Abstract
Reperfusion pulmonary edema (RPE) is a common complication after pulmonary endarterectomy (PEA) in patients with chronic thromboembolic pulmonary hypertension (CTEPH). However, the precise mechanisms underlying the development of RPE remain unclear. To evaluate the effects of pulmonary vasculopathy on RPE, the severity of the pulmonary arteriopathies and venopathies of lung tissues biopsied during PEA were pathologically quantified in 33 CTEPH patients. The severity of RPE was classified from grade 0 (no RPE) to 4 (death due to RPE) based on the arterial oxygen tension/inspiratory oxygen fraction (P/F ratio) and necessity of respiratory management. Among the 33 patients (27 women; mean age = 63.3 years), 17 (51.5%) patients developed RPE. The severity of pulmonary arteriopathy (obstruction ratio) correlated with the grade of RPE (r = 0.576, P = 0.0005). The obstruction ratio also correlated with the P/F ratio (r = −0.543, P = 0.001) and the perioperative mean pulmonary arterial pressure (r = 0.445, P = 0.009). Multivariate logistic regression analysis revealed that the obstruction ratio was a significant independent determinant for the development of RPE (odds ratio = 15.7; 95% confidence interval = 2.29–108.00, P = 0.005). In conclusion, pulmonary arteriopathy could be a determinant of the development and severity of RPE after PEA.
Chronic thromboembolic pulmonary hypertension (CTEPH) is a subtype of pulmonary hypertension (PH) characterized by occlusion of pulmonary arteries due to chronic thromboemboli.1,2 Pulmonary endarterectomy (PEA) surgery, which is the main treatment for CTEPH patients, removes the chronic thrombi from the pulmonary arteries. 1 Reperfusion pulmonary edema (RPE) is a major complication of PEA. RPE is lung edema in the lung area in which the blood perfusion improved after PEA. 3 The occurrence of RPE is 8–91%,3–7 which varies by criteria of RPE.4,7 Patients with severe RPE often require prolonged mechanical ventilation and hospitalization in the intensive care unit (ICU).3–6 The mechanisms of RPE appear to include endothelial damage and enhanced vascular permeability related to reperfusion after ischemia.4,7 However, the detailed mechanisms underlying the development of RPE remain unclear.3,4
Clinically, the presence of severe PH before PEA and residual PH after PEA are considered risks factors for the development of RPE. 8 We previously reported a close association between pulmonary arteriopathy and residual PH. 9 Therefore, we hypothesized that pulmonary arteriopathy could be associated with the development of RPE. The purpose of this study was to evaluate whether pulmonary arteriopathy or venopathy is associated with the development of RPE and to understand the pathogenesis of RPE.
Methods
The Ethics Committee of Chiba University approved this study (approval no. 1221). All patients provided written informed consent.
Participants
Between December 2011 and August 2016, 51 CTEPH patients underwent PEA at Chiba University Hospital. Lung biopsy was performed in 35 patients. Of the 35 patients, two were excluded from the study because the specimens were too small for detailed pathological analysis. Thus, clinical and pathological data from 33 patients were retrospectively analyzed. Determination of a definitive diagnosis and an examination including right heart catheterization were performed at the preoperative period and a complete examination was performed one month and one year after PEA in all patients, as previously reported.9,10 The hemodynamic data soon after PEA were collected from the patients' ICU records. The hemodynamic data were measured using CCOmbo Catheters (774F75, Edwards Lifesciences, Irvine, CA, USA). The mean pulmonary arterial pressure (mPAP) in the ICU and continuous cardiac output was defined as the pressure at the onset of RPE in patients with RPE or the pressure 5–6 h after PEA in patients without RPE.
Pulmonary endarterectomy
PEA was performed by KI and MM at Chiba University Hospital. The indications for PEA were as follows 10 : (1) mPAP ≥ 25 mmHg; (2) pulmonary vascular resistance (PVR) > 300 dyne·s·cm−5; (3) World Health Organization (WHO) functional class ≥ 2; (4) technically operable; (5) no significant co-morbidity; and (6) full understanding of the benefits and the risks. The Jamieson's classification was determined during the operation by two experienced cardiovascular surgeons (KI and MM), as previously reported. 11
Reperfusion pulmonary edema
We diagnosed RPE based on the definition described by Jenkins, et al. 3 : (1) arterial oxygen tension/inspiratory oxygen fraction (P/F ratio) < 300; (2) presence of infiltrative shadow on a chest radiograph in a reperfused lung area after PEA; and (3) no obvious cause of the symptoms, such as pulmonary bleeding, atelectasis, or pneumonia. The P/F ratio analyzed in this study was the value at the diagnosis of RPE in patients with RPE or 5–6 h after PEA in those without RPE. Two trained respiratory physicians (TJ-S and AN) interpreted the chest radiography images. The weighed kappa coefficient of the inter-observer agreement was 0.9495. Anesthetists checked whether airway bleeding exist by bronchoscopy during PEA. In the current study, no patient had relevant airway bleeding. The severity of RPE was graded based on the P/F ratio and the required respiratory management, and defined as follows: grade 0 = absence of RPE; grade 1 = presence of RPE, only typical respiratory management required; grade 2 = presence of RPE, respiratory management with positive end-expiratory pressure (non-invasive positive-pressure ventilation and/or nasal high flow) required; grade 3 = presence of RPE, prolonged artificial ventilation or extracorporeal membrane oxygenation required; and grade 4 = presence of RPE related to perioperative death. To prevent reperfusion lung edema, we used low dose of catecholamine to avoid high cardiac output and maintained water balance negative with aggressive use of diuretics. The patients were extubated as early as possible if oxygenation was maintained with PaO2 > 80 mmHg. The extubation was typically conducted on postoperative day 1 and then oxygen was supplied by venturi masks or nasal cannulas. Non-invasive positive-pressure ventilation was applied if PaO2 > 60 mmHg was not achieved with the venturi mask. The requirement of other respiratory management depended on the decision of the cardiovascular surgeons and clinicians in the ICU.
