Abstract
Heart failure due to diastolic dysfunction and pulmonary hypertension are frequent comorbid conditions with significant morbidity and mortality. Identifying the presence and etiology of diastolic dysfunction in the setting of pulmonary hypertension remains challenging despite profound therapeutic and prognostic implications. Additionally, there is little guidance in identifying and parsing etiology of diastolic dysfunction in patients found to have pulmonary hypertension. This review discusses the complex interplay between left ventricular diastolic dysfunction and pulmonary hypertension. With an explicit focus on the use of echocardiography for determination of diastolic dysfunction and etiology of pulmonary hypertension, this review also provides a comprehensive review of the literature and provides a framework by which to assess diastolic dysfunction echocardiographically in the setting of pulmonary hypertension.
Introduction
With a growing incidence and prevalence worldwide, heart failure remains a condition with limited treatment options and poor prognosis.1,2 Left ventricular diastolic dysfunction, defined by impaired relaxation of the myocardium, is a hallmark of heart failure in patients who present with reduced ejection fraction (HFrEF) or preserved ejection fraction (HFpEF). 3 The presence of even asymptomatic diastolic dysfunction portends a worse prognosis for patients regardless of the underlying etiology.4–6 Pulmonary hypertension frequently coexists with left heart diastolic dysfunction.7–10 Since both pulmonary hypertension and diastolic dysfunction can be a primary cause of symptoms such as exertional dyspnea, orthopnea, paroxysmal nocturnal dyspnea, and exercise intolerance, distinguishing the primary cause of symptoms and pathology carries significant prognostic and therapeutic implications.
A key determinant in the primary cause of symptoms in patients with both diastolic dysfunction and pulmonary hypertension is the evaluation of left atrial filling pressures and left ventricular end-diastolic pressures (collectively referred to as left sided filling pressures).8,11–13 Invasive cardiac catheterization with or without provocation by exercise or fluid challenge is the gold standard for measuring left-sided filling pressures; however, it can be associated with misclassification of patients with concomitant pulmonary hypertension due to technical/interpretative errors or lack of evidence of elevated filling pressures in the absence of provocative maneuvers.5,12,14,15 Non-invasive assessment of left sided filling pressures allows for a more global structural assessment of the heart to provide additional clues that may aid in the differentiation of the primary cause of symptoms. In addition, non-invasive approaches eliminate periprocedural risk, potentially reducing health care costs and allowing for a potential method for serial monitoring of response to therapies. While recent guidelines have created a more simplified and unified approach to the non-invasive assessment of diastolic function in echocardiography with a focus on a disease-oriented approach, there is limited guidance on and data supporting the accurate assessment of diastolic function in pulmonary hypertension. 3
