Abstract
Pulmonary hypertension is a condition with high morbidity and mortality. Resting transthoracic echocardiography is a pivotal diagnostic and screening test for pulmonary hypertension. The role of exercise stress echocardiography in the diagnosis of pulmonary hypertension is not well-established. We studied right ventricular size changes during exercise using exercise stress echocardiography to assess differences between normal and pulmonary hypertension patients and evaluate test safety, feasibility, and reproducibility. Healthy control and pulmonary hypertension patients performed recumbent exercise using a bicycle ergometer. Experienced echocardiography sonographers recorded the following resting and peak exercise right ventricular parameters using the apical four chamber view: end-diastolic area; end-systolic area; mid-diameter; basal diameter; and longitudinal diameter. Two cardiologists masked to clinical information subsequently analyzed the recordings. Parameters with acceptable inter-rater reliability were analyzed for statistical differences between the normal and pulmonary hypertension patient groups and their association with pulmonary hypertension. We enrolled 38 healthy controls and 40 pulmonary hypertension patients. Exercise stress echocardiography testing was found to be safe and feasible. Right ventricular size parameters were all readily obtainable and all had acceptable inter-observer reliability except for right ventricular longitudinal diameter. During exercise, healthy controls demonstrated a decrease in right ventricular end-systolic area, end-diastolic area, mid-diameter, and basal diameter (P < 0.05). Conversely, pulmonary hypertension patients demonstrated an increase in right ventricular end-systolic area, end-diastolic area, and mid-diameter (P < 0.05). These changes were unaffected by multivariate corrections. The sensitivity for pulmonary hypertension of an increase in right ventricular size was 97.2% with a negative predictive value of 95.2%. The ROC C-statistic for increase in right ventricular size was 0.93. This transient exertional dilation (TED) of the right ventricle is observed in pulmonary hypertension patients but not in healthy controls. Recumbent right ventricular exercise stress echocardiography is a feasible and safe diagnostic test for pulmonary hypertension which warrants additional study.
Introduction
Pulmonary arterial hypertension (PAH) is a disease with high morbidity and mortality that affects about 15 persons per million (1). Despite recent advances in diagnosis and therapy, substantial delays remains from onset of symptoms to diagnosis of PAH persist and mortality is nearly 15% at one year (2,3).
Resting transthoracic echocardiography is a fundamental diagnostic and screening test for pulmonary hypertension (PH) although right heart catherization (RHC) remains the gold standard for diagnosis. Right ventricular (RV) parameters obtained at rest by two-dimensional (2D), M-mode, and Doppler echocardiography have been validated extensively and recently reviewed (4–6). Resting echocardiographic parameters with diagnostic value in PH include right ventricular systolic pressure (RVSP), tricuspid regurgitation jet velocity, RV outflow tract acceleration time, PA diastolic pressure by pulmonary regurgitation-end velocity, tricuspid annular systolic myocardial velocity, RV isovolumic relaxation time, right atrial and RV dilation, and RV myocardial performance index (7–10).
There currently are limited data on RV physiology during exercise and the role of exercise stress echocardiography (ESE) in the diagnosis of PH is much less well established. It has been shown that pulmonary arterial systolic pressure (or RVSP) increases modestly during exercise in “normal” individuals and significantly more so in athletes (11–13). Patients with PH exhibit a greater increase in RVSP compared with the healthy population (14–16). There is no single upper limit of normal for RVSP during exercise as individuals may have an elevation of RVSP due to abnormal pulmonary vascular resistance (e.g. due to PH) or high pulmonary vascular flow (e.g. high performing athletic individuals) (17,18).
Recent studies have focused on ESE indirect measurements of PVR through mean pulmonary arterial pressure (mPAP)-to-workload (mPAP/W) and mPAP-to-cardiac output (mPAP/CO) ratios and evaluated these against similar RHC measurements (19,20). Other parameters such as tricuspid annular plane systolic excursion (TAPSE) and tricuspid annular systolic velocity have been found to be reliable measures of RV response during exercise (21). However, limitations to obtaining these measurements include feasibility, variability of baseline normal values among different populations, inter-rater reliability, and reproducibility of measurements. We hypothesized that recording observed changes in RV morphology during exercise might be simpler and more reproducible measures of pulmonary vascular pathophysiology.
We sought to study the right ventricle during recumbent exercise stress echocardiography with the goals of: (1) evaluating test safety, feasibility, and reproducibility; (2) quantifying changes in echocardiographic RV size parameters during exercise in healthy controls compared to a cohort of well-defined PH patients; and (3) assessing the potential diagnostic utility of those changes in identifying PH.
Methods
Study population and design
This study was approved by the Maine Medical Center Institutional Review Board (IRB: 958764-3,959072-2). Individuals were aged 18–65 years, capable of exercising on a recumbent bicycle, and could understand and freely give informed consent. Healthy controls were recruited from our surrounding geographic region. Activity levels for healthy individuals were defined per American Heart Association (AHA) 2007 guidelines as minimal (less than the recommended 150 min of moderate aerobic activity weekly), moderate (150 min of moderate aerobic activity weekly), or intense (>150 min of moderate aerobic activity weekly) (22–25). Individuals with PH confirmed by a historic RHC were recruited through the Maine Pulmonary Hypertension Program. PH individuals were required to be in WHO Functional Class (FC) groups I–III and on a stable medical regimen.
Exclusion criteria were pregnant women, prisoners, current acute illness, current tobacco use, chronic alcohol abuse (>3 drinks/day for men and >2 drinks/day for women), and history of illicit drug use. Additional exclusion criteria for healthy controls were any previously known chronic cardiopulmonary medical conditions and use of any chronic prescription medications except oral contraceptives. Additional exclusion criteria for PH patients were WHO FC IV, unstable PH symptoms, and any history of exertional angina or syncope.
Exercise protocol
All exercise tests were performed on a semi-recumbent cycle ergometer (Medical Positoning, Kansas City, MO, USA). The ergometer table was tilted to 20° to optimize echocardiography image acquisition. Resting echocardiography was first obtained and all individuals were then exercised at incremental stages of 25 W workloads every 3 min. Exercise echocardiographic images were recorded during peak exercise. Safety measures were followed per ACC/AHA guidelines (26). Tests were terminated early for chest pain, fatigue, light headedness, dyspnea, hypoxemia, or significant EKG changes. The electrocardiogram (EKG) and pulse oxygen saturation were continuously monitored. Heart rate (HR) and blood pressure (BP) were recorded at each stage. A test was considered complete when a rate-pressure product (RPP) of 20,000 mmHg/min was reached. Individuals were observed during post-procedure recovery until their HR was <100 beats per minute, exertional symptoms had resolved, and there were no diagnostic EKG changes.