Lung biopsy
Lung specimens were biopsied from the right middle lobe or left lingular segment during PEA. Lung biopsy was limited to a single sample for safety and ethical reasons. In this study, pneumothorax that was potentially associated with the lung biopsy occurred in two of 33 patients (6.1%); both patients improved within a few days following thoracic drainage. The biopsied lung tissue was prepared based on methods described previously.9,10 In total, 781 slices were prepared, stained using the Elastica-van Gieson method, and analyzed pathologically, as described in the following section.
Quantification of pulmonary arteriopathy and venopathy
Two trained investigators (TJ-S and HI-U) conducted a pathological evaluation of pulmonary arteriopathy and venopathy based on methods described previously.9,10 All pulmonary muscular arteries with adjacent bronchioles were recorded. The inclusion criteria of pulmonary arteries in the current study were as follows: (1) the horizontal-to-vertical ratio of these vessels was <1:2; and (2) the vascular diameter was <300 µm. The ratios of the intimal and medial area to the vascular area of the pulmonary arteries were evaluated in all arteries and the obstruction ratio of each patient was defined as the average of the ratios. Pulmonary veins were identified based on the following criteria: (1) vessels running into the interlobular septa; and (2) vessels with thick external elastic layers and unclear internal elastic layers. The extent of remodeling of each pulmonary vein (PV) was scored as follows: 0 = almost normal; 1 = slight; 2 = mild (<50% obstruction); 3 = moderate (>50% obstruction and/or muscularization); and 4 = severe (> 80% obstruction). The PV score of each patient was defined as the average of the scores.
Quantification of segmental pulmonary thrombi
The extent of the segmental pulmonary arterial thrombi was evaluated using enhanced computed tomography (CT) based on methods described previously. 10 Each segmental pulmonary artery was scored as follows: 0 = no thrombi; 1 = narrowed but contrast medium passed; or 2 = obstructed. The average of the scores obtained from each patient was defined as the segmental obstruction index. Two trained investigators (TJ-S and AN) examined a total of 91 CT images in a blind manner. There was a significant correlation between the scores of the two investigators (r = 0.888, P = 9 × 10−32).
Quantification of bronchial arteries
Recently, it was reported that transcatheter occlusion of the bronchial arteries (BAs) before PEA might prevent RPE. 12 In order to evaluate the relationship between the enlargement of the BAs and the development of RPE, the size of the bronchial arteries was quantified using enhanced CT scans based on the methods used in our previous study. 13 The diameter of each BA was measured at its origin and the cross-sectional area was calculated. The sum of the cross-sectional areas in each patient was defined as the total area of the BAs.
Statistical analysis
Continuous variables are described as the mean ± standard deviation unless otherwise stated. Correlations between variables were evaluated using Spearman's rank correlation coefficient. The time-dependent change of the variables was evaluated using the Wilcoxon signed rank test. Differences between three or more groups were evaluated using the Kruskal–Wallis test, adjusted using the Bonferroni method for multiple comparisons. Logistic regression analysis was performed to evaluate the factors associated with the development of RPE. A P value < 0.05 was considered significant. All data were analyzed using EZR (ver. 1.37, Saitama Medical Center, Jichi Medical University, Saitama, Japan). 14
Results
Patient characteristics
The characteristics of the 33 patients are shown in Table 1. One patient died 15 days after PEA due to sudden respiratory failure. Though the cause of death was unclear, there was no RPE during the perioperative period. mPAPs for the four periods are shown below: before PEA = 46.3 ± 10.7 mmHg; in the ICU = 28.5 ± 7.2 mmHg; one month after PEA = 25.7 ± 8.6 mmHg; and one year after PEA = 26.5 ± 8.5 mmHg. In comparison with the PAP before PEA, the mPAP significantly decreased at all of the following time points: in the ICU (P = 1.4 × 10−9); one month after PEA (P = 5.8 × 10−11); and one year after PEA (P = 1.8 × 10−11) (Fig. 1a). However, there was no statistical difference in the mPAP between the three periods after PEA: in the ICU versus one month after PEA (P = 0.3); in the ICU versus one year after PEA (P = 0.6); and one month and one year after PEA (P = 1.0) (Supplemental Table). The mPAP was positively correlated with those of one month and one year after PEA: one month after PEA (r = 0.652, P = 0.001); and one year after PEA (r = 0.611, P = 0.0003) (Fig. 1b and 1c).