Thus, the following review aims to further discuss this complex interplay between diastolic dysfunction and pulmonary hypertension, as well as provide a framework by which diastolic function can more accurately be estimated in the setting of pulmonary hypertension.
Complex interplay between left ventricular diastolic function and pulmonary vascular function
While many previous studies have focused on the complex coupling between the right ventricle and pulmonary artery that is well studied in the setting of pulmonary hypertension,16,17 there also exists a complex interaction between the left ventricle and pulmonary circulation.
A number of studies have reviewed the role of left ventricular impaired relaxation resulting in passive congestion and chronic pulmonary vascular changes.18,19 Other studies have also shown that the chronic pulmonary vascular changes, whether primary or secondary, can themselves impair diastolic relaxation of the myocardium globally.8,18,20–22 Additionally, the presence of pulmonary hypertension is associated with progressive structural changes on both the right and left side of the heart.23,24
Pulmonary hypertension as a consequence of left ventricular diastolic dysfunction
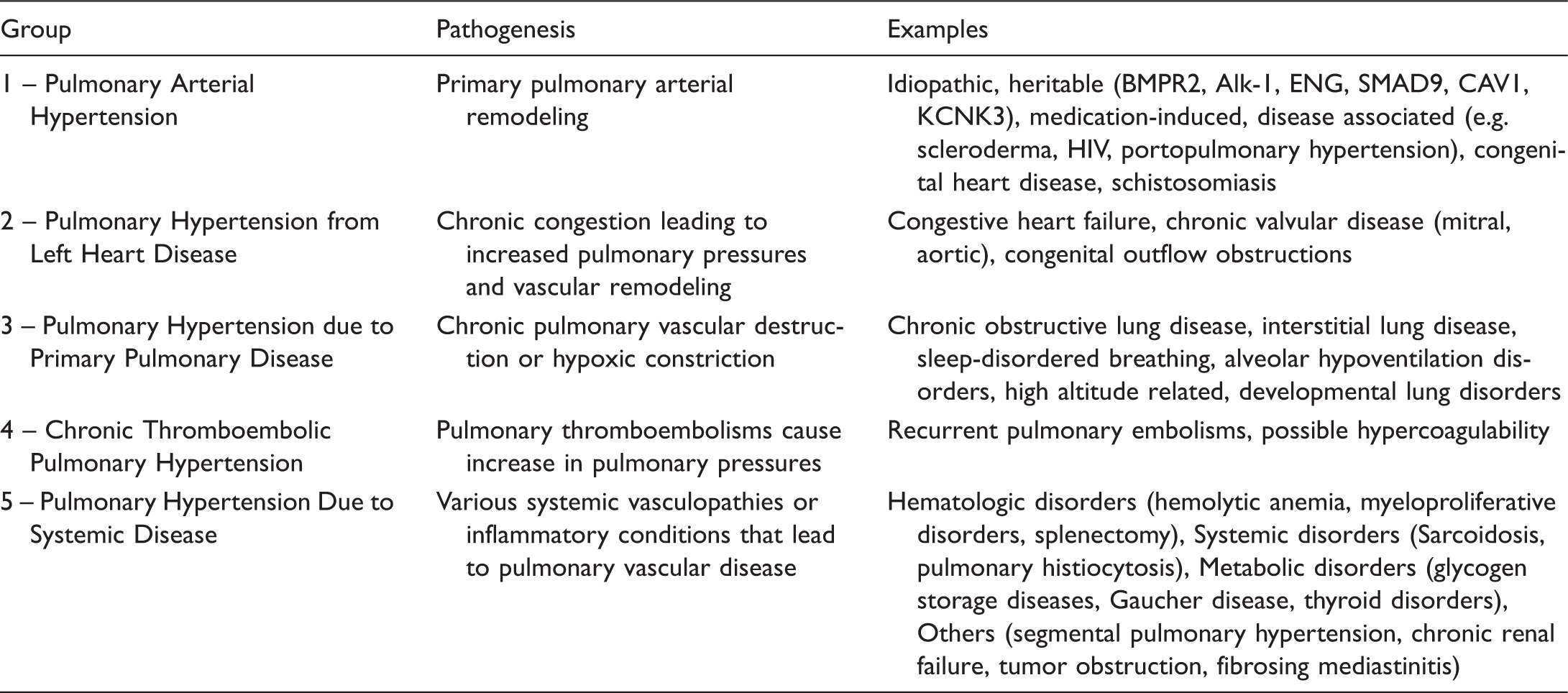
The most commonly understood mechanism by which left ventricular diastolic dysfunction and pulmonary hypertension coexist is in the setting of pulmonary hypertension secondary to left sided heart disease. Pulmonary hypertension is classically divided into five groups by the World Health Organization (WHO) (Table 1),
25
with group 2 representing patients with pulmonary hypertension due to elevated left heart filling pressures that are thought to be the primary cause of pulmonary hypertension. Defined invasively by a measured mean pulmonary artery pressure (PAP) ≥ 25 mmHg and a mean pulmonary arterial wedge pressure (PAWP) > 15 mmHg at rest, group 2 PH patients are further subdivided into patients with isolated post-capillary pulmonary hypertension (Ipc-PH) and combined pre- and post-capillary pulmonary hypertension (Cpc-PH).
26
The latter condition is characterized by the development of pulmonary vascular remodeling leading to increased pulmonary vascular resistance,
27