Echocardiography protocol
A Phillips IE 33 Ultrasound (Phillips Corporation, Andover, MA, USA) was used for imaging. A staff cardiologist, cardiac sonographer, and exercise technician were present for each test. All echocardiographic studies were performed by an experienced sonographer. Resting and exercise echocardiographic 2D, M-mode, and Doppler recordings were obtained using a S 5-1 multi-Hertz phased array transducer as previously described in standard fashion (4,27,28). Resting cardiac images were first recorded, using the apical four-chamber view. RV imaging focused on maximizing the RV size and maintaining the entire RV in view. Once the resting images were acquired and optimal probe positioning noted, the test participant began to exercise per protocol. During recumbent exercise, individuals were instructed to inform the exercise staff as they felt they were approaching their maximal ability and echocardiographic images were again acquired during this point in exercise along with recording the BP, HR, and wattage achieved.
Following exercise, two independent cardiologists masked to clinical information subsequently analyzed the recordings. The RV echocardiographic parameters were chosen based on the American Society of Echocardiography guidelines and included morphometric parameters of RV basal, middle, and longitudinal diameters (RVBD, RVMD, and RVLD, respectively), RV end-diastolic and end-systolic areas (RVEDA and RVESA, respectively). Analyzing the apical four-chamber view, the right ventricle was focused on to obtain the maximum size measurements. Tracings of RV internal diameters avoided trabeculations and excluded the moderator band. To obtain the RVLD, the distance from the tricuspid annulus to the RV apex was measured in diastole. To obtain the RVMD, the distance from middle of the interventricular septum (inferoseptum) to the opposing wall (RV lateral) was measured at the level of the papillary muscles in diastole. To obtain the RVBD, the distance from the inferoseptum to the RV lateral wall was measured at the level of the basal one-third of the ventricular cavity during diastole. Tricuspid regurgitation velocity (TRV) and tricuspid annular plane systolic excursion (TAPSE) were obtained as previously described (4).
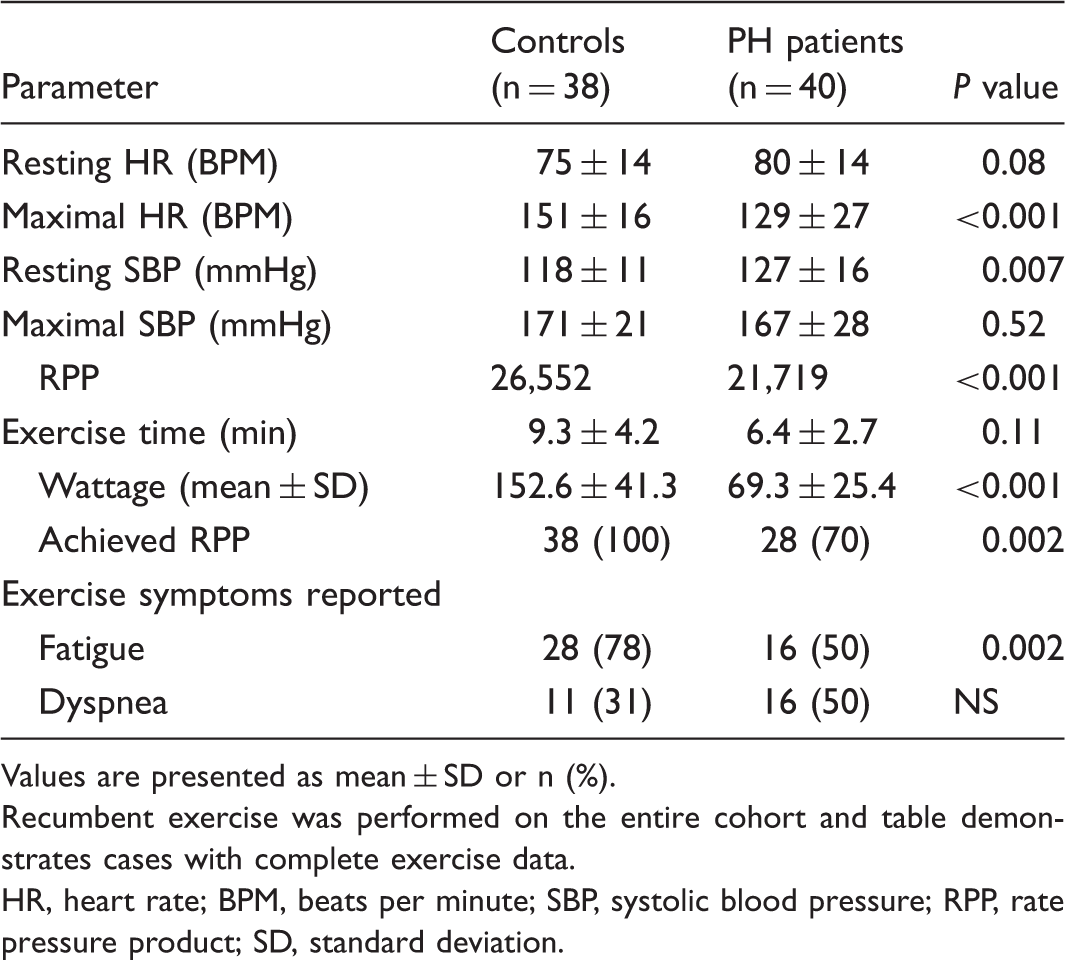
Statistical analysis
Normally distributed continuous variables are presented as mean ± standard deviation and group differences evaluated using Student’s t-test for statistical significance (defined as P < 0.05). Non-normally distributed continuous data are presented as medians and ranges and group differences analyzed by Mann–Whitney U test for statistical significance (defined as P < 0.05). Ordinal data are presented as mode and ranges. The intra-class correlation coefficient (ICC) was used to assess inter-rater reliability for continuous measures and the Kappa statistic to assess inter-rater reliability for categorical measures. Only echocardiographic parameters with acceptable inter-rater reliability were analyzed to determine statistical differences. Receiver operating characteristic (ROC) curves were generated to assess the relative ability of each exercise echocardiographic RV size parameter to predict PH.
Results
Demographics and clinical characteristics
Study population clinical demographics.

Values are presented as n (%) or mean ± SD.
AHA, American Heart Association; APAH, associated pulmonary arterial hypertension; BMI, body mass index; ERA, endothelin receptor antagonist; HPAP, hereditary pulmonary arterial hypertension; IPAH, idiopathic pulmonary arterial hypertension; WHO, World Health Organization; PDE5I, phosphodiesterase-5 inhibitor; PG, prostacyclin agonist (parenteral epoprostenol or treprostinil); SD, standard deviation; FC, functional class; 6MWD, 6-min walking distance; RAP, right atrial pressure; mPAP, mean pulmonary arterial pressure; PCWP, pulmonary capillary wedge pressure; CO, cardiac output.
