Abstract
Arrhythmias are increasingly recognized as serious, end-stage complications of pre-capillary pulmonary hypertension, including pulmonary arterial hypertension (PAH) and chronic thromboembolic pulmonary hypertension (CTEPH). Although arrhythmias contribute to symptoms, morbidity, in-hospital mortality, and possibly sudden death in PAH/CTEPH, there remains a paucity of epidemiologic, pathophysiologic, and outcome data to guide management of these patients. This review summarizes the most current evidence on the topic: from the molecular mechanisms driving arrhythmia in the hypertrophied or failing right heart, to the clinical aspects of epidemiology, diagnosis, and management.
Keywords
Introduction
Pulmonary hypertension (PH) is defined as an increase in mean pulmonary artery pressure (mPAP) to ≥25 mmHg at rest as assessed by right heart catheterization (RHC). As outlined in the most recent European Society of Cardiology (ESC)/European Respiratory Society (ERS) guidelines, PH is subdivided into groups 1–5: group 1 (pulmonary artery hypertension [PAH] including idiopathic, heritable, toxin-induced, or associated with other conditions); group 2 (PH due to left heart disease); group 3 (PH due to lung diseases and/or hypoxia); group 4 (chronic thromboembolic PH [CTEPH]); and, lastly, group 5 (PH with unclear or multifactorial mechanisms). 1 Estimated five-year survival with PH is in the range of 38–59%, 2 depending on the underlying etiology, with group 3 PH currently having the worst prognosis. 3 In a recent population-based epidemiologic study of PH patients, a diagnosis of any form of PH was associated with a sevenfold increase in standardized mortality rate. 4
The primary cause of death in PAH is thought to be right heart failure, occurring as a direct consequence of elevated PAP, although in some studies approximately 50% of patients died from another cause, with PH as a contributing factor.5–7 Arrhythmias, such as sinus tachycardia, atrial tachycardia, atrial fibrillation (AF), atrial flutter (Afl), sinus bradycardia, ventricular tachycardia (VT), and ventricular fibrillation (VF), have been recognized as serious, end-stage complications of PAH and CTEPH. 8 Despite evidence that these arrhythmias contribute to symptom burden, morbidity, in-hospital mortality, and possibly sudden death,8–12 there remains scant data regarding the epidemiology, pathophysiology, and outcome of PAH patients with arrhythmia.
In this review, we explore the maladaptive and arrhythmogenic response of the right heart to group 1 and group 4 PH. We discuss the current patterns of clinical management, noting where these are evidence-based, and consider options for management of arrhythmia in PH. We also identify knowledge gaps and propose future directions.
While arrhythmia has been identified to coexist in all subgroups of PH, we will focus mainly on group 1 PH (PAH) and CTEPH in this review for two reasons. First, the majority of basic science studies pertaining to this topic have been conducted in PAH animal models and most clinical studies have predominantly included patients with PAH (group 1) and/or CTEPH (group 4). Second, the other subgroups (notably groups 2 and 3) have distinct etiology and pathophysiology, and therefore likely have differences in the mechanism of arrhythmogenesis, types of arrhythmia, and in the incidence and outcomes of these arrhythmias. For clarity and simplicity of text, we will refer to the group 1 (PAH) and group 4 (CTEPH) patients collectively as PAH/CTEPH, unless otherwise noted.
The arrhythmogenic substrate of the right heart in pulmonary hypertension
A number of potential mechanisms have been identified as contributing to arrhythmia susceptibility in patients with elevated PAPs and pressure- and volume-overloaded right atrium and ventricle. One of the earliest studies noted vascular degeneration and infarction in the sinus and AV node and sudden death in patients with what was then called primary PH (now referred to as idiopathic PAH [IPAH]). 12 In subsequent decades, a more granular mechanistic exploration has unfolded, revealing complex alterations in structure, electrophysiology, metabolism, and signaling pathways in the right heart.
Autonomic nervous system
The autonomic nervous system plays a key role in the development and progression of PAH and right heart failure 13 and has been implicated in pathogenesis of arrhythmia and sudden cardiac death (SCD). 14 Sympathetic overdrive in PAH is manifested by decreased heart rate variability, a blunted baroreflex, and poor exercise capacity, and is associated with associated with worse clinical status and prognosis.15–17 Increased sympathetic activity has also been correlated with premature ventricular contractions and ventricular arrhythmia in PAH patients. 18 Iodine-123-metaiodobenzylguanidine (123I-mIBG) myocardial imaging, a technique used to evaluate cardiac sympathetic nervous activity using single-photon emission computed tomography (SPECT), supports the aforementioned findings. Uptake of 123I-mIBG, a stable, modified form of guanethidine, occurs via the uptake-1 mechanism that normally uptakes norepinephrine. 19 By comparing activity at 3-h scans to those at 30 min, one can assess washout of the mIBG, which is a measure of the retained NE within sympathetic neurons. When the sympathetic system is activated there is a reduction of pre-synaptic norepinephrine uptake, manifest as lower retention of mIBG. A low heart to mediastinal (HMR) ratio of mIBG (≤ 1.2) in late images predicts event-free survival in left heart failure. 20 Increasing mPAP correlates with decreased mIBG activity in the right ventricle (RV), indicative of increased RV sympathetic activity. This decreased mIBG activity is associated with worse cumulative survival in PAH patients.21–23 Additionally, there is evidence of adrenergic remodeling in the RV, including downregulation and desensitization of β1-adrenergic receptors, as well as downregulation of α–adrenergic and dopaminergic receptors.24,25 In PAH patients with RV failure, there is also decreased adenylyl cyclase responsiveness to β-agonists and depletion of norepinephrine,26,27 with a loss of the ability to augment catecholamine levels with exercise. 28
The intrinsic and extrinsic cardiac nervous systems have been implicated in the susceptibility to supraventricular arrhythmia (SVA) in PAH. In a recent study, Huang et al. studied atrial arrhythmia using a canine model in which PAH is induced in beagles using a single injection of 2 mg/kg of dehydromonocrotaline followed by an eight-week interval of observation while PAH develops. 29 The dogs with PAH were more vulnerable than controls to induced AF/AFl. Histopathology demonstrated increased densities of sympathetic nerves and β-1 autonomic receptors in the right atrium, in contrast to the downregulation of β-adrenergic receptors in the failing RV.25,27 By systematically ablating the ganglionic plexi, which contain cholinergic and adrenergic neurons affecting the atrial myocardium, this reduced susceptibility to atrial arrhythmia, thereby demonstrating a pivotal role for the adrenergic component of the autonomic nervous system in regulating vulnerability to atrial arrhythmia in PAH.