Group 2 PH is the most prevalent form of pulmonary hypertension in developed countries and is rapidly becoming the most prevalent form of pulmonary hypertension worldwide,11,25,28 and studies utilizing provocative maneuvers such as exercise or fluid challenge with right heart catheterization suggest that the true prevalence of group 2 PH may be underestimated due to misclassification (Figs. 1 and 2).11,12,29 A number of studies have now evaluated the prevalence of occult post-capillary pulmonary hypertension in patients who are initially diagnosed with pulmonary arterial hypertension (PAH). In one study of 287 patients who all underwent fluid challenge for evaluation of pulmonary hypertension, 22% of them were found to have elevated PAWP ≥ 15 mmHg after fluid challenge.
11
In another cohort of 53 patients with scleroderma-associated pulmonary hypertension, 45% of patients had evidence of occult post-capillary pulmonary hypertension after fluid challenge.
30
However, when using a stricter threshold for an abnormal fluid challenge, D’Alto et al. found, in their cohort of 190 patients, that about 7% of patients would be reclassified as having post-capillary pulmonary hypertension.
29
All in all, the presence of patients with occult post-capillary pulmonary hypertension has decreased the accuracy of current estimates of prevalence. Notably, current guidelines do not yet routinely recommend provocative challenge for identifying patients with occult post-capillary pulmonary hypertension.
Right heart catheterization in a patient with pulmonary arterial hypertension shows normal RAP (a), elevated pulmonary pressures (b), normal PAWP at rest (c), and no change in PAWP with fluid challenge (d). The same patient on echocardiogram shows evidence of right atrial and ventricular enlargement, septal bowing, elevated tricuspid regurgitation velocity, normal mitral in-flow velocity pattern, and normal mitral annular tissue doppler velocity patterns (e). Right heart catheterization in a patient with inducible post-capillary pulmonary hypertension shows normal RAP (a), elevated pulmonary pressures (b), and normal PAWP at rest (c). Notably, there is a marked increase in PAWP with fluid challenge (d). Echocardiogram shows evidence of a left ventricle larger than right, left atrial enlargement, normal septal shape, elevated tricuspid regurgitation velocity, abnormal mitral in-flow velocity pattern, and abnormal mitral annular tissue doppler velocity patterns (e). Pulmonary hypertension group classifications, according to the World Health Organization.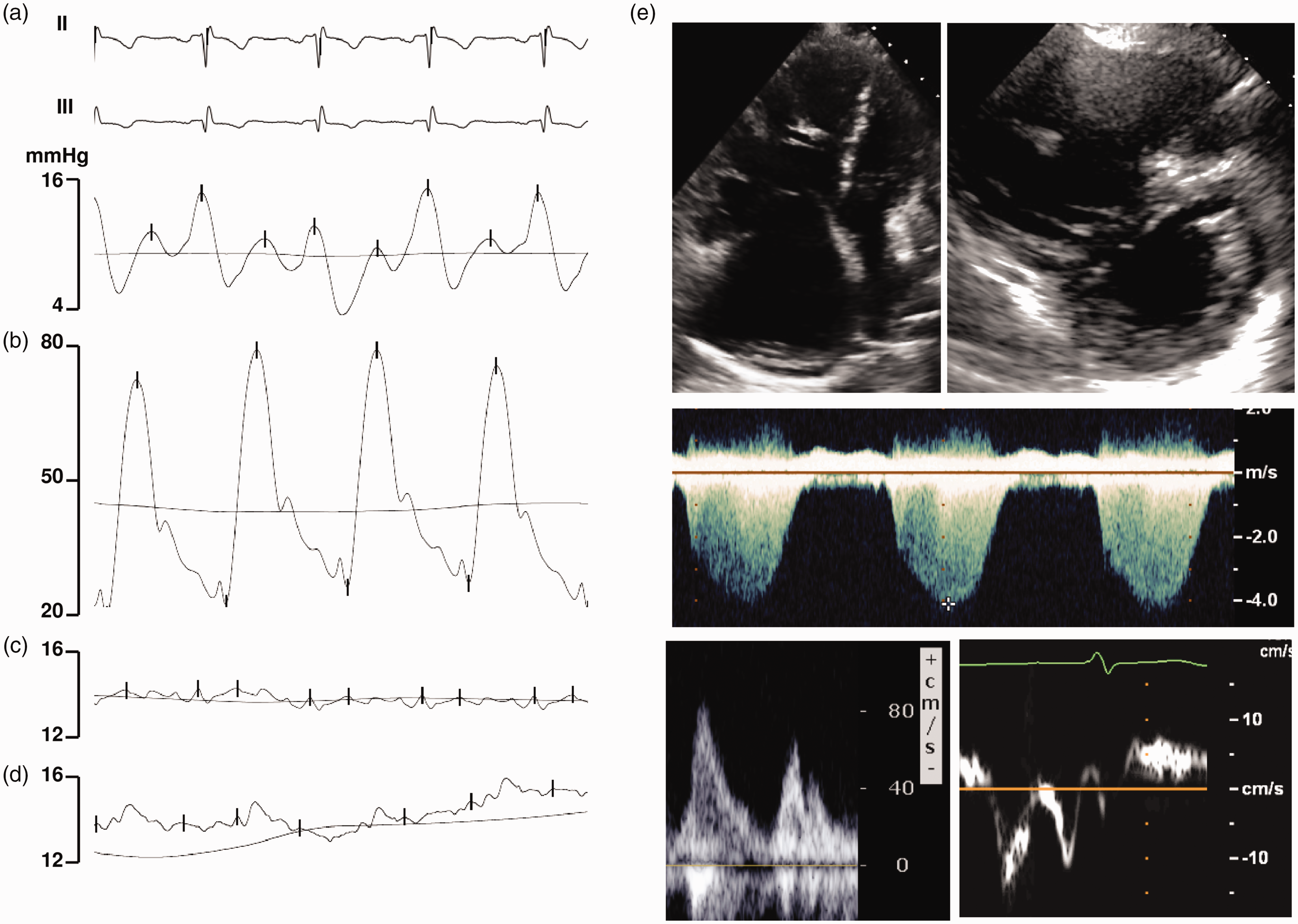
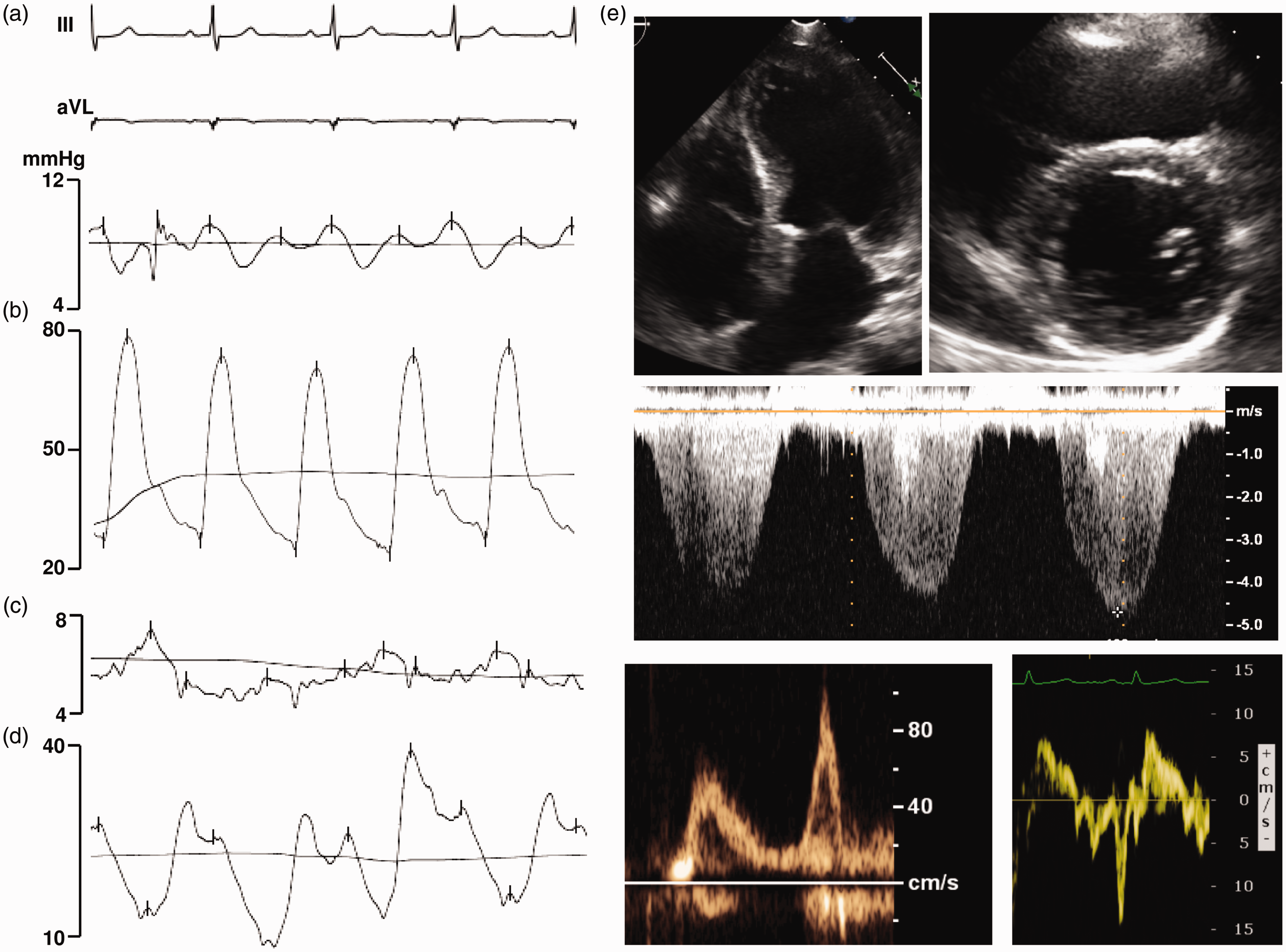
The prevalence is further affected by the accepted threshold of 25 mmHg. There is substantial data suggesting that patients with “borderline” pulmonary hypertension have a higher risk of mortality and hospitalization.31–35 Many of these patients have increased left sided filling pressures by cardiac catheterization, 32 and left atrial and ventricular structural changes, 11 suggesting left ventricular diastolic dysfunction as the driver of pathology.