All but three of the PH patients had PAH and were on PAH-specific therapy (Table 1). Those receiving PAH therapy were treated for 1–44 months at the time of ESE testing. None of the PH patients required supplemental oxygen at rest or for exercise. Test participant demographics, WHO FC, and AHA exercise assignments, six-minute walking distance (6MWD) tests, and RHC hemodynamic findings from the most recent catheterization are shown in Table 1.
Recumbent exercise stress echocardiogram safety and feasibility
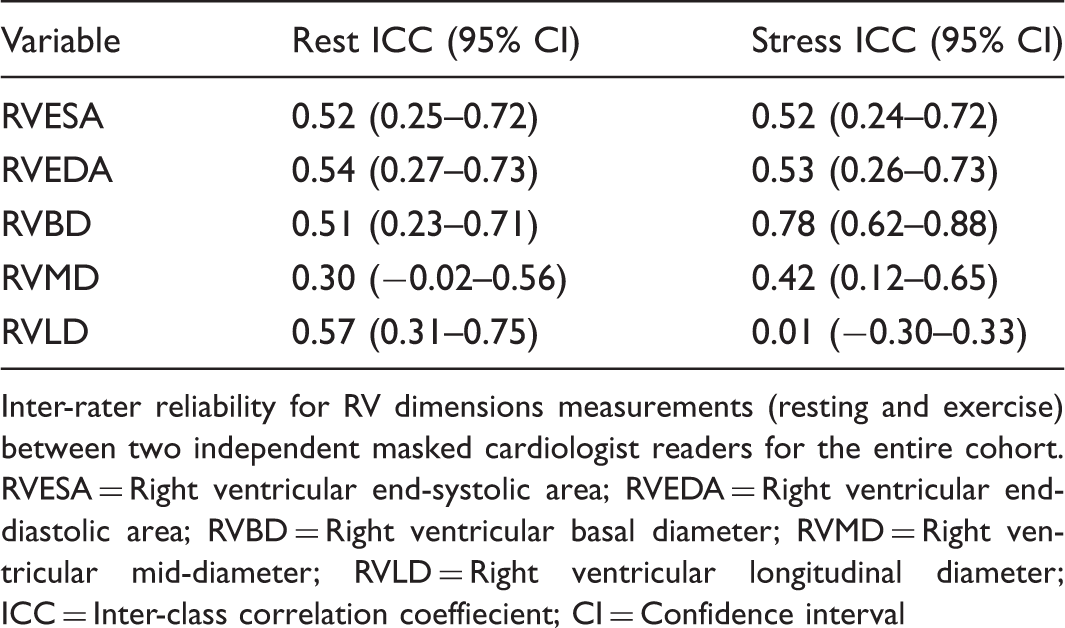
All participants were able to exercise per protocol. The targeted RPP was achieved by 28/40 PH patients and 38/38 controls. No tests were stopped early for chest pain, hypoxemia, EKG changes, or hemodynamic instability. No serious adverse events were encountered. Individuals exercised for up to 15 min and all testing procedures were completed within 1 h. Exercise time appeared less in the PH group while RPP and wattage attained were significantly lower (Table 2). We were able to adequately obtain 90% of the sought RV echocardiographic parameters in PH patients and 95% in controls. There were no significant differences in the ability to acquire adequate images between resting and exercise states. Representative echocardiographic images of the cardiac four-chamber view during rest and exercise focusing on the right ventricle are shown in Fig. 1. The inter-observer reliability between two independent readers’ measurements for the RV size parameters during exercise was acceptable for each RV parameter with the exception of RV longitudinal diameter (Table 3).
Measurements of RVESA and RVEDA during rest and exercise. Typical four-chamber echocardiographic images for a representative subject with PH of RV systole (a) and diastole (b) at rest. Peak exercise images of RV systole (c) and RV diastole (d) are notable for RV dilation relative to the resting state. Recumbent exercise testing safety and feasibility. Values are presented as mean ± SD or n (%). Recumbent exercise was performed on the entire cohort and table demonstrates cases with complete exercise data. HR, heart rate; BPM, beats per minute; SBP, systolic blood pressure; RPP, rate pressure product; SD, standard deviation. Intra-class correlation coefficients (ICCs) for inter-rater reliability for RV size parameters at rest and during exercise. Inter-rater reliability for RV dimensions measurements (resting and exercise) between two independent masked cardiologist readers for the entire cohort. RVESA = Right ventricular end-systolic area; RVEDA = Right ventricular end-diastolic area; RVBD = Right ventricular basal diameter; RVMD = Right ventricular mid-diameter; RVLD = Right ventricular longitudinal diameter; ICC = Inter-class correlation coeffiecient; CI = Confidence interval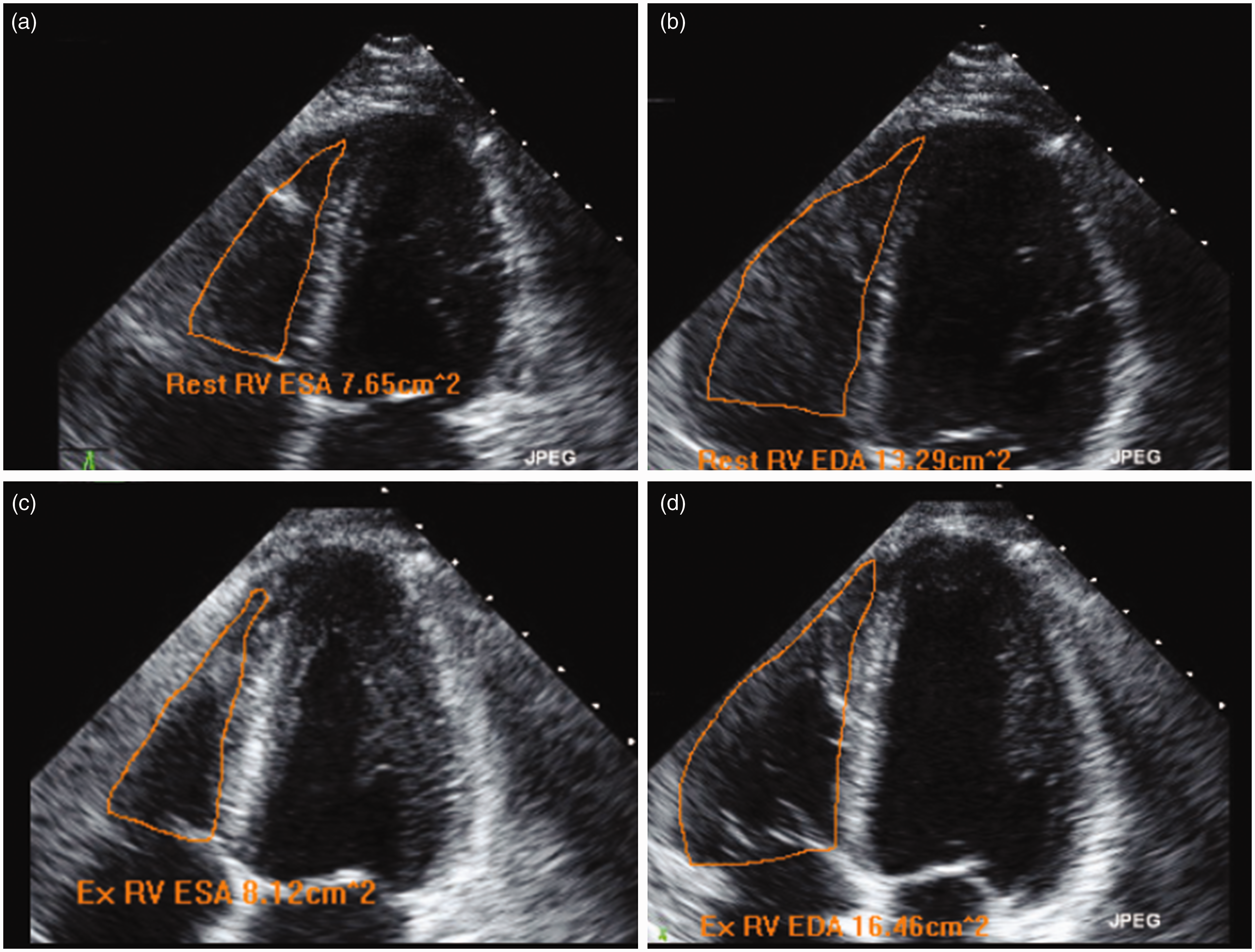
Changes in RV parameters during recumbent exercise
RV size parameters at rest and during exercise in healthy controls and PH patients.
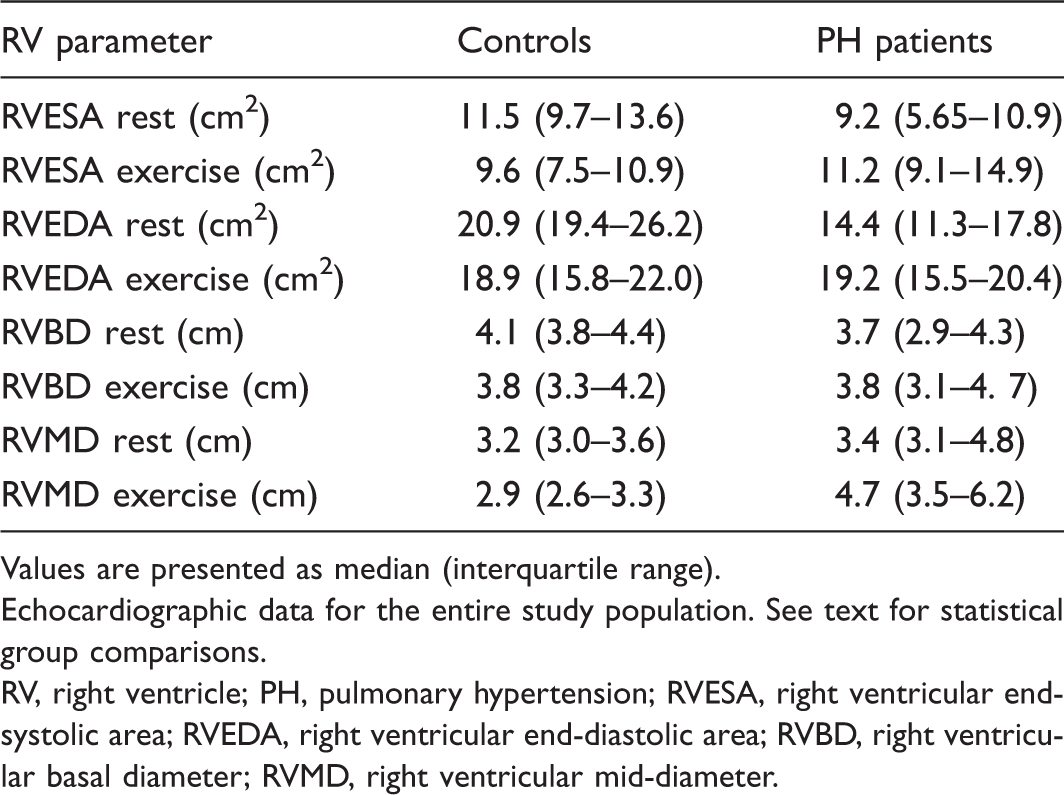
Values are presented as median (interquartile range).
Echocardiographic data for the entire study population. See text for statistical group comparisons.
RV, right ventricle; PH, pulmonary hypertension; RVESA, right ventricular end-systolic area; RVEDA, right ventricular end-diastolic area; RVBD, right ventricular basal diameter; RVMD, right ventricular mid-diameter.
During exercise, healthy controls demonstrated a significant decrease in RVESA, RVEDA, RVMD, and RVBD (P < 0.05). Conversely, PH patients demonstrated a significant increase in RVESA, RVEDA, and RVMD (P < 0.05), which we term “transient exertion dilation” (TED). At peak exercise, PH patients had a significantly larger RVMD measurement compared to controls (P < 0.0001). There were no significant differences in the absolute RV dimensional measurements between the two groups for the other parameters at peak exercise. These RV parameter changes during exercise were unaffected by adjustments for height, BSA, and wattage achieved. Box plots for exercise-related changes in each RV size parameter are shown in Fig. 2. Two-dimensional representations of healthy control and PH RV size changes during peak exercise are presented in Fig. 3.