Mean pulmonary arterial pressure (mPAP) before and after pulmonary and endarterectomy (PEA). (a) The change of mPAP before and after PEA. *P < 0.05 vs. preoperative mPAP. (b) The correlation between the mPAP in the intensive care unit (ICU) and 1 month after PEA (r = 0.562, P = 0.001). (c) The correlation between the mPAP in the ICU and one year after PEA (r = 0.611, P = 0.0003). Patient characteristics. PEA, pulmonary endarterectomy; mPAP, mean pulmonary arterial pressure; PVR, pulmonary vascular resistance; CI, cardiac index; PAWP, pulmonary arterial wedge pressure; ICU, intensive care unit; PV, pulmonary vein; BA, bronchial artery; SGOI, segmental obstruction index.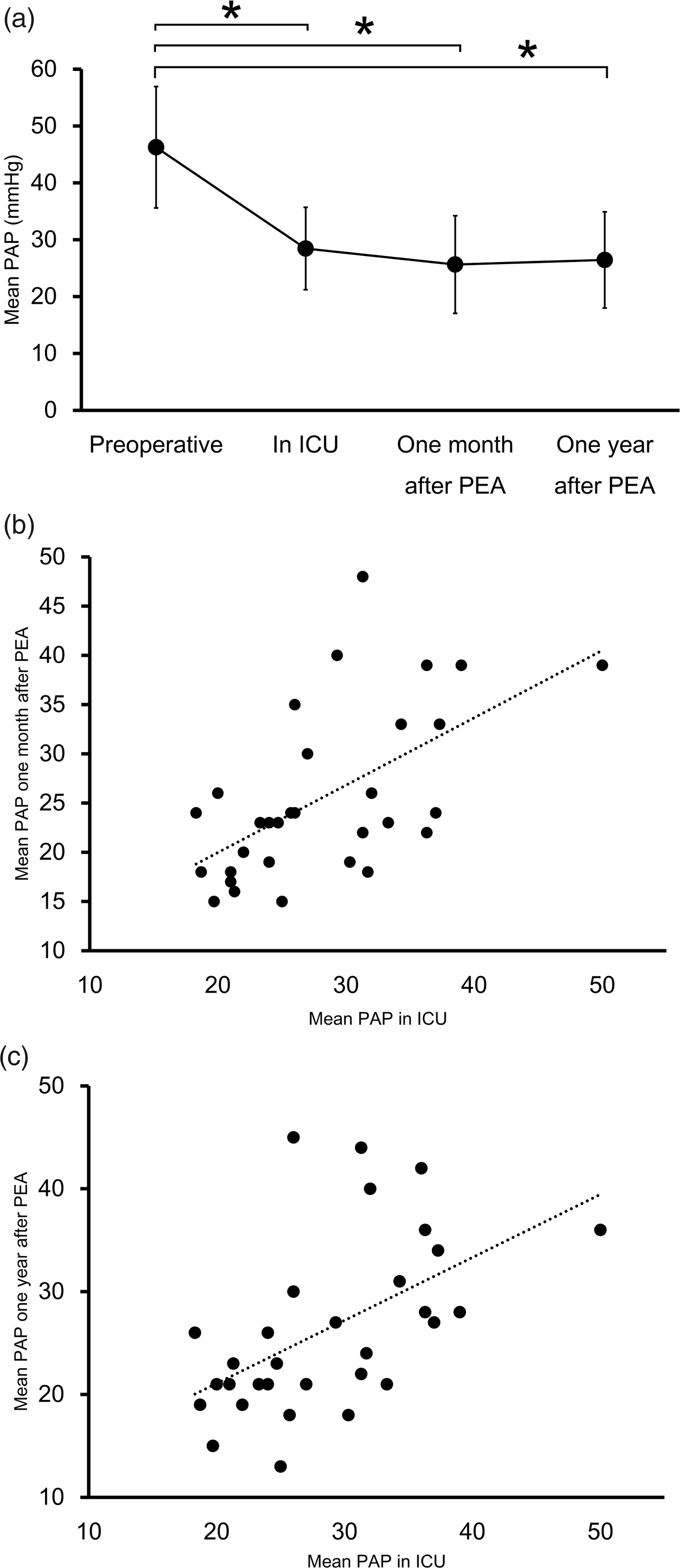

Reperfusion pulmonary edema
All infiltrations were found in lung areas from which chronic thrombi were removed. RPE occurred in 17 of 33 cases (51.5%). The distribution of the RPE grade was as follows: grade 0 = 16 cases (48.5%); grade 1 = two cases (6.1%); grade 2 = 10 cases (30.3%); grade 3 = five cases (15.2%); and no patients had grade 4 RPE (Fig. 2a). The onset of RPE occurred on the day of PEA in 16 of 17 patients (94.1%) and on postoperative day 1 in one patient (5.9%). The mPAP in the ICU was positively correlated with the RPE grade (r = 0.657, P = 0.00003, Fig. 2b).
