Abstract
We show that spironolactone use was associated with an increased rate of all-cause hospitalizations, but no difference in hospitalizations for heart failure or pulmonary arterial hypertension, in patients with World Health Organization Group 1 pulmonary arterial hypertension. A possible reason for this finding is confounding from retrospective study design.
There is a global deficit of knowledge for effective therapies targeting the renin-angiotensin-aldosterone system (RAAS) in patients with pulmonary arterial hypertension (PAH). Preclinical research shows a role of systemic and local RAAS modulation in the pathogenesis of right ventricular failure and pulmonary vascular remodeling associated with PAH. 1 Experimental models have further demonstrated that aldosterone receptor antagonists (ARAs) improve pulmonary hemodynamics and pulmonary vascular remodeling. 2 Additionally, ARA-mediated natriuresis and mild diuresis may provide another proposed rationale for the benefit of use in PAH patients. 2
Despite these proposed benefits, little research exists evaluating the clinical benefit of ARAs in patients with PAH. A post-hoc analysis of the ARIES-1 and ARIES-2 trials evaluated patients with World Health Organization (WHO) Group 1 PAH randomized to ambrisentan 10 mg/day or placebo in whom spironolactone use was reported. The addition of spironolactone to ambrisentan resulted in a statistically non-significant improvement in 6-minute walk distance, plasma B-type natriuretic peptide concentrations, and WHO functional classification. 3
Patients with PAH are at risk for development of right heart failure (RHF) and therapies that potentially target the right ventricle could be of particular benefit. While no clinical research exists regarding the effect of ARAs on RHF to date, these agents have demonstrated unequivocal benefit in patients with left heart failure with a reduced ejection fraction.4,5 The present study compared the rates of all-cause hospitalization in PAH patients receiving versus not receiving ARAs in addition to evidence-based PAH medication therapy.
We performed a single-center, retrospective review approved by our Institutional Review Board. All patients aged at least 18 years with WHO Group 1 PAH and a PAH clinic visit with the University of North Carolina (UNC) Pulmonary Hypertension Program between 1 June 2016 and 31 May 2017 were reviewed. We defined the index PAH clinic visit as the most recent documented PAH clinic visit in the study period. Patients were identified using a comprehensive electronic list maintained by the UNC Pulmonary Hypertension Program. Patients were excluded if an ARA was initiated or discontinued within the six months preceding the index PAH clinic visit, an additional class of PAH specific therapy was initiated within the three months preceding the index PAH clinic visit, left ventricular ejection fraction was ≤40% on echocardiogram, or they had concomitant presence of WHO Groups 2 through 5. Patients receiving ARA therapy (ARA group) were compared to those not receiving ARA therapy (control group) during the treatment period, defined as the six months preceding the index clinic visit.
All data were collected from the electronic medical record (EMR). Clinical information collected include WHO Group 1 PAH subtype; WHO functional classification at time of index clinic visit; concomitant therapy with PAH specific medications, calcium channel blocker, beta blocker, angiotensin converting enzyme inhibitor, angiotensin receptor blocker, digoxin, or loop diuretic; right ventricular (RV) function on echocardiogram (RV dilation, RV contractile dysfunction, severity of RV contractile dysfunction); hemodynamic parameters (pulmonary vascular resistance, cardiac index, and right atrial pressure); 6-MWD; and NT-proBNP. RV function represents data from the most recent echocardiogram and hemodynamic parameters represent data from the most recent right heart catheterization within six months and one year of the index clinic visit, respectively.
The primary outcome was the rate of all-cause hospitalization during the treatment period. Admissions to hospitals other than UNC Medical Center were included if documented in the EMR. A subgroup analysis was conducted evaluating the primary outcome in patients stratified by WHO functional class.
Baseline characteristics.
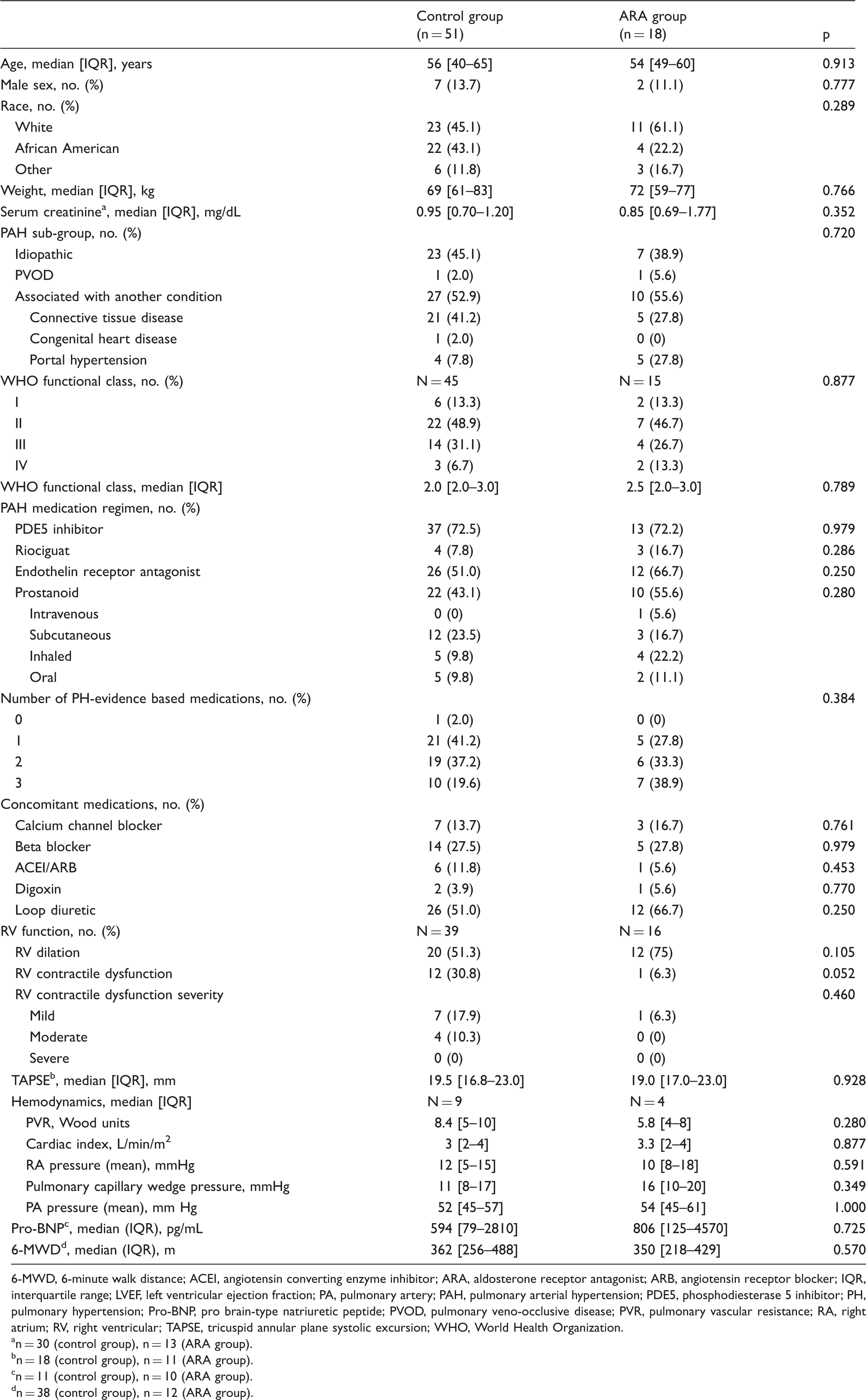
6-MWD, 6-minute walk distance; ACEI, angiotensin converting enzyme inhibitor; ARA, aldosterone receptor antagonist; ARB, angiotensin receptor blocker; IQR, interquartile range; LVEF, left ventricular ejection fraction; PA, pulmonary artery; PAH, pulmonary arterial hypertension; PDE5, phosphodiesterase 5 inhibitor; PH, pulmonary hypertension; Pro-BNP, pro brain-type natriuretic peptide; PVOD, pulmonary veno-occlusive disease; PVR, pulmonary vascular resistance; RA, right atrium; RV, right ventricular; TAPSE, tricuspid annular plane systolic excursion; WHO, World Health Organization.
n = 30 (control group), n = 13 (ARA group).
n = 18 (control group), n = 11 (ARA group).
n = 11 (control group), n = 10 (ARA group).
n = 38 (control group), n = 12 (ARA group).
There was a higher rate of all-cause hospitalization during the treatment period in the ARA group compared to the control group (61.1% vs. 21.6%; p = 0.002). However, there was no difference in the rates of hospitalization for worsening heart failure or pulmonary hypertension between groups (22.2% ARA group vs. 13.7% control group; p = 0.397). In a sub-group of patients with documented WHO functional class III or IV at the time of the index clinic visit (n = 23), there was a higher rate of all-cause hospitalization in the ARA group compared to the control group (6/6 (100%) vs. 7/17 (41.2%); p = 0.012). Similarly, in patients with documented WHO functional class I or II at the time of the index clinic visit, there was a higher rate of all-cause hospitalization in the ARA group compared to the control group (4/9 (44.4%) vs. 3/28 (10.7%); p = 0.025). These findings of the sub-group analysis are consistent with the primary outcome of the overall study population. There was no difference between groups in the total number of hospitalizations per patient (1.91 ± 1.37 ARA group vs. 1.91 ± 1.04 control group; p = 1.000) or in the number of hospitalizations for worsening HF or PAH per patient (0.64 ± 1.02 ARA group vs. 1.27 ± 1.27 control group; p = 0.211).
Limitations of this study include single-center retrospective design, which introduced the possibility for bias by indication, and small sample size. Additionally, trends in differences observed in key baseline characteristics, including a non-significantly higher rate of loop diuretic use and higher pulmonary capillary wedge pressure in the ARA group compared to the control group may have contributed to our findings. This would support the possibility raised by Maron et al. 3 that ARA use may represent a marker for PAH disease severity. Finally, the study design prevented us from evaluating changes in functional criteria, including WHO functional class and 6-MWD, over the study period.
There are currently two prospective randomized placebo-controlled trials ongoing to evaluate the potential benefit of spironolactone in patients with chronic stable RHF and those with stable PAH without RHF, respectively.6.7 STAR-HF is evaluating whether spironolactone is associated with a significant reduction in RV ventricular wall stress, as reflected by a reduction in serum NT-proBNP, in approximately 30 patients with chronic stable right HF. 6 The latter study is evaluating the effect of spironolactone on exercise capacity, clinical worsening, and vascular inflammation in approximately 70 patients with idiopathic or disease-associated PAH. 7
Overall, we found a significantly higher rate of all-cause hospitalization in patients with Group 1 PAH in the ARA group compared to the control group. Results from larger, prospective studies such as those currently ongoing are needed to further evaluate the clinical benefit of ARAs in PAH patients.
Footnotes
Author contributions
MEC: Participated in research design, performance of research, data analysis, and writing of the paper. LTD, MMC, BPM, and LJR-J
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