Electrical remodeling
Stretch and fibrosis of the right atrium constitute an arrhythmogenic substrate, which combined with electrical remodeling due to changes in expression and function of ion channels in cardiomyocytes, increases susceptibility to and propagation of arrhythmia.30–33 An electrophysiology study comparing the right atrium of eight patients with longstanding IPAH versus 16 matched controls demonstrated prolongation of sinus node recovery time and reduction in tissue voltage with electrically silent areas in the atrium. There was also slowing of atrial conduction and activation times and an increase in areas of complex fractionated activity (critical sites for AF perpetuation).34,35 These factors are associated with increased susceptibility to induction AF, as stimulated by either increased automaticity (ectopy) or single and multiple re-entrant pathways.30,36
Right ventricular hypertrophy (RVH) and fibrosis are seen in patients with PAH/CTEPH and are associated with increased all-cause mortality. 37 Downregulation of inward rectifying potassium channels and slowed ventricular conduction (secondary to decreased gap junction expression, specifically connexin-43 remodeling, and fibrosis) impairs depolarization and repolarization in RVH. 38 Similar changes also cause arrhythmogenic early and late after- depolarizations and increase susceptibility to spontaneous or induced ventricular arrhythmia in models of RVH and left ventricular hypertrophy (LVH).39–41 Changes in Ca2+ handling has also been implicated in the prolongation and dispersion of action potential duration in rat models of PH.42,43 In aggregate, these active and passive changes in the electrical system in the RV predispose to creation of reentrant circuits and arrhythmia.44,45 Structural remodeling, for instance myocyte fiber angle disarray, can also change electrical conduction leading to prolonged action potential duration and arrhythmia vulnerability, as seen in the monocrotaline-PAH rat model. 46
Ischemia
Ischemia is a known pro-arrhythmic factor in the left ventricle;47,48 likewise, RV ischemia is thought to contribute to arrhythmia vulnerability in PAH/CTEPH. Elevated troponin levels in PAH patients reflect this ischemia and are associated with increased mortality, although not specifically with arrhythmia. 49 The genesis of ischemia in RVH is multifactorial, reflecting some combination of changes in epicardial coronary artery perfusion pressure (reduced perfusion pressure in the right coronary artery due to elevated right ventricular systolic pressure [RVSP], 50 left main coronary artery compression 51 ) and microvascular abnormalities (impaired angiogenesis manifest as capillary rarefaction related to decreased angiogenic gene expression).52–54 Secondary ischemia-related metabolic changes in the maladaptive RV, such as induction of increased rates of uncoupled glycolysis, are thought to perpetuate the vicious cycle of ischemia. 54
Electrocardiogram (ECG) abnormalities
Changes in the ECG are common in patients with PAH/CTEPH; however, there is controversy regarding the clinical significance of these abnormalities, and therefore uncertainty regarding the utility of screening electrocardiography. Kanemoto et al. 11 reviewed 171 ECGs of 101 patients with PAH and found “arrhythmia” in 17.8% of all cases, with a higher prevalence (33.9%) among the deceased cohort. Three abnormalities accounted for 70% of all observed arrhythmias: sinus tachycardia (38%); sinus bradycardia (18%); and first-degree A-V block (15%); with sinus tachycardia being significantly associated with mortality. A more contemporary investigation examined the progression of ECG changes in 50 PAH patients and found a significant increase in median heart rate, PR interval, QRS duration, R/S ratio in lead V1, and QTc duration in the ECG recorded closest to death, compared to the ECG recorded at the time of initial diagnosis. No PAH patient retained a normal ECG close to death. 55
As previously discussed, sinus tachycardia likely reflects autonomic dysregulation. 15 In addition, in patients with RV failure and PH, stroke volume may be fixed (and low) and sinus tachycardia is likely a reactive mechanism to maintain cardiac output. Loss of heart rate variability is a marker of autonomic dysfunction that reflects tonic activation of the autonomic nervous system and may also be a homeostatic attempt to maintain cardiac output. Heart rate variability is blunted during exercise testing of PAH patients; a decrease in heart rate variability at rest parallels the severity of PH in PAH, as measured by mPAP.17,56 Sinus tachycardia and decreased heart rate variability may be useful predictors of susceptibility to SVA.
In left heart disease and other non-cardiac conditions, prolongation of the QT interval on ECG is described as a precursor to ventricular arrhythmia and is associated with increased mortality and sudden death.8,57–64 In PAH, longer QTc and QTc dispersion are correlated with increasing mPAP. 65 In an age- and sex-matched cohort study of patients receiving PAH-specific therapy, both QRS duration and QTc interval were prolonged in 202 PAH patients versus 100 controls. A QTc duration ≥480 ms was an independent predictor of mortality, even in the subgroup of PAH and CTEPH. 66 There was no difference in serum potassium to explain the prolongation of QTc, as might be expected with aggressive diuretic use. QTc prolongation correlated with impaired RV function, increased RV mass (measured by cardiac MRI), and poor prognosis.
In animal models of PAH, QT prolongation is associated with longer action potential duration and a proarrhythmic state. 43 In the monocrotaline-rodent PAH model, QT prolongation is associated with decreased expression of repolarizing, voltage-gated (Kv) potassium channels (Kv1.2, Kv1.5, and Kv4.2) in the RV myocytes and is reversed with metabolic therapies, such as the pyruvate dehydrogenase kinase inhibitor, dichloroacetate. 38 The etiology of prolonged QTc in patients with PH has not been clearly defined but may be a result of chronic ischemia in RVH and/or changes in the sympathetic nervous system, as discussed previously.38,67 Extrapolation from other disease processes would suggest an that such ionic remodeling and QTc prolongation would increase susceptibility to ventricular arrhythmia; however, this has not been directly studied in PAH/CTEPH patients and presents an opportunity for future investigation.
QRS prolongation has also been an area of active study in PAH. QRS duration has been found to be longer in patients with PH compared to controls and is associated with indices of decreased RV function. 66 More specifically, prolonged QRS duration can contribute to RV dysynchrony, which is frequent in PAH and is an independent predictor of clinical worsening.68,69
Supraventricular arrhythmias
SVAs are the most prevalent rhythm disturbance observed in patients with PAH/CTEPH and they occur with greater incidence than in the general population.1,70–72 In this discussion of SVA, we are referring specifically to AF, AFl, atrioventricular nodal reentrant tachycardia (AVNRT), or atrial tachycardia, and not to sinus tachycardia. The onset of SVA often signals progressive right-sided cardiac dysfunction and frequently precipitates clinical decompensation by eliminating the atrial kick which is important to diastolic RV filling in the hypertrophied RV.8,73,74 Four retrospective and three prospective studies have examined the incidence and clinical relevance of SVA in PAH, in some cases including patients with CTEPH and in one study including patients with group 3 PH.8,75–79 The details of these studies are outlined in Tables 1 and 2. A graphical inter-study comparison of SVA frequency is depicted in Fig. 1. The seven studies are all small, with substantial limitations in methodology, but taken together, highlight six important points in regard to SVA and PAH/CTEPH.