Reperfusion pulmonary edema (RPE) grades. (a) The RPE grades of the 33 patients. (b) Scatter diagram of the RPE grade and mPAP after pulmonary endarterectomy. The mPAP was positively correlated with the RPE grade (r = 0.657, P = 0.00003).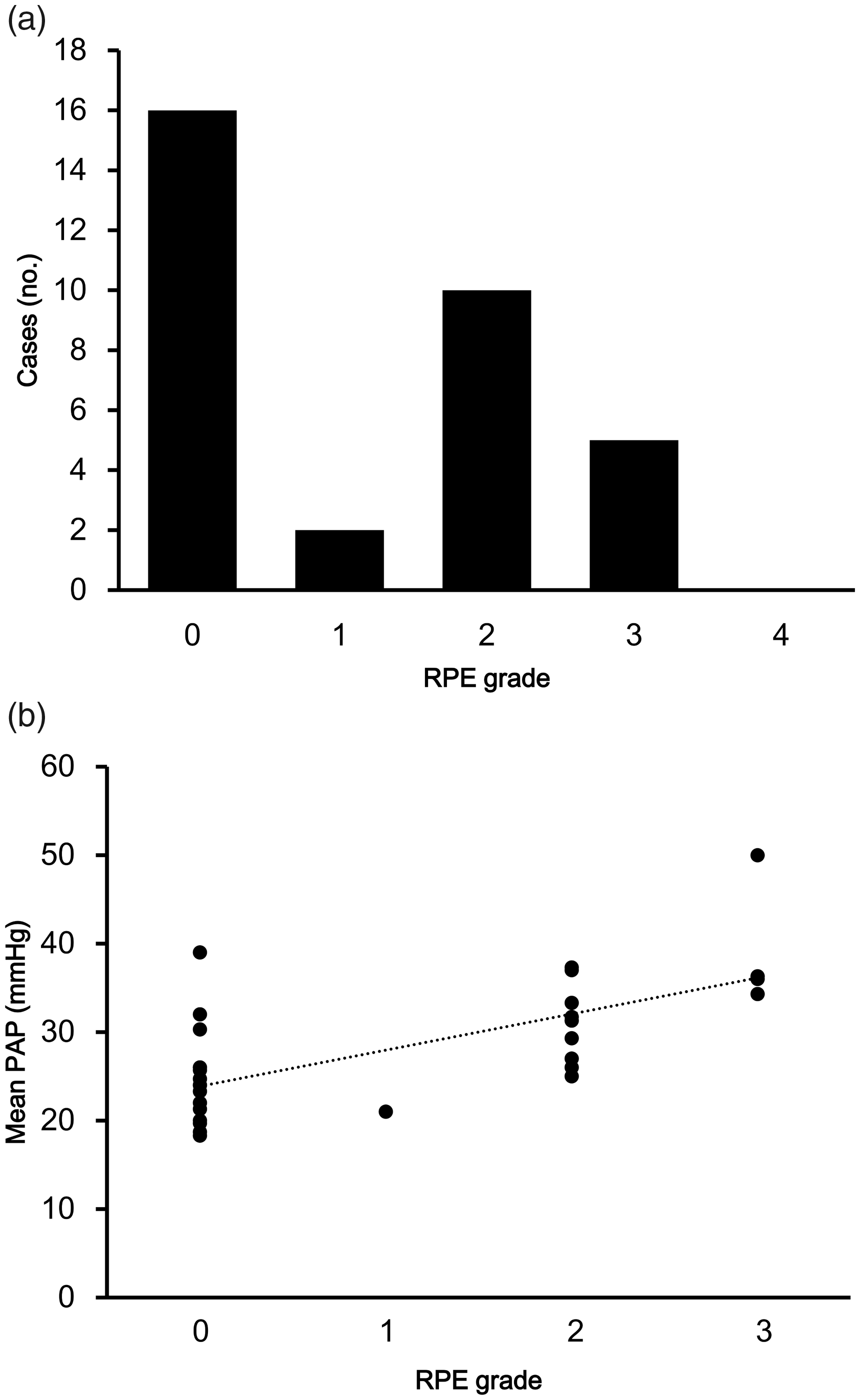
Pulmonary arteriopathy and venopathy
Pulmonary arteriopathy and venopathy were observed in all 33 cases (Fig. 3). In this study, 406 PAs (12.3 per patient and 0.52 per section) and 6382 PVs (193 per patient and 8.2 per section) were examined in 781 slides from 33 cases. The average PA diameter was 170.3 ± 59.8 µm (range = 52.1–297 µm) and the average obstruction ratio was 0.850 ± 0.139. The distribution of the PV scores was as follows: 0 = 25.9 ± 19.5%; 1 = 44.7 ± 12.8%; 2 = 23.9 ± 14.1%; 3 = 4.4 ± 5.1%; and 4 = 1.0 ± 2.6%. The average PV score was 1.10 ± 0.41%. Pathological findings, including pulmonary edema or capillary changes such as pulmonary capillary hemangiomatosis, were not found in any cases.
Pulmonary arteriopathies and venopathies. (a, b) Pulmonary arteriopathies: (a) severe pulmonary arteriopathy with a thickened neo-intima and medial layer – the lumen is narrowed and the obstruction ratio is 0.994; (b) mild pulmonary arteriopathy – the obstruction ratio is 0.745. (c–g) Pulmonary