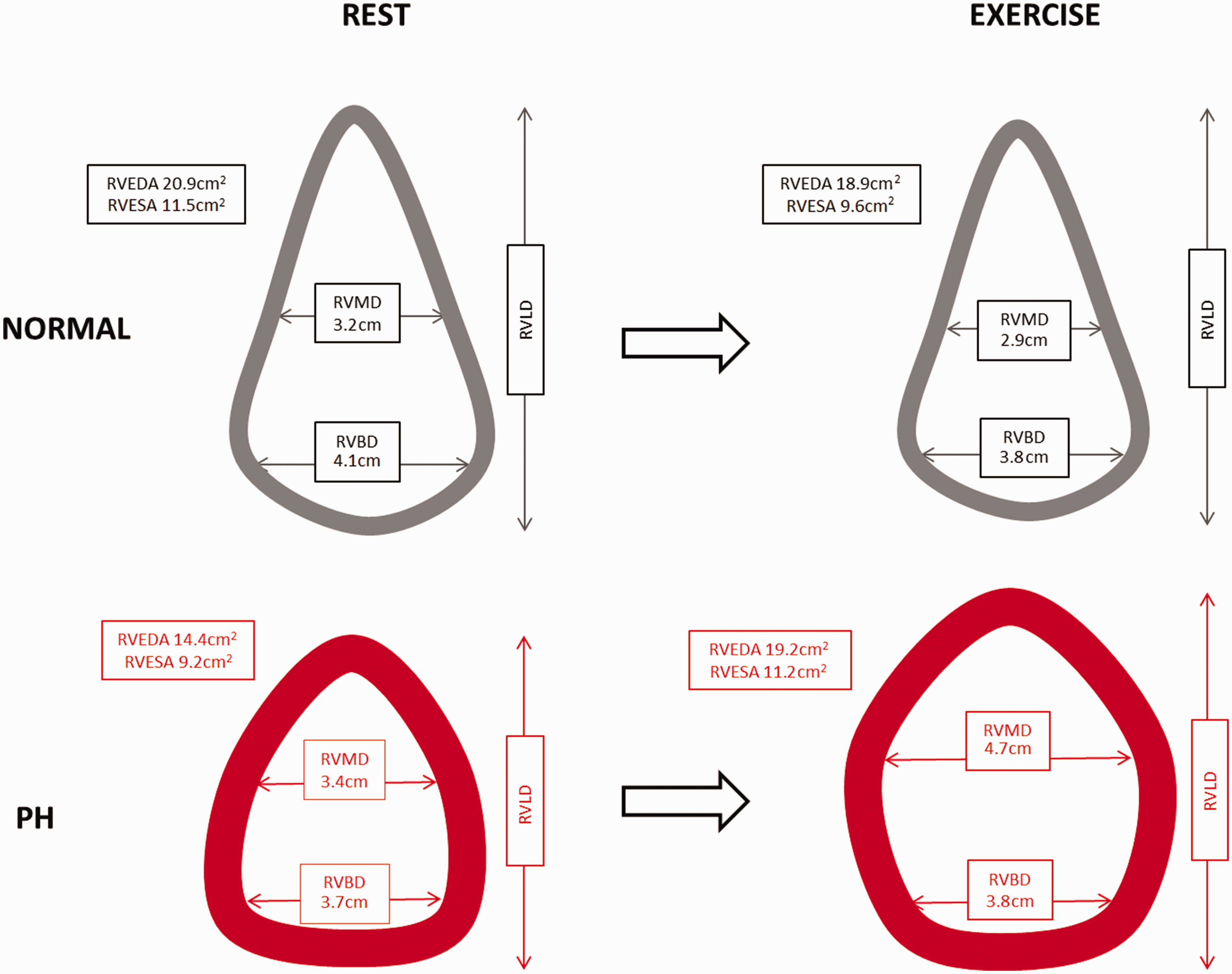
Boxplot representation of changes in RV size parameters during exercise in normal and PH patients. Normal individuals (gray boxes) generally demonstrate a reduction in RV size measurements during exercise while PH patients (red boxes) generally demonstrate an increase in RV size measurements during exercise. RVESA, right ventricular end-systolic area; RVEDA, right ventricular end-diastolic area; RVBD, right ventricular basal diameter; RVMD, right ventricular mid-diameter. Median changes in RV size parameters from rest to maximal exercise in healthy controls and PH patients. Healthy controls (gray figures) and PH patients (red figures) show opposite directional median size changes during exercise as measured by recumbent ESE. Note all size dimensions decrease in controls while all increase in PH patients. RV, right ventricle; PH, pulmonary hypertension; RVESA, right ventricular end-systolic area; RVEDA, right ventricular end-diastolic area; RVBD, right ventricular basal diameter; RVMD, right ventricular mid-diameter; RVLD, right ventricular longitudinal diameter (data not shown). Numbers represent median values for each reproducible parameter. See Fig. 2 for additional details.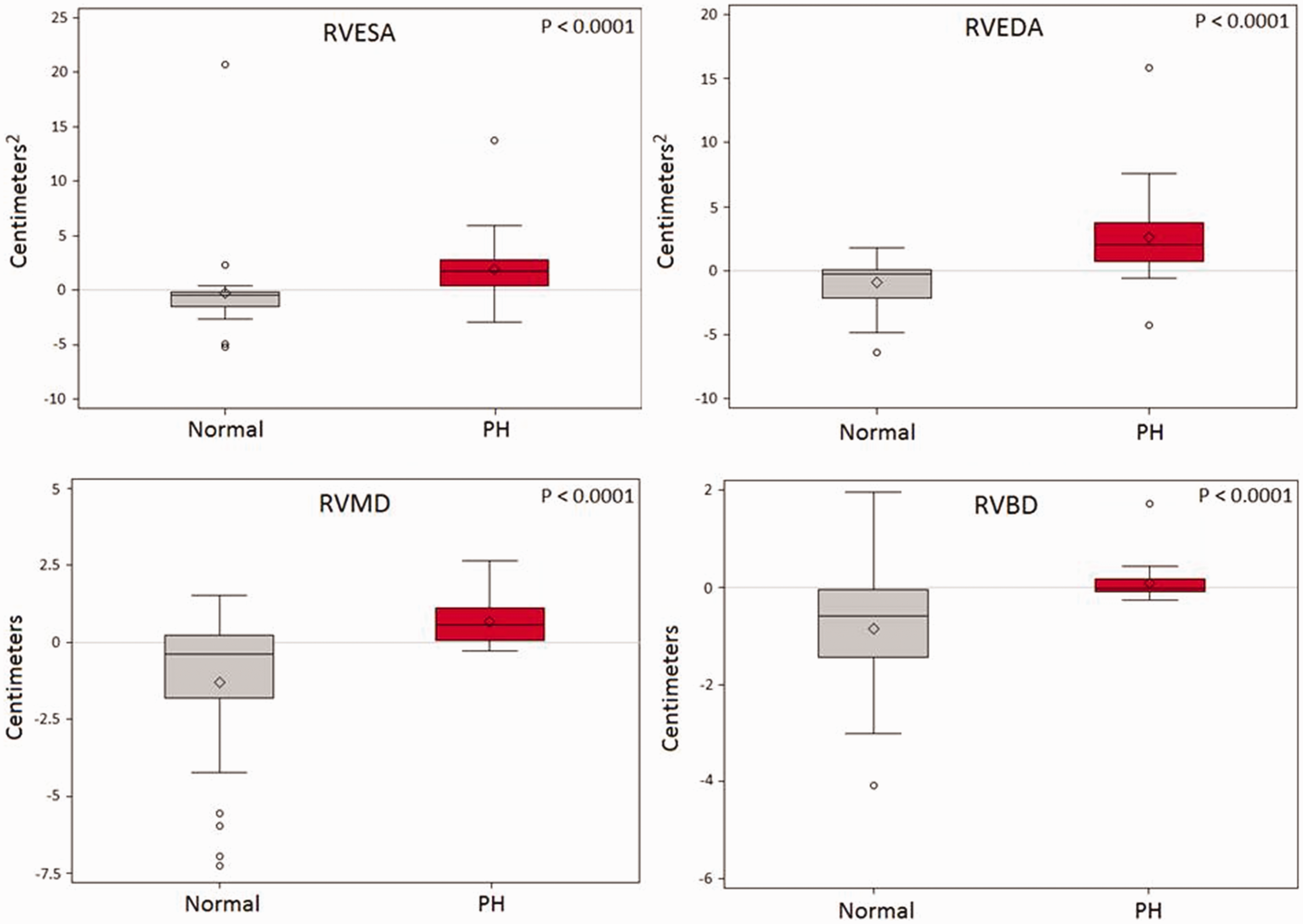
The resting TRV in the control cohort was 2.0 ± 0.36 m/s and 3.4 ± 0.97 m/s in the PH cohort (P < 0.001). The exercise TRV in the control cohort was 2.36 ± 0.45 m/s and 4.5 ± 1.4 in the PH cohort (P < 0.001). The change in TRV between rest and exercise was significantly greater in the PH group (P < 0.001). These data support that RV afterload is higher at rest in the PH cohort (as expected) but also rises disproportionately in PH individuals during exercise.
