia Apostolopoulou, Michalis Lignos, Alexandra Frogoudaki, Loukianos Rallidis, Christos Pappas, Athanasios Pappas, Roza Vrettou, John Lekakis, Efstathios Ιliodromitis, Pelagia Katsimbri, Dimitrios Boumpas, Theofanis Karageorgas, Antonios Fanouriakis, Panagiotis Vlachoyiannopoulos, Spyridon Papiris, Efrosini Manali, Andrianna Papaioannou, Aikaterini Vlami, Panagiotis Lymperopoulos, Stylianos Argentos, Maria Mademli, Nikolaos Kelekis, Athanasios Tzioufas, Ioannis Armeniakos, Trapali Xeni Geronikola, Konstantina Ntai, Vasiliki Tsangkari, Elli Mitrakou, and Marina Kontogiorgi
Cardiology-Pediatric Cardiology Department, Onassis Cardiac Surgery Center, Athens: George Athanasopoulos, George Karatasakis, Genovefa Kolovou, Maria Koutelou, Katerina Marathia, Irene Mastorakou, Stamatios Adamopoulos, Eftychia Sbarouni, Athanasios Dritsas, Sofia Thomopoulou, Ioannis Iakovou, Ioannis Kriaras, Athanasios Theodorakos, Olga Karapanagiotou, Stamatia Kampanarou, Spyridoula Katsilouli, Evangelos Leontiades, Panagiota Georgiadou, and Varvara Papadopoulou
Cardiology Department, Hippokration General Hospital, Athens: Eleutherios Pashalidis and Ioannis Leontitsis
Department of Thoracic Medicine, University Hospital of Heraklion, Heraklion, Crete, Greece: Georgios Prinianakis, Alexandros Patrianakos, Dimitrios Georgopoulos, Eumorfia Kondili, Nikolaos Tzanakis, Michalis Fanaridis, Markella Antonogiannaki, and Maria Bolaki
2nd Department of Cardiology, University of Ioannina Medical School, University Hospital of Ioannina: Aris Bechlioulis and Lampros Michalis
Cardiology Department, Democritus University of Thrace, Alexandroupolis, Greece: Stavros Konstantinides
Cardiothoracic Surgery Department, St. Luke’s Clinic, Panorama, Thessaloniki: Antonios Pitsis
Fourth Department of Internal Medicine, Hippokration General Hospital, School of Medicine, Aristotle University of Thessaloniki, Thessaloniki: Theodoros Dimitroulas
Ethical approval
The HOPE Registry received ethical approval from the Institutional Review Board of every participating center (AHEPA University Hospital, Thessaloniki; Mediterraneo Hospital, Athens; Attikon University General Hospital, Athens; Onassis Cardiac Surgery Center, Athens; Hippokration University General Hospital, Athens; Heraklion University Hospital, Crete; University Hospital of Ioannina; “G. Papanikolaou” Hospital, Thessaloniki; and University Hospital of Alexandroupolis) according to the Declaration of Helsinki, and all patients provided written informed consent for their inclusion in the study.
Authors’ contribution
Alexandra Arvanitaki and Maria Boutsikou contributed equally to the design of this work, the acquisition, analysis, and interpretation of data, and drafted and revised the article for important intellectual content. Athanasios Manginas made a substantial contribution to the concept and design of this work and revised the article critically for important intellectual content. The rest of the authors contributed to the acquisition of patients’ data and revised the article critically for important intellectual content.
Conflict of interest
Alexandra Arvanitaki and Maria Boutsikou were financially supported from the Hellenic Society for the Study of Pulmonary Hypertension. Anastasia Anthi reports receiving honoraria and consultancy fees from Actelion Pharmaceuticals Hellas, Bayer, ELPEN, GSK, MSD, Lilly, and United Therapeutics. Eftychia Demerouti is an advisory board member for Actelion Pharmaceuticals Hellas, MSD Hellas, and GlaxoSmithKline and an honorarium speaker for Actelion Pharmaceutical Hellas and MSD Hellas. George Giannakoulas reports receiving honoraria and consultancy fees from Actelion Pharmaceuticals Hellas, Bayer, ELPEN, Galenica-Ferrer, GSK, Pfizer, Lilly, MSD, and United Therapeutics. Haralambos Karvounis reports receiving honoraria and consultancy fees from Actelion Pharmaceuticals Hellas, Bayer, ELPEN, Galenica-Ferrer, GSK, Pfizer, Lilly, and MSD. Ioanna Mitrouska reports receiving honoraria and consultancy fees from Actelion Pharmaceuticals Hellas, Bayer, ELPEN, MSD, and GSK. Katerina K. Naka reports lecture fees from Novartis, Medtronic, and Abbott, while she serves as an investigator in randomized clinical trials and/or registries sponsored by Novartis, Merck, Amgen, BMS, Boehringer Ingelheim, Pfizer, and Actelion Pharmaceutical Hellas, outside the submitted work. Stylianos E. Orfanos reports receiving research grants and/or honoraria and/or consultancy fees from Actelion Pharmaceutical Hellas, Bayer, ELPEN, Galenica-Ferrer, GSK, MSD, Pharmaserve Lilly, PharmaSwiss, Pfizer, and United Therapeutics. Georgia Pitsiou reports receiving honoraria and consultancy fees from Actelion Pharmaceutical Hellas, Bayer, ELPEN, MSD, and GSK. Ioannis Stanopoulos reports receiving honoraria and consultancy fees from Actelion Pharmaceuticals Hellas. Iraklis Tsangaris reports receiving honoraria and consultancy fees from Actelion Pharmaceuticals Hellas, Bayer, ELPEN, GSK, Pfizer, Lilly, and United Therapeutics. Athanasios Manginas reports consultation fees from Astra Zeneca, Bayer, ELPEN, Actelion Pharmaceuticals Hellas, and MSD. The rest of the authors report no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