venopathies: (c) pulmonary vein (PV) score 0; (d) PV score 1; (e) PV score 2; (f) PV score 3 (muscularized PV); and (g) PV score 4 (pulmonary veno-occlusive disease-like). All slides were stained using the Elastica-van Gieson method. Scale bars: 50 µm.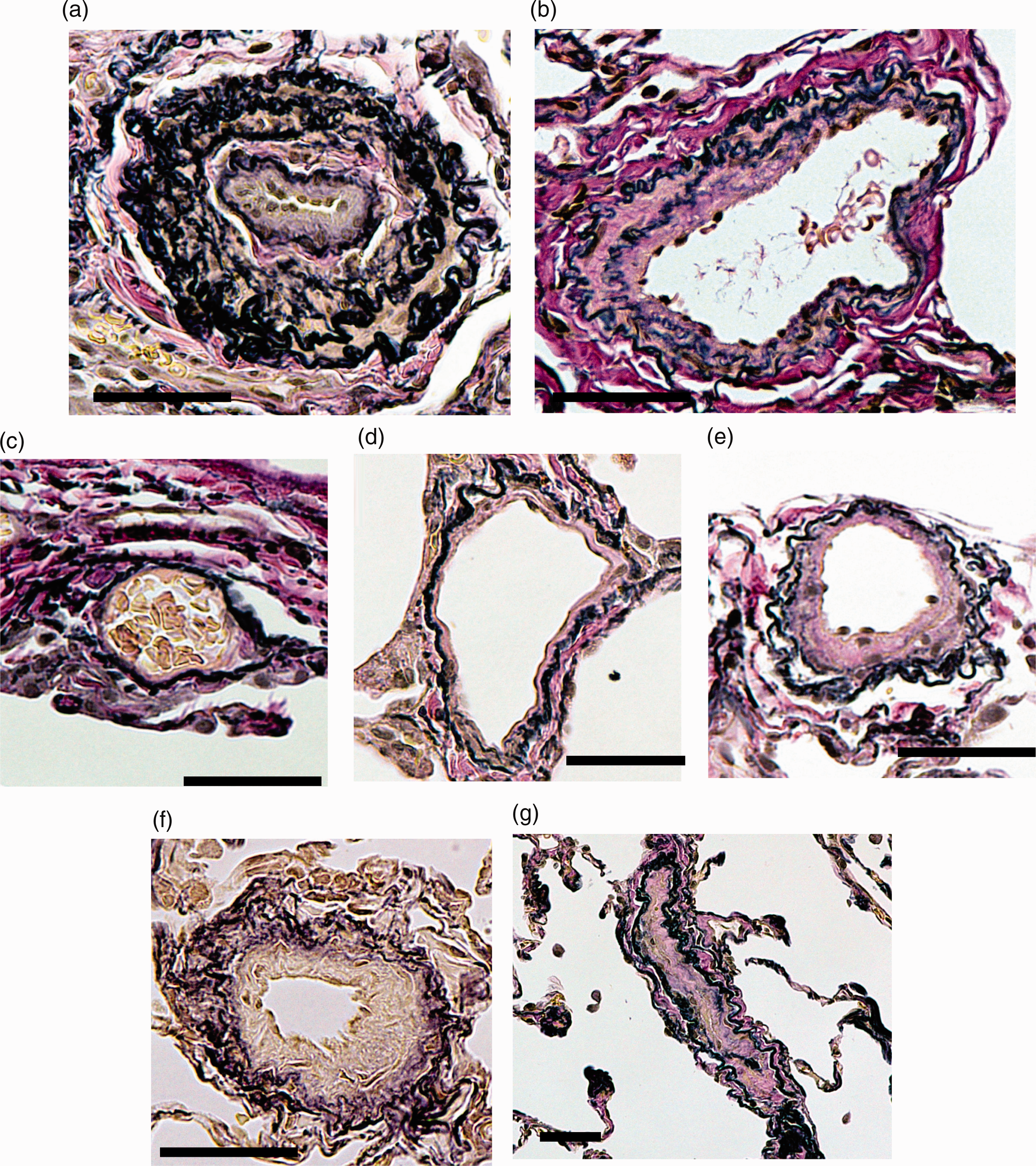
Hemodynamic change after PEA and the effect of pulmonary arteriopathy
The obstruction ratio was positively correlated with the mPAP obtained in the ICU (r = 0.445, P = 0.009; Fig 4) as well as the mPAP before, one month after, and one year after PEA: before PEA (r = 0.499, P = 0.003); one month after PEA (r = 0.433, P = 0.02); and one year after PEA (r = 0.498, P = 0.004).
Correlation between pulmonary arteriopathy and reperfusion pulmonary edema (RPE). (a) The obstruction ratio was positively correlated with the severity of RPE (r = 0.576, P = 0.0005). (b) The obstruction ratio was negatively correlated with the arterial oxygen tension/inspiratory oxygen fraction (P/F ratio) (r = –0.543, P = 0.001). (c) The obstruction ratio was positively correlated with the perioperative mPAP (r = 0.445, P = 0.009).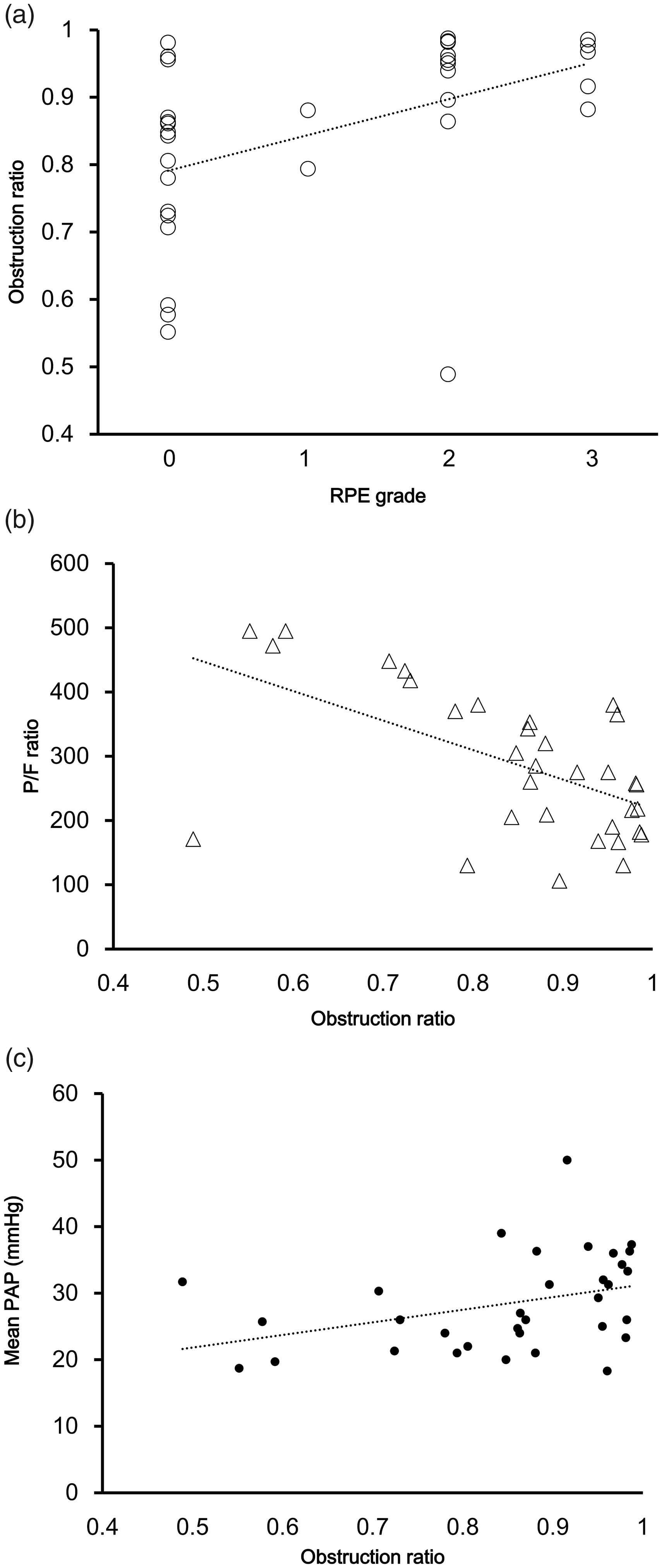
The relationship between pulmonary arteriopathy and the severity of RPE
Comparison of patient characteristics among RPE grades.