Impaired left ventricular diastolic filling as a result of PAH
It has been noted that a subset of patients who are diagnosed with PAH also exhibit left ventricular diastolic dysfunction. Two possible mechanisms have been proposed to contribute to this pathogenesis.
One mechanism that has been proposed is impaired diastolic filling of the left ventricle as a consequence of significant pre-capillary pulmonary hypertension. This occurs due to ventricular interdependence, a phenomenon in which left and right ventricular volumes/pressures are interdependent due to the presence of an interventricular septum and pericardial sac that limits volume expansion of the heart.20–22,36–38 Under normal conditions, this interdependence is minimal and without clinical consequences, but significant pulmonary hypertension can significantly impair left ventricular diastolic filling by significantly altering the geometry of the left ventricle due to septal bowing from a pressure and volume overloaded right ventricle (Fig. 3). Notably, impaired ventricular filling as a result of enhanced ventricular interdependence is functionally distinct from intrinsic impaired myocardial relaxation. In addition, prior studies investigating this method have shown up to a 3 mmHg increase in PAWP.14,29,39,40 Thus, while it is important to consider these changes in filling pressures when determining the etiology of pulmonary hypertension, ventricular interdependence may not be the sole cause of significant left ventricular diastolic dysfunction in patients with PAH.
Due to ventricular interdependence of the right and left ventricle, pressure and volume overload in the setting of pulmonary arterial hypertension can lead to impaired diastolic filling of the left ventricle. This results in a pressure volume relationship under normal conditions (1) that shifts to increased pressure for any given volume (2).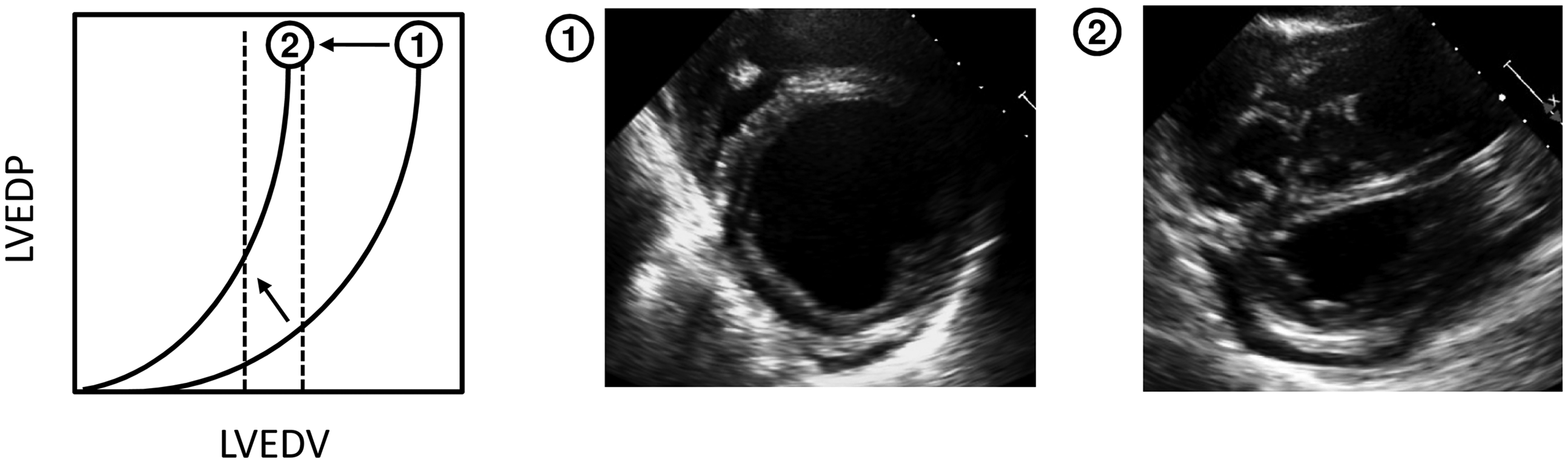
The second mechanism that has been proposed in various studies is common genetic pathogenesis that gives rise to both pulmonary vascular remodeling and cardiomyopathy. This is supported by a number of studies that have found contractile deficits in isolated myocytes from the left ventricle and septum of patients with idiopathic PAH (IPAH) as well as systemic sclerosis-associated PAH.41,42 In addition, in a cohort of patients with IPAH, heritable PAH, connective tissue disease PAH, and chronic thromboembolic disease-related PAH, non-invasive assessment of LV diastolic function by cardiac magnetic resonance imaging (cMRI) suggested global impairment in LV diastolic function in patients with PAH compared with controls. 43 While it is possible that diastolic dysfunction is an acquired change in the LV of patients with PAH, LV diastolic impairment has also been identified in children with PAH by echo. 44 While cardiac manifestations of PAH have been studied in patients in certain forms of PAH, 37 the overlap between diastolic cardiomyopathy and PAH remains incompletely understood.
Non-invasive evaluation of diastolic function by echocardiography
Non-invasive evaluation of diastolic function is possible through multiple imaging modalities. 45 In particular, cardiac MRI is increasingly being utilized for assessment of cardiac structure and function. This has been discussed extensively in a number of recent review articles,46,47 although not studied specifically in the setting of pulmonary hypertension. The present review focuses on echocardiography as it is currently the most commonly used method for assessing diastolic function non-invasively.