The resting FAC in the healthy control cohort was 0.46 ± 0.09 and 0.40 ± 0.11 in the PH cohort (P < 0.05). The exercise FAC in the control cohort was 0.49 ± 0.09 and 0.37 ± 0.10 in the PH cohort (P < 0.001). The change in FAC between rest and exercise was not statistically different between rest and exercise in either group.
The resting TAPSE in the control cohort was 2.6 ± 0.50 cm versus 2.3 ± 0.48 cm in the PH cohort (P < 0.01). The exercise TAPSE in the control cohort was 4.0 ± 3.2 cm versus 2.4 ± 0.6 in the PH cohort (P < 0.01). There was a significant increase in TAPSE during exercise in the control group (P < 0.01); however, this was not seen in the PH cohort. These data show PH subjects have a limited ability to augment RV ejection fraction during exercise in the setting of increasing RV afterload.
Recumbent RV exercise stress echocardiography test characteristics in detecting pulmonary hypertension
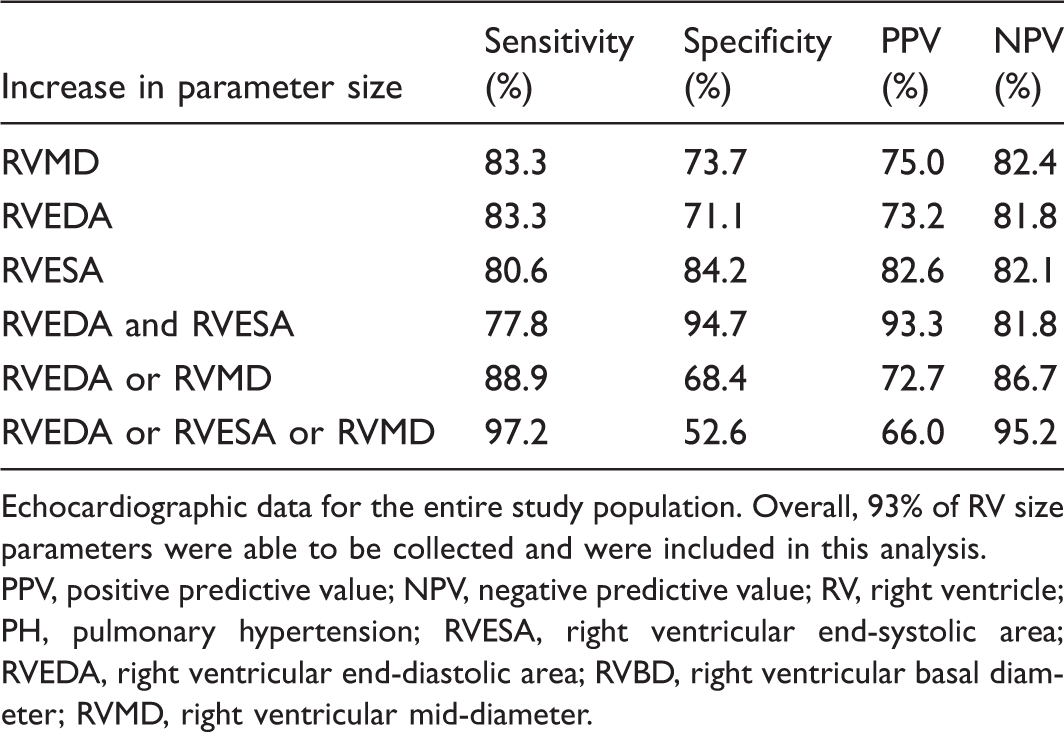
The overall sensitivity of TED (increase in any of the RV parameters) to detect pulmonary hypertension was 97.2% with a negative predictive value of 95.2%. The specificity of an increase in right ventricular EDA and ESA was 94.7% with a positive predictive value of 93.3%. A sensitivity-specificity analysis for RV parameters is shown in Table 5. The ROC curve C-statistic for an increase of any RV parameter to detect PH was 0.93 (Fig. 4).