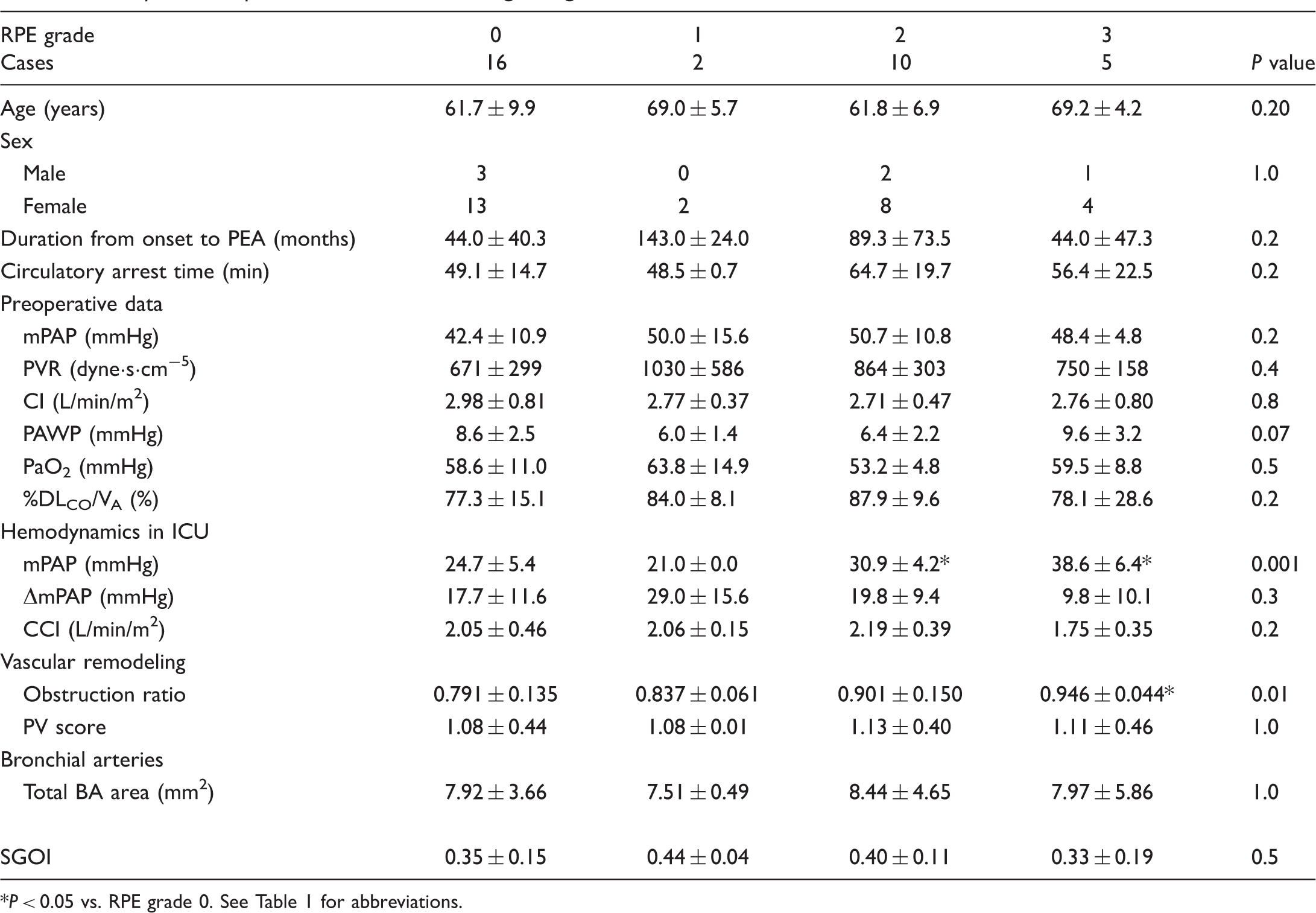
P < 0.05 vs. RPE grade 0. See Table 1 for abbreviations.