Recommendations for echocardiographic determination of diastolic function have recently been updated, with increased emphasis on high specificity of diagnosis and reproducibility of measures.
3
The four recommended variables that are reproducibly associated with diastolic dysfunction are the mitral annular tissue Doppler velocity e’ (septal e’ < 7 cm/sec and/or lateral e’ < 10 cm/sec), left atrial maximal volume index (> 34 ml/m2), average mitral in-flow velocity to annular tissue doppler velocity ratio (E/e’ > 14), and peak tricuspid regurgitant velocity (>2.8 m/s). Notably, the recommended cutoffs are based on prior studies showing high specificity for diastolic dysfunction.
48
Further characterization of the severity, or grade, of diastolic dysfunction is then based on the number of abnormal parameters as well as the E velocity and E/A ratio (Fig. 4).
Determination of diastolic function and left atrial pressure estimation by echocardiogram, adapted from the American Society of Echocardiography 2016 guidelines. LAP: left atrial pressure estimate.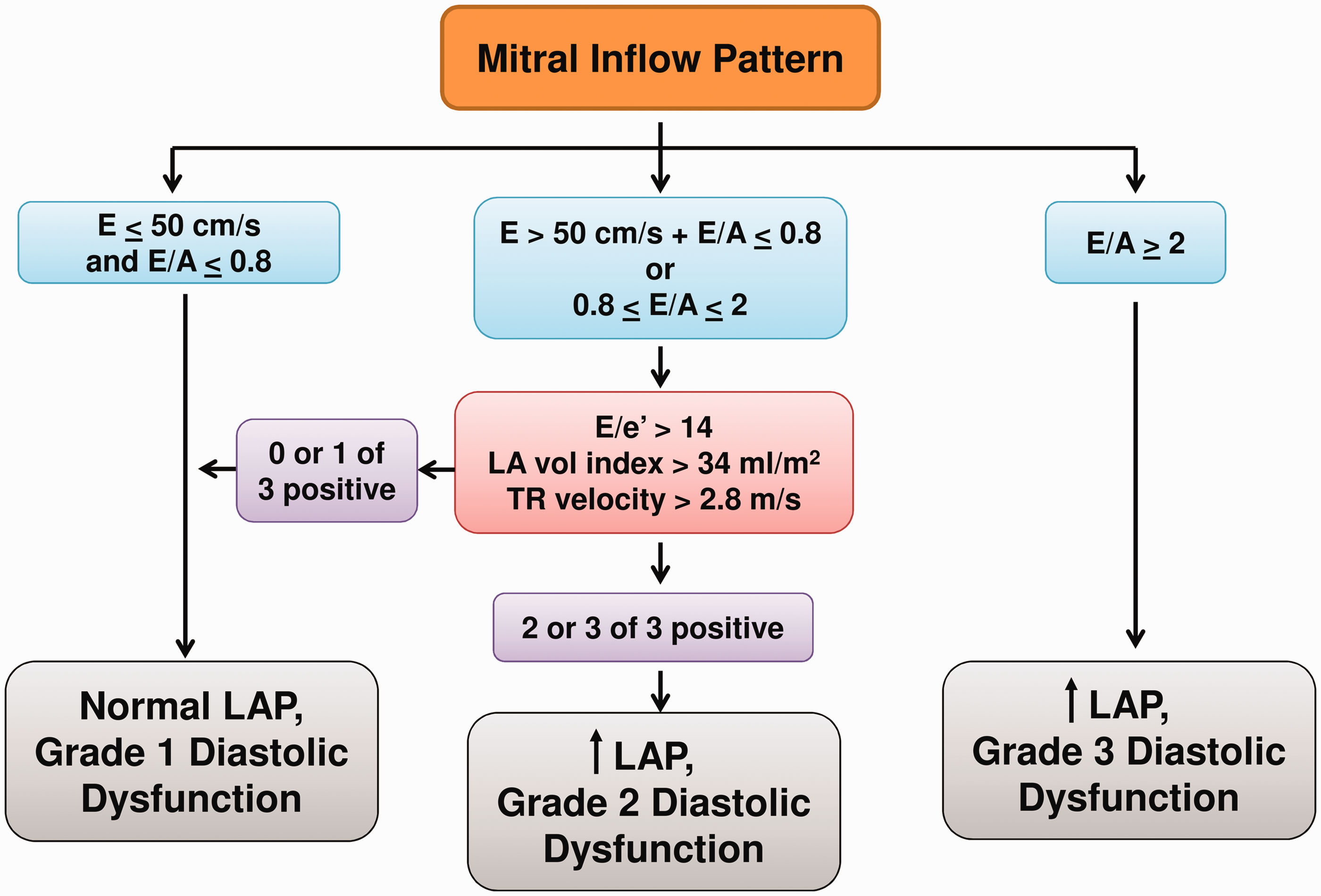
Prior algorithms proposed for non-invasive estimation of diastolic function.