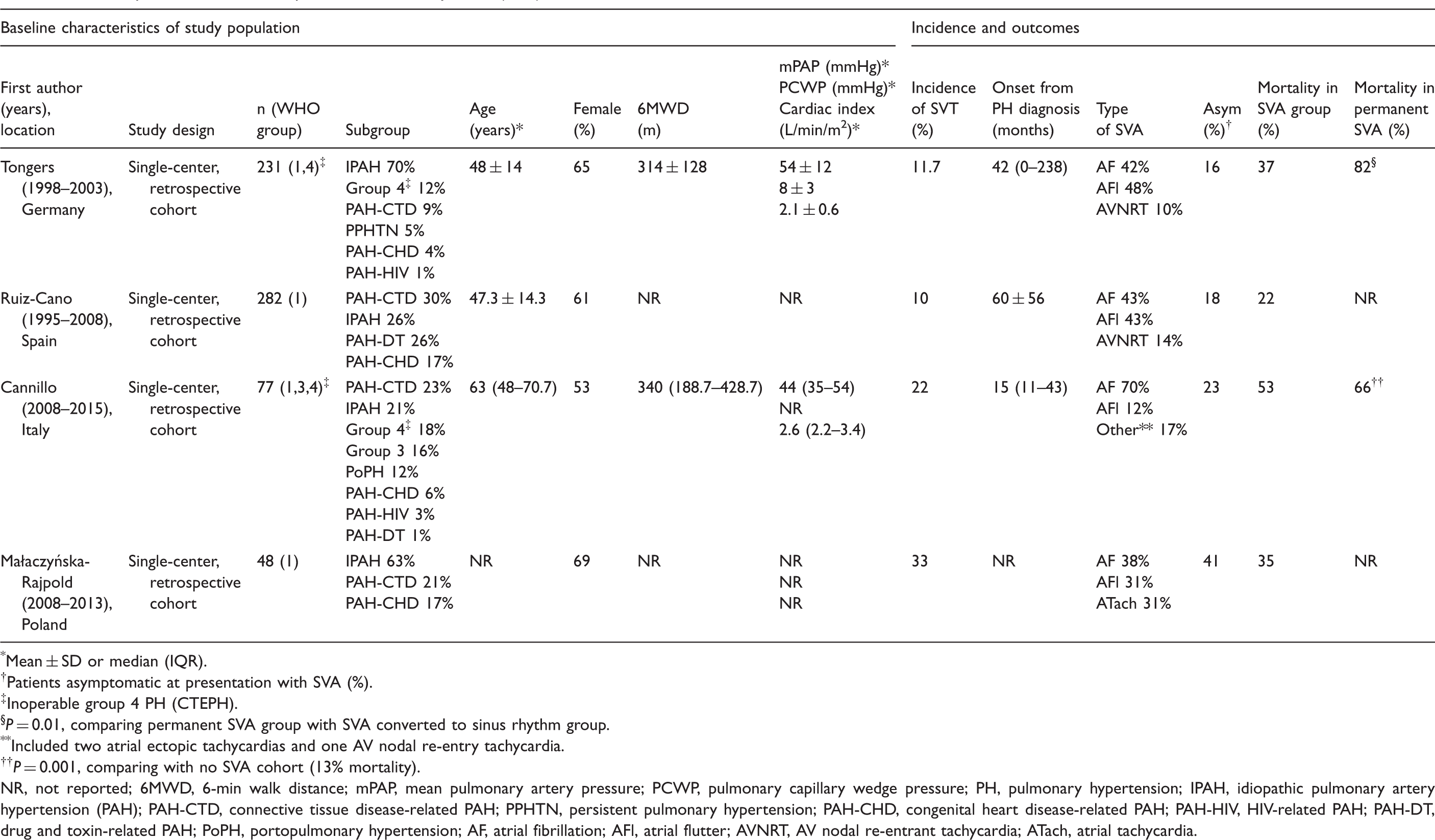
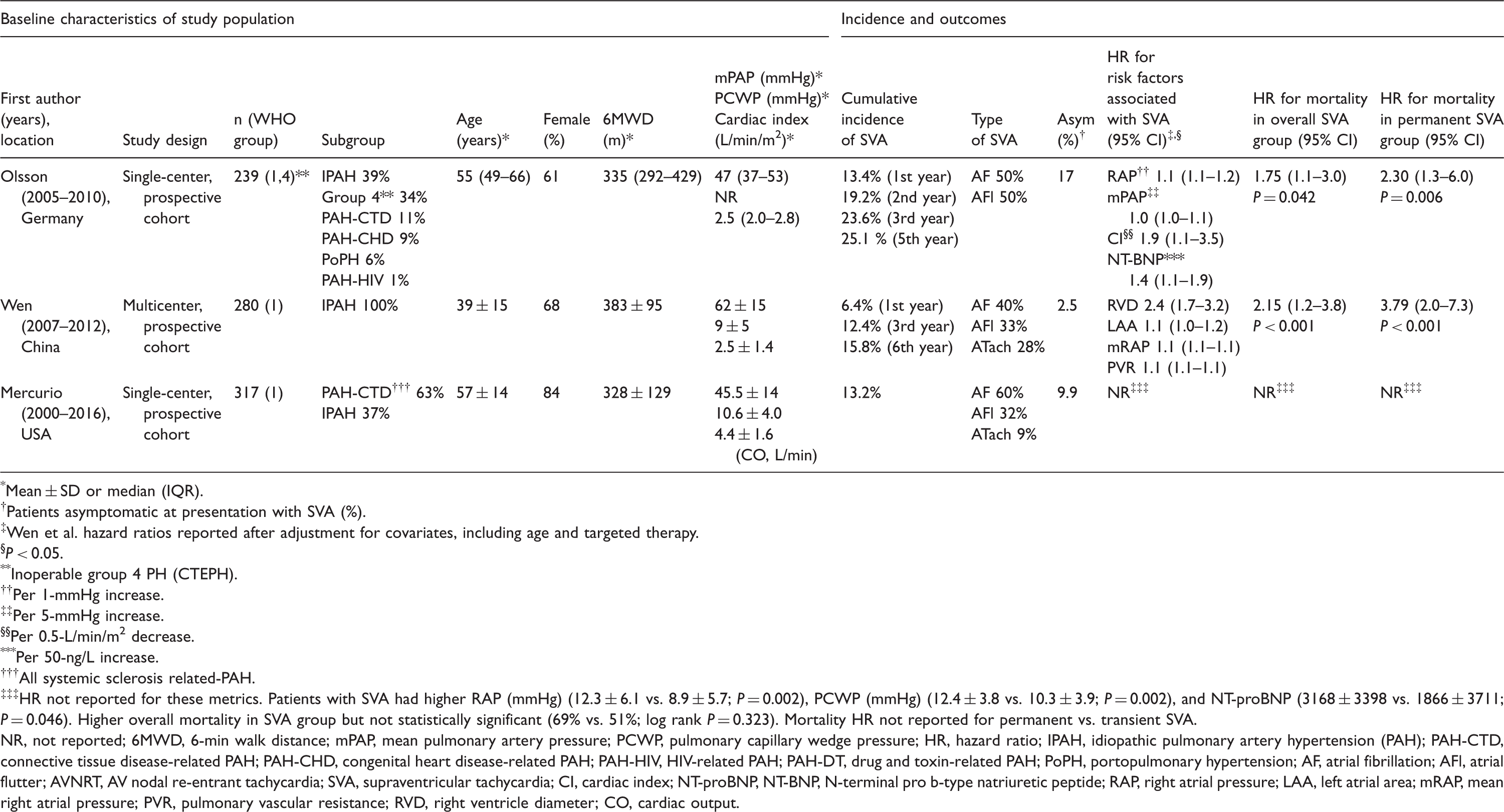
Overall frequency and subtypes of SVA (by study) in PAH/CTEPH. Subtypes: atrial fibrillation, atrial flutter, or other atrial tachycardia. SVA, supraventricular arrhythmia. Retrospective studies of supraventricular arrhythmia (SVA) in PAH/CTEPH. *Mean ± SD or median (IQR). †Patients asymptomatic at presentation with SVA (%). ‡Inoperable group 4 PH (CTEPH). §P = 0.01, comparing permanent SVA group with SVA converted to sinus rhythm group. **Included two atrial ectopic tachycardias and one AV nodal re-entry tachycardia. ††P = 0.001, comparing with no SVA cohort (13% mortality). NR, not reported; 6MWD, 6-min walk distance; mPAP, mean pulmonary artery pressure; PCWP, pulmonary capillary wedge pressure; PH, pulmonary hypertension; IPAH, idiopathic pulmonary artery hypertension (PAH); PAH-CTD, connective tissue disease-related PAH; PPHTN, persistent pulmonary hypertension; PAH-CHD, congenital heart disease-related PAH; PAH-HIV, HIV-related PAH; PAH-DT, drug and toxin-related PAH; PoPH, portopulmonary hypertension; AF, atrial fibrillation; AFl, atrial flutter; AVNRT, AV nodal re-entrant tachycardia; ATach, atrial tachycardia. Prospective studies of supraventricular arrhythmia (SVA) in PAH/CTEPH. *Mean ± SD or median (IQR). †Patients asymptomatic at presentation with SVA (%). ‡Wen et al. hazard ratios reported after adjustment for covariates, including age and targeted therapy. §P < 0.05. **Inoperable group 4 PH (CTEPH). ††Per 1-mmHg increase. ‡‡Per 5-mmHg increase. §§Per 0.5-L/min/m
2
decrease. ***Per 50-ng/L increase. †††All systemic sclerosis related-PAH. ‡‡‡HR not reported for these metrics. Patients with SVA had higher RAP (mmHg) (12.3 ± 6.1 vs. 8.9 ± 5.7; P = 0.002), PCWP (mmHg) (12.4 ± 3.8 vs. 10.3 ± 3.9; P = 0.002), and NT-proBNP (3168 ± 3398 vs. 1866 ± 3711; P = 0.046). Higher overall mortality in SVA group but not statistically significant (69% vs. 51%; log rank P = 0.323). Mortality HR not reported for permanent vs. transient SVA. NR, not reported; 6MWD, 6-min walk