Determinant for developing RPE
Logistic regression analysis for the development of RPE.
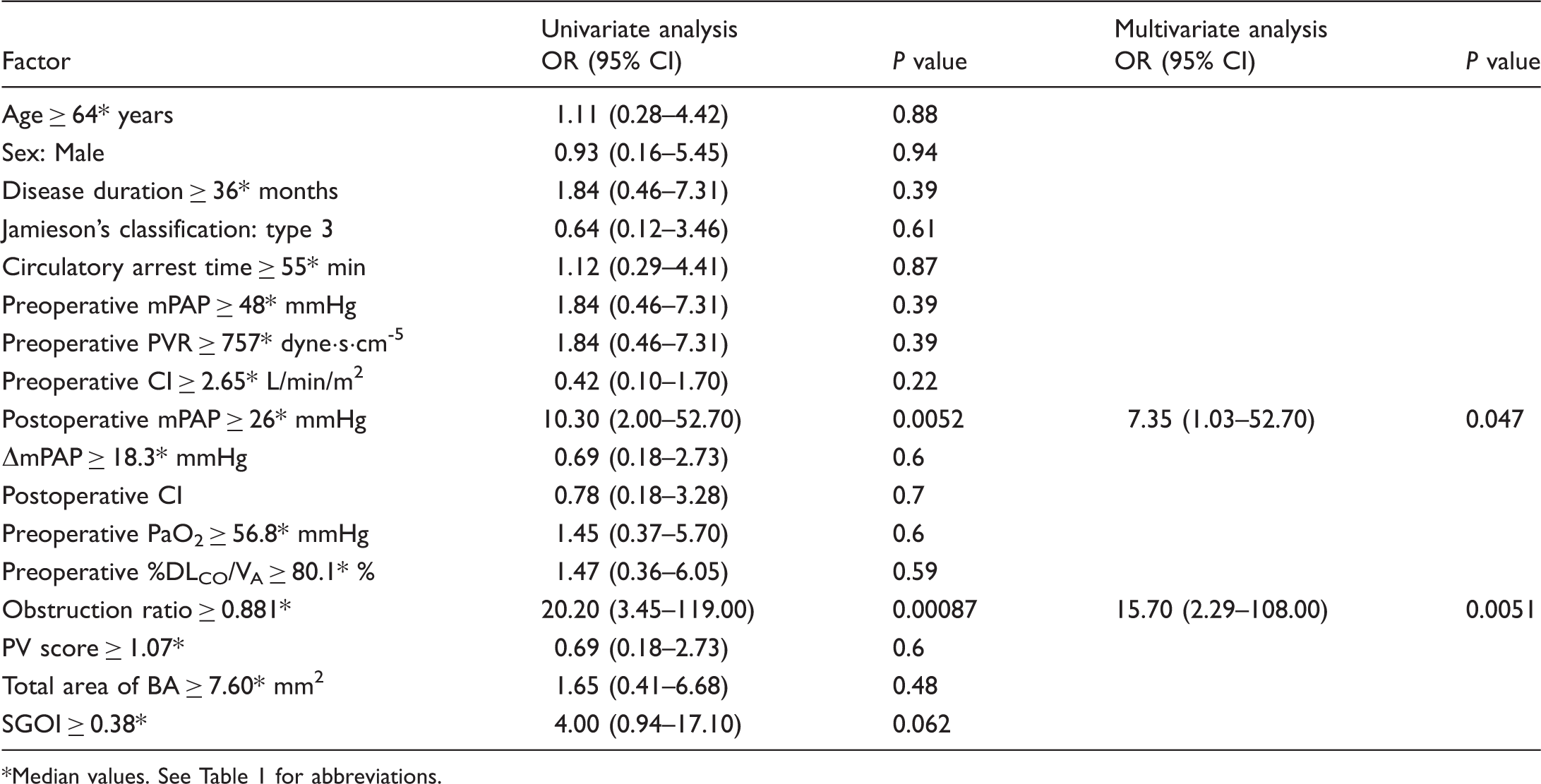
Median values. See Table 1 for abbreviations.
Discussion
In this study, we evaluated the clinical and pathological factors associated with the development of RPE. We found that the obstruction ratio was a determinant for the development of RPE. In addition, the obstruction ratio was significantly correlated with the severity of RPE, P/F ratio, and mPAP after PEA. The preoperative clinical variables, including the total area of the BAs, were not associated with the development or severity of RPE.
In the current study, the obstruction ratio was found to be a predictor of RPE, independent of the mPAP values. There are two types of PH after PEA 3 that can influence the development of PEA. Reversible PH after PEA can result from pulmonary vasoconstriction due to mechanical and/or reperfusion injury; 3 this commonly improves within 72 h after PEA. 15 Residual PH, which can be irreversible and persistent, 3 may result from severe pulmonary arteriopathy. 9 In the current study, mPAP in the ICU was positively correlated with the RPE grade and did not improve one month or one year after PEA. The long-term data of the current and previous study showed that the obstruction ratio was closely related with poor hemodynamics both soon after PEA and at one month and one year after PEA. 10 Thus, it was supposed that residual PH due to severe pulmonary arteriopathy could be a key factor in RPE development. Consistent with our findings, Stephan et al. reported that poor improvement of PVR just after PEA and a lower P/F ratio were correlated with severe exudation of extravascular lung water. 7 Reperfusion to ischemic lung tissue could cause increased shear stress and endothelial damage, resulting in extravasation.16,17 It could be that the residual PH associated with severe pulmonary arteriopathy induces increased sheer stress and lung injury following PEA. Persistent hypoxemia after PEA, partly due to ventilation/perfusion mismatch and impaired diffusion capacity, would be associated with severe pulmonary arteriopathy. 10 Therefore, the exudation of extravascular lung water showing infiltration in the lung area aggravates the ventilation/perfusion mismatch and diffusion capacity, thereby inducing an impaired P/F ratio after PEA.