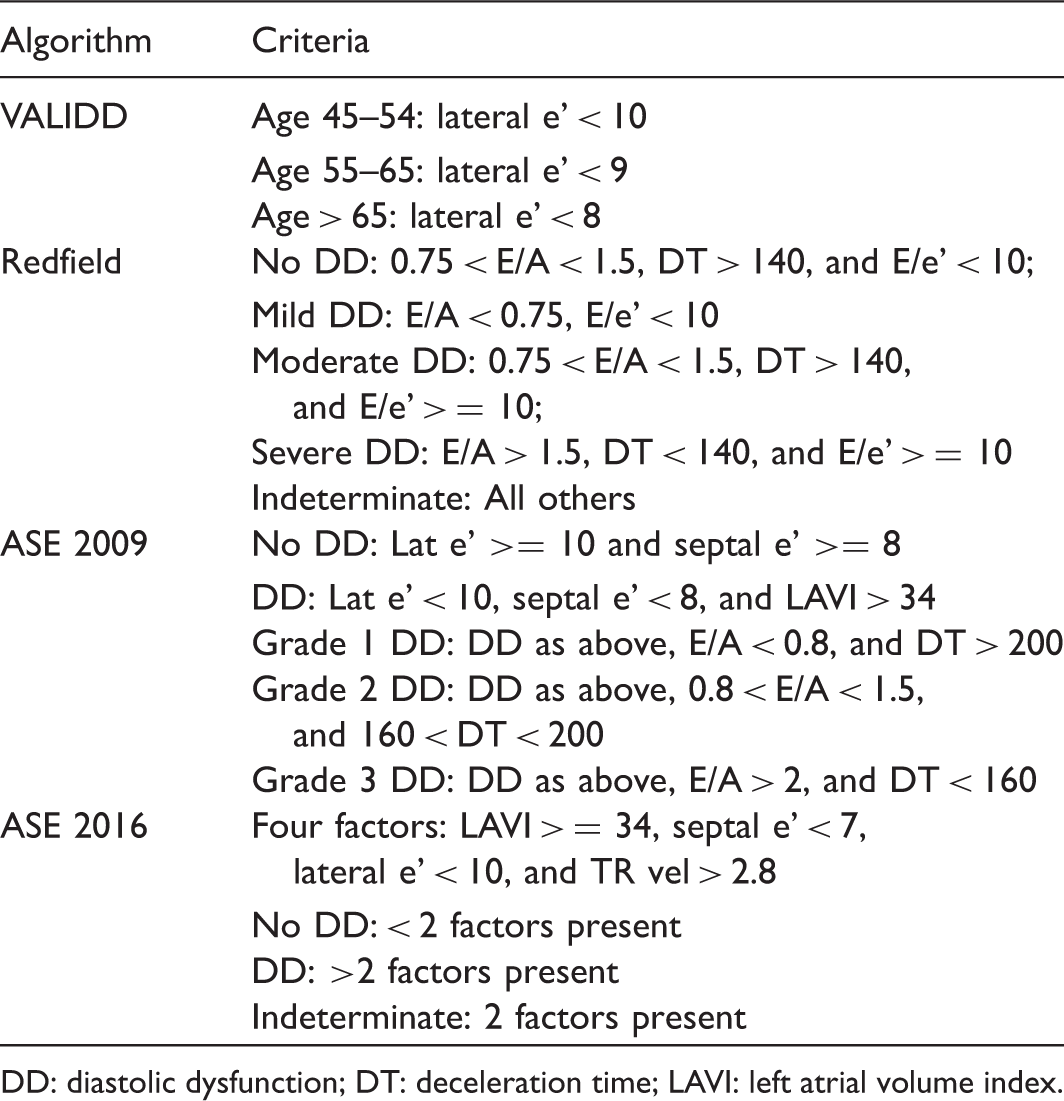
DD: diastolic dysfunction; DT: deceleration time; LAVI: left atrial volume index.
Pitfalls in diastolic function assessment: tricuspid regurgitation velocity
Although used in current algorithms to identify the presence or absence of diastolic dysfunction, tricuspid regurgitant velocity, when detectable, is elevated in all etiologies of pulmonary hypertension, thus limiting its specificity.8,18,20–22,35 The classic approach to non-invasive estimation of pulmonary pressures is the use of a tricuspid regurgitant velocity using Bernoulli’s equation. When added to an estimation of the right atrial pressure by assessment of the inferior vena cava (IVC), this pressure reasonably approximates invasively measured pulmonary artery systolic pressure.59,60 When simultaneously measured and interpreted by readers with high inter-reader reproducibility, the correlation between TR velocity derived and invasively measured PA systolic pressures is high.59,61,62 However, in clinical populations, this correlation declines to 0.6–0.8.63–65 This is thought to be due to a number of pitfalls in the use of tricuspid regurgitant velocity as the sole determinant of pulmonary artery pressures. In conditions of a compensated right ventricle in the setting of high pulmonary and right ventricular pressures, and in conditions of equalization of pressures between the right atrium and right ventricle, tricuspid regurgitant velocity may be artificially low, and may underestimate the degree of pulmonary hypertension. Additionally, the estimation of pulmonary pressures is dependent on accurate assessment of the right atrial pressure by IVC assessment, which has diagnostic limitations. The presence of any primary right sided valvular disease can further affect accurate assessment of pulmonary pressures by this method. Finally, the TR velocity envelope is often incomplete or “cut-off” in severe TR, leading to underestimation of PA systolic pressure.63,65–68
Pitfalls in diastolic function assessment: left atrial pressure estimation in pulmonary hypertension
One of the seminal studies that formulated the basis of the guidelines for assessment of diastolic dysfunction due to pulmonary hypertension studied 70 patients with idiopathic pulmonary hypertension and 35 normal control patients who underwent simultaneous right heart catheterization and echocardiography. 69 The study importantly noted the difference between groups in the ratio of mitral in-flow E velocity and mitral annular tissue velocity e’ (E/e’) if the tissue doppler velocity was measured on the septal or lateral aspect of the mitral annulus. Ruan et al. showed that the septal E/e’ was universally reduced in patients with idiopathic pulmonary hypertension when compared with controls, but lateral E/e’ was no different. 69 They also suggested that lateral E/e’ correlated well with a normal or reduced PAWP, although correlation coefficient was only 0.45. On the basis of this study, the most recent diastolic function guidelines suggest using only lateral E/e’ in the presence of pulmonary hypertension to determine cardiac vs non-cardiac etiology. 3 While a seminal study in the non-invasive evaluation of diastolic function in pulmonary hypertension, this study has a number of limitations. All patients had idiopathic pulmonary hypertension, and their delineation did not account for subsequent observations that a subset of these patients actually have diastolic dysfunction when subjected to provocative maneuvers. 11 In addition, the correlation, which was modest at best based on a coefficient of 0.45, correlated highly with low or normal PAWP. Thus, the study did not shed light on the accuracy of lateral E/e’ in estimating an abnormal left sided filling pressure in pulmonary hypertension.