distance; mPAP, mean pulmonary artery pressure; PCWP, pulmonary capillary wedge pressure; HR, hazard ratio; IPAH, idiopathic pulmonary artery hypertension (PAH); PAH-CTD, connective tissue disease-related PAH; PAH-CHD, congenital heart disease-related PAH; PAH-HIV, HIV-related PAH; PAH-DT, drug and toxin-related PAH; PoPH, portopulmonary hypertension; AF, atrial fibrillation; AFl, atrial flutter; AVNRT, AV nodal re-entrant tachycardia; SVA, supraventricular tachycardia; CI, cardiac index; NT-proBNP, NT-BNP, N-terminal pro b-type natriuretic peptide; RAP, right atrial pressure; LAA, left atrial area; mRAP, mean right atrial pressure; PVR, pulmonary vascular resistance; RVD, right ventricle diameter; CO, cardiac output.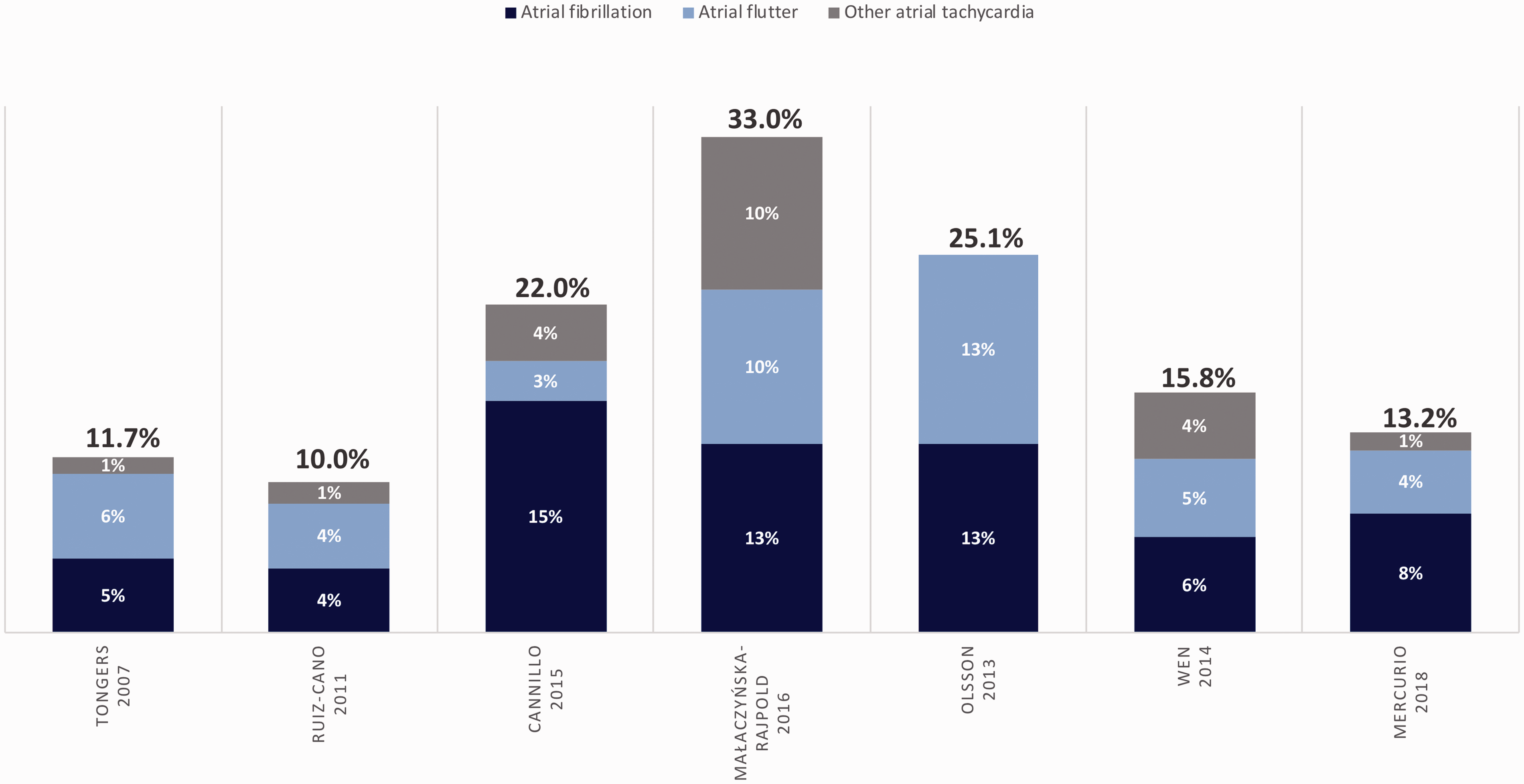
1. There is no clear difference in baseline characteristics predicting SVA susceptibility
Baseline characteristics do not reliably predict which patients will develop SVA (Tables 1 and 2). However, the findings most correlated with development of SVA include elevated right atrial pressure (RAP)75,76,78,80 and markers of overall cardiac dysfunction, such as decreased cardiac index/output, increased RV diameter, and elevated NT-proBNP.76,78,80 Only in a minority of studies were pulmonary vascular resistance (PVR) and mPAP associated with incident SVA,76,78 perhaps suggesting it is the adaptation of the heart to increased afterload, rather than the afterload itself, that predicts development of SVA. There is biologic plausibility to this finding, given that the maladaptive response of the right heart is thought to predispose to atrial arrhythmias.29,30
Differences in non-invasive measures of disease severity, such as 6-minute walk distance (6MWD) and World Health Organization (WHO) functional class (FC), did not predict development of SVA in any of the studies. This is unexpected but may be a consequence of the overall homogeneity of the included cohorts, which largely included patients with poor functional state, or the relatively small sample size with less sensitive and more subjective measures.