Whether bronchial artery transformation is related to the development of RPE was unclear in our study. It was hypothesized that the enlargement of the BAs might be related to the development of RPE in the current study. It was shown that the BAs of CTEPH patients are enlarged 13 and that shunts between the pulmonary and bronchial arteries exist. 18 It was reported that the cross-sectional area measured by using CT images could be positively correlated with the shunt blood volume 19 and the collateral blood flow increased in patients with CTEPH compared with the levels in pulmonary arterial hypertension patients or healthy volunteers.19,20 Gan et al. suggested that preoperative occlusion of enlarged BAs might prevent patients from developing RPE. 12 In contrast to those reports, enlargement of the BAs was not found to be associated with the development or severity of RPE in the current study. The reason for this discrepancy remains unclear. The role of BAs occlusion before PEA in the prevention of RPE has not been determined; therefore, further investigations in this area are essential.
Pulmonary venopathy was not related to the development of RPE in this study. Pulmonary veno-occlusive disease is a subtype of PH characterized by the severe remodeling of PVs. 21 Increased blood flow caused by specific pulmonary vasodilators could induce pulmonary edema in patients with pulmonary veno-occlusive disease.21,22 In the current study, approximately 95% of PVs in CTEPH patients remained normal or mild. This may explain the limited effects of the PVs on the development of RPE.
The present study had several limitations. First, this study was conducted with a small sample size at a single institution and all participants were Japanese. Second, respiratory management depended on the decision of the cardiac surgeons and staff in the ICU. This might have affected the severity of RPE. Third, since lung biopsy is an invasive procedure, we do not recommend routine lung biopsy before PEA. In this study, we felt that the alternative methods for evaluating the pulmonary microcirculation were needed. Fourth, the number of PEA cases in our institution was relatively small. It was reported that the number of PEAs per year could be related to the postoperative prognosis; 23 therefore, the relative lack of experience at our institution might have affected the occurrence and severity of RPE. Fifth, the obstruction ratio might vary by the amount of occlusion in the proximal pulmonary arteries. It was reported that remodeled pulmonary arteries were identified not only in the open vascular area but also in the occluded area.18,24 With additional analysis, there was no significant correlation between the SGOI at the biopsied segments and the obstruction ratio (r = 0.03, P = 0.8; data not shown.). Finally, the resected lung specimens were small and only one specimen was obtained from each patient. It is unclear whether small samples may reflect the condition of the entire lung. Dorfmüller et al. described that remodeled pulmonary arteries were identified on all of the 170 slides randomly selected from the peripheral lung area of CTEPH patients. 18 In this study, we felt that the random sampling of pulmonary arteries using the single lung biopsy specimens could provide important information. Despite these limitations, we believe that our analysis is able to partially explain the mechanisms involved in the development of RPE after PEA. As a future treatment strategy, it seems that the reverse remodeling of pulmonary arteries before PEA might help to reduce the development of RPE.
In conclusion, severe pulmonary arteriopathy could be a key factor in the development and severity of RPE after PEA.
Supplemental Material
Supplemental material for Involvement of pulmonary arteriopathy in the development and severity of reperfusion pulmonary edema after pulmonary endarterectomy
Supplemental Material for Involvement of pulmonary arteriopathy in the development and severity of reperfusion pulmonary edema after pulmonary endarterectomy by Takayuki Jujo Sanada, Nobuhiro Tanabe, Hatsue Ishibashi-Ueda, Keiichi Ishida, Akira Naito, Seiichiro Sakao, Rika Suda, Hajime Kasai, Rintaro Nishimura, Toshihiko Sugiura, Ayako Shigeta, Yu Taniguchi, Masahisa Masuda and Koichiro Tatsumi in Pulmonary Circulation
Footnotes
Conflict of interest
TJ is a member of an endowed department sponsored by Actelion Pharmaceuticals Ltd.; NT is a member of an endowed department sponsored by Actelion Pharmaceuticals Ltd. and received lecture honoraria from Bayer AG, Daiichi-Sankyo Company, Limited, Actelion Pharmaceuticals Ltd. and Nippon Shinyaku Co., Ltd; AN is a member of the Joint Collaborative Department with Teijin Pharma, Ltd. and Ono Pharmaceutical, Ltd.; SS received honoraria for lectures from Nippon Shinyaku Co., Ltd, Bayer, Actelion Pharmaceuticals, and Pfizer. AS is a member of an endowed department sponsored by Actelion Pharmaceuticals Ltd.; YT received a research grant from Actelion Pharmaceuticals Ltd. and lecture honoraria from Nippon Shinyaku Co., Ltd; and KT received lecture honoraria from Actelion Pharmaceuticals Ltd. The other authors had no potential conflicts of interest.
Funding
This study was supported by the Japanese Ministry of Health, Labour and Welfare research grants specifically designated to the Respiratory Failure Research Group and Cardiovascular Diseases and the Pulmonary Hypertension Research Group from the Japan Agency for Medical Research and Development (No. 16ek0109127h0002). The funder had no role in the study’s design, collection of data or their analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