ROC curve of RV TED observed during exercise echocardiography. The ROC curve analysis of increase in any of RVMD, RVEDA, RVESA, or RVLD size parameters to detect PH. RVESA, right ventricular end-systolic area; RVEDA, right ventricular end-diastolic area; RVBD, right ventricular basal diameter; RVMD, right ventricular mid-diameter; RVLD, right ventricular longitudinal diameter. Sensitivity, specificity, PPV, and NPV of increases in RV size parameters during exercise for identifying PH. Echocardiographic data for the entire study population. Overall, 93% of RV size parameters were able to be collected and were included in this analysis. PPV, positive predictive value; NPV, negative predictive value; RV, right ventricle; PH, pulmonary hypertension; RVESA, right ventricular end-systolic area; RVEDA, right ventricular end-diastolic area; RVBD, right ventricular basal diameter; RVMD, right ventricular mid-diameter.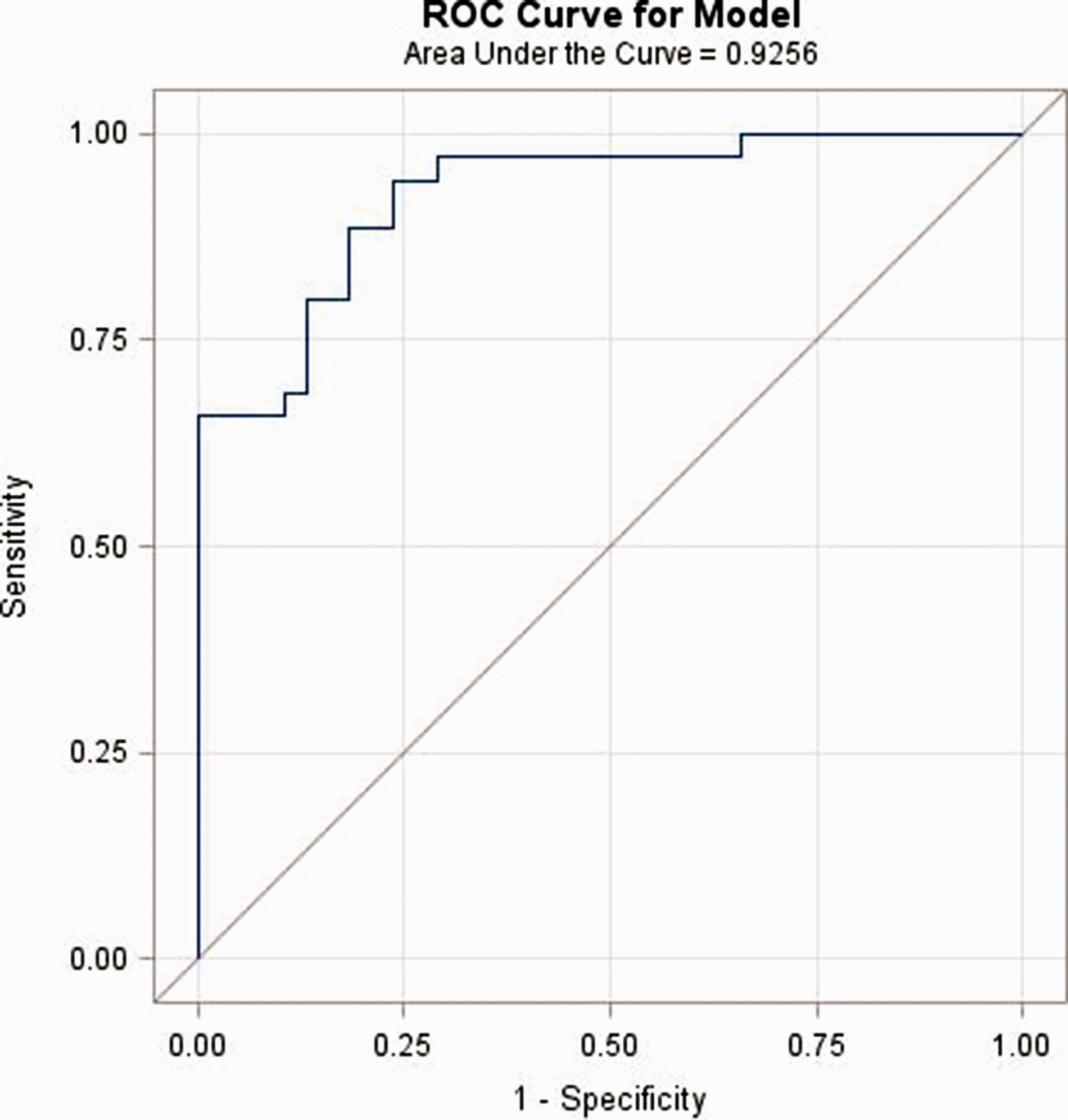
An analysis of the PH patients with normal resting RVSP (n = 14) versus those with elevated resting RVSP (n = 26) showed RV TED was unrelated to the severity of PH. Our PH patients with normal resting echocardiographic pulmonary pressures (resting TRV < 2.9 m/s) had RV TED at a similar frequency and magnitude as PH patients with resting TRV > 2.9 m/s (see Suppl. Figs. S1–S4). Reviewing all individuals with a normal resting RVSP, RV TED had a calculated sensitivity of 0.87 (95% confidence interval [CI] = 0.56–0.97) and specificity of 0.97 (95% CI = 0.84–0.99) for PH. Positive and negative predictive values would be determined by the prevalence of PH within the specific population being screened.
Discussion
A major focus of PH patient advocacy organizations and clinicians is earlier diagnosis for patients affected by PH. Although RHC remains the gold standard for diagnosis, transthoracic echocardiography is a critical diagnostic and screening tool. Resting Doppler echocardiography has been extensively studied and validated (6,25,27–32). The most recent European Society of Cardiology/European Respiratory Society guidelines recommend the use of resting echocardiography as a screening tool for patients with suspected PH (31).
Despite widespread use of resting Doppler echocardiography in PH screening, the test has demonstrated inaccuracies in estimating PAPs in patients being evaluated for PH. There is significant overestimation and underestimation of RVSP in up to 20–25% of cases. Errors commonly result from suboptimal tricuspid regurgitant jet Doppler imaging and inaccurate right atrial pressure (RAP) estimations (29). To further increase diagnostic accuracy, it is recommended that additional resting echocardiographic parameters be taken into consideration including RV/LV basal diameter ratio, flattening of the interventricular septum, RV outflow tract acceleration time (RVOT), PA diastolic pressure by PR-end velocity, tricuspid annular systolic myocardial (Sm) velocity, RV isovolumic relaxation time, RA and RV dilation, and RV myocardial performance index (7–10). A difficulty facing clinicians in the practice of PH is the relative strengths and limitations of each of these tests have not been studied in a comparative fashion.
The role of ESE in PH diagnosis has not been established as there are significantly fewer studies on exercise imaging of the right ventricle. Prior studies of right heart exercise have examined changes in TRV, TAPSE, mPAP, mPAP-to-workload (mPAP/W) and mPAP-to-cardiac output (mPAP/CO) ratios (11,15,16,21,33–40). Recently, mPAP/W and mPAP/CO ratios were correlated against RHC measurements and found to be reliable (19,20). However, these measurements are often limited due to baseline physiological variability and the interdependency of pulmonary flow and pressure with cardiac output (41). Additionally, combining multiple separate echocardiographic measures will naturally compound the inherent errors of each individual measurement in the final results, limiting reproducibility.