Likewise, in most studies, the baseline PAH therapy and co-morbidities were no different between PAH/CTEPH patients with SVA versus those without. An exception is the study by Mercurio et al., who found a significantly increased frequency of thyroid disease (defined as hypo- or hyperthyroidism, or radiographic thyroid nodules) in the SVA group compared to those without SVA (69% vs. 43%). 80 The clinical significance of thyroid disease, which is prevalent in PAH, was not explored in the other studies and may be an underrecognized contributing factor to development of arrhythmia. 81
2. SVA often precipitates clinical decompensation
Onset of SVA often correlates with a clinically relevant decompensation. Whether the SVA triggers the clinical decompensation or is the result of right heart failure is not always clear; however, in many cases, clinical decompensation onsets abruptly with an episode of SVA and conversely resolves with restoration of sinus rhythm. The majority of patients reported symptoms at time of SVA onset (59–98%). Across all studies, objective and standardized measures of disease severity consistently worsened after development of SVA. The reported measures included worsening of New York Heart Association (NYHA) FC and a decrease in 6MWD. One study reported an increase in PAH-specific therapy in 46% of patients with SVA, 82 while another noted that arrhythmia led to admission to the intensive care unit and vasopressor use in approximately 30% of SVA episodes. 80
3. Some SVAs are subclinical and detection may require a screening ECG or ambulatory monitor
While the majority of patients do have symptoms at SVA onset (mean ∼80%), up to 41% of episodes are asymptomatic, with their arrhythmia identified only via screening ECG or ambulatory monitor. With the finding that SVA is associated with increased mortality in patients with PAH/CTEPH, the role for surveillance of PAH/CTEPH patients by means of routine ECG, Holter monitors, or loop recorders warrants further study.
4. Restoration of sinus rhythm reverses decompensation
Restoration of sinus rhythm coincides with the reversal of adverse effects of an episode of SVA. Atrioventricular synchrony in sinus rhythm increases cardiac output and is particularly important when there is diastolic dysfunction. Thus, it is likely the active, synchronous atrial loading of the ventricles that results from sinus rhythm increases cardiac output in PAH patients. Restoring sinus rhythm should therefore be strongly considered in patients with PH. However, as will be discussed in the management section, there is a lack of randomized clinical trial data comparing rate versus rhythm control in PAH/CTEPH patients. When reported, restoration of sinus rhythm was associated with objectively confirmed clinical improvement. For example, in one study, at the time of SVA onset, 6MWD decreased from 362 ± 114 m to 258 ± 147 m, returning to a value near baseline (345 ± 137 m) 6–12 weeks after restoration of sinus rhythm. Over the same time period, NT-proBNP decreased from 5926 ± 4648 ng/L at the time of SVA onset to 3360 ± 2804 ng/L after correction. 76
5. Permanent SVA is associated with increased risk of death
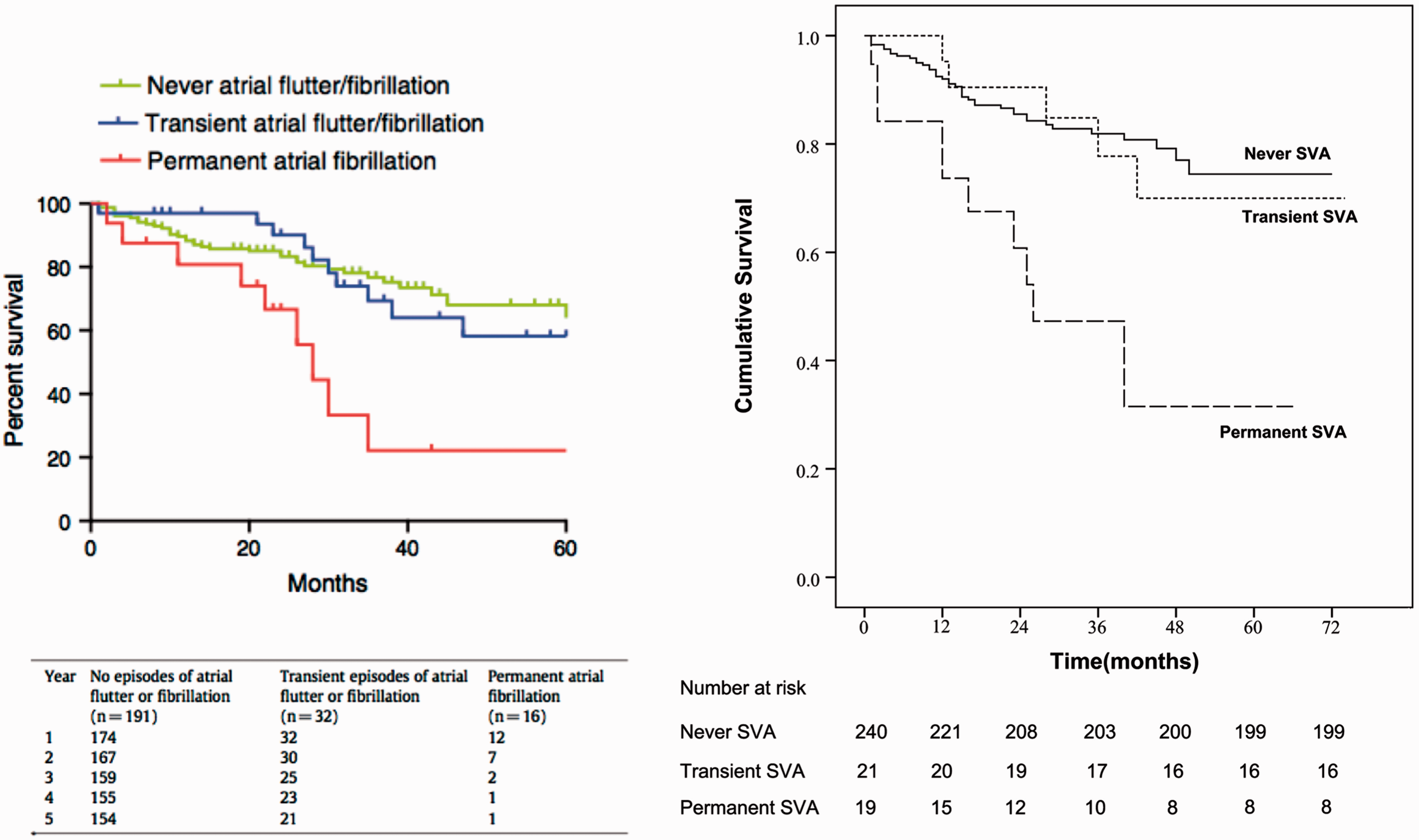
SVA is associated with increased mortality, driven primarily by permanent (as compared to transient) SVA. In the four retrospective studies, mortality was in the range of 22–53% in the SVA cohort compared to 0–13% in those patients without SVA.10,75,79,82 Similarly, two of the prospective studies report elevated hazard ratios (HR) for mortality in those who develop SVA (HR = 1.75–2.15).76,78 A third study describes an increased mortality of 69% in the SVA group compared to 51% in those without SVA, although this was not statistically significant on survival analysis.