Recently, Leung et al. has evaluated the accuracy of the 2016 ASE guidelines in estimating elevated resting left sided filling pressures in the setting of suspected pulmonary arterial hypertension. 58 Over 8 years, 94 patients were identified with resting echocardiogram and invasive right heart catheterization within 3 months of each other. The recommend algorithm for estimating elevated left sided filling pressures had a sensitivity of 89.5% and negative predictive value of 81.8% in identifying elevated PAWP, but 100% sensitivity and negative predictive value in identifying elevated LVEDP. However, the algorithm had low specificity (39% for PAWP and 32% for LVEDP). 58 Similar to the study by Ruan and colleagues, Leung et al. relied on resting invasive measurements and did not account for patients in whom provocative maneuvers uncovered occult diastolic dysfunction.11,14 Thus, while promising, future work including further refinement of recommend cutoffs and prospective validation in other cohorts is necessary.
Refined algorithms for distinction of pre- and post-capillary pulmonary hypertension
Various non-invasive algorithms for differentiating pre- and post-capillary pulmonary hypertension.
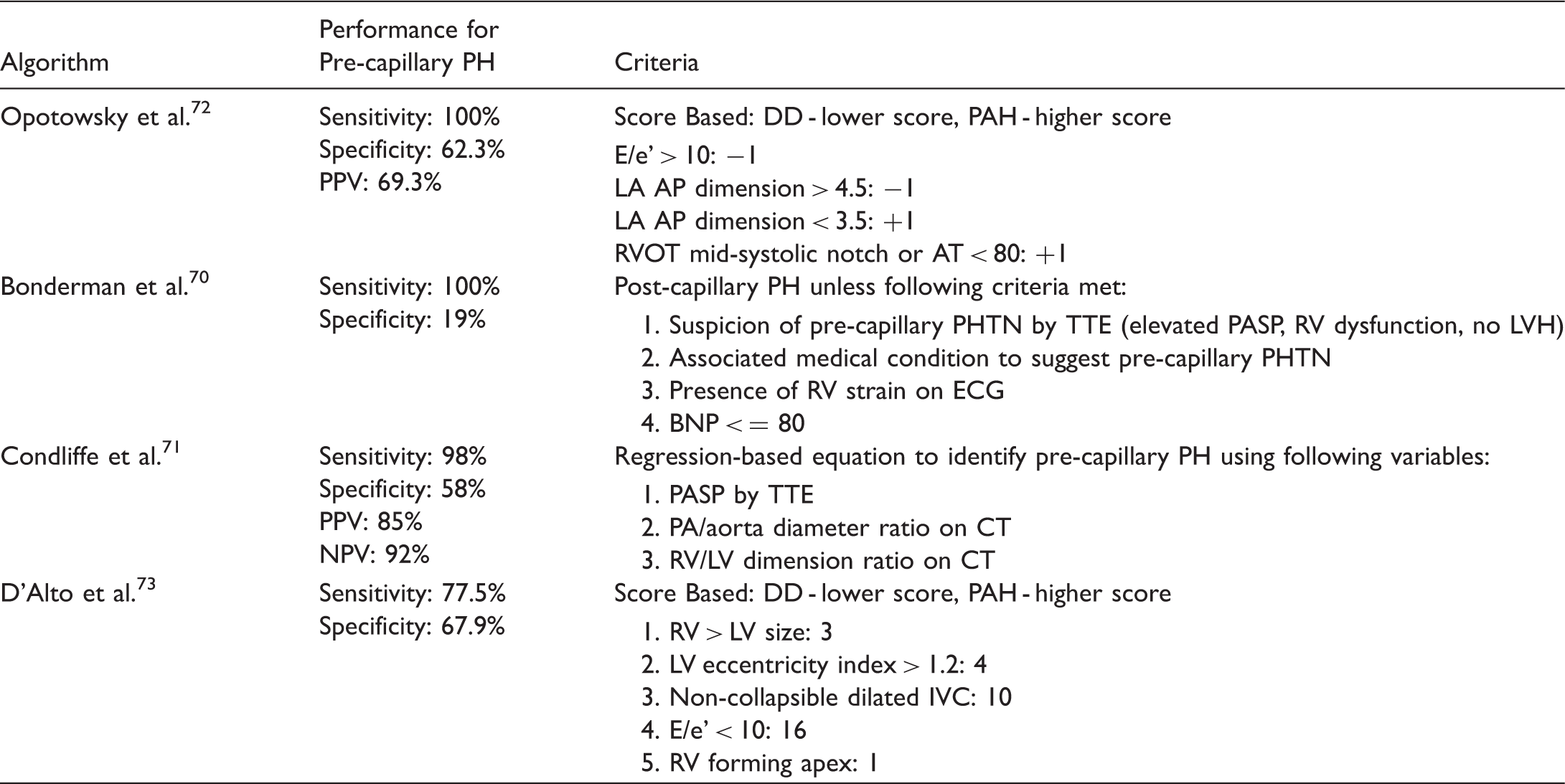
Bonderman et al. identified a non-invasive algorithm that utilized the tricuspid annular plane systolic excursion (TAPSE), visual inspection of right ventricular function, and inferior vena cava (IVC) diameter as echocardiographic distinguishing factors of pulmonary arterial hypertension. 70 When combined with electrocardiographic (ECG) findings of right heart strain and biomarker evidence of elevated NT-pro B-natriuretic peptide levels, the sensitivity for pulmonary arterial hypertension was 100% but specificity was only 19.3%. 70 Condliffe et al. utilized a combination of the tricuspid regurgitant velocity and measured diameters of the RV, LV, aorta, and pulmonary artery by computed tomography (CT) as a method to distinguish pulmonary arterial hypertension in patients with scleroderma. 71
Opotowsky et al. developed a non-invasive algorithm for identifying the subset of patients with elevated pulmonary pressures by echocardiogram that likely have PAH. Using an estimation of the left atrial pressure (LAP) based on E/e’ ratio, left atrial (LA) dimension, pulmonary artery acceleration time, and the presence or absence of mid-systolic notching in the right ventricular outflow tract (RVOT) pulse wave doppler signal, they were able to identify patients with PAH with 100% sensitivity and 62.3% specificity. 72
An alternative non-invasive algorithm using echocardiography reported by D’Alto et al. used the identification of the apex forming ventricle, left ventricle eccentricity index, the presence or absence of a dilated IVC, and E/e’ ratio to predict pre- vs post-capillary pulmonary hypertension. 73 Based on these factors, D’Alto et al. reported a sensitivity of 77.5% and specificity of 67.9% for pulmonary arterial hypertension. When compared with the Opotowsky algorithm within their cohort of patients, D’Alto’s algorithm showed improved performance (area under ROC 0.756 vs 0.645, P = 0.0021). 73
Notably, many of the listed algorithms focused on structural echocardiographic findings and did not rely on variables used in the measure of diastolic function, with the exception of measures of E/e’ and left atrial dimension. Many of the measured variables, such as the determination of RV forming apex and eccentricity index, are not routine measures made in everyday clinical practice and may limit the widespread adoption of these algorithms into everyday practice. Most importantly, the algorithms above focus on identifying the presence of resting pulmonary arterial hypertension. As a significant subset of patients identified initially as pulmonary arterial hypertension likely have inducible post-capillary pulmonary hypertension upon provocative testing, more investigation will be needed to evaluate these and other algorithms in the growing cohort of patients with inducible pulmonary hypertension.11,12
Emerging modalities for assessment of diastolic function: strain imaging and diastolic stress testing