Acute right ventricular dilation during exercise in PH patients was first demonstrated by ESE in single case reports in 2004 and 2007 (42,43). This RV dilation was observed to resolve within 40 s of stopping exercise. Ours is the first study to systematically characterize RV morphologic changes using ESE in healthy controls and PH patients. In this pilot study, we have demonstrated that ESE is feasible and safe. All of our participants completed the test without adverse events and >90% of the sought RV echocardiographic parameters were obtained. Imaging acquisition was reproducible and demonstrated acceptable inter-observer reliability between independent cardiologist readers for key measures. Observing directional changes in RV size parameters during exercise (as opposed to obtaining specific RV size measurements) is simple, repeatable, and has high test reliability.
We sought to determine if RV exertional dilation is a pathologic or physiologic response to exercise. Claessen et al. previously studied eight healthy individuals at rest and during exercise using real-time cardiac magnetic resonance to study RV and LV volumes (44). They found that both RVEDV and RVESV of healthy individuals declined when performing exercise > 25 W, with greater declines seen in RVESV than RVEDV. While there were demonstrable respiratory variations in RV volumes between inspiration (larger) and expiration (smaller) termed the “respiratory pump” effect, these respiratory variations were significantly smaller than exercise effects. We were also able to demonstrate reduction in RV diameters and cross-sectional areas during exercise in normal controls using recumbent ESE. A second cardiac MRI study by Jaijee et al. to assess RV responses to exercise in PAH showed similar findings to ours. They studied 14 PAH patients and 34 healthy controls using exercise MRI and also showed significant exercise-induced increases in RVEDV and RVESV indexes in the PAH patients and corresponding decreases in RV volumes in the controls (45). Transient exertional dilation (TED) of the right ventricle is a pathologic finding related to increased pulmonary afterload (exercise TRV or RVSP) coupled with RV functional limitations in contractility (reduced exercise FAC and TAPSE). Conversely, healthy controls demonstrate a decrease in RV size measurements during exercise as a result of reduced diastolic filling time and increased contractility.
As an overall screening tool for PH, the absence of RV TED exhibited a high sensitivity of 97% (negative predictive value = 95%). Additionally, finding an increase in both RVEDA and RVESA had carried a high specificity of 95% for diagnosis of PH (positive predictive value = 93%). Recumbent exercise-stress RV echocardiography could have a role in evaluating patients with unexplained exertional dyspnea and nondiagnostic resting echocardiographic findings (e.g. resting RVSP near the upper limits of normal with normal RV size and function, or those with abnormal RV size and function with unmeasurable TRV/RVSP). Although we did not study individuals with unexplained exertional dyspnea, we did note that RV TED was not related to the resting RVSP in the PH patients. PH patients with normal RVSP were found to have RV TED at the same frequency and magnitude as those with elevated resting RVSP. However, we note that these individuals may have had other signs of PH on resting echocardiograms.
Another problematic area in using RV ESE to screen for PH is in evaluation of athletic individuals in whom RVSP may rise significantly during exercise. To examine this issue, our “high performers” healthy controls were reviewed (defined as those with a peak exercise wattage ≥200 W, or with the highest exercise RVSP, or highest increase in RVSP during exercise). These subjects rarely demonstrated exertional RV dilation and RV TED was not more frequent in this group compared with the remainder of the healthy control group (see Supplemental Table S1). The specificity and sensitivity of RV ESE was enhanced by combining multiple RV measurements to assess for RV TED in this situation.
Overall, the diagnostic sensitivity and specificity of RV TED for PH is well in line with the performance characteristics of other clinically used screening tests. Demonstration of RV TED supports an invasive hemodynamic assessment of pulmonary hemodynamics should be considered. Conversely, the absence of RV TED during such a study is reassuring to both the patient and clinician that significant PH is unlikely to be present.
There were several limitations to our study. Firstly, most of our PH patients had diagnostic group 1 PH (PAH) and few other etiologies of PH were included in this study. We have no data in WHO Group 2, 3, or 4 patients and cannot yet say if these finding are specific to precapillary PH. There are no current studies evaluating right heart parameter comparing pre- and post-capillary PH cohorts using exercise echocardiography. Determination of LVEDP at rest (and often during exercise) remains of critical importance. Future studies of RV ESE comparing HFpEF and PH/PAH individuals are needed.
Secondly, our PH patients had well established PH and it is unclear to what extent RV TED is seen in exercise-induced PH. However, as noted above, exercise-related RV dilation appears independent of PH severity and is very rarely observed in high performing healthy individuals.
Finally, our finding of smaller RVESA and RVEDA measurements at rest in the PH patients compared with the healthy controls was surprising. However, Jaijee et al. also found smaller resting RVEDV index in the PAH patients than in the controls (88.1 ± 2.5 mL/m2 vs. 89.6 ± 2.7 mL/m2) by cardiac MRI (45). The PAH patients had a slightly larger RVESV index at rest but with significantly wider confidence intervals for each (40.1 ± 9.5 vs. 38.4 ± 8.5 mL/m2). This finding could be explained by longstanding PH individuals having reduced inner RV chamber size due to RVH. Additionally, all individuals cycle in a semi-recumbent position with legs partially elevated which will increase RV preload. This increased preload might account for the slightly larger RVEDV in healthy participants due to higher RV compliance compared with the PH individuals. We noted that the baseline RV dimensions were not related to presence or absence of RV TED and advocate focusing on RV dimension directional changes over absolute size measurements when using ESE.
In conclusion, recumbent ESE is a feasible and safe outpatient diagnostic test. Recumbent RV ESE produces reliable RV size measurements in a practical timeframe when performed by trained personnel using a standardized protocol and instrumentation. We believe recumbent exercise stress RV echocardiography requires additional study in a wider diversity of individuals and test standardization before clinical adoption, however holds promise as a diagnostic testing modality for pulmonary hypertension.
Supplemental Material
Supplemental material for Pulmonary hypertension subjects exhibit right ventricular transient exertional dilation during supine exercise stress echocardiography
Supplemental Material for Pulmonary hypertension subjects exhibit right ventricular transient exertional dilation during supine exercise stress echocardiography by Rama El-Yafawi, David Rancourt, Melkon Hacobian, Dennis Atherton, Mylan C. Cohen and Joel A. Wirth in Pulmonary Circulation
Footnotes
Acknowledgments
The authors thank Lesya Belusov (posthumously) and Julie Hopkins for assistance with data acquisition and John Dzio-Dzio for assistance with figure preparation and data analysis.
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Maine Medical Center Research Institute.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