80
The effect of permanent SVA on mortality is quite striking, as depicted in the survival curves in Fig. 2. The increase in mortality was almost entirely attributable to development of permanent SVA (HR = 2.3–3.8), as compared to transient or no SVA.76,78
6. Atrial fibrillation is the most common cause of permanent SVA in PAH/CTEPH
The most common cause of permanent SVA is AF (accounting for 42–70% of all cases). However, Afl is also prevalent, occurring in 12–50% of the series reviewed (Fig. 1). Less common SVAs include AVNRT and atrial tachycardia. The high incidence of AF has clinical implications given that restoration of sinus rhythm is more difficult in AF compared to other SVA, and that permanent SVA is associated with increased mortality. Clinicians should regard the development of permanent AF as a serious adverse event in a patient with PAH/CTEPH. Addition of permanent SVA to contemporary mortality prediction models (for example, REVEAL registry risk assessment) may help with prognostication, as it appears to portend a significant decline in survival.1,83–86
Additional large-scale investigations are needed to prospectively characterize the incidence of SVA in PH. While onset of SVA appears to be associated with increased mortality, it is not clear if the SVA is causal of deterioration or simply a result of worsening clinical condition. Unfortunately, given the nature of the disease process and the invasiveness required to obtain hemodynamic data, the frequent monitoring required to address this question may require these data be obtained as a part of large clinical trials or international registries. To date, the incidence/prevalence of SVA have not been reported in prospective or large-scale studies.2,85,87,88
Management
Guidelines
Recommendations for the management of SVAs in PAH and CTEPH.

Rate and rhythm control
Management strategies and success rates of therapy for SVA in PAH/CTEPH.
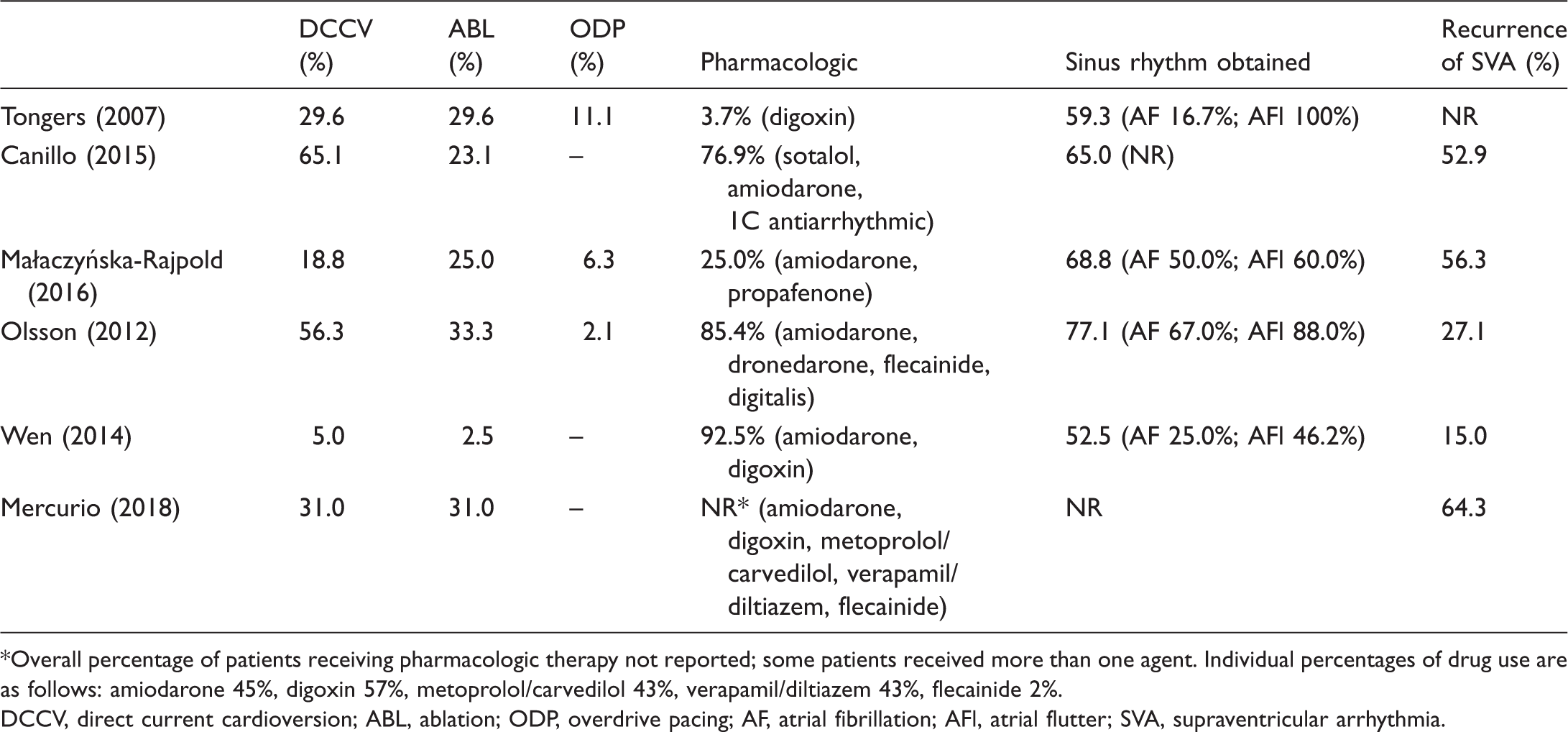
Overall percentage of patients receiving pharmacologic therapy not reported; some patients received more than one agent. Individual percentages of drug use are as follows: amiodarone 45%, digoxin 57%, metoprolol/carvedilol 43%, verapamil/diltiazem 43%, flecainide 2%.
DCCV, direct current cardioversion; ABL, ablation; ODP, overdrive pacing; AF, atrial fibrillation; AFl, atrial flutter; SVA, supraventricular arrhythmia.