Strain imaging is increasingly used for assessment of subclinical cardiac dysfunction in various patient populations.74–76 There has also been an emerging role for strain imaging in quantifying and identifying regions of diastolic function in the myocardium. 77 Measure of strain by ultrasonography is thought to be a surrogate measure of deformation, and strain rate is a surrogate measure of rate of deformation. In a study by Kasner et al. of 33 patients who underwent strain imaging echocardiography and invasive catheterization for pressure-volume loop analysis simultaneously, LV global strain rate was reduced in patients with diastolic dysfunction compared with controls. Additionally, LV global strain rate during early diastole and isovolumic relaxation correlated with invasively measured left ventricular relaxation constant τ, LV end diastolic pressure, and LV stiffness constant β. 77 Strain imaging in pulmonary hypertension has been used to study the mechanics of the right ventricle and response to therapy,78–80 but this technology has not been used to study the left ventricle or distinguish pre- vs post-capillary pulmonary hypertension in the literature.
Similarly, strain imaging has been utilized to study atrial mechanics as a method by which to identify patients with abnormal cardiovascular hemodynamics. In a patient study of 69 patients, which included both patients in sinus rhythm and with atrial fibrillation, speckle tracking echocardiography (STE) was utilized to evaluate contractile function of the left atrium and compared with invasive left and right heart catheterization-derived measurements of intracardiac pressures. 81 While no differences were noted in patients with atrial fibrillation, atrial peak positive strain of less than 16% in patients in sinus rhythm was significantly predictive of a PAWP ≥ 18 mmHg, and more predictive than non-invasive estimates such as E/e’. This has further been studied in 49 patients, 45 of whom had pulmonary hypertension (22 of them with pulmonary arterial hypertension, 19 with left sided heart disease as the cause of pulmonary hypertension, 1 with chronic thromboembolic pulmonary hypertension). 82 Left atrial strain rate was used to derive a non-invasive estimate of PAWP, and the resulting PVR correlated strongly with catheter-derived measurements (sensitivity 85%, specificity 74%), suggesting that left atrial strain can be a useful measure of left sided filling pressures and pulmonary vascular resistance. While very promising, current limitations of this technology remain the lack of uniform algorithms for measuring strain as well as a lack of consensus on various cutoffs for determining normal vs abnormal strain.
Based on previous studies showing that provocative maneuvers such as fluid challenge or exercise can identify patients with occult post-capillary pulmonary hypertension that otherwise would have been classified as having pulmonary arterial hypertension,11,12 non-invasive stress testing for evaluation of diastolic function has previously been studied in the setting of pulmonary hypertension. 7 Studies have shown that stress, via modalities such as exercise, can elicit changes in estimated pulmonary artery pressures, 83 tricuspid regurgitation, right and left ventricular functional changes, 84 and changes in Doppler based estimates of left ventricular filling pressures. 85 However, the application of this approach to the distinction of pre- and post-capillary pulmonary hypertension remains an area of investigation. Limitations of this technology remain technical challenges in obtaining accurate data and feasibility of upscaling this technology to serve as a broad screening tool for all patients with pulmonary hypertension.
Conclusions
Distinction of pulmonary hypertension due to left ventricular diastolic dysfunction and pulmonary arterial hypertension has both prognostic and therapeutic implications. Non-invasive methods to differentiate these sub populations offers the opportunity for better screening and monitoring of therapeutic efficacy without the risks of invasive procedures. Despite multiple non-invasive algorithms proposed for identification for distinguish pre- and post-capillary pulmonary hypertension, future studies are necessary to evaluate these algorithms in the growing subset of patients who have evidence of diastolic dysfunction only after provocation. With the growing prevalence of pulmonary hypertension worldwide and the increasing therapeutic implications of identification of the subtype of pulmonary hypertension in patients, reliable non-invasive identification of diastolic dysfunction in pulmonary hypertension may have profound impact on diagnosis and management.
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
National Institutes of Health T32-HL007411 (Agrawal). American Heart Association 13FTF16070002 (Brittain).