A variety of pharmacologic strategies were used for SVA (Table 4) including digoxin, amiodarone, sotalol, dronedarone, and class 1C antiarrhythmic agents (flecainide, propafenone). Medical therapy for SVA in PAH/CTEPH is limited by concern for drug–drug interactions with antiarrhythmics and PAH-specific therapies, as well as adverse effects. For example, bosentan (an endothelin receptor antagonist [ERA]) is an inducer of CYP3A4 and may reduce levels of amiodarone or dronedarone.90,91 Many antiarrhythmic drugs have antihypertensive effects based on mechanism of action and may cause systemic hypotension when used concomitantly with phosphodiesterase inhibitors (sildenafil, tadalafil) or prostacyclins (epoprostenol). 90 The use of beta-adrenergic blockers in PH is also controversial. There is risk of provoking heart failure or circulatory collapse, based on their well-established negative inotropic and chronotropic effects.92,93 More contemporary data have started to study the safety and efficacy of beta-blockers in patients with PAH who are free from congestion/heart failure,94–96 but the use of beta-blockers for improvement of RV function and/or survival remains a research question and is not standard of care. In contrast, the use of short acting beta-blockers may be helpful in restoring sinus rhythm or regulating heart rate in patients with acute SVA.
Amiodarone (both intravenous and oral administration) was the most frequently reported antiarrhythmic agent for medical cardioversion and/or maintenance of sinus rhythm. While amiodarone is typically well-tolerated in the short term, the long-term effects may compound morbidity in patients with PAH/CTEPH. 97 In the studies of SVA in PAH/CTEPH, AFl and other atrial arrhythmias, such as AVNRT, were more readily converted to and maintained in sinus rhythm with amiodarone in short- and intermediate-term follow-up compared to AF.10,76,79,82
Electrophysiology studies with catheter ablation are safe, feasible, and successful in patients with PH.98–102 In a retrospective analysis of 22 patients with AFl and PAH/CTEPH, Showkathali et al. showed that AFl ablation at the cavotricuspid isthmus was successfully performed without complications in all patients and was associated with a statistically significant improvement in FC. 98 The relative success of this interventional strategy can be attributed to the right-sided location of typical flutter or re-entrant pathways, allowing avoidance of trans-septal puncture thereby reducing procedural risks of stroke and bleeding.
Little is known about left heart interventions in PAH/CTEPH patients and it is unclear whether, or to what degree, left-sided pathways contribute to arrhythmia in this cohort. Were pulmonary veins and left atrial pathways to prove important in PAH/CTEPH-related SVA, caution would be required in terms of catheter-based therapeutics. There is uncertainty regarding the effect of pulmonary vein manipulation on pre-capillary pulmonary pressure.75,79 Pulmonary vein isolation is more invasive compared to catheter-based interventions in the right heart and concerns exist regarding the risk of anesthesia and intubation in patients with PAH/CTEPH. In general, intubation should be avoided in PAH patients due to the problematic effect of sedation on cardiac function and vasodilation leading to hemodynamic collapse. 103 In-hospital mortality in PAH patients requiring vasopressor and mechanical ventilation for RV failure approaches 100%, 104 justifiably increasing hesitancy to perform more invasive (and potentially low-yield) ablation procedures.
Anticoagulation
Since the use of anticoagulation in CTEPH is routine management, anticoagulation for SVA would not alter therapy; however, there is increasing recognition of the absence of benefit and potential for harm with anticoagulation in some forms of PAH.105,106 This highlights the value of more carefully establishing the risks and benefits of anticoagulation for arrhythmia in PAH patients.
In recent guidelines, oral anticoagulation is considered harmful in patients with associated-PAH but is recommended in idiopathic, heritable, or anorexigen-induced PAH (expert consensus 89 /Grade IIb 1 ). It is uncertain whether these recommendations should change in the event of permanent atrial fibrillation and it is unknown if the current risk stratification models (such as CHA2DS2-VASc) are valid in PAH/CTEPH. 107 Only the prospective studies by Olsson et al., Wen et al., and Mercurio et al. specifically comment on the frequency of anticoagulation therapy in the examined PAH/CTEPH patients: Olsson et al., 92% overall on oral anticoagulants; 76 Wen et al., 73% in SVA cohort, overall use not reported; 78 and Mercurio et al., 36% of SVA cohort, overall use not reported. 80 These differences likely reflect international practice pattern variation and inclusion criteria, e.g. the Olsson study included CTEPH while the others did not. There is no apparent difference in risk of developing SVA or subsequent morbidity or mortality after onset of arrhythmia based on the use of anticoagulation;76,78 however, vascular outcomes such as stroke were not specifically addressed. Unfortunately, neither of the recent publications that examined the utility of anticoagulation in PAH (COMPERA and REVEAL) comment on atrial arrhythmias,87,108 the presence of which might alter the benefits of anticoagulation. With up to 41% of patients experiencing asymptomatic atrial arrhythmias (Tables 1 and 2), a study assessing incremental benefit in morbidity with anticoagulation would require screening ECG or ambulatory monitoring to capture all potentially relevant SVAs.
Ventricular arrhythmia
Ventricular tachyarrhythmias (VT or VF) are thought to cause the majority of SCDs in adult patients. 109 Registry data demonstrate that most PAH patients die from progressive right heart failure or SCD.3,110 Since SCD often occurs outside the hospital, the cause of sudden death in PAH patients has not been well elucidated. The basis of SCD is hypothesized to include arrhythmia, circulatory collapse, compression of left main coronary artery, or pulmonary artery rupture or dissection. 111 In contrast to advanced LV failure, malignant ventricular arrhythmias are not frequently observed in PAH, despite plausible pathophysiologic changes in the ventricular tissue (fibrosis) and QTc prolongation. VF has been demonstrated in a monocrotaline-induced PAH rat model, 41 but clinical investigations have only identified VT/VF in a minority of PAH patients with SCD (8%).641
Non-sustained ventricular tachycardia (NSVT) was recently identified to be more prevalent than previously thought in a cohort of non-group 2 PH patients. 112 Nonetheless, in a follow-up study, NSVT was not correlated with increased mortality in PAH/CTEPH. 113 Since routine and sustained rhythm monitoring in PAH/CTEPH is not a standard clinical practice, the possibility that VT/VF contributes to a higher proportion of SCD cannot be excluded.
Antiarrhythmic and device therapy for SCD
There are currently no data to show that patients with PAH/CTEPH and RV dysfunction benefit from defibrillator implantation to prevent SCD. The guidelines and management for arrhythmia in left heart failure may not apply to RV failure, due to differences in the underlying disease processes. Just as medications that are beneficial for group 1 PAH have been shown to be harmful or ineffective in group 2 PH (and vice versa), strategies to prevent and treat SCD may also differ. Practice guidelines for prevention of SCD in PAH state that “antiarrhythmic therapy is not indicated for prevention of SCD in patients with PAH,” and, further, that patients may be at “high risk during surgical procedures, such as ICD implantation.” 111 ICD implantation is often offered to non-PH patients who have a cardiac arrest secondary to documented VT or VF,111,114 but clinical judgment must be employed regarding use of ICDs or prophylactic antiarrhythmic agents in PAH/CTEPH, given the absence of any clinical trials demonstrating benefit in this cohort.
Likewise, the role of cardiac resynchronization therapy (CRT) in right heart failure due to PAH/CTEPH remains undefined.115,116 Ventricular dyssynchrony is observed in progressive RV failure associated with PAH, often visualized as paradoxical septal movement caused by RV pressure/volume overload that compresses the left ventricle.117,118 This dyssynchrony is thought to hinder LV diastolic filling resulting in decreased LV stroke volume. 119 RV free wall pacing with resynchronization improves RV function in a computer simulation model of PAH, an animal model of PAH, and clinically in a small cohort of patients with CTEPH.120–122 However, further investigation is needed to confirm whether CRT is a valid therapeutic strategy in PAH/CTEPH patients with RV failure. Of note, in left heart failure, CRT has not been shown to be beneficial in patients with right bundle branch block or with QRS intervals of <160 ms, two features that are common in patients with RV failure from PH. This raises some skepticism about the potential role CRT in PAH/CTEPH.
Conclusion
In summary, arrhythmias are an increasingly recognized cause of morbidity and mortality in patients with PAH/CTEPH.
123
Despite ongoing research, there remain numerous gaps in knowledge, which limit a clinician's ability to optimally identify and manage arrhythmia in PAH/CTEPH (Table 5). SVAs, including AF and Afl, are more common than ventricular arrhythmias and, if sustained, portend clinical decompensation and increased mortality (Fig. 3). Improved screening and standardized management of SVA has the potential to improve outcomes in PAH patients. Randomized trials assessing rhythm versus rate control strategies and evaluating the role of anticoagulation are needed. Ventricular arrhythmias, while rare, can cause SCD in PAH/CTEPH. In contrast to left heart failure, Grade I evidence is lacking regarding the value of ICDs for primary prevention of such arrhythmias in PAH with RV failure. A large-scale surveillance study for sudden death etiology, perhaps using PH registries, could provide needed insight on the magnitude of arrhythmias and help to guide further recommendations for prevention and management.
Schematic illustration outlining the development and consequence of arrhythmia in PH. CRT, cardiac resynchronization therapy; ICD, implanted cardiac defibrillator; VT, ventricular tachycardia; VF, ventricular fibrillation; fib, fibrillation; 6WMD, 6-min walk distance; WHO FC, World Health Organization functional class; BNP, brain natriuretic peptide; NT-proBNP, N-terminus-pro brain natriuretic peptide. Opportunities for further investigation in arrhythmia and pulmonary hypertension. ECG, electrocardiogram; RV, right ventricular; ICD, implantable cardiac defibrillator; SVA, supraventricular arrhythmia.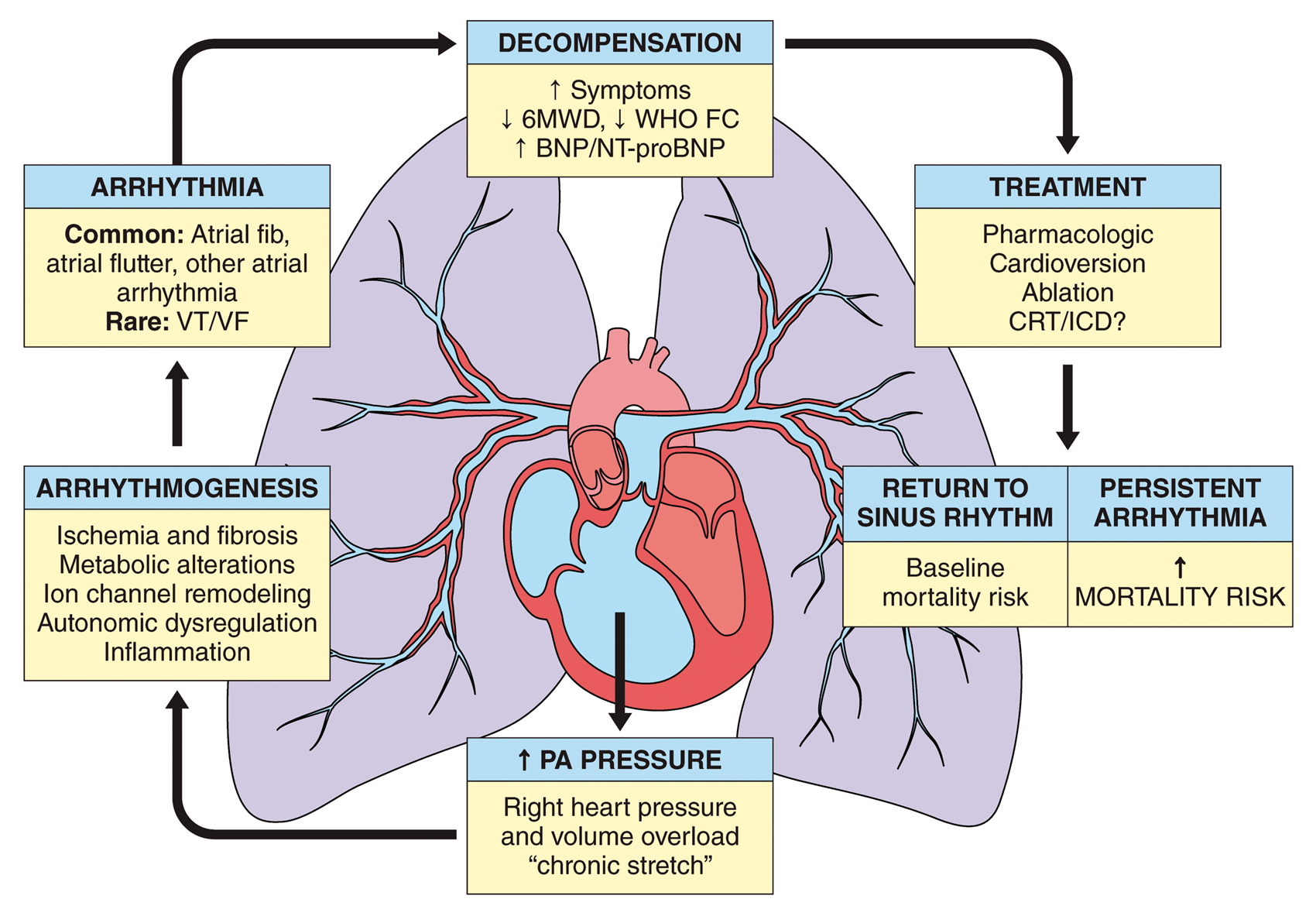

Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
SLA and his research are supported by a CIHR Foundation Grant (NIH-RO1-HL071115, 1RC1HL099462), a Tier 1 Canada Research Chair in Mitochondrial Dynamics, the William J Henderson Foundation, the CIHR Vascular Network, the Canadian Vascular Network, and the Queen’s Cardiopulmonary Unit (QCPU). JJR and his research are supported by grants from the Reagan Corporation and The Cushman Family